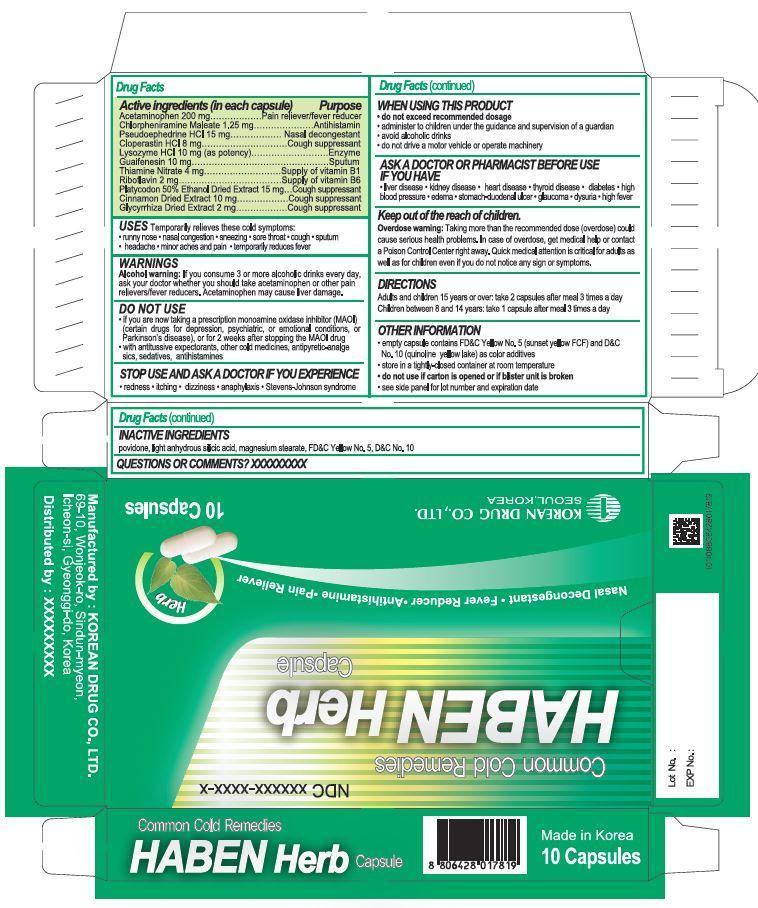
Drug Facts
569cfe1e-a9ed-4c85-94e7-1f42d5b1897a
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
chlorpheniramine maleate, acetaminophen, pseudoephedrine hydrochloride, cloperastin hydrochloride, lysozyme hydrochloride, guaifenesin, thiamine nitrate, riboflavin, platycodon 50% ethanol dried extract, cinnamon dried extract, glycyrrhize dried extract
Medication Information
Warnings and Precautions
[DO NOT USE]
if you are hypersensitive to this drug and any component of this drug
if you have a history of asthma after taking this drug, other cold drugs or antipyreticanalgesics
in infants under 3 months of age
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for
depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after
stopping the MAOI drug
with antitussive expectorants, other cold medicines, antipyretic-analgesics, sedatives,
antihistamines(certain drugs for rhinitis, preventive drugs for travel sickness, allergies)
[ASK A DOCTOR OR PHARMACIST BEFORE USE]
in infants or children under 15 years infected with chickenpox or influenza
in infants under 2 years of age only when it is necessary. Inform to the guardian/s and
monitor closely while administering
if you are genetically prone to uticaria, contact dermatitis, bronchial asthma, allergic rhinitis,
migraine, food allergy
If you have allergic reactions (e.g., fever, rash, arthralgia, asthma, itch, etc.) to this drug and
any component of this drug
if you have liver disease, kidney disease, heart disease, thyroid disease, diabetes, high blood
pressure, edema, stomach-duodenal ulcer, glaucoma, dysuria, high fever
if you are elderly or week
if you have bleeding , ache and discomfort in stomach
If (possibly) pregnant or breast-feeding
If you are taking a prescription drug for diabetes, goat, arthritis, anticoagulants and steroids
if you have cough by smoking, asthma, chronic bronchitis, emphysema, cough accompanied
with excessive sputum, fever, rash, persistent headache and cough lasting more than 1 week,
recurrent cough or chronic cough
[STOP USE AND ASK A DOCTOR IF]
you experience any of the following symptoms:
rash, redness, itching, nausea, vomiting, loss of appetite, constipation, edema, dysuria, severe
or persistent thirst, dizziness, anxiety, shivering, insomnia
you experience any of the following severe symptoms:
anaphylaxis (e.g., immediate rash, edema, shortness of breath along with paling of face,
cold sweat, cold hands and feet)
Stevens-Johnson syndrome (mucocutaneous syndrome) or Lyell syndrome (toxic epidermal
necrosis) (e.g., rash and redness accompanied with high fever, blisters as in burn over the
body or mucous membranes of mouth or eyes) asthma
liver dysfunction (e.g., drowsiness, jaundice)
interstitial pneumonia (e.g., shortness of breath, fever accompanied with cough)
aldosteronism: long-term continuous use of preparations containing over than 1g as licorice
(daily maximum dose) may cause hypokalemia, increased blood pressure, fluid retention of
sodium, edema, weight gain, aldosteronism. Observe closely (blood potassium concentration)
and stop taking this drug and when you have above symptoms.
myopathy may rarely occur as a result of hypokalemia. Observe closely and stop taking this
drug when you appear symptoms such as weakness, limb spasms, and numbness.
[WHEN USING THIS PRODUCT]
do not exceed recommended dosage
do not take for a long period of time
administer to children under the guidance and supervision of a guardian
avoid alcoholic drinks
do not drive a motor vehicle or operate machinery
barbiturates, tricyclic antidepressants, and alcohol may reduce the ability to metabolize large
quantities of acetaminophen, thereby increasing the plasma half-life of acetaminophen
myopathy may occur due to aldosteronism or hypokalemia when administered with drugs
containing potassium, licorice, glycyrrhizin acid or its salts, loop diuretics (furosemide,
ethacrynic acid) or thiazide diuretic (trichlormethiazide).
Indications and Usage
Adults and children 15 years or over: take 2 capsules after meal 3 times a day
Children between 8 and 14 years: take 1 capsule after meal 3 times a day
Dosage and Administration
for oral administration
Description
chlorpheniramine maleate, acetaminophen, pseudoephedrine hydrochloride, cloperastin hydrochloride, lysozyme hydrochloride, guaifenesin, thiamine nitrate, riboflavin, platycodon 50% ethanol dried extract, cinnamon dried extract, glycyrrhize dried extract
Section 50565-1
keep out of reach of the children
Section 51727-6
povidone, light anhydrous silicic acid, magnesium stearate
Section 51945-4
Section 55105-1
- temporarily relieves these cold symptomes
runny nose, nasal congestion, sneezing, sore throat, cough, sputum, headache, minor aches and pain
- temporarily reduces fever
Section 55106-9
chlorpheniramine maleate, acetaminophen, pseudoephedrine hydrochloride, cloperastin hydrochloride, lysozyme hydrochloride, guaifenesin, thiamine nitrate, riboflavin, platycodon 50% ethanol dried extract, cinnamon dried extract, glycyrrhize dried extract
Structured Label Content
Indications and Usage (34067-9)
Adults and children 15 years or over: take 2 capsules after meal 3 times a day
Children between 8 and 14 years: take 1 capsule after meal 3 times a day
Dosage and Administration (34068-7)
for oral administration
Warnings and Precautions (34071-1)
[DO NOT USE]
if you are hypersensitive to this drug and any component of this drug
if you have a history of asthma after taking this drug, other cold drugs or antipyreticanalgesics
in infants under 3 months of age
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for
depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after
stopping the MAOI drug
with antitussive expectorants, other cold medicines, antipyretic-analgesics, sedatives,
antihistamines(certain drugs for rhinitis, preventive drugs for travel sickness, allergies)
[ASK A DOCTOR OR PHARMACIST BEFORE USE]
in infants or children under 15 years infected with chickenpox or influenza
in infants under 2 years of age only when it is necessary. Inform to the guardian/s and
monitor closely while administering
if you are genetically prone to uticaria, contact dermatitis, bronchial asthma, allergic rhinitis,
migraine, food allergy
If you have allergic reactions (e.g., fever, rash, arthralgia, asthma, itch, etc.) to this drug and
any component of this drug
if you have liver disease, kidney disease, heart disease, thyroid disease, diabetes, high blood
pressure, edema, stomach-duodenal ulcer, glaucoma, dysuria, high fever
if you are elderly or week
if you have bleeding , ache and discomfort in stomach
If (possibly) pregnant or breast-feeding
If you are taking a prescription drug for diabetes, goat, arthritis, anticoagulants and steroids
if you have cough by smoking, asthma, chronic bronchitis, emphysema, cough accompanied
with excessive sputum, fever, rash, persistent headache and cough lasting more than 1 week,
recurrent cough or chronic cough
[STOP USE AND ASK A DOCTOR IF]
you experience any of the following symptoms:
rash, redness, itching, nausea, vomiting, loss of appetite, constipation, edema, dysuria, severe
or persistent thirst, dizziness, anxiety, shivering, insomnia
you experience any of the following severe symptoms:
anaphylaxis (e.g., immediate rash, edema, shortness of breath along with paling of face,
cold sweat, cold hands and feet)
Stevens-Johnson syndrome (mucocutaneous syndrome) or Lyell syndrome (toxic epidermal
necrosis) (e.g., rash and redness accompanied with high fever, blisters as in burn over the
body or mucous membranes of mouth or eyes) asthma
liver dysfunction (e.g., drowsiness, jaundice)
interstitial pneumonia (e.g., shortness of breath, fever accompanied with cough)
aldosteronism: long-term continuous use of preparations containing over than 1g as licorice
(daily maximum dose) may cause hypokalemia, increased blood pressure, fluid retention of
sodium, edema, weight gain, aldosteronism. Observe closely (blood potassium concentration)
and stop taking this drug and when you have above symptoms.
myopathy may rarely occur as a result of hypokalemia. Observe closely and stop taking this
drug when you appear symptoms such as weakness, limb spasms, and numbness.
[WHEN USING THIS PRODUCT]
do not exceed recommended dosage
do not take for a long period of time
administer to children under the guidance and supervision of a guardian
avoid alcoholic drinks
do not drive a motor vehicle or operate machinery
barbiturates, tricyclic antidepressants, and alcohol may reduce the ability to metabolize large
quantities of acetaminophen, thereby increasing the plasma half-life of acetaminophen
myopathy may occur due to aldosteronism or hypokalemia when administered with drugs
containing potassium, licorice, glycyrrhizin acid or its salts, loop diuretics (furosemide,
ethacrynic acid) or thiazide diuretic (trichlormethiazide).
Section 50565-1 (50565-1)
keep out of reach of the children
Section 51727-6 (51727-6)
povidone, light anhydrous silicic acid, magnesium stearate
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
- temporarily relieves these cold symptomes
runny nose, nasal congestion, sneezing, sore throat, cough, sputum, headache, minor aches and pain
- temporarily reduces fever
Section 55106-9 (55106-9)
chlorpheniramine maleate, acetaminophen, pseudoephedrine hydrochloride, cloperastin hydrochloride, lysozyme hydrochloride, guaifenesin, thiamine nitrate, riboflavin, platycodon 50% ethanol dried extract, cinnamon dried extract, glycyrrhize dried extract
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:49.966174 · Updated: 2026-03-14T22:53:48.902596