These Highlights Do Not Include All The Information Needed To Use Sildenafil Safely And Effectively. See Full Prescribing Information For Sildenafil.
56a962e8-de00-4a97-8904-0b1bd217dd83
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Sildenafil is indicated for the treatment of pulmonary arterial hypertension (WHO Group I) in adults to improve exercise ability and delay clinical worsening. The delay in clinical worsening was demonstrated when sildenafil was added to background epoprostenol therapy [see Clinical Studies (14) ] . Studies establishing effectiveness were short-term (12 to 16 weeks), and included predominately patients with New York Heart Association (NYHA) Functional Class II–III symptoms and idiopathic etiology (71%) or associated with connective tissue disease (CTD) (25%).
Indications and Usage
Sildenafil is indicated for the treatment of pulmonary arterial hypertension (WHO Group I) in adults to improve exercise ability and delay clinical worsening. The delay in clinical worsening was demonstrated when sildenafil was added to background epoprostenol therapy [see Clinical Studies (14) ] . Studies establishing effectiveness were short-term (12 to 16 weeks), and included predominately patients with New York Heart Association (NYHA) Functional Class II–III symptoms and idiopathic etiology (71%) or associated with connective tissue disease (CTD) (25%).
Dosage and Administration
1. Tablets and oral suspension: 5 mg or 20 mg three times a day, 4–6 hours apart; see full prescribing information for reconstitution instructions for the Powder for Oral Suspension ( 2.1 , 2.2 )
Warnings and Precautions
• Increased mortality with increasing doses in pediatric patients. Not recommended for use in pediatric patients. ( 5.1 ) • Vasodilation effects may be more common in patients with hypotension or on antihypertensive therapy. ( 5.2 ) • Use in pulmonary veno-occlusive disease may cause pulmonary edema and is not recommended. ( 5.3 ) • Hearing or visual impairment: Seek medical attention if sudden decrease or loss of vision or hearing occurs. ( 5.5 , 5.6 ) • Pulmonary hypertension secondary to sickle cell disease: sildenafil may cause serious vaso-occlusive crises. ( 5.9 )
Contraindications
Sildenafil is contraindicated in patients with: • Concomitant use of organic nitrates in any form, either regularly or intermittently, because of the greater risk of hypotension [see Warnings and Precautions (5.2) ] . • Concomitant use of riociguat, a guanylate cyclase stimulator. PDE-5 inhibitors, including sildenafil, may potentiate the hypotensive effects of riociguat. • Known hypersensitivity to sildenafil or any component of the tablet or oral suspension. Hypersensitivity, including anaphylactic reaction, anaphylactic shock and anaphylactoid reaction, has been reported in association with the use of sildenafil.
Adverse Reactions
The following serious adverse events are discussed elsewhere in the labeling: • Mortality with pediatric use [see Warnings and Precautions (5.1) and Use in Specific Populations (8.4) ] • Hypotension [see Warnings and Precautions (5.2) ] • Vision loss [see Warnings and Precautions (5.5) ] • Hearing loss [see Warnings and Precautions (5.6) ] • Priapism [see Warnings and Precautions (5.8) ] • Vaso-occlusive crisis [see Warnings and Precautions (5.9) ]
Drug Interactions
• Concomitant alpha-blockers or amlodipine: Note additive blood pressure lowering effects. ( 7 ) • Use with ritonavir and other potent CYP3A inhibitors: Not recommended. ( 7 , 12.3 ) • Concomitant PDE-5 inhibitors: Avoid use with Viagra or other PDE-5 inhibitors. ( 5.7 )
Patient Information
Sildenafil tablets Sildenafil oral suspension Read this Patient Information before you start taking sildenafil and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or treatment. If you have any questions about sildenafil, ask your doctor or pharmacist. What is the most important information I should know about sildenafil? Never take sildenafil with any nitrate or guanylate cyclase stimulator medicines. Your blood pressure could drop quickly to an unsafe level. Nitrate medicines include: • Medicines that treat chest pain (angina) • Nitroglycerin in any form including tablets, patches, sprays, and ointments • Isosorbide mononitrate or dinitrate • Street drugs called "poppers" (amyl nitrate or nitrite) Guanylate cyclase stimulators include: 1. Riociguat (Adempas) Ask your doctor or pharmacist if you are not sure if you are taking a nitrate or a guanylate cyclase stimulator medicine. What is sildenafil? Sildenafil is a prescription medicine used in adults to treat pulmonary arterial hypertension (PAH). With PAH, the blood pressure in your lungs is too high. Your heart has to work hard to pump blood into your lungs. Sildenafil improves the ability to exercise and can slow down worsening changes in your physical condition. • Sildenafil is not for use in children • Adding sildenafil to another medication used to treat PAH bosentan (Tracleer ® ), does not result in improvement in your ability to exercise. Sildenafil contains the same medicine as VIAGRA ® (sildenafil), which is used to treat erectile dysfunction (impotence). Do not take sildenafil with VIAGRA or other PDE-5 inhibitors. Who should not take Sildenafil? Do not take sildenafil if you: • take nitrate medicines. See " What is the most important information I should know about sildenafil? " • take guanylate cyclase stimulator medicines. See " What is the most important information I should know about sildenafil? " • are allergic to sildenafil or any other ingredient in sildenafil tablets or oral suspension. See " What are the ingredients in sildenafil tablets and oral suspension? " at the end of this leaflet. What should I tell my doctor before taking sildenafil? Tell your doctor about all of your medical conditions, including if you • have heart problems such as angina (chest pain), heart failure, irregular heartbeats, or have had a heart attack • have a disease called pulmonary veno-occlusive disease (PVOD) • have high or low blood pressure or blood circulation problems • have an eye problem called retinitis pigmentosa • have or had loss of sight in one or both eyes • have any problem with the shape of your penis or Peyronie's disease • have any blood cell problems such as sickle cell anemia • have a stomach ulcer or any bleeding problems • are pregnant or planning to become pregnant. It is not known if sildenafil could harm your unborn baby. • are breastfeeding. Sildenafil passes into your breast milk, it is not known if it could harm your baby. Tell your doctor about all of the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal products. Sildenafil and certain other medicines can cause side effects if you take them together. The doses of some of your medicines may need to be adjusted while you take sildenafil. Especially tell your doctor if you take • Nitrate medicines. See " What is the most important information I should know about sildenafil? " • Riociguat (Adempas). See " What is the most important information I should know about sildenafil? " • Ritonavir (Norvir ® ) or other medicines used to treat HIV infection • Ketoconazole (Nizoral ® ) • Itraconazole (Sporanox) • High blood pressure medicine Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist when you get a new medicine. How should I take sildenafil? 2. Take sildenafil exactly as your doctor tells you. Sildenafil may be prescribed to you as • Sildenafil tablets or sildenafil oral suspension • Take sildenafil tablet or oral suspension 3 times a day about 4 to 6 hours apart. • Take sildenafil tablets or oral suspension at the same times every day. • Sildenafil oral suspension will be mixed for you by your pharmacist. Do not mix sildenafil oral suspension with other medicine or flavoring. Shake well for at least 10 seconds before each dose. • If you miss a dose, take it as soon as you remember. If it is close to your next dose, skip the missed dose, and take your next dose at the regular time. • Do not take more than one dose of sildenafil at a time. • Do not change your dose or stop taking sildenafil on your own. Talk to your doctor first. • If you take too much sildenafil, call your doctor or go to the nearest hospital emergency room. What are the possible side effects of sildenafil? • low blood pressure. Low blood pressure may cause you to feel faint or dizzy. Lie down if you feel faint or dizzy. • more shortness of breath than usual. Tell your doctor if you get more short of breath after you start sildenafil. More shortness of breath than usual may be due to your underlying medical condition. • decreased eyesight or loss of sight in one or both eyes (NAION). If you notice a sudden decrease or loss of eyesight, talk to your doctor right away. • sudden decrease or loss of hearing. If you notice a sudden decrease or loss of hearing, talk to your doctor right away. It is not possible to determine whether these events are related directly to this class of oral medicines, including sildenafil, or to other diseases or medicines, to other factors, or to a combination of factors. • heart attack, stroke, irregular heartbeats, and death. Most of these happened in men who already had heart problems. • erections that last several hours. If you have an erection that lasts more than 4 hours, get medical help right away. If it is not treated right away, priapism can permanently damage your penis. The most common side effects with sildenafil include: Nosebleed, headache, upset stomach, getting red or hot in the face (flushing), trouble sleeping, as well as fever, erection increased, respiratory infection, nausea, vomiting, bronchitis, pharyngitis, runny nose, and pneumonia in children. Tell your doctor if you have any side effect that bothers you or doesn't go away. These are not all the possible side effects of sildenafil. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. How should I store sildenafil? • Store sildenafil tablets at controlled room temperature, between 20°C – 25°C (68°F to 77°F). • Store sildenafil reconstituted oral suspension below 30°C (86°F) or in a refrigerator between 2°C to 8°C (36°F to 46°F). • Do not freeze sildenafil oral suspension. • Throw away sildenafil oral suspension after 60 days. • Keep sildenafil and all medicines away from children. General information about sildenafil Medicines are sometimes prescribed for purposes that are not in the patient leaflet. Do not use sildenafil for a condition for which it was not prescribed. Do not give sildenafil to other people, even if they have the same symptoms you have. It could harm them. This patient leaflet summarizes the most important information about sildenafil. If you would like more information about sildenafil talk with your doctor. You can ask your doctor or pharmacist for information about sildenafil that is written for health professionals. For more information go to www.greenstonellc.com or call 1-800-438-1985. What are the ingredients in sildenafil tablets and oral suspension? Sildenafil tablets Active ingredients: sildenafil citrate Inactive ingredients: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, magnesium stearate, hypromellose, titanium dioxide, lactose monohydrate, and triacetin Sildenafil for oral suspension Active ingredients: sildenafil citrate Inactive ingredients: sorbitol, citric acid anhydrous, sucralose, sodium citrate dihydrate, xanthan gum, titanium dioxide, sodium benzoate, colloidal silicon dioxide anhydrous, and grape flavor This product's label may have been updated. For current full prescribing information, please visit www.greenstonellc.com. This Patient Information has been approved by the U.S. Food and Drug Administration. LAB-0563-7.0 Revised: February 2020
Medication Information
Warnings and Precautions
• Increased mortality with increasing doses in pediatric patients. Not recommended for use in pediatric patients. ( 5.1 ) • Vasodilation effects may be more common in patients with hypotension or on antihypertensive therapy. ( 5.2 ) • Use in pulmonary veno-occlusive disease may cause pulmonary edema and is not recommended. ( 5.3 ) • Hearing or visual impairment: Seek medical attention if sudden decrease or loss of vision or hearing occurs. ( 5.5 , 5.6 ) • Pulmonary hypertension secondary to sickle cell disease: sildenafil may cause serious vaso-occlusive crises. ( 5.9 )
Indications and Usage
Sildenafil is indicated for the treatment of pulmonary arterial hypertension (WHO Group I) in adults to improve exercise ability and delay clinical worsening. The delay in clinical worsening was demonstrated when sildenafil was added to background epoprostenol therapy [see Clinical Studies (14) ] . Studies establishing effectiveness were short-term (12 to 16 weeks), and included predominately patients with New York Heart Association (NYHA) Functional Class II–III symptoms and idiopathic etiology (71%) or associated with connective tissue disease (CTD) (25%).
Dosage and Administration
1. Tablets and oral suspension: 5 mg or 20 mg three times a day, 4–6 hours apart; see full prescribing information for reconstitution instructions for the Powder for Oral Suspension ( 2.1 , 2.2 )
Contraindications
Sildenafil is contraindicated in patients with: • Concomitant use of organic nitrates in any form, either regularly or intermittently, because of the greater risk of hypotension [see Warnings and Precautions (5.2) ] . • Concomitant use of riociguat, a guanylate cyclase stimulator. PDE-5 inhibitors, including sildenafil, may potentiate the hypotensive effects of riociguat. • Known hypersensitivity to sildenafil or any component of the tablet or oral suspension. Hypersensitivity, including anaphylactic reaction, anaphylactic shock and anaphylactoid reaction, has been reported in association with the use of sildenafil.
Adverse Reactions
The following serious adverse events are discussed elsewhere in the labeling: • Mortality with pediatric use [see Warnings and Precautions (5.1) and Use in Specific Populations (8.4) ] • Hypotension [see Warnings and Precautions (5.2) ] • Vision loss [see Warnings and Precautions (5.5) ] • Hearing loss [see Warnings and Precautions (5.6) ] • Priapism [see Warnings and Precautions (5.8) ] • Vaso-occlusive crisis [see Warnings and Precautions (5.9) ]
Drug Interactions
• Concomitant alpha-blockers or amlodipine: Note additive blood pressure lowering effects. ( 7 ) • Use with ritonavir and other potent CYP3A inhibitors: Not recommended. ( 7 , 12.3 ) • Concomitant PDE-5 inhibitors: Avoid use with Viagra or other PDE-5 inhibitors. ( 5.7 )
Patient Information
Sildenafil tablets
Sildenafil oral suspension
Read this Patient Information before you start taking sildenafil and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or treatment. If you have any questions about sildenafil, ask your doctor or pharmacist.
What is the most important information I should know about sildenafil?
Never take sildenafil with any nitrate or guanylate cyclase stimulator medicines. Your blood pressure could drop quickly to an unsafe level.
Nitrate medicines include:
-
•Medicines that treat chest pain (angina)
-
•Nitroglycerin in any form including tablets, patches, sprays, and ointments
-
•Isosorbide mononitrate or dinitrate
-
•Street drugs called "poppers" (amyl nitrate or nitrite)
Guanylate cyclase stimulators include:
-
1.Riociguat (Adempas)
Ask your doctor or pharmacist if you are not sure if you are taking a nitrate or a guanylate cyclase stimulator medicine.
What is sildenafil?
Sildenafil is a prescription medicine used in adults to treat pulmonary arterial hypertension (PAH). With PAH, the blood pressure in your lungs is too high. Your heart has to work hard to pump blood into your lungs.
Sildenafil improves the ability to exercise and can slow down worsening changes in your physical condition.
-
•Sildenafil is not for use in children
-
•Adding sildenafil to another medication used to treat PAH bosentan (Tracleer®), does not result in improvement in your ability to exercise.
Sildenafil contains the same medicine as VIAGRA® (sildenafil), which is used to treat erectile dysfunction (impotence). Do not take sildenafil with VIAGRA or other PDE-5 inhibitors.
Who should not take Sildenafil?
Do not take sildenafil if you:
-
•take nitrate medicines. See " What is the most important information I should know about sildenafil? "
-
•take guanylate cyclase stimulator medicines. See " What is the most important information I should know about sildenafil? "
-
•are allergic to sildenafil or any other ingredient in sildenafil tablets or oral suspension. See " What are the ingredients in sildenafil tablets and oral suspension?" at the end of this leaflet.
What should I tell my doctor before taking sildenafil?
Tell your doctor about all of your medical conditions, including if you
-
•have heart problems such as angina (chest pain), heart failure, irregular heartbeats, or have had a heart attack
-
•have a disease called pulmonary veno-occlusive disease (PVOD)
-
•have high or low blood pressure or blood circulation problems
-
•have an eye problem called retinitis pigmentosa
-
•have or had loss of sight in one or both eyes
-
•have any problem with the shape of your penis or Peyronie's disease
-
•have any blood cell problems such as sickle cell anemia
-
•have a stomach ulcer or any bleeding problems
-
•are pregnant or planning to become pregnant. It is not known if sildenafil could harm your unborn baby.
-
•are breastfeeding. Sildenafil passes into your breast milk, it is not known if it could harm your baby.
Tell your doctor about all of the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal products. Sildenafil and certain other medicines can cause side effects if you take them together. The doses of some of your medicines may need to be adjusted while you take sildenafil.
Especially tell your doctor if you take
-
•Nitrate medicines. See " What is the most important information I should know about sildenafil? "
-
•Riociguat (Adempas). See " What is the most important information I should know about sildenafil? "
-
•Ritonavir (Norvir®) or other medicines used to treat HIV infection
-
•Ketoconazole (Nizoral®)
-
•Itraconazole (Sporanox)
-
•High blood pressure medicine
Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist when you get a new medicine.
How should I take sildenafil?
-
2.Take sildenafil exactly as your doctor tells you.
Sildenafil may be prescribed to you as
-
•Sildenafil tablets or sildenafil oral suspension
-
•Take sildenafil tablet or oral suspension 3 times a day about 4 to 6 hours apart.
-
•Take sildenafil tablets or oral suspension at the same times every day.
-
•Sildenafil oral suspension will be mixed for you by your pharmacist. Do not mix sildenafil oral suspension with other medicine or flavoring. Shake well for at least 10 seconds before each dose.
-
•If you miss a dose, take it as soon as you remember. If it is close to your next dose, skip the missed dose, and take your next dose at the regular time.
-
•Do not take more than one dose of sildenafil at a time.
-
•Do not change your dose or stop taking sildenafil on your own. Talk to your doctor first.
-
•If you take too much sildenafil, call your doctor or go to the nearest hospital emergency room.
What are the possible side effects of sildenafil?
-
•low blood pressure. Low blood pressure may cause you to feel faint or dizzy. Lie down if you feel faint or dizzy.
-
•more shortness of breath than usual. Tell your doctor if you get more short of breath after you start sildenafil. More shortness of breath than usual may be due to your underlying medical condition.
-
•decreased eyesight or loss of sight in one or both eyes (NAION). If you notice a sudden decrease or loss of eyesight, talk to your doctor right away.
-
•sudden decrease or loss of hearing. If you notice a sudden decrease or loss of hearing, talk to your doctor right away. It is not possible to determine whether these events are related directly to this class of oral medicines, including sildenafil, or to other diseases or medicines, to other factors, or to a combination of factors.
-
•heart attack, stroke, irregular heartbeats, and death. Most of these happened in men who already had heart problems.
-
•erections that last several hours. If you have an erection that lasts more than 4 hours, get medical help right away. If it is not treated right away, priapism can permanently damage your penis.
The most common side effects with sildenafil include:
Nosebleed, headache, upset stomach, getting red or hot in the face (flushing), trouble sleeping, as well as fever, erection increased, respiratory infection, nausea, vomiting, bronchitis, pharyngitis, runny nose, and pneumonia in children.
Tell your doctor if you have any side effect that bothers you or doesn't go away.
These are not all the possible side effects of sildenafil. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store sildenafil?
-
•Store sildenafil tablets at controlled room temperature, between 20°C – 25°C (68°F to 77°F).
-
•Store sildenafil reconstituted oral suspension below 30°C (86°F) or in a refrigerator between 2°C to 8°C (36°F to 46°F).
-
•Do not freeze sildenafil oral suspension.
-
•Throw away sildenafil oral suspension after 60 days.
-
•Keep sildenafil and all medicines away from children.
General information about sildenafil
Medicines are sometimes prescribed for purposes that are not in the patient leaflet. Do not use sildenafil for a condition for which it was not prescribed. Do not give sildenafil to other people, even if they have the same symptoms you have. It could harm them.
This patient leaflet summarizes the most important information about sildenafil. If you would like more information about sildenafil talk with your doctor. You can ask your doctor or pharmacist for information about sildenafil that is written for health professionals.
For more information go to www.greenstonellc.com or call 1-800-438-1985.
What are the ingredients in sildenafil tablets and oral suspension?
Sildenafil tablets
Active ingredients: sildenafil citrate
Inactive ingredients: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, magnesium stearate, hypromellose, titanium dioxide, lactose monohydrate, and triacetin
Sildenafil for oral suspension
Active ingredients: sildenafil citrate
Inactive ingredients: sorbitol, citric acid anhydrous, sucralose, sodium citrate dihydrate, xanthan gum, titanium dioxide, sodium benzoate, colloidal silicon dioxide anhydrous, and grape flavor
This product's label may have been updated. For current full prescribing information, please visit www.greenstonellc.com.
This Patient Information has been approved by the U.S. Food and Drug Administration.
LAB-0563-7.0
Revised: February 2020
Description
Sildenafil is indicated for the treatment of pulmonary arterial hypertension (WHO Group I) in adults to improve exercise ability and delay clinical worsening. The delay in clinical worsening was demonstrated when sildenafil was added to background epoprostenol therapy [see Clinical Studies (14) ] . Studies establishing effectiveness were short-term (12 to 16 weeks), and included predominately patients with New York Heart Association (NYHA) Functional Class II–III symptoms and idiopathic etiology (71%) or associated with connective tissue disease (CTD) (25%).
Section 42229-5
Limitation of Use: Adding sildenafil to bosentan therapy does not result in any beneficial effect on exercise capacity [see Clinical Studies (14)].
Section 44425-7
Recommended Storage for Sildenafil Tablets: Store at controlled room temperature 20°C – 25°C (68°F – 77°F); excursions permitted to 15°C – 30°C (59°F – 86°F) [see USP Controlled Room Temperature].
5.8 Priapism
Use sildenafil with caution in patients with anatomical deformation of the penis (e.g., angulation, cavernosal fibrosis, or Peyronie's disease) or in patients who have conditions, which may predispose them to priapism (e.g., sickle cell anemia, multiple myeloma, or leukemia). In the event of an erection that persists longer than 4 hours, the patient should seek immediate medical assistance. If priapism (painful erection greater than 6 hours in duration) is not treated immediately, penile tissue damage and permanent loss of potency could result.
10 Overdosage
In studies with healthy volunteers of single doses up to 800 mg, adverse events were similar to those seen at lower doses but rates and severities were increased.
In cases of overdose, standard supportive measures should be adopted as required. Renal dialysis is not expected to accelerate clearance as sildenafil is highly bound to plasma proteins and it is not eliminated in the urine.
5.4 Epistaxis
The incidence of epistaxis was 13% in patients taking sildenafil with PAH secondary to CTD. This effect was not seen in idiopathic PAH (sildenafil 3%, placebo 2%) patients. The incidence of epistaxis was also higher in sildenafil-treated patients with a concomitant oral vitamin K antagonist (9% versus 2% in those not treated with concomitant vitamin K antagonist).
The safety of sildenafil is unknown in patients with bleeding disorders or active peptic ulceration.
11 Description
Sildenafil, phosphodiesterase-5 (PDE-5) inhibitor, is the citrate salt of sildenafil, a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type-5 (PDE-5). Sildenafil is also marketed as VIAGRA® for erectile dysfunction.
Sildenafil citrate is designated chemically as 1-[[3-(6,7-dihydro-1-methyl-7-oxo-3-propyl-1H-pyrazolo [4,3-d] pyrimidin-5-yl)-4-ethoxyphenyl] sulfonyl]-4-methylpiperazine citrate and has the following structural formula:
Sildenafil citrate is a white to off-white crystalline powder with a solubility of 3.5 mg/mL in water and a molecular weight of 666.7.
Sildenafil Tablets: Sildenafil is formulated as white, film-coated round tablets for oral administration. Each tablet contains sildenafil citrate equivalent to 20 mg of sildenafil. In addition to the active ingredient, sildenafil citrate, each tablet contains the following inactive ingredients: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, magnesium stearate, hypromellose, titanium dioxide, lactose monohydrate, and triacetin.
Sildenafil for Oral Suspension: Sildenafil is supplied as white to off-white powders containing 1.57 g of sildenafil citrate (equivalent to 1.12 g sildenafil) in an amber glass bottle intended for reconstitution. Following reconstitution with 90 mL water, the volume of the oral suspension is 112 mL and the oral suspension contains 10 mg/mL sildenafil. The inactive ingredients include sorbitol, citric acid anhydrous, sucralose, sodium citrate dihydrate, xanthan gum, titanium dioxide, sodium benzoate, colloidal silicon dioxide anhydrous and grape flavor. In addition to the bottle, a press-in bottle adapter and an oral dosing syringe (with 0.5 mL and 2 mL dose markings) are provided.
5.2 Hypotension
Sildenafil has vasodilatory properties, resulting in mild and transient decreases in blood pressure. Before prescribing sildenafil, carefully consider whether patients with certain underlying conditions could be adversely affected by such vasodilatory effects (e.g., patients on antihypertensive therapy or with resting hypotension [BP less than 90/50], fluid depletion, severe left ventricular outflow obstruction, or autonomic dysfunction). Monitor blood pressure when co-administering blood pressure lowering drugs with sildenafil.
5.5 Visual Loss
When used to treat erectile dysfunction, non-arteritic anterior ischemic optic neuropathy (NAION), a cause of decreased vision including permanent loss of vision, has been reported postmarketing in temporal association with the use of phosphodiesterase type 5 (PDE-5) inhibitors, including sildenafil. Most, but not all, of these patients had underlying anatomic or vascular risk factors for developing NAION, including but not necessarily limited to: low cup to disc ratio ("crowded disc"), age over 50, diabetes, hypertension, coronary artery disease, hyperlipidemia and smoking. Based on published literature, the annual incidence of NAION is 2.5–11.8 cases per 100,000 males aged ≥ 50 per year in the general population.
An observational case-crossover study evaluated the risk of NAION when PDE-5 inhibitor use, as a class, occurred immediately before NAION onset (within 5 half-lives), compared to PDE-5 inhibitor use in a prior time period. The results suggest an approximate 2-fold increase in the risk of NAION, with a risk estimate of 2.15 (95% CI 1.06, 4.34). A similar study reported a consistent result, with a risk estimate of 2.27 (95% CI 0.99, 5.20). Other risk factors for NAION, such as the presence of "crowded" optic disc, may have contributed to the occurrence of NAION in these studies.
Neither the rare postmarketing reports, nor the association of PDE-5 inhibitor use and NAION in the observational studies, substantiate a causal relationship between PDE-5 inhibitor use and NAION [see Adverse Reactions (6.2].
Advise patients to seek immediate medical attention in the event of a sudden loss of vision in one or both eyes while taking PDE-5 inhibitors, including sildenafil. Physicians should also discuss the increased risk of NAION with patients who have already experienced NAION in one eye, including whether such individuals could be adversely affected by use of vasodilators, such as PDE-5 inhibitors.
There are no controlled clinical data on the safety or efficacy of sildenafil in patients with retinitis pigmentosa, a minority whom have genetic disorders of retinal phosphodiesterases. Prescribe sildenafil with caution in these patients.
5.6 Hearing Loss
Cases of sudden decrease or loss of hearing, which may be accompanied by tinnitus and dizziness, have been reported in temporal association with the use of PDE-5 inhibitors, including sildenafil. In some of the cases, medical conditions and other factors were reported that may have played a role. In many cases, medical follow-up information was limited. It is not possible to determine whether these reported events are related directly to the use of sildenafil, to the patient's underlying risk factors for hearing loss, a combination of these factors, or to other factors.
Advise patients to seek prompt medical attention in the event of sudden decrease or loss of hearing while taking PDE-5 inhibitors, including sildenafil.
8.4 Pediatric Use
In a randomized, double-blind, multi-center, placebo-controlled, parallel-group, dose-ranging study, 234 patients with PAH, aged 1 to 17 years, body weight greater than or equal to 8 kg, were randomized, on the basis of body weight, to three dose levels of sildenafil, or placebo, for 16 weeks of treatment. Most patients had mild to moderate symptoms at baseline: WHO Functional Class I (32%), II (51%), III (15%), or IV (0.4%). One-third of patients had primary PAH; two-thirds had secondary PAH (systemic-to-pulmonary shunt in 37%; surgical repair in 30%). Sixty-two percent of patients were female. Drug or placebo was administered three times a day.
The primary objective of the study was to assess the effect of sildenafil on exercise capacity as measured by cardiopulmonary exercise testing in pediatric patients developmentally able to perform the test (n = 115). Administration of sildenafil did not result in a statistically significant improvement in exercise capacity in those patients. No patients died during the 16-week controlled study.
After completing the 16-week controlled study, a patient originally randomized to sildenafil remained on his/her dose of sildenafil or, if originally randomized to placebo, was randomized to low-, medium-, or high-dose sildenafil. After all patients completed 16 weeks of follow-up in the controlled study, the blind was broken and doses were adjusted as clinically indicated. Patients treated with sildenafil were followed for a median of 4.6 years (range 2 days to 8.6 years). Mortality during the long-term study, by originally assigned dose, is shown in Figure 6:
Figure 6. Kaplan-Meier Plot of Mortality by Sildenafil Dose
During the study, there were 42 reported deaths, with 37 of these deaths reported prior to a decision to titrate subjects to a lower dosage because of a finding of increased mortality with increasing sildenafil doses. For the survival analysis which included 37 deaths, the hazard ratio for high dose compared to low dose was 3.9, p=0.007. Causes of death were typical of patients with PAH. Use of sildenafil, particularly chronic use, is not recommended in children.
8.5 Geriatric Use
Clinical studies of sildenafil did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)].
4 Contraindications
Sildenafil is contraindicated in patients with:
-
•Concomitant use of organic nitrates in any form, either regularly or intermittently, because of the greater risk of hypotension [see Warnings and Precautions (5.2)].
-
•Concomitant use of riociguat, a guanylate cyclase stimulator. PDE-5 inhibitors, including sildenafil, may potentiate the hypotensive effects of riociguat.
-
•Known hypersensitivity to sildenafil or any component of the tablet or oral suspension. Hypersensitivity, including anaphylactic reaction, anaphylactic shock and anaphylactoid reaction, has been reported in association with the use of sildenafil.
6 Adverse Reactions
The following serious adverse events are discussed elsewhere in the labeling:
-
•Mortality with pediatric use [see Warnings and Precautions (5.1) and Use in Specific Populations (8.4)]
-
•Hypotension [see Warnings and Precautions (5.2)]
-
•Vision loss [see Warnings and Precautions (5.5)]
-
•Hearing loss [see Warnings and Precautions (5.6)]
-
•Priapism [see Warnings and Precautions (5.8)]
-
•Vaso-occlusive crisis [see Warnings and Precautions (5.9)]
7 Drug Interactions
Instructions for Use
Sildenafil oral suspension
Read this Instructions for Use for sildenafil oral suspension before you start taking and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
Important information:
-
•Your pharmacist should tell you how to measure sildenafil oral suspension by using the oral syringe provided in the pack.
-
•Sildenafil oral suspension should only be given using the oral syringe supplied with each pack.
-
•Sildenafil for oral suspension should not be mixed with any other medicine or flavoring.
Supplies you will need to take sildenafil oral suspension:
-
6.Bottle of sildenafil oral suspension with syringe adaptor fitted in neck of bottle
-
7.Oral syringe (as supplied by pharmacist). (See Figure A)
-
1.Shake the bottle of sildenafil oral suspension for 10 seconds before each use. (See Figure B)
-
2.Remove the cap. Open the bottle by pushing downward on the cap and twisting it in the direction of the arrow (counter-clockwise). (See Figure B)
-
3.Insert the tip of the oral syringe into the adaptor while the bottle is upright, on a flat surface. Fully push down (depress) the plunger of the syringe. (See Figure C)
-
4.Turn the bottle upside down while holding the oral syringe in place. Slowly pull back the plunger of the oral syringe until the bottom of the plunger is even with the graduation markings on the syringe for the prescribed dose for you. Take your dose of sildenafil oral suspension exactly as prescribed by your doctor. If air bubbles can be seen, slowly push the oral suspension in the syringe back into the bottle. Repeat steps 3 and 4. (See Figure D)
-
5.Turn the bottle back upright with the oral syringe still in place. Remove the oral syringe from the bottle by pulling straight up on the barrel of the oral dosing syringe. (See Figure E)
-
6.Put the tip of the oral syringe into your mouth. Point the tip of the oral syringe towards the inside of the cheek. Slowly push down the plunger of the oral syringe. (See Figure F)
-
7.Replace the cap on the bottle, leaving the bottle adaptor in place. Wash the oral syringe as instructed below.
-
8.The syringe should be washed after each dose. Pull the plunger out of the barrel and rinse both parts with water. (See Figure G)
-
9.Dry all parts with a clean paper towel. Push the plunger back into the barrel. Store the syringe with sildenafil oral suspension in a clean safe place.
Administer sildenafil oral suspension using the oral syringe supplied with each pack. Refer to the patient instructions for use for more detailed instructions for use. Discard any unused suspension after the expiration date written on the bottle.
How should I store sildenafil?
-
•Store sildenafil reconstituted oral suspension below 30°C (86°F) or in a refrigerator between 2°C to 8°C (36°F to 46°F).
-
•Do not freeze sildenafil oral suspension
-
•Throw away (discard) sildenafil oral suspension after 60 days.
-
•Keep sildenafil and all medicines away from children.
This Instruction for Use has been approved by the U.S. Food and Drug Administration.
This product's label may have been updated. For current full prescribing information, please visit www.greenstonellc.com
LAB-0959-2.0
Revised: February 2020
1 Indications and Usage
Sildenafil is indicated for the treatment of pulmonary arterial hypertension (WHO Group I) in adults to improve exercise ability and delay clinical worsening. The delay in clinical worsening was demonstrated when sildenafil was added to background epoprostenol therapy [see Clinical Studies (14)].
Studies establishing effectiveness were short-term (12 to 16 weeks), and included predominately patients with New York Heart Association (NYHA) Functional Class II–III symptoms and idiopathic etiology (71%) or associated with connective tissue disease (CTD) (25%).
12.1 Mechanism of Action
Sildenafil is an inhibitor of cGMP specific phosphodiesterase type-5 (PDE-5) in the smooth muscle of the pulmonary vasculature, where PDE-5 is responsible for degradation of cGMP. Sildenafil, therefore, increases cGMP within pulmonary vascular smooth muscle cells resulting in relaxation. In patients with PAH, this can lead to vasodilation of the pulmonary vascular bed and, to a lesser degree, vasodilatation in the systemic circulation.
Studies in vitro have shown that sildenafil is selective for PDE-5. Its effect is more potent on PDE-5 than on other known phosphodiesterases (10-fold for PDE6, greater than 80-fold for PDE1, greater than 700-fold for PDE2, PDE3, PDE4, PDE7, PDE8, PDE9, PDE10, and PDE11). The approximately 4,000-fold selectivity for PDE-5 versus PDE3 is important because PDE3 is involved in control of cardiac contractility. Sildenafil is only about 10-fold as potent for PDE-5 compared to PDE6, an enzyme found in the retina and involved in the phototransduction pathway of the retina. This lower selectivity is thought to be the basis for abnormalities related to color vision observed with higher doses or plasma levels [see Clinical Pharmacology (12.2)].
In addition to pulmonary vascular smooth muscle and the corpus cavernosum, PDE-5 is also found in other tissues including vascular and visceral smooth muscle and in platelets. The inhibition of PDE-5 in these tissues by sildenafil may be the basis for the enhanced platelet anti-aggregatory activity of nitric oxide observed in vitro, and the mild peripheral arterial-venous dilatation in vivo.
5 Warnings and Precautions
-
•Increased mortality with increasing doses in pediatric patients. Not recommended for use in pediatric patients. (5.1)
-
•Vasodilation effects may be more common in patients with hypotension or on antihypertensive therapy. (5.2)
-
•Use in pulmonary veno-occlusive disease may cause pulmonary edema and is not recommended. (5.3)
-
•Hearing or visual impairment: Seek medical attention if sudden decrease or loss of vision or hearing occurs. (5.5, 5.6)
-
•Pulmonary hypertension secondary to sickle cell disease: sildenafil may cause serious vaso-occlusive crises. (5.9)
2 Dosage and Administration
3 Dosage Forms and Strengths
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of sildenafil (marketed for both PAH and erectile dysfunction). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Safety data of sildenafil in adults were obtained from the 12-week, placebo-controlled clinical study (Study 1) and an open-label extension study in 277 sildenafil-treated patients with PAH, WHO Group I [see Clinical Studies (14)].
The overall frequency of discontinuation in sildenafil-treated patients on 20 mg three times a day was 3% and was the same for the placebo group.
In Study 1, the adverse reactions that were reported by at least 3% of sildenafil-treated patients (20 mg three times a day) and were more frequent in sildenafil-treated patients than in placebo-treated patients, are shown in Table 1. Adverse reactions were generally transient and mild to moderate in nature.
|
Placebo, %
(n = 70) |
Sildenafil 20 mg three times a day, %
(n = 69) |
Placebo-Subtracted, % | |
|---|---|---|---|
|
Epistaxis |
1 |
9 |
8 |
|
Headache |
39 |
46 |
7 |
|
Dyspepsia |
7 |
13 |
6 |
|
Flushing |
4 |
10 |
6 |
|
Insomnia |
1 |
7 |
6 |
|
Erythema |
1 |
6 |
5 |
|
Dyspnea exacerbated |
3 |
7 |
4 |
|
Rhinitis |
0 |
4 |
4 |
|
Diarrhea |
6 |
9 |
3 |
|
Myalgia |
4 |
7 |
3 |
|
Pyrexia |
3 |
6 |
3 |
|
Gastritis |
0 |
3 |
3 |
|
Sinusitis |
0 |
3 |
3 |
|
Paresthesia |
0 |
3 |
3 |
At doses higher than the recommended 20 mg three times a day, there was a greater incidence of some adverse reactions including flushing, diarrhea, myalgia and visual disturbances. Visual disturbances were identified as mild and transient, and were predominately color-tinge to vision, but also increased sensitivity to light or blurred vision.
The incidence of retinal hemorrhage with sildenafil 20 mg three times a day was 1.4% versus 0% placebo and for all sildenafil doses studied was 1.9% versus 0% placebo. The incidence of eye hemorrhage at both 20 mg three times a day and at all doses studied was 1.4% for sildenafil versus 1.4% for placebo. The patients experiencing these reactions had risk factors for hemorrhage including concurrent anticoagulant therapy.
In a placebo-controlled fixed dose titration study (Study 2) of sildenafil (starting with recommended dose of 20 mg and increased to 40 mg and then 80 mg all three times a day) as an adjunct to intravenous epoprostenol in patients with PAH, the adverse reactions that were more frequent in the sildenafil + epoprostenol group than in the epoprostenol group (greater than 6% difference) are shown in Table 2 [see Clinical Studies (14)].
|
Sildenafil + Epoprostenol
(n = 134) |
Epoprostenol
(n = 131) |
(Sildenafil + Epoprostenol) minus Epoprostenol | |
|---|---|---|---|
|
Headache |
57 |
34 |
23 |
|
Edema includes peripheral edema
|
25 |
13 |
14 |
|
Dyspepsia |
16 |
2 |
14 |
|
Pain in extremity |
17 |
6 |
11 |
|
Diarrhea |
25 |
18 |
7 |
|
Nausea |
25 |
18 |
7 |
|
Nasal congestion |
9 |
2 |
7 |
5.1 Mortality With Pediatric Use
In a long-term trial in pediatric patients with PAH, an increase in mortality with increasing sildenafil dose was observed. Deaths were first observed after about 1 year and causes of death were typical of patients with PAH. Use of sildenafil, particularly chronic use, is not recommended in children [see Use in Specific Populations (8.4)].
17 Patient Counseling Information
See FDA-approved patient labeling (Patient Information)
-
•Inform patients of contraindication of sildenafil with regular and/or intermittent use of organic nitrates.
-
•Inform patients that sildenafil is also marketed as VIAGRA for erectile dysfunction. Advise patients taking sildenafil not to take VIAGRA or other PDE-5 inhibitors.
-
•Advise patients to seek immediate medical attention for a sudden loss of vision in one or both eyes while taking sildenafil. Such an event may be a sign of NAION.
-
•Advise patients to seek prompt medical attention in the event of sudden decrease or loss of hearing while taking sildenafil. These events may be accompanied by tinnitus and dizziness.
8.7 Patients With Renal Impairment
No dose adjustment is required (including severe impairment CLcr < 30 mL/min) [see Clinical Pharmacology (12.3)].
8.6 Patients With Hepatic Impairment
No dose adjustment for mild to moderate impairment is required. Severe impairment has not been studied [see Clinical Pharmacology (12.3)].
2.1 Sildenafil Tablets and Oral Suspension
The recommended dose of sildenafil is 5 mg or 20 mg three times a day. Administer sildenafil doses 4–6 hours apart.
In the clinical trial no greater efficacy was achieved with the use of higher doses. Treatment with doses higher than 20 mg three times a day is not recommended.
5.7 Combination With Other Pde 5 Inhibitors
Sildenafil is also marketed as VIAGRA®. The safety and efficacy of combinations of sildenafil with VIAGRA or other PDE-5 inhibitors have not been studied. Inform patients taking sildenafil not to take VIAGRA or other PDE-5 inhibitors.
Principal Display Panel 10 Mg/ml Bottle Label
NDC 59762-2058-1
GREENSTONE® BRAND
sildenafil
for oral suspension
10 mg/mL
Shake Well Before Each Use
For Oral Use Only
112 mL following Reconstitution
Rx only
Principal Display Panel 10 Mg/ml Bottle Carton
NDC 59762-2058-1
GREENSTONE® BRAND
sildenafil
for oral suspension
10 mg/mL
For Oral Use Only
Shake Well Before Each Use
112 mL following Reconstitution
Rx only
Grape Flavored
5.3 Worsening Pulmonary Vascular Occlusive Disease
Pulmonary vasodilators may significantly worsen the cardiovascular status of patients with pulmonary veno-occlusive disease (PVOD). Since there are no clinical data on administration of sildenafil to patients with veno-occlusive disease, administration of sildenafil to such patients is not recommended. Should signs of pulmonary edema occur when sildenafil is administered, consider the possibility of associated PVOD.
Principal Display Panel 20 Mg Tablet Bottle Label
NDC 59762-0033-1
90 Tablets
GREENSTONE® BRAND
sildenafil
tablets
20 mg*
Rx only
2.2 Reconstitution of the Powder for Oral Suspension
Note: Reconstitute the contents of the bottle with a total volume of 90 mL (60 mL followed by 30 mL). Refer to the detailed instructions below.
-
1.Tap the bottle to release the powder.
-
2.Remove the cap.
-
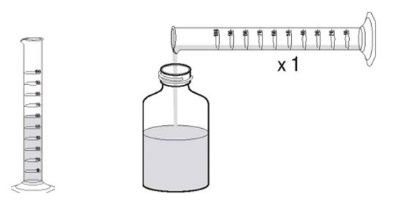
3.Accurately measure out 60 mL of water and pour the water into the bottle. (Figure 1)
-
4.Replace the cap and shake the bottle vigorously for a minimum of 30 seconds. (Figure 2)
-
5.Remove the cap.
-
6.Accurately measure out another 30 mL of water and add this to the bottle. You should always add a total of 90 mL of water irrespective of the dose prescribed. (Figure 3)
-
7.Replace the cap and shake the bottle vigorously for a minimum of 30 seconds. (Figure 4)
-
8.Remove the cap.
-
9.Press the bottle adaptor into the neck of the bottle (as shown on Figure 5, below). The adaptor is provided so that you can fill the oral syringe with medicine from the bottle. Replace the cap on the bottle.
-
10.Write the expiration date of the reconstituted oral suspension on the bottle label (the expiration date of the reconstituted oral suspension is 60 days from the date of reconstitution).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Sildenafil was not carcinogenic when administered to rats for up to 24 months at 60 mg/kg/day, a dose resulting in total systemic exposure (AUC) to unbound sildenafil and its major metabolite 33- and 37-times, for male and female rats respectively, the human exposure at the RHD of 20 mg three times a day. Sildenafil was not carcinogenic when administered to male and female mice for up to 21 and 18 months, respectively, at doses up to a maximally tolerated level of 10 mg/kg/day, a dose equivalent to the RHD on a mg/m2 basis.
Sildenafil was negative in in vitro bacterial and Chinese hamster ovary cell assays to detect mutagenicity, and in vitro human lymphocytes and in vivo mouse micronucleus assays to detect clastogenicity.
There was no impairment of fertility in male or female rats given up to 60 mg sildenafil/kg/day, a dose producing a total systemic exposure (AUC) to unbound sildenafil and its major metabolite of 19- and 38- times for males and females, respectively, the human exposure at the RHD of 20 mg three times a day.
5.9 Vaso Occlusive Crisis in Patients With Pulmonary Hypertension Secondary to Sickle Cell Anemia
In a small, prematurely terminated study of patients with pulmonary hypertension (PH) secondary to sickle cell disease, vaso-occlusive crises requiring hospitalization were more commonly reported by patients who received sildenafil than by those randomized to placebo. The effectiveness and safety of sildenafil in the treatment of PAH secondary to sickle cell anemia has not been established.
Structured Label Content
Section 42229-5 (42229-5)
Limitation of Use: Adding sildenafil to bosentan therapy does not result in any beneficial effect on exercise capacity [see Clinical Studies (14)].
Section 44425-7 (44425-7)
Recommended Storage for Sildenafil Tablets: Store at controlled room temperature 20°C – 25°C (68°F – 77°F); excursions permitted to 15°C – 30°C (59°F – 86°F) [see USP Controlled Room Temperature].
5.8 Priapism
Use sildenafil with caution in patients with anatomical deformation of the penis (e.g., angulation, cavernosal fibrosis, or Peyronie's disease) or in patients who have conditions, which may predispose them to priapism (e.g., sickle cell anemia, multiple myeloma, or leukemia). In the event of an erection that persists longer than 4 hours, the patient should seek immediate medical assistance. If priapism (painful erection greater than 6 hours in duration) is not treated immediately, penile tissue damage and permanent loss of potency could result.
10 Overdosage (10 OVERDOSAGE)
In studies with healthy volunteers of single doses up to 800 mg, adverse events were similar to those seen at lower doses but rates and severities were increased.
In cases of overdose, standard supportive measures should be adopted as required. Renal dialysis is not expected to accelerate clearance as sildenafil is highly bound to plasma proteins and it is not eliminated in the urine.
5.4 Epistaxis
The incidence of epistaxis was 13% in patients taking sildenafil with PAH secondary to CTD. This effect was not seen in idiopathic PAH (sildenafil 3%, placebo 2%) patients. The incidence of epistaxis was also higher in sildenafil-treated patients with a concomitant oral vitamin K antagonist (9% versus 2% in those not treated with concomitant vitamin K antagonist).
The safety of sildenafil is unknown in patients with bleeding disorders or active peptic ulceration.
11 Description (11 DESCRIPTION)
Sildenafil, phosphodiesterase-5 (PDE-5) inhibitor, is the citrate salt of sildenafil, a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type-5 (PDE-5). Sildenafil is also marketed as VIAGRA® for erectile dysfunction.
Sildenafil citrate is designated chemically as 1-[[3-(6,7-dihydro-1-methyl-7-oxo-3-propyl-1H-pyrazolo [4,3-d] pyrimidin-5-yl)-4-ethoxyphenyl] sulfonyl]-4-methylpiperazine citrate and has the following structural formula:
Sildenafil citrate is a white to off-white crystalline powder with a solubility of 3.5 mg/mL in water and a molecular weight of 666.7.
Sildenafil Tablets: Sildenafil is formulated as white, film-coated round tablets for oral administration. Each tablet contains sildenafil citrate equivalent to 20 mg of sildenafil. In addition to the active ingredient, sildenafil citrate, each tablet contains the following inactive ingredients: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, magnesium stearate, hypromellose, titanium dioxide, lactose monohydrate, and triacetin.
Sildenafil for Oral Suspension: Sildenafil is supplied as white to off-white powders containing 1.57 g of sildenafil citrate (equivalent to 1.12 g sildenafil) in an amber glass bottle intended for reconstitution. Following reconstitution with 90 mL water, the volume of the oral suspension is 112 mL and the oral suspension contains 10 mg/mL sildenafil. The inactive ingredients include sorbitol, citric acid anhydrous, sucralose, sodium citrate dihydrate, xanthan gum, titanium dioxide, sodium benzoate, colloidal silicon dioxide anhydrous and grape flavor. In addition to the bottle, a press-in bottle adapter and an oral dosing syringe (with 0.5 mL and 2 mL dose markings) are provided.
5.2 Hypotension
Sildenafil has vasodilatory properties, resulting in mild and transient decreases in blood pressure. Before prescribing sildenafil, carefully consider whether patients with certain underlying conditions could be adversely affected by such vasodilatory effects (e.g., patients on antihypertensive therapy or with resting hypotension [BP less than 90/50], fluid depletion, severe left ventricular outflow obstruction, or autonomic dysfunction). Monitor blood pressure when co-administering blood pressure lowering drugs with sildenafil.
5.5 Visual Loss
When used to treat erectile dysfunction, non-arteritic anterior ischemic optic neuropathy (NAION), a cause of decreased vision including permanent loss of vision, has been reported postmarketing in temporal association with the use of phosphodiesterase type 5 (PDE-5) inhibitors, including sildenafil. Most, but not all, of these patients had underlying anatomic or vascular risk factors for developing NAION, including but not necessarily limited to: low cup to disc ratio ("crowded disc"), age over 50, diabetes, hypertension, coronary artery disease, hyperlipidemia and smoking. Based on published literature, the annual incidence of NAION is 2.5–11.8 cases per 100,000 males aged ≥ 50 per year in the general population.
An observational case-crossover study evaluated the risk of NAION when PDE-5 inhibitor use, as a class, occurred immediately before NAION onset (within 5 half-lives), compared to PDE-5 inhibitor use in a prior time period. The results suggest an approximate 2-fold increase in the risk of NAION, with a risk estimate of 2.15 (95% CI 1.06, 4.34). A similar study reported a consistent result, with a risk estimate of 2.27 (95% CI 0.99, 5.20). Other risk factors for NAION, such as the presence of "crowded" optic disc, may have contributed to the occurrence of NAION in these studies.
Neither the rare postmarketing reports, nor the association of PDE-5 inhibitor use and NAION in the observational studies, substantiate a causal relationship between PDE-5 inhibitor use and NAION [see Adverse Reactions (6.2].
Advise patients to seek immediate medical attention in the event of a sudden loss of vision in one or both eyes while taking PDE-5 inhibitors, including sildenafil. Physicians should also discuss the increased risk of NAION with patients who have already experienced NAION in one eye, including whether such individuals could be adversely affected by use of vasodilators, such as PDE-5 inhibitors.
There are no controlled clinical data on the safety or efficacy of sildenafil in patients with retinitis pigmentosa, a minority whom have genetic disorders of retinal phosphodiesterases. Prescribe sildenafil with caution in these patients.
5.6 Hearing Loss
Cases of sudden decrease or loss of hearing, which may be accompanied by tinnitus and dizziness, have been reported in temporal association with the use of PDE-5 inhibitors, including sildenafil. In some of the cases, medical conditions and other factors were reported that may have played a role. In many cases, medical follow-up information was limited. It is not possible to determine whether these reported events are related directly to the use of sildenafil, to the patient's underlying risk factors for hearing loss, a combination of these factors, or to other factors.
Advise patients to seek prompt medical attention in the event of sudden decrease or loss of hearing while taking PDE-5 inhibitors, including sildenafil.
8.4 Pediatric Use
In a randomized, double-blind, multi-center, placebo-controlled, parallel-group, dose-ranging study, 234 patients with PAH, aged 1 to 17 years, body weight greater than or equal to 8 kg, were randomized, on the basis of body weight, to three dose levels of sildenafil, or placebo, for 16 weeks of treatment. Most patients had mild to moderate symptoms at baseline: WHO Functional Class I (32%), II (51%), III (15%), or IV (0.4%). One-third of patients had primary PAH; two-thirds had secondary PAH (systemic-to-pulmonary shunt in 37%; surgical repair in 30%). Sixty-two percent of patients were female. Drug or placebo was administered three times a day.
The primary objective of the study was to assess the effect of sildenafil on exercise capacity as measured by cardiopulmonary exercise testing in pediatric patients developmentally able to perform the test (n = 115). Administration of sildenafil did not result in a statistically significant improvement in exercise capacity in those patients. No patients died during the 16-week controlled study.
After completing the 16-week controlled study, a patient originally randomized to sildenafil remained on his/her dose of sildenafil or, if originally randomized to placebo, was randomized to low-, medium-, or high-dose sildenafil. After all patients completed 16 weeks of follow-up in the controlled study, the blind was broken and doses were adjusted as clinically indicated. Patients treated with sildenafil were followed for a median of 4.6 years (range 2 days to 8.6 years). Mortality during the long-term study, by originally assigned dose, is shown in Figure 6:
Figure 6. Kaplan-Meier Plot of Mortality by Sildenafil Dose
During the study, there were 42 reported deaths, with 37 of these deaths reported prior to a decision to titrate subjects to a lower dosage because of a finding of increased mortality with increasing sildenafil doses. For the survival analysis which included 37 deaths, the hazard ratio for high dose compared to low dose was 3.9, p=0.007. Causes of death were typical of patients with PAH. Use of sildenafil, particularly chronic use, is not recommended in children.
8.5 Geriatric Use
Clinical studies of sildenafil did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
Sildenafil is contraindicated in patients with:
-
•Concomitant use of organic nitrates in any form, either regularly or intermittently, because of the greater risk of hypotension [see Warnings and Precautions (5.2)].
-
•Concomitant use of riociguat, a guanylate cyclase stimulator. PDE-5 inhibitors, including sildenafil, may potentiate the hypotensive effects of riociguat.
-
•Known hypersensitivity to sildenafil or any component of the tablet or oral suspension. Hypersensitivity, including anaphylactic reaction, anaphylactic shock and anaphylactoid reaction, has been reported in association with the use of sildenafil.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse events are discussed elsewhere in the labeling:
-
•Mortality with pediatric use [see Warnings and Precautions (5.1) and Use in Specific Populations (8.4)]
-
•Hypotension [see Warnings and Precautions (5.2)]
-
•Vision loss [see Warnings and Precautions (5.5)]
-
•Hearing loss [see Warnings and Precautions (5.6)]
-
•Priapism [see Warnings and Precautions (5.8)]
-
•Vaso-occlusive crisis [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Patient Information (PATIENT INFORMATION)
Sildenafil tablets
Sildenafil oral suspension
Read this Patient Information before you start taking sildenafil and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or treatment. If you have any questions about sildenafil, ask your doctor or pharmacist.
What is the most important information I should know about sildenafil?
Never take sildenafil with any nitrate or guanylate cyclase stimulator medicines. Your blood pressure could drop quickly to an unsafe level.
Nitrate medicines include:
-
•Medicines that treat chest pain (angina)
-
•Nitroglycerin in any form including tablets, patches, sprays, and ointments
-
•Isosorbide mononitrate or dinitrate
-
•Street drugs called "poppers" (amyl nitrate or nitrite)
Guanylate cyclase stimulators include:
-
1.Riociguat (Adempas)
Ask your doctor or pharmacist if you are not sure if you are taking a nitrate or a guanylate cyclase stimulator medicine.
What is sildenafil?
Sildenafil is a prescription medicine used in adults to treat pulmonary arterial hypertension (PAH). With PAH, the blood pressure in your lungs is too high. Your heart has to work hard to pump blood into your lungs.
Sildenafil improves the ability to exercise and can slow down worsening changes in your physical condition.
-
•Sildenafil is not for use in children
-
•Adding sildenafil to another medication used to treat PAH bosentan (Tracleer®), does not result in improvement in your ability to exercise.
Sildenafil contains the same medicine as VIAGRA® (sildenafil), which is used to treat erectile dysfunction (impotence). Do not take sildenafil with VIAGRA or other PDE-5 inhibitors.
Who should not take Sildenafil?
Do not take sildenafil if you:
-
•take nitrate medicines. See " What is the most important information I should know about sildenafil? "
-
•take guanylate cyclase stimulator medicines. See " What is the most important information I should know about sildenafil? "
-
•are allergic to sildenafil or any other ingredient in sildenafil tablets or oral suspension. See " What are the ingredients in sildenafil tablets and oral suspension?" at the end of this leaflet.
What should I tell my doctor before taking sildenafil?
Tell your doctor about all of your medical conditions, including if you
-
•have heart problems such as angina (chest pain), heart failure, irregular heartbeats, or have had a heart attack
-
•have a disease called pulmonary veno-occlusive disease (PVOD)
-
•have high or low blood pressure or blood circulation problems
-
•have an eye problem called retinitis pigmentosa
-
•have or had loss of sight in one or both eyes
-
•have any problem with the shape of your penis or Peyronie's disease
-
•have any blood cell problems such as sickle cell anemia
-
•have a stomach ulcer or any bleeding problems
-
•are pregnant or planning to become pregnant. It is not known if sildenafil could harm your unborn baby.
-
•are breastfeeding. Sildenafil passes into your breast milk, it is not known if it could harm your baby.
Tell your doctor about all of the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal products. Sildenafil and certain other medicines can cause side effects if you take them together. The doses of some of your medicines may need to be adjusted while you take sildenafil.
Especially tell your doctor if you take
-
•Nitrate medicines. See " What is the most important information I should know about sildenafil? "
-
•Riociguat (Adempas). See " What is the most important information I should know about sildenafil? "
-
•Ritonavir (Norvir®) or other medicines used to treat HIV infection
-
•Ketoconazole (Nizoral®)
-
•Itraconazole (Sporanox)
-
•High blood pressure medicine
Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist when you get a new medicine.
How should I take sildenafil?
-
2.Take sildenafil exactly as your doctor tells you.
Sildenafil may be prescribed to you as
-
•Sildenafil tablets or sildenafil oral suspension
-
•Take sildenafil tablet or oral suspension 3 times a day about 4 to 6 hours apart.
-
•Take sildenafil tablets or oral suspension at the same times every day.
-
•Sildenafil oral suspension will be mixed for you by your pharmacist. Do not mix sildenafil oral suspension with other medicine or flavoring. Shake well for at least 10 seconds before each dose.
-
•If you miss a dose, take it as soon as you remember. If it is close to your next dose, skip the missed dose, and take your next dose at the regular time.
-
•Do not take more than one dose of sildenafil at a time.
-
•Do not change your dose or stop taking sildenafil on your own. Talk to your doctor first.
-
•If you take too much sildenafil, call your doctor or go to the nearest hospital emergency room.
What are the possible side effects of sildenafil?
-
•low blood pressure. Low blood pressure may cause you to feel faint or dizzy. Lie down if you feel faint or dizzy.
-
•more shortness of breath than usual. Tell your doctor if you get more short of breath after you start sildenafil. More shortness of breath than usual may be due to your underlying medical condition.
-
•decreased eyesight or loss of sight in one or both eyes (NAION). If you notice a sudden decrease or loss of eyesight, talk to your doctor right away.
-
•sudden decrease or loss of hearing. If you notice a sudden decrease or loss of hearing, talk to your doctor right away. It is not possible to determine whether these events are related directly to this class of oral medicines, including sildenafil, or to other diseases or medicines, to other factors, or to a combination of factors.
-
•heart attack, stroke, irregular heartbeats, and death. Most of these happened in men who already had heart problems.
-
•erections that last several hours. If you have an erection that lasts more than 4 hours, get medical help right away. If it is not treated right away, priapism can permanently damage your penis.
The most common side effects with sildenafil include:
Nosebleed, headache, upset stomach, getting red or hot in the face (flushing), trouble sleeping, as well as fever, erection increased, respiratory infection, nausea, vomiting, bronchitis, pharyngitis, runny nose, and pneumonia in children.
Tell your doctor if you have any side effect that bothers you or doesn't go away.
These are not all the possible side effects of sildenafil. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store sildenafil?
-
•Store sildenafil tablets at controlled room temperature, between 20°C – 25°C (68°F to 77°F).
-
•Store sildenafil reconstituted oral suspension below 30°C (86°F) or in a refrigerator between 2°C to 8°C (36°F to 46°F).
-
•Do not freeze sildenafil oral suspension.
-
•Throw away sildenafil oral suspension after 60 days.
-
•Keep sildenafil and all medicines away from children.
General information about sildenafil
Medicines are sometimes prescribed for purposes that are not in the patient leaflet. Do not use sildenafil for a condition for which it was not prescribed. Do not give sildenafil to other people, even if they have the same symptoms you have. It could harm them.
This patient leaflet summarizes the most important information about sildenafil. If you would like more information about sildenafil talk with your doctor. You can ask your doctor or pharmacist for information about sildenafil that is written for health professionals.
For more information go to www.greenstonellc.com or call 1-800-438-1985.
What are the ingredients in sildenafil tablets and oral suspension?
Sildenafil tablets
Active ingredients: sildenafil citrate
Inactive ingredients: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, magnesium stearate, hypromellose, titanium dioxide, lactose monohydrate, and triacetin
Sildenafil for oral suspension
Active ingredients: sildenafil citrate
Inactive ingredients: sorbitol, citric acid anhydrous, sucralose, sodium citrate dihydrate, xanthan gum, titanium dioxide, sodium benzoate, colloidal silicon dioxide anhydrous, and grape flavor
This product's label may have been updated. For current full prescribing information, please visit www.greenstonellc.com.
This Patient Information has been approved by the U.S. Food and Drug Administration.
LAB-0563-7.0
Revised: February 2020
Instructions for Use
Sildenafil oral suspension
Read this Instructions for Use for sildenafil oral suspension before you start taking and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
Important information:
-
•Your pharmacist should tell you how to measure sildenafil oral suspension by using the oral syringe provided in the pack.
-
•Sildenafil oral suspension should only be given using the oral syringe supplied with each pack.
-
•Sildenafil for oral suspension should not be mixed with any other medicine or flavoring.
Supplies you will need to take sildenafil oral suspension:
-
6.Bottle of sildenafil oral suspension with syringe adaptor fitted in neck of bottle
-
7.Oral syringe (as supplied by pharmacist). (See Figure A)
-
1.Shake the bottle of sildenafil oral suspension for 10 seconds before each use. (See Figure B)
-
2.Remove the cap. Open the bottle by pushing downward on the cap and twisting it in the direction of the arrow (counter-clockwise). (See Figure B)
-
3.Insert the tip of the oral syringe into the adaptor while the bottle is upright, on a flat surface. Fully push down (depress) the plunger of the syringe. (See Figure C)
-
4.Turn the bottle upside down while holding the oral syringe in place. Slowly pull back the plunger of the oral syringe until the bottom of the plunger is even with the graduation markings on the syringe for the prescribed dose for you. Take your dose of sildenafil oral suspension exactly as prescribed by your doctor. If air bubbles can be seen, slowly push the oral suspension in the syringe back into the bottle. Repeat steps 3 and 4. (See Figure D)
-
5.Turn the bottle back upright with the oral syringe still in place. Remove the oral syringe from the bottle by pulling straight up on the barrel of the oral dosing syringe. (See Figure E)
-
6.Put the tip of the oral syringe into your mouth. Point the tip of the oral syringe towards the inside of the cheek. Slowly push down the plunger of the oral syringe. (See Figure F)
-
7.Replace the cap on the bottle, leaving the bottle adaptor in place. Wash the oral syringe as instructed below.
-
8.The syringe should be washed after each dose. Pull the plunger out of the barrel and rinse both parts with water. (See Figure G)
-
9.Dry all parts with a clean paper towel. Push the plunger back into the barrel. Store the syringe with sildenafil oral suspension in a clean safe place.
Administer sildenafil oral suspension using the oral syringe supplied with each pack. Refer to the patient instructions for use for more detailed instructions for use. Discard any unused suspension after the expiration date written on the bottle.
How should I store sildenafil?
-
•Store sildenafil reconstituted oral suspension below 30°C (86°F) or in a refrigerator between 2°C to 8°C (36°F to 46°F).
-
•Do not freeze sildenafil oral suspension
-
•Throw away (discard) sildenafil oral suspension after 60 days.
-
•Keep sildenafil and all medicines away from children.
This Instruction for Use has been approved by the U.S. Food and Drug Administration.
This product's label may have been updated. For current full prescribing information, please visit www.greenstonellc.com
LAB-0959-2.0
Revised: February 2020
1 Indications and Usage (1 INDICATIONS AND USAGE)
Sildenafil is indicated for the treatment of pulmonary arterial hypertension (WHO Group I) in adults to improve exercise ability and delay clinical worsening. The delay in clinical worsening was demonstrated when sildenafil was added to background epoprostenol therapy [see Clinical Studies (14)].
Studies establishing effectiveness were short-term (12 to 16 weeks), and included predominately patients with New York Heart Association (NYHA) Functional Class II–III symptoms and idiopathic etiology (71%) or associated with connective tissue disease (CTD) (25%).
12.1 Mechanism of Action
Sildenafil is an inhibitor of cGMP specific phosphodiesterase type-5 (PDE-5) in the smooth muscle of the pulmonary vasculature, where PDE-5 is responsible for degradation of cGMP. Sildenafil, therefore, increases cGMP within pulmonary vascular smooth muscle cells resulting in relaxation. In patients with PAH, this can lead to vasodilation of the pulmonary vascular bed and, to a lesser degree, vasodilatation in the systemic circulation.
Studies in vitro have shown that sildenafil is selective for PDE-5. Its effect is more potent on PDE-5 than on other known phosphodiesterases (10-fold for PDE6, greater than 80-fold for PDE1, greater than 700-fold for PDE2, PDE3, PDE4, PDE7, PDE8, PDE9, PDE10, and PDE11). The approximately 4,000-fold selectivity for PDE-5 versus PDE3 is important because PDE3 is involved in control of cardiac contractility. Sildenafil is only about 10-fold as potent for PDE-5 compared to PDE6, an enzyme found in the retina and involved in the phototransduction pathway of the retina. This lower selectivity is thought to be the basis for abnormalities related to color vision observed with higher doses or plasma levels [see Clinical Pharmacology (12.2)].
In addition to pulmonary vascular smooth muscle and the corpus cavernosum, PDE-5 is also found in other tissues including vascular and visceral smooth muscle and in platelets. The inhibition of PDE-5 in these tissues by sildenafil may be the basis for the enhanced platelet anti-aggregatory activity of nitric oxide observed in vitro, and the mild peripheral arterial-venous dilatation in vivo.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Increased mortality with increasing doses in pediatric patients. Not recommended for use in pediatric patients. (5.1)
-
•Vasodilation effects may be more common in patients with hypotension or on antihypertensive therapy. (5.2)
-
•Use in pulmonary veno-occlusive disease may cause pulmonary edema and is not recommended. (5.3)
-
•Hearing or visual impairment: Seek medical attention if sudden decrease or loss of vision or hearing occurs. (5.5, 5.6)
-
•Pulmonary hypertension secondary to sickle cell disease: sildenafil may cause serious vaso-occlusive crises. (5.9)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of sildenafil (marketed for both PAH and erectile dysfunction). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Safety data of sildenafil in adults were obtained from the 12-week, placebo-controlled clinical study (Study 1) and an open-label extension study in 277 sildenafil-treated patients with PAH, WHO Group I [see Clinical Studies (14)].
The overall frequency of discontinuation in sildenafil-treated patients on 20 mg three times a day was 3% and was the same for the placebo group.
In Study 1, the adverse reactions that were reported by at least 3% of sildenafil-treated patients (20 mg three times a day) and were more frequent in sildenafil-treated patients than in placebo-treated patients, are shown in Table 1. Adverse reactions were generally transient and mild to moderate in nature.
|
Placebo, %
(n = 70) |
Sildenafil 20 mg three times a day, %
(n = 69) |
Placebo-Subtracted, % | |
|---|---|---|---|
|
Epistaxis |
1 |
9 |
8 |
|
Headache |
39 |
46 |
7 |
|
Dyspepsia |
7 |
13 |
6 |
|
Flushing |
4 |
10 |
6 |
|
Insomnia |
1 |
7 |
6 |
|
Erythema |
1 |
6 |
5 |
|
Dyspnea exacerbated |
3 |
7 |
4 |
|
Rhinitis |
0 |
4 |
4 |
|
Diarrhea |
6 |
9 |
3 |
|
Myalgia |
4 |
7 |
3 |
|
Pyrexia |
3 |
6 |
3 |
|
Gastritis |
0 |
3 |
3 |
|
Sinusitis |
0 |
3 |
3 |
|
Paresthesia |
0 |
3 |
3 |
At doses higher than the recommended 20 mg three times a day, there was a greater incidence of some adverse reactions including flushing, diarrhea, myalgia and visual disturbances. Visual disturbances were identified as mild and transient, and were predominately color-tinge to vision, but also increased sensitivity to light or blurred vision.
The incidence of retinal hemorrhage with sildenafil 20 mg three times a day was 1.4% versus 0% placebo and for all sildenafil doses studied was 1.9% versus 0% placebo. The incidence of eye hemorrhage at both 20 mg three times a day and at all doses studied was 1.4% for sildenafil versus 1.4% for placebo. The patients experiencing these reactions had risk factors for hemorrhage including concurrent anticoagulant therapy.
In a placebo-controlled fixed dose titration study (Study 2) of sildenafil (starting with recommended dose of 20 mg and increased to 40 mg and then 80 mg all three times a day) as an adjunct to intravenous epoprostenol in patients with PAH, the adverse reactions that were more frequent in the sildenafil + epoprostenol group than in the epoprostenol group (greater than 6% difference) are shown in Table 2 [see Clinical Studies (14)].
|
Sildenafil + Epoprostenol
(n = 134) |
Epoprostenol
(n = 131) |
(Sildenafil + Epoprostenol) minus Epoprostenol | |
|---|---|---|---|
|
Headache |
57 |
34 |
23 |
|
Edema includes peripheral edema
|
25 |
13 |
14 |
|
Dyspepsia |
16 |
2 |
14 |
|
Pain in extremity |
17 |
6 |
11 |
|
Diarrhea |
25 |
18 |
7 |
|
Nausea |
25 |
18 |
7 |
|
Nasal congestion |
9 |
2 |
7 |
5.1 Mortality With Pediatric Use (5.1 Mortality with Pediatric Use)
In a long-term trial in pediatric patients with PAH, an increase in mortality with increasing sildenafil dose was observed. Deaths were first observed after about 1 year and causes of death were typical of patients with PAH. Use of sildenafil, particularly chronic use, is not recommended in children [see Use in Specific Populations (8.4)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
See FDA-approved patient labeling (Patient Information)
-
•Inform patients of contraindication of sildenafil with regular and/or intermittent use of organic nitrates.
-
•Inform patients that sildenafil is also marketed as VIAGRA for erectile dysfunction. Advise patients taking sildenafil not to take VIAGRA or other PDE-5 inhibitors.
-
•Advise patients to seek immediate medical attention for a sudden loss of vision in one or both eyes while taking sildenafil. Such an event may be a sign of NAION.
-
•Advise patients to seek prompt medical attention in the event of sudden decrease or loss of hearing while taking sildenafil. These events may be accompanied by tinnitus and dizziness.
8.7 Patients With Renal Impairment (8.7 Patients with Renal Impairment)
No dose adjustment is required (including severe impairment CLcr < 30 mL/min) [see Clinical Pharmacology (12.3)].
8.6 Patients With Hepatic Impairment (8.6 Patients with Hepatic Impairment)
No dose adjustment for mild to moderate impairment is required. Severe impairment has not been studied [see Clinical Pharmacology (12.3)].
2.1 Sildenafil Tablets and Oral Suspension
The recommended dose of sildenafil is 5 mg or 20 mg three times a day. Administer sildenafil doses 4–6 hours apart.
In the clinical trial no greater efficacy was achieved with the use of higher doses. Treatment with doses higher than 20 mg three times a day is not recommended.
5.7 Combination With Other Pde 5 Inhibitors (5.7 Combination with other PDE-5 inhibitors)
Sildenafil is also marketed as VIAGRA®. The safety and efficacy of combinations of sildenafil with VIAGRA or other PDE-5 inhibitors have not been studied. Inform patients taking sildenafil not to take VIAGRA or other PDE-5 inhibitors.
Principal Display Panel 10 Mg/ml Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg/mL Bottle Label)
NDC 59762-2058-1
GREENSTONE® BRAND
sildenafil
for oral suspension
10 mg/mL
Shake Well Before Each Use
For Oral Use Only
112 mL following Reconstitution
Rx only
Principal Display Panel 10 Mg/ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 10 mg/mL Bottle Carton)
NDC 59762-2058-1
GREENSTONE® BRAND
sildenafil
for oral suspension
10 mg/mL
For Oral Use Only
Shake Well Before Each Use
112 mL following Reconstitution
Rx only
Grape Flavored
5.3 Worsening Pulmonary Vascular Occlusive Disease
Pulmonary vasodilators may significantly worsen the cardiovascular status of patients with pulmonary veno-occlusive disease (PVOD). Since there are no clinical data on administration of sildenafil to patients with veno-occlusive disease, administration of sildenafil to such patients is not recommended. Should signs of pulmonary edema occur when sildenafil is administered, consider the possibility of associated PVOD.
Principal Display Panel 20 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label)
NDC 59762-0033-1
90 Tablets
GREENSTONE® BRAND
sildenafil
tablets
20 mg*
Rx only
2.2 Reconstitution of the Powder for Oral Suspension
Note: Reconstitute the contents of the bottle with a total volume of 90 mL (60 mL followed by 30 mL). Refer to the detailed instructions below.
-
1.Tap the bottle to release the powder.
-
2.Remove the cap.
-
3.Accurately measure out 60 mL of water and pour the water into the bottle. (Figure 1)
-
4.Replace the cap and shake the bottle vigorously for a minimum of 30 seconds. (Figure 2)
-
5.Remove the cap.
-
6.Accurately measure out another 30 mL of water and add this to the bottle. You should always add a total of 90 mL of water irrespective of the dose prescribed. (Figure 3)
-
7.Replace the cap and shake the bottle vigorously for a minimum of 30 seconds. (Figure 4)
-
8.Remove the cap.
-
9.Press the bottle adaptor into the neck of the bottle (as shown on Figure 5, below). The adaptor is provided so that you can fill the oral syringe with medicine from the bottle. Replace the cap on the bottle.
-
10.Write the expiration date of the reconstituted oral suspension on the bottle label (the expiration date of the reconstituted oral suspension is 60 days from the date of reconstitution).
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Sildenafil was not carcinogenic when administered to rats for up to 24 months at 60 mg/kg/day, a dose resulting in total systemic exposure (AUC) to unbound sildenafil and its major metabolite 33- and 37-times, for male and female rats respectively, the human exposure at the RHD of 20 mg three times a day. Sildenafil was not carcinogenic when administered to male and female mice for up to 21 and 18 months, respectively, at doses up to a maximally tolerated level of 10 mg/kg/day, a dose equivalent to the RHD on a mg/m2 basis.
Sildenafil was negative in in vitro bacterial and Chinese hamster ovary cell assays to detect mutagenicity, and in vitro human lymphocytes and in vivo mouse micronucleus assays to detect clastogenicity.
There was no impairment of fertility in male or female rats given up to 60 mg sildenafil/kg/day, a dose producing a total systemic exposure (AUC) to unbound sildenafil and its major metabolite of 19- and 38- times for males and females, respectively, the human exposure at the RHD of 20 mg three times a day.
5.9 Vaso Occlusive Crisis in Patients With Pulmonary Hypertension Secondary to Sickle Cell Anemia (5.9 Vaso-occlusive Crisis in Patients with Pulmonary Hypertension Secondary to Sickle Cell Anemia)
In a small, prematurely terminated study of patients with pulmonary hypertension (PH) secondary to sickle cell disease, vaso-occlusive crises requiring hospitalization were more commonly reported by patients who received sildenafil than by those randomized to placebo. The effectiveness and safety of sildenafil in the treatment of PAH secondary to sickle cell anemia has not been established.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:57.845 · Updated: 2026-03-14T22:26:02.784287