55c0f077-e365-1d95-e054-00144ff8d46c
34390-5
HUMAN OTC DRUG LABEL
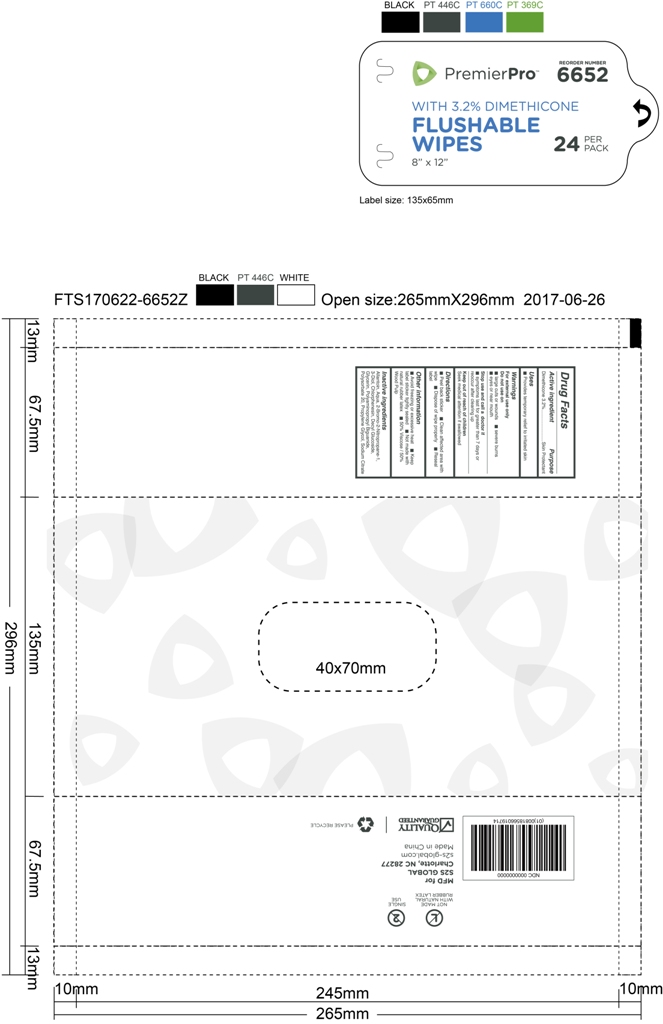
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dimethicone 3.2%
Purpose
Skin Protectant
Medication Information
Purpose
Skin Protectant
Description
Dimethicone 3.2%
Use
provides temporary relief to irritated skin
Section 50565-1
Keep out of reach of children ,seek medical attention if swallowed
Section 51945-4
Warning
For external use only
Do not use on
Large cuts or wounds
severe burns
eyes or near mouth
stop use and call a doctor if
symptoms last for greater than 7 days or reoccur after clearing up
Directions
peel back sticker
clean affected area with wipe
dispose of wipe properly
reseal label
Active Ingredient
Dimethicone 3.2%
Inactive Ingredient
Aqua
Glycerin
Propylene Glycol
Polyaminopropyl Biguanide
Polysorbate 20
Allantoin
Chlorphenesin
Decyl Glucoside
2-Bromo-2-Nitropropane-1,3-Diol
Sodium Citrate
Structured Label Content
Use
provides temporary relief to irritated skin
Section 50565-1 (50565-1)
Keep out of reach of children ,seek medical attention if swallowed
Section 51945-4 (51945-4)
Purpose
Skin Protectant
Warning
For external use only
Do not use on
Large cuts or wounds
severe burns
eyes or near mouth
stop use and call a doctor if
symptoms last for greater than 7 days or reoccur after clearing up
Directions
peel back sticker
clean affected area with wipe
dispose of wipe properly
reseal label
Active Ingredient
Dimethicone 3.2%
Inactive Ingredient (INACTIVE INGREDIENT)
Aqua
Glycerin
Propylene Glycol
Polyaminopropyl Biguanide
Polysorbate 20
Allantoin
Chlorphenesin
Decyl Glucoside
2-Bromo-2-Nitropropane-1,3-Diol
Sodium Citrate
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:29.163469 · Updated: 2026-03-14T22:54:59.525683