Drug Facts
55e65545-9869-413b-8239-bd2d43fe36d8
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethanol 60% by volume
Purpose
Antispetic handwash
Medication Information
Purpose
Antispetic handwash
Description
Ethanol 60% by volume
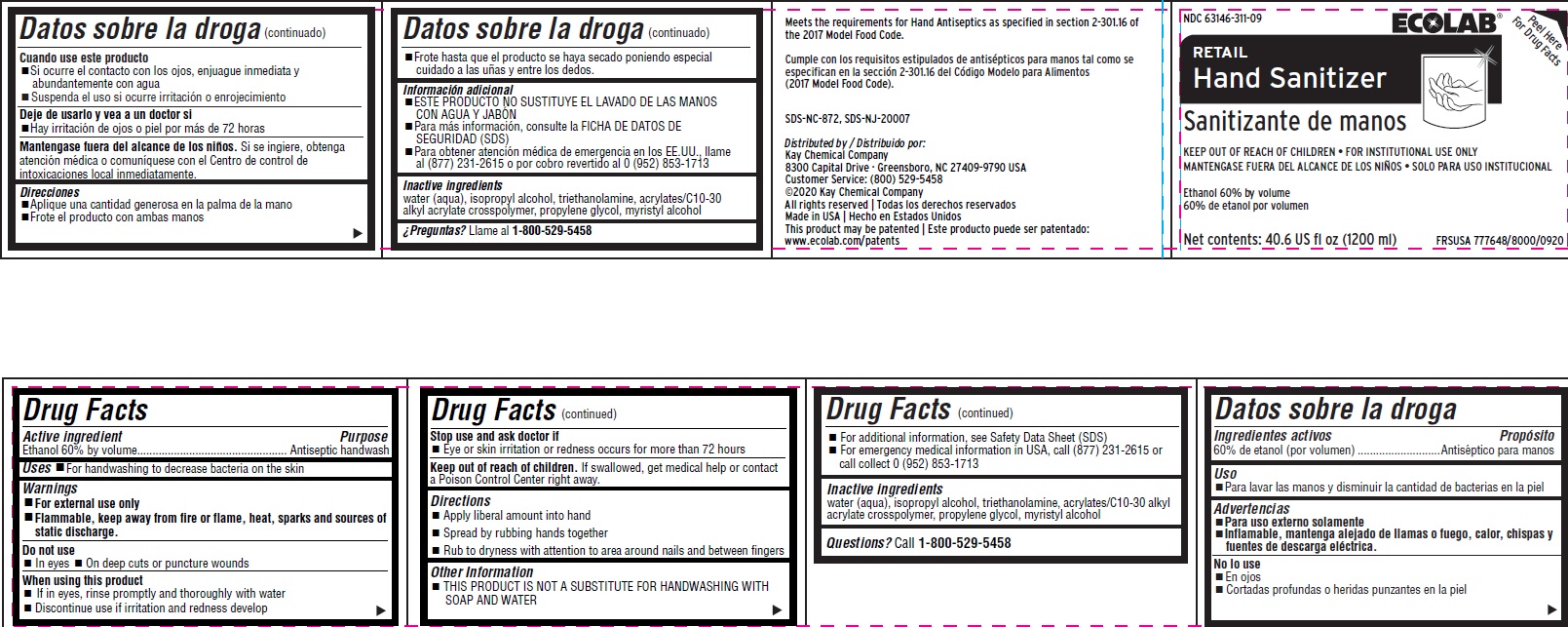
Uses
- For handwashing to decrease bacteria on the skin
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50569-3
Stop use and ask a doctor if
- Eye or skin irritation or redenss occurs for more than 72 hours
Section 51727-6
Inactive ingredients water (aqua), isopropyl alcohol, triethanolamine, acrylates/C10-30 alkyl acrylate crosspolymer, propylene glycol, myristyl alcohol
Section 53413-1
Questions? Call 1-800-529-5458
Warnings
- For external use only
- FLAMMABLE, keep away from fire or flame, heat, sparks and sources of static discharge
Directions
- Apply liberal amount into hand
- Spread by rubbing hands together
- Rub to dryness with attention to are around nails and between fingers
Do Not Use
- In eyes
- On deep cuts or puncture wounds
Active Ingredient
Ethanol 60% by volume
Other Information
- For additional information, see Safety Data Sheet (SDS)
- For emergency medical information in USA, call (877) 231-2615 or call collect 0 (952) 853-1713
When Using This Product
- If in eyes, rinse promptly and thoroughly with water
- Discontinue use if irritation and rendess develop
Principal Display Panel and Representative Label
RETAIL
Hand Sanitizer
Sanitizante de manos
KEEP OUT OF REACH OF CHILDREN • FOR INSTITUTIONAL USE ONLY
MANTENGASE FUERA DEL ALCANCE DE LOS NIÑOS • SOLO PARA USO INSTITUCIONAL
Ethanol 60% by volume
60% de etanol por volume
Net contents: 40.6 US fl oz (1200 ml)
FRSUSA 777648/8000/0920
Meets the requirements for Hand Antiseptics as specified in section 2-301.16 of the 2017 Model Food Code.
Cumple con los requisitos estipulados de antisépticos para manos tal como se especifican en la sección 2-301.16 del Código Modelo para Alimentos
(2017 Model Food Code).
SDS-NC-872, SDS-NJ-20007
Distributed by / Distribuido por:
Kay Chemical Company
8300 Capital Drive · Greensboro, NC 27409-9790 USA
Customer Service: (800) 529-5458
©2020 Kay Chemical Company
All rights reserved | Todas los derechos reservados
Made in USA | Hecho en Estados Unidos
This product may be patented | Este producto puede ser patentado:
Structured Label Content
Uses
- For handwashing to decrease bacteria on the skin
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50569-3 (50569-3)
Stop use and ask a doctor if
- Eye or skin irritation or redenss occurs for more than 72 hours
Section 51727-6 (51727-6)
Inactive ingredients water (aqua), isopropyl alcohol, triethanolamine, acrylates/C10-30 alkyl acrylate crosspolymer, propylene glycol, myristyl alcohol
Section 53413-1 (53413-1)
Questions? Call 1-800-529-5458
Purpose
Antispetic handwash
Warnings
- For external use only
- FLAMMABLE, keep away from fire or flame, heat, sparks and sources of static discharge
Directions
- Apply liberal amount into hand
- Spread by rubbing hands together
- Rub to dryness with attention to are around nails and between fingers
Do Not Use (Do not use)
- In eyes
- On deep cuts or puncture wounds
Active Ingredient (Active ingredient)
Ethanol 60% by volume
Other Information (Other information)
- For additional information, see Safety Data Sheet (SDS)
- For emergency medical information in USA, call (877) 231-2615 or call collect 0 (952) 853-1713
When Using This Product (When using this product)
- If in eyes, rinse promptly and thoroughly with water
- Discontinue use if irritation and rendess develop
Principal Display Panel and Representative Label (Principal display panel and representative label)
RETAIL
Hand Sanitizer
Sanitizante de manos
KEEP OUT OF REACH OF CHILDREN • FOR INSTITUTIONAL USE ONLY
MANTENGASE FUERA DEL ALCANCE DE LOS NIÑOS • SOLO PARA USO INSTITUCIONAL
Ethanol 60% by volume
60% de etanol por volume
Net contents: 40.6 US fl oz (1200 ml)
FRSUSA 777648/8000/0920
Meets the requirements for Hand Antiseptics as specified in section 2-301.16 of the 2017 Model Food Code.
Cumple con los requisitos estipulados de antisépticos para manos tal como se especifican en la sección 2-301.16 del Código Modelo para Alimentos
(2017 Model Food Code).
SDS-NC-872, SDS-NJ-20007
Distributed by / Distribuido por:
Kay Chemical Company
8300 Capital Drive · Greensboro, NC 27409-9790 USA
Customer Service: (800) 529-5458
©2020 Kay Chemical Company
All rights reserved | Todas los derechos reservados
Made in USA | Hecho en Estados Unidos
This product may be patented | Este producto puede ser patentado:
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:56.561041 · Updated: 2026-03-14T23:11:59.437858