These Highlights Do Not Include All The Information Needed To Use Doxepin Tablets Safely And Effectively. See Full Prescribing Information For Doxepin Tablets.
554ab06f-6420-42b3-89d1-d97554c237d8
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Doxepin tablets is indicated for the treatment of insomnia characterized by difficulty with sleep maintenance. The clinical trials performed in support of efficacy were up to 3 months in duration .
Indications and Usage
Doxepin tablets is indicated for the treatment of insomnia characterized by difficulty with sleep maintenance. The clinical trials performed in support of efficacy were up to 3 months in duration .
Dosage and Administration
The dose of doxepin tablets should be individualized.
Warnings and Precautions
Need to Evaluate for Co-morbid Diagnoses: Reevaluate if insomnia persists after 7 to 10 days of use. ( 5.1 ) Abnormal thinking, behavioral changes, complex behaviors: May include "Sleep-driving" and hallucinations. Immediately evaluate any new onset behavioral changes. ( 5.2 ) Depression: Worsening of depression or suicidal thinking may occur. Prescribe the least amount feasible to avoid intentional overdose. ( 5.3 ) CNS-depressant effects: Use can impair alertness and motor coordination. Avoid engaging in hazardous activities such as operating a motor vehicle or heavy machinery after taking drug. ( 5.4 ) Do not use with alcohol. ( 5.4 , 7.3 ) Potential additive effects when used in combination with CNS depressants or sedating antihistamines. Dose reduction may be needed. ( 5.4 , 7.4 ) Patients with severe sleep apnea: Doxepin is ordinarily not recommended for use in this population. ( 8.7 )
Contraindications
• Hypersensitivity to doxepin hydrochloride, inactive ingredients, or other dibenzoxepines. (4.1) • Co-administration with Monoamine Oxidase Inhibitors (MAOIs): Do not administer if patient is taking MAOIs or has used MAOIs within the past two weeks. (4.2) • Untreated narrow angle glaucoma or severe urinary retention. (4.3)
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of labeling: Abnormal thinking and behavioral changes [see Warnings and Precautions (5.2) ]. Suicide risk and worsening of depression [see Warnings and Precautions (5.3) ]. CNS Depressant effects [see Warnings and Precautions (5.4) ].
Drug Interactions
MAO inhibitors: Doxepin should not be administered in patients on MAOIs within the past two weeks. ( 4.2 ) Cimetidine: Increases exposure to doxepin. ( 7.2 ) Alcohol: Sedative effects may be increased with doxepin. ( 7.3 , 5.4 ) CNS Depressants and Sedating Antihistamines: Sedative effects may be increased with doxepin. ( 7.4 , 5.4 ) Tolazamide: A case of severe hypoglycemia has been reported. ( 7.5 )
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from light.
How Supplied
Doxepin 3 mg tablets are oval shaped, blue, identified with debossed markings of "T" & "S" on one side and blank on the other side. NDC 51672-4246-2 Bottle of 30 tablets Doxepin 6 mg tablets are oval shaped, green, and are identified with debossed markings of "T" & "S" on one side and "6" on the other side: NDC 51672-4247-2 Bottle of 30 tablets
Medication Information
Warnings and Precautions
Need to Evaluate for Co-morbid Diagnoses: Reevaluate if insomnia persists after 7 to 10 days of use. ( 5.1 ) Abnormal thinking, behavioral changes, complex behaviors: May include "Sleep-driving" and hallucinations. Immediately evaluate any new onset behavioral changes. ( 5.2 ) Depression: Worsening of depression or suicidal thinking may occur. Prescribe the least amount feasible to avoid intentional overdose. ( 5.3 ) CNS-depressant effects: Use can impair alertness and motor coordination. Avoid engaging in hazardous activities such as operating a motor vehicle or heavy machinery after taking drug. ( 5.4 ) Do not use with alcohol. ( 5.4 , 7.3 ) Potential additive effects when used in combination with CNS depressants or sedating antihistamines. Dose reduction may be needed. ( 5.4 , 7.4 ) Patients with severe sleep apnea: Doxepin is ordinarily not recommended for use in this population. ( 8.7 )
Indications and Usage
Doxepin tablets is indicated for the treatment of insomnia characterized by difficulty with sleep maintenance. The clinical trials performed in support of efficacy were up to 3 months in duration .
Dosage and Administration
The dose of doxepin tablets should be individualized.
Contraindications
• Hypersensitivity to doxepin hydrochloride, inactive ingredients, or other dibenzoxepines. (4.1) • Co-administration with Monoamine Oxidase Inhibitors (MAOIs): Do not administer if patient is taking MAOIs or has used MAOIs within the past two weeks. (4.2) • Untreated narrow angle glaucoma or severe urinary retention. (4.3)
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of labeling: Abnormal thinking and behavioral changes [see Warnings and Precautions (5.2) ]. Suicide risk and worsening of depression [see Warnings and Precautions (5.3) ]. CNS Depressant effects [see Warnings and Precautions (5.4) ].
Drug Interactions
MAO inhibitors: Doxepin should not be administered in patients on MAOIs within the past two weeks. ( 4.2 ) Cimetidine: Increases exposure to doxepin. ( 7.2 ) Alcohol: Sedative effects may be increased with doxepin. ( 7.3 , 5.4 ) CNS Depressants and Sedating Antihistamines: Sedative effects may be increased with doxepin. ( 7.4 , 5.4 ) Tolazamide: A case of severe hypoglycemia has been reported. ( 7.5 )
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from light.
How Supplied
Doxepin 3 mg tablets are oval shaped, blue, identified with debossed markings of "T" & "S" on one side and blank on the other side. NDC 51672-4246-2 Bottle of 30 tablets Doxepin 6 mg tablets are oval shaped, green, and are identified with debossed markings of "T" & "S" on one side and "6" on the other side: NDC 51672-4247-2 Bottle of 30 tablets
Description
Doxepin tablets is indicated for the treatment of insomnia characterized by difficulty with sleep maintenance. The clinical trials performed in support of efficacy were up to 3 months in duration .
Section 42229-5
Associated with Discontinuation of Treatment
The percentage of subjects discontinuing Phase 1, 2, and 3 trials for an adverse reaction was 0.6% in the placebo group compared to 0.4%, 1%, and 0.7% in the doxepin 1 mg, 3 mg, and 6 mg groups, respectively. No reaction that resulted in discontinuation occurred at a rate greater than 0.5%.
Section 42231-1
|
Dispense with Medication Guide available at: https://www.sunpharma.com/usa/products MEDICATION GUIDE Doxepin (dox'e pin) Tablets What is the most important information I should know about doxepin tablets? Doxepin Tablets can cause serious side effects including: After taking doxepin tablets, you may get up out of bed while not being fully awake and do an activity that you do not know you are doing. The next morning, you may not remember that you did anything during the night. You have a higher chance for doing these activities if you drink alcohol or take other medicines that make you sleepy with doxepin tablets. Reported activities include:
Stop taking doxepin tablets and call your healthcare provider right away if you find out that you have done any of the above activities after taking doxepin tablets. Important:
Take doxepin tablets 30 minutes before bedtime. After taking doxepin tablets, you should only do activities needed to get ready for bed. |
|
What are doxepin tablets? Doxepin tablets is a prescription medicine used to treat adults who have trouble staying asleep. It is not known if doxepin tablets is safe and effective in children. |
|
|
Before taking doxepin tablets, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all of the medicines you take including prescription and over-the-counter medicines, vitamins and herbal supplements. Doxepin tablets and other medicines may affect each other causing side effects. Doxepin tablets may affect the way other medicines work, and other medicines may affect how doxepin tablets works. Especially tell your healthcare provider if you take:
Know the medicines you take. Keep a list of your medicines with you to show your healthcare provider and pharmacist each time you get a new medicine. |
|
How should I take doxepin tablets?
|
|
What should I avoid during treatment with doxepin tablets?
|
|
What are the possible side effects of doxepin tablets? Doxepin tablets can cause serious side effects including:
The most common side effects of doxepin tablets include:
Doxepin tablets may cause fertility problems in females and males, which may affect your ability to have children. Talk to your healthcare provider if you have concerns about fertility. These are not all of the possible side effects of doxepin tablets. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store doxepin tablets?
Keep doxepin tablets and all medicines out of the reach of children. |
|
General Information about the safe and effective use of doxepin tablets. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use doxepin tablets for a condition for which it was not prescribed. Do not give doxepin tablets to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about doxepin tablets that is written for healthcare professionals. |
|
What are the ingredients in doxepin tablets? Active Ingredient: doxepin hydrochloride Inactive Ingredients: colloidal silicone dioxide, lactose anhydrous, magnesium stearate and microcrystalline cellulose. The 3 mg tablet also contains FD&C Blue No. 1. The 6 mg tablet also contains FD&C Yellow No. 10 and FD&C Blue No. 1 Mfd. by: Taro Pharmaceutical Industries Ltd. Haifa Bay, Israel 2624761 Dist. by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532 For more information, contact Taro Pharmaceuticals U.S.A., at 1-866-923-4914. |
9.2 Abuse
Doxepin is not associated with abuse potential in animals or in humans. Physicians should carefully evaluate patients for history of drug abuse and follow such patients closely, observing them for signs of misuse or abuse of doxepin (e.g., incrementation of dose, drug-seeking behavior).
7.3 Alcohol
When taken with doxepin, the sedative effects of alcohol may be potentiated [see Warnings and Precautions (5.2, 5.4)].
10 Overdosage
Doxepin is routinely administered for indications other than insomnia at doses 10-to 50-fold higher than the highest recommended dose of doxepin.
The signs and symptoms associated with doxepin use at doses several-fold higher than the maximum recommended dose (Excessive dose) of doxepin for the treatment of insomnia are described [see Overdosage (10.1)] , as are signs and symptoms associated with higher multiples of the maximum recommended dose (Critical overdose) [see Overdosage (10.2)] .
7.2 Cimetidine
Doxepin exposure is doubled with concomitant administration of cimetidine, a nonspecific inhibitor of CYP isozymes. A maximum dose of 3 mg is recommended in adults and elderly when cimetidine is co-administered with Doxepin [see Clinical Pharmacology (12.3)]
7.5 Tolazamide
A case of severe hypoglycemia has been reported in a type II diabetic patient maintained on tolazamide (1 g/day) 11 days after the addition of oral doxepin (75 mg/day).
9.3 Dependence
In a brief assessment of adverse events observed during discontinuation of doxepin following chronic administration, no symptoms indicative of a withdrawal syndrome were observed. Thus, doxepin does not appear to produce physical dependence.
11. Description
Doxepin tablets are available in 3 mg and 6 mg strength for oral administration. Each tablet contains 3.39 mg or 6.78 mg doxepin hydrochloride USP, equivalent to 3 mg and 6 mg of doxepin, respectively.
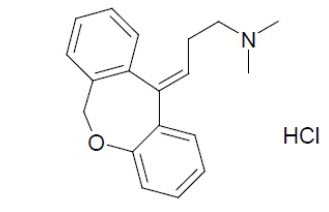
Chemically, doxepin hydrochloride USP is an (E) and (Z) geometric, isomeric mixture of 1 propanamine, 3-Dibenz[ b,e]oxepin-11(6 H)ylidene- N,N-dimethyl 1-Propanamine, Hydrochloride. It has the following structure formula:
Doxepin hydrochloride USP appears as white or almost white crystalline powder, that is freely soluble in water. It has a molecular weight of 315.84 g/mole and molecular formula of C 19H 21NO∙HCl.
Each doxepin tablet includes the following inactive ingredients: colloidal silicone dioxide, lactose anhydrous, magnesium stearate and microcrystalline cellulose. The 3 mg tablet also contains FD&C Blue No. 1. The 6 mg tablet also contains FD&C Yellow No. 10 and FD&C Blue No. 1
16.1 How Supplied
Doxepin 3 mg tablets are oval shaped, blue, identified with debossed markings of "T" & "S" on one side and blank on the other side.
| NDC 51672-4246-2 | Bottle of 30 tablets |
Doxepin 6 mg tablets are oval shaped, green, and are identified with debossed markings of "T" & "S" on one side and "6" on the other side:
| NDC 51672-4247-2 | Bottle of 30 tablets |
8.4 Pediatric Use
The safety and effectiveness of doxepin in pediatric patients have not been evaluated .
8.5 Geriatric Use
A total of 362 subjects who were ≥ 65 years and 86 subjects who were ≥ 75 years received doxepin in controlled clinical studies. No overall differences in safety or effectiveness were observed between these subjects and younger adult subjects. Greater sensitivity of some older individuals cannot be ruled out. Sleep-promoting drugs may cause confusion and over-sedation in the elderly. A starting dose of 3 mg is recommended in this population and evaluation prior to considering dose escalation is recommended [see Dosage and Administration (2.2)].
2.3 Administration
Doxepin tablets should be taken within 30 minutes of bedtime.
To minimize the potential for next day effects, doxepin tablets should not be taken within 3 hours of a meal [see Clinical Pharmacology (12.3)].
The total doxepin tablets dose should not exceed 6 mg per day.
4 Contraindications
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of labeling:
- Abnormal thinking and behavioral changes [see Warnings and Precautions (5.2)].
- Suicide risk and worsening of depression [see Warnings and Precautions (5.3)].
- CNS Depressant effects [see Warnings and Precautions (5.4)].
7 Drug Interactions
2.1 Dosing in Adults
The recommended dose of doxepin tablets for adults is 6 mg once daily. A 3 mg once daily dose may be appropriate for some patients, if clinically indicated.
4.1 Hypersensitivity
Doxepin is contraindicated in individuals who have shown hypersensitivity to doxepin HCl, any of its inactive ingredients, or other dibenzoxepines.
12.2 Pharmacodynamics
Doxepin has high binding affinity to the H1 receptor (Ki < 1 nM).
1 Indications and Usage
Doxepin tablets is indicated for the treatment of insomnia characterized by difficulty with sleep maintenance. The clinical trials performed in support of efficacy were up to 3 months in duration .
12.1 Mechanism of Action
The mechanism of action of doxepin in sleep maintenance is unclear; however, doxepin's effect could be mediated through antagonism of the H1 receptor.
9.1 Controlled Substance
Doxepin is not a controlled substance.
16.2 Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from light.
2.2 Dosing in the Elderly
The recommended starting dose of doxepin tablets in elderly patients (≥ 65 years old) is 3 mg once daily. The daily dose can be increased to 6 mg, if clinically indicated.
5 Warnings and Precautions
5.4 Cns Depressant Effects
After taking doxepin, patients should confine their activities to those necessary to prepare for bed. Patients should avoid engaging in hazardous activities, such as operating a motor vehicle or heavy machinery, at night after taking doxepin, and should be cautioned about potential impairment in the performance of such activities that may occur the day following ingestion.
When taken with doxepin, the sedative effects of alcoholic beverages, sedating antihistamines, and other CNS depressants may be potentiated [see Warnings and Precautions (5.2) and Drug Interactions (7.3, 7.4)] . Patients should not consume alcohol with doxepin
[see Warnings and Precautions (5.2) and Drug Interactions (7.3)].
Patients should be cautioned about potential additive effects of doxepin used in combination with CNS depressants or sedating antihistamines [see Warnings and Precautions (5.2) and Drug Interactions (7.4)].
10.3 Recommended Management
As management of overdose is complex and changing, it is recommended that the physician contact a poison control center for current information on treatment. In addition, the possibility of a multiple drug ingestion should be considered.
If an overdose is suspected, an ECG should be obtained and cardiac monitoring should be initiated immediately. The patient's airway should be protected, an intravenous line should be established, and gastric decontamination should be initiated. A minimum of six hours of observation with cardiac monitoring and observation for signs of CNS or respiratory depression, hypotension, cardiac dysrhythmias and/or conduction blocks, and seizures is strongly advised. If signs of toxicity occur at any time during this period, extended monitoring is recommended. There are case reports of patients succumbing to fatal dysrhythmias late after overdose; these patients had clinical evidence of significant poisoning prior to death and most received inadequate gastrointestinal decontamination. Monitoring of plasma drug levels should not guide management of the patient.
2 Dosage and Administration
The dose of doxepin tablets should be individualized.
3 Dosage Forms and Strengths
Doxepin is an immediate-release, oval-shaped, tablet for oral administration available in strengths of 3 mg and 6 mg. The tablets for the 3 mg are blue and are identified with debossed markings of "T" & "S" on one side and blank on the other side. The 6 mg are green and are identified with debossed markings of "T" & "S" on one side and "6" on the other side. Doxepin tablets are not scored.
7.1 Cytochrome P450 Isozymes
Doxepin is primarily metabolized by hepatic cytochrome P450 isozymes CYP2C19 and CYP2D6, and to a lesser extent, by CYP1A2 and CYP2C9. Inhibitors of these isozymes may increase the exposure of doxepin. Doxepin is not an inhibitor of any CYP isozymes at therapeutically relevant concentrations. The ability of doxepin to induce CYP isozymes is not known.
8 Use in Specific Populations
6.1 Clinical Trials Experience
The pre-marketing development program for doxepin included doxepin HCl exposures in 1017 subjects (580 insomnia patients and 437 healthy subjects) from 12 studies conducted in the United States. 863 of these subjects (580 insomnia patients and 283 healthy subjects) participated in six randomized, placebo-controlled efficacy studies with Doxepin doses of 1 mg, 3 mg, and 6 mg for up to 3-months in duration.
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. However, data from the doxepin studies provide the physician with a basis for estimating the relative contributions of drug and non-drug factors to adverse reaction incidence rates in the populations studied.
14.1 Controlled Clinical Trials
The efficacy of doxepin for improving sleep maintenance was supported by six randomized, double-blind studies up to 3 months in duration that included 1,423 subjects, 18 to 93 years of age, with chronic (N=858) or transient (N=565) insomnia. Doxepin tablets were evaluated at doses of 1 mg, 3 mg, and 6 mg relative to placebo in inpatient (sleep laboratory) and outpatient settings.
The primary efficacy measures for assessment of sleep maintenance were the objective and subjective time spent awake after sleep onset (respectively, objective Wake After Sleep Onset [WASO] and subjective WASO). Subjects in studies of chronic insomnia were required to have at least a 3-month history of insomnia.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
4.3 Glaucoma and Urinary Retention
Doxepin is contraindicated in individuals with untreated narrow angle glaucoma or severe urinary retention.
8.7 Use in Patients With Sleep Apnea
Doxepin has not been studied in patients with obstructive sleep apnea. Since hypnotics have the capacity to depress respiratory drive, precautions should be taken if doxepin is prescribed to patients with compromised respiratory function. In patients with severe sleep apnea, doxepin is ordinarily not recommended for use.
10.1 Signs and Symptoms of Excessive Doses
The following adverse effects have been associated with use of doxepin at doses higher than 6 mg.
Anticholinergic Effects:constipation and urinary retention.
Central Nervous System:disorientation, hallucinations, numbness, paresthesias, extrapyramidal symptoms, seizures, tardive dyskinesia.
Cardiovascular:hypotension.
Gastrointestinal:aphthous stomatitis, indigestion.
Endocrine:raised libido, testicular swelling, gynecomastia in males, enlargement of breasts and galactorrhea in the female, raising or lowering of blood sugar levels, and syndrome of inappropriate antidiuretic hormone secretion.
Other:tinnitus, weight gain, sweating, flushing, jaundice, alopecia, exacerbation of asthma, and hyperpyrexia (in association with chlorpromazine).
5.1 Need to Evaluate for Comorbid Diagnoses
Because sleep disturbances may be the presenting manifestation of a physical and/or psychiatric disorder, symptomatic treatment of insomnia should be initiated only after careful evaluation of the patient. The failure of insomnia to remit after 7 to 10 days of treatment may indicate the presence of a primary psychiatric and/or medical illness that should be evaluated.Exacerbation of insomnia or the emergence of new cognitive or behavioral abnormalities may be the consequence of an unrecognized psychiatric or physical disorder. Such findings have emerged during the course of treatment with hypnotic drugs.
8.6 Use in Patients With Hepatic Impairment
Patients with hepatic impairment may display higher doxepin concentrations than healthy individuals. Initiate doxepin treatment with 3 mg in patients with hepatic impairment and monitor closely for adverse daytime effects. [see Clinical Pharmacology (12.3)]
10.2 Signs and Symptoms of Critical Overdose
Manifestations of doxepin critical overdose include: cardiac dysrhythmias, severe hypotension, convulsions, and CNS depression including coma. Electrocardiogram changes, particularly in QRS axis or width, are clinically significant indicators of tricyclic compound toxicity. Other signs of overdose may include, but are not limited to: confusion, disturbed concentration, transient visual hallucinations, dilated pupils, agitation, hyperactive reflexes, stupor, drowsiness, muscle rigidity, vomiting, hypothermia, hyperpyrexia.
5.2 Abnormal Thinking and Behavioral Changes
Complex behaviors such as "sleep-driving" (i.e., driving while not fully awake after ingestion of a hypnotic, with amnesia for the event) have been reported with hypnotics. These events can occur in hypnotic-naive as well as in hypnotic-experienced persons. Although behaviors such as "sleep-driving" may occur with hypnotics alone at therapeutic doses, the use of alcohol and other CNS depressants with hypnotics appears to increase the risk of such behaviors, as does the use of hypnotics at doses exceeding the maximum recommended dose. Due to the risk to the patient and the community, discontinuation of doxepin should be strongly considered for patients who report a "sleep-driving" episode. Other complex behaviors (e.g., preparing and eating food, making phone calls, or having sex) have been reported in patients who are not fully awake after taking a hypnotic. As with "sleep-driving", patients usually do not remember these events. Amnesia, anxiety and other neuro-psychiatric symptoms may occur unpredictably.
5.3 Suicide Risk and Worsening of Depression
In primarily depressed patients, worsening of depression, including suicidal thoughts and actions (including completed suicides), has been reported in association with the use of hypnotics.
Doxepin, the active ingredient in doxepin tablets, is an antidepressant at doses 10-to 100-fold higher than in doxepin tablets. Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Risk from the lower dose of doxepin in doxepin tablets cannot be excluded.
It can rarely be determined with certainty whether a particular instance of the abnormal behaviors listed above is drug induced, spontaneous in origin, or a result of an underlying psychiatric or physical disorder. Nonetheless, the emergence of any new behavioral sign or symptom of concern requires careful and immediate evaluation.
7.4 Cns Depressants and Sedating Antihistamines
When taken with doxepin, the sedative effects of sedating antihistamines and CNS depressants may be potentiated [see Warnings and Precautions (5.2, 5.4)].
Principal Display Panel 3 Mg Tablet Bottle Label
NDC 51672-4246-2
30 Tablets
Doxepin
Tablets
3 mg*
Rx only
Principal Display Panel 6 Mg Tablet Bottle Label
NDC 51672-4247-2
30 Tablets
Doxepin
Tablets
6 mg*
Rx only
4.2 Co Administration With Monoamine Oxidase Inhibitors (maois)
Serious side effects and even death have been reported following the concomitant use of certain drugs with MAO inhibitors. Do not administer doxepin if patient is currently on MAOIs or has MAOIs within the past two weeks. The exact length of time may vary depending on the particular MAOI dosage and duration of treatment.
6.3 Other Reactions Observed During the Pre Marketing Evaluation of Doxepin
Doxepin was administered to 1017 subjects in clinical trials in the United States. Treatment-emergent adverse reactions recorded by clinical investigators were standardized using a modified MedDRA dictionary of preferred terms. The following is a list of MedDRA terms that reflect treatment-emergent adverse reactions reported by subjects treated with doxepin.
Adverse reactions are further categorized by body system and listed in order of decreasing frequency according to the following definitions: Frequent adverse reactions are those that occurred on one or more occasions in at least 1/100 subjects; Infrequent adverse reactions are those that occurred in fewer than 1/100 subjects and more than 1/1000 subjects. Rare adverse reactions are those that occurred in fewer than 1/1000 subjects. Adverse reactions that are listed in Table 1 are not included in the following listing of frequent, infrequent, and rare AEs.
Blood and Lymphatic System Disorders:Infrequent: anemia; Rare: thrombocythemia.
Cardiac Disorders:Rare: atrioventricular block, palpitations, tachycardia, ventricular extrasystoles.
Ear and Labyrinth Disorders:Rare: ear pain, hypoacusis, motion sickness, tinnitus, tympanic membrane perforation.
Eye Disorders:Infrequent: eye redness, vision blurred; Rare: blepharospasm, diplopia, eye pain, lacrimation decreased.
Gastrointestinal Disorders:Infrequent: abdominal pain, dry mouth, gastroesophageal reflux disease, vomiting; Rare: dyspepsia, constipation, gingival recession, haematochezia, lip blister.
General Disorders and Administration Site Conditions: Infrequent: asthenia, chest pain, fatigue; Rare: chills, gait abnormal, edema peripheral.
Hepatobiliary Disorders:Rare: hyperbilirubinemia.
Immune System Disorders:Rare: hypersensitivity.
Infections and Infestations: Infrequent: bronchitis, fungal infection, laryngitis, sinusitis, tooth infection, urinary tract infection, viral infection; Rare: cellulitis staphylococcal, eye infection, folliculitis, gastroenteritis viral, herpes zoster, infective tenosynovitis, influenza, lower respiratory tract infection, onychomycosis, pharyngitis, pneumonia.
Injury, Poisoning and Procedural Complications:Infrequent: back injury, fall, joint sprain; Rare: bone fracture, skin laceration.
Investigations:Infrequent: blood glucose increased; Rare: alanine aminotransferase increased, blood pressure decreased, blood pressure increased, electrocardiogram ST-T segment abnormal, electrocardiogram QRS complex abnormal, heart rate decreased, neutrophil count decreased, QRS axis abnormal, transaminases increased.
Metabolism and Nutrition Disorders:Infrequent: anorexia, decreased appetite, hyperkalemia, hypermagnesemia, increased appetite; Rare: hypokalemia.
Musculoskeletal and Connective Tissue Disorders:Infrequent: arthralgia, back pain, myalgia, neck pain, pain in extremity; Rare: joint range of motion decreased, muscle cramp, sensation of heaviness.
Neoplasms Benign, Malignant and Unspecified (Including Cysts and Polyps): Rare: lung adenocarcinoma stage I, malignant melanoma.
Nervous System Disorders: Frequent: dizziness; Infrequent: dysgeusia, lethargy, parasthesia, syncope; Rare: ageusia, ataxia, cerebrovascular accident, disturbance in attention, migraine, sleep paralysis, syncope vasovagal, tremor.
Psychiatric Disorders: Infrequent: abnormal dreams, adjustment disorder, anxiety, depression; Rare: confusional state, elevated mood, insomnia, libido decreased, nightmare.
Reproductive System and Breast Disorders: Rare: breast cyst, dysmenorrhea.
Renal and Urinary Disorders: Rare: dysuria, enuresis, hemoglobinuria, nocturia.
Respiratory, Thoracic and Mediastinal Disorders: Infrequent: nasal congestion, pharyngolaryngeal pain, sinus congestion, wheezing; Rare: cough, crackles lung, nasopharyngeal disorder, rhinorrhea, dyspnea.
Skin and Subcutaneous Tissue Disorders: Infrequent: skin irritation; Rare: cold sweat, dermatitis, erythema, hyperhidrosis, pruritis, rash, rosacea.
Surgical and Medical Procedures: Rare: arthrodesis.
Vascular Disorders: Infrequent: pallor; Rare: blood pressure inadequately controlled, hematoma, hot flush.
In addition, the reactions below have been reported for other tricyclics and may be idiosyncratic (not related to dose).
Allergic:photosensitization, skin rash.
Hematologic:agranulocytosis, eosinophilia, leukopenia, purpura, thrombocytopenia
Structured Label Content
Section 42229-5 (42229-5)
Associated with Discontinuation of Treatment
The percentage of subjects discontinuing Phase 1, 2, and 3 trials for an adverse reaction was 0.6% in the placebo group compared to 0.4%, 1%, and 0.7% in the doxepin 1 mg, 3 mg, and 6 mg groups, respectively. No reaction that resulted in discontinuation occurred at a rate greater than 0.5%.
Section 42231-1 (42231-1)
|
Dispense with Medication Guide available at: https://www.sunpharma.com/usa/products MEDICATION GUIDE Doxepin (dox'e pin) Tablets What is the most important information I should know about doxepin tablets? Doxepin Tablets can cause serious side effects including: After taking doxepin tablets, you may get up out of bed while not being fully awake and do an activity that you do not know you are doing. The next morning, you may not remember that you did anything during the night. You have a higher chance for doing these activities if you drink alcohol or take other medicines that make you sleepy with doxepin tablets. Reported activities include:
Stop taking doxepin tablets and call your healthcare provider right away if you find out that you have done any of the above activities after taking doxepin tablets. Important:
Take doxepin tablets 30 minutes before bedtime. After taking doxepin tablets, you should only do activities needed to get ready for bed. |
|
What are doxepin tablets? Doxepin tablets is a prescription medicine used to treat adults who have trouble staying asleep. It is not known if doxepin tablets is safe and effective in children. |
|
|
Before taking doxepin tablets, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all of the medicines you take including prescription and over-the-counter medicines, vitamins and herbal supplements. Doxepin tablets and other medicines may affect each other causing side effects. Doxepin tablets may affect the way other medicines work, and other medicines may affect how doxepin tablets works. Especially tell your healthcare provider if you take:
Know the medicines you take. Keep a list of your medicines with you to show your healthcare provider and pharmacist each time you get a new medicine. |
|
How should I take doxepin tablets?
|
|
What should I avoid during treatment with doxepin tablets?
|
|
What are the possible side effects of doxepin tablets? Doxepin tablets can cause serious side effects including:
The most common side effects of doxepin tablets include:
Doxepin tablets may cause fertility problems in females and males, which may affect your ability to have children. Talk to your healthcare provider if you have concerns about fertility. These are not all of the possible side effects of doxepin tablets. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store doxepin tablets?
Keep doxepin tablets and all medicines out of the reach of children. |
|
General Information about the safe and effective use of doxepin tablets. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use doxepin tablets for a condition for which it was not prescribed. Do not give doxepin tablets to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about doxepin tablets that is written for healthcare professionals. |
|
What are the ingredients in doxepin tablets? Active Ingredient: doxepin hydrochloride Inactive Ingredients: colloidal silicone dioxide, lactose anhydrous, magnesium stearate and microcrystalline cellulose. The 3 mg tablet also contains FD&C Blue No. 1. The 6 mg tablet also contains FD&C Yellow No. 10 and FD&C Blue No. 1 Mfd. by: Taro Pharmaceutical Industries Ltd. Haifa Bay, Israel 2624761 Dist. by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532 For more information, contact Taro Pharmaceuticals U.S.A., at 1-866-923-4914. |
9.2 Abuse
Doxepin is not associated with abuse potential in animals or in humans. Physicians should carefully evaluate patients for history of drug abuse and follow such patients closely, observing them for signs of misuse or abuse of doxepin (e.g., incrementation of dose, drug-seeking behavior).
7.3 Alcohol
When taken with doxepin, the sedative effects of alcohol may be potentiated [see Warnings and Precautions (5.2, 5.4)].
10 Overdosage (10 OVERDOSAGE)
Doxepin is routinely administered for indications other than insomnia at doses 10-to 50-fold higher than the highest recommended dose of doxepin.
The signs and symptoms associated with doxepin use at doses several-fold higher than the maximum recommended dose (Excessive dose) of doxepin for the treatment of insomnia are described [see Overdosage (10.1)] , as are signs and symptoms associated with higher multiples of the maximum recommended dose (Critical overdose) [see Overdosage (10.2)] .
7.2 Cimetidine
Doxepin exposure is doubled with concomitant administration of cimetidine, a nonspecific inhibitor of CYP isozymes. A maximum dose of 3 mg is recommended in adults and elderly when cimetidine is co-administered with Doxepin [see Clinical Pharmacology (12.3)]
7.5 Tolazamide
A case of severe hypoglycemia has been reported in a type II diabetic patient maintained on tolazamide (1 g/day) 11 days after the addition of oral doxepin (75 mg/day).
9.3 Dependence
In a brief assessment of adverse events observed during discontinuation of doxepin following chronic administration, no symptoms indicative of a withdrawal syndrome were observed. Thus, doxepin does not appear to produce physical dependence.
11. Description (11. DESCRIPTION)
Doxepin tablets are available in 3 mg and 6 mg strength for oral administration. Each tablet contains 3.39 mg or 6.78 mg doxepin hydrochloride USP, equivalent to 3 mg and 6 mg of doxepin, respectively.
Chemically, doxepin hydrochloride USP is an (E) and (Z) geometric, isomeric mixture of 1 propanamine, 3-Dibenz[ b,e]oxepin-11(6 H)ylidene- N,N-dimethyl 1-Propanamine, Hydrochloride. It has the following structure formula:
Doxepin hydrochloride USP appears as white or almost white crystalline powder, that is freely soluble in water. It has a molecular weight of 315.84 g/mole and molecular formula of C 19H 21NO∙HCl.
Each doxepin tablet includes the following inactive ingredients: colloidal silicone dioxide, lactose anhydrous, magnesium stearate and microcrystalline cellulose. The 3 mg tablet also contains FD&C Blue No. 1. The 6 mg tablet also contains FD&C Yellow No. 10 and FD&C Blue No. 1
16.1 How Supplied
Doxepin 3 mg tablets are oval shaped, blue, identified with debossed markings of "T" & "S" on one side and blank on the other side.
| NDC 51672-4246-2 | Bottle of 30 tablets |
Doxepin 6 mg tablets are oval shaped, green, and are identified with debossed markings of "T" & "S" on one side and "6" on the other side:
| NDC 51672-4247-2 | Bottle of 30 tablets |
8.4 Pediatric Use
The safety and effectiveness of doxepin in pediatric patients have not been evaluated .
8.5 Geriatric Use
A total of 362 subjects who were ≥ 65 years and 86 subjects who were ≥ 75 years received doxepin in controlled clinical studies. No overall differences in safety or effectiveness were observed between these subjects and younger adult subjects. Greater sensitivity of some older individuals cannot be ruled out. Sleep-promoting drugs may cause confusion and over-sedation in the elderly. A starting dose of 3 mg is recommended in this population and evaluation prior to considering dose escalation is recommended [see Dosage and Administration (2.2)].
2.3 Administration
Doxepin tablets should be taken within 30 minutes of bedtime.
To minimize the potential for next day effects, doxepin tablets should not be taken within 3 hours of a meal [see Clinical Pharmacology (12.3)].
The total doxepin tablets dose should not exceed 6 mg per day.
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in greater detail in other sections of labeling:
- Abnormal thinking and behavioral changes [see Warnings and Precautions (5.2)].
- Suicide risk and worsening of depression [see Warnings and Precautions (5.3)].
- CNS Depressant effects [see Warnings and Precautions (5.4)].
7 Drug Interactions (7 DRUG INTERACTIONS)
2.1 Dosing in Adults
The recommended dose of doxepin tablets for adults is 6 mg once daily. A 3 mg once daily dose may be appropriate for some patients, if clinically indicated.
4.1 Hypersensitivity
Doxepin is contraindicated in individuals who have shown hypersensitivity to doxepin HCl, any of its inactive ingredients, or other dibenzoxepines.
12.2 Pharmacodynamics
Doxepin has high binding affinity to the H1 receptor (Ki < 1 nM).
1 Indications and Usage (1 INDICATIONS AND USAGE)
Doxepin tablets is indicated for the treatment of insomnia characterized by difficulty with sleep maintenance. The clinical trials performed in support of efficacy were up to 3 months in duration .
12.1 Mechanism of Action
The mechanism of action of doxepin in sleep maintenance is unclear; however, doxepin's effect could be mediated through antagonism of the H1 receptor.
9.1 Controlled Substance
Doxepin is not a controlled substance.
16.2 Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from light.
2.2 Dosing in the Elderly
The recommended starting dose of doxepin tablets in elderly patients (≥ 65 years old) is 3 mg once daily. The daily dose can be increased to 6 mg, if clinically indicated.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
5.4 Cns Depressant Effects (5.4 CNS Depressant Effects)
After taking doxepin, patients should confine their activities to those necessary to prepare for bed. Patients should avoid engaging in hazardous activities, such as operating a motor vehicle or heavy machinery, at night after taking doxepin, and should be cautioned about potential impairment in the performance of such activities that may occur the day following ingestion.
When taken with doxepin, the sedative effects of alcoholic beverages, sedating antihistamines, and other CNS depressants may be potentiated [see Warnings and Precautions (5.2) and Drug Interactions (7.3, 7.4)] . Patients should not consume alcohol with doxepin
[see Warnings and Precautions (5.2) and Drug Interactions (7.3)].
Patients should be cautioned about potential additive effects of doxepin used in combination with CNS depressants or sedating antihistamines [see Warnings and Precautions (5.2) and Drug Interactions (7.4)].
10.3 Recommended Management
As management of overdose is complex and changing, it is recommended that the physician contact a poison control center for current information on treatment. In addition, the possibility of a multiple drug ingestion should be considered.
If an overdose is suspected, an ECG should be obtained and cardiac monitoring should be initiated immediately. The patient's airway should be protected, an intravenous line should be established, and gastric decontamination should be initiated. A minimum of six hours of observation with cardiac monitoring and observation for signs of CNS or respiratory depression, hypotension, cardiac dysrhythmias and/or conduction blocks, and seizures is strongly advised. If signs of toxicity occur at any time during this period, extended monitoring is recommended. There are case reports of patients succumbing to fatal dysrhythmias late after overdose; these patients had clinical evidence of significant poisoning prior to death and most received inadequate gastrointestinal decontamination. Monitoring of plasma drug levels should not guide management of the patient.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The dose of doxepin tablets should be individualized.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Doxepin is an immediate-release, oval-shaped, tablet for oral administration available in strengths of 3 mg and 6 mg. The tablets for the 3 mg are blue and are identified with debossed markings of "T" & "S" on one side and blank on the other side. The 6 mg are green and are identified with debossed markings of "T" & "S" on one side and "6" on the other side. Doxepin tablets are not scored.
7.1 Cytochrome P450 Isozymes
Doxepin is primarily metabolized by hepatic cytochrome P450 isozymes CYP2C19 and CYP2D6, and to a lesser extent, by CYP1A2 and CYP2C9. Inhibitors of these isozymes may increase the exposure of doxepin. Doxepin is not an inhibitor of any CYP isozymes at therapeutically relevant concentrations. The ability of doxepin to induce CYP isozymes is not known.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
The pre-marketing development program for doxepin included doxepin HCl exposures in 1017 subjects (580 insomnia patients and 437 healthy subjects) from 12 studies conducted in the United States. 863 of these subjects (580 insomnia patients and 283 healthy subjects) participated in six randomized, placebo-controlled efficacy studies with Doxepin doses of 1 mg, 3 mg, and 6 mg for up to 3-months in duration.
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. However, data from the doxepin studies provide the physician with a basis for estimating the relative contributions of drug and non-drug factors to adverse reaction incidence rates in the populations studied.
14.1 Controlled Clinical Trials
The efficacy of doxepin for improving sleep maintenance was supported by six randomized, double-blind studies up to 3 months in duration that included 1,423 subjects, 18 to 93 years of age, with chronic (N=858) or transient (N=565) insomnia. Doxepin tablets were evaluated at doses of 1 mg, 3 mg, and 6 mg relative to placebo in inpatient (sleep laboratory) and outpatient settings.
The primary efficacy measures for assessment of sleep maintenance were the objective and subjective time spent awake after sleep onset (respectively, objective Wake After Sleep Onset [WASO] and subjective WASO). Subjects in studies of chronic insomnia were required to have at least a 3-month history of insomnia.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
4.3 Glaucoma and Urinary Retention
Doxepin is contraindicated in individuals with untreated narrow angle glaucoma or severe urinary retention.
8.7 Use in Patients With Sleep Apnea (8.7 Use in Patients with Sleep Apnea)
Doxepin has not been studied in patients with obstructive sleep apnea. Since hypnotics have the capacity to depress respiratory drive, precautions should be taken if doxepin is prescribed to patients with compromised respiratory function. In patients with severe sleep apnea, doxepin is ordinarily not recommended for use.
10.1 Signs and Symptoms of Excessive Doses
The following adverse effects have been associated with use of doxepin at doses higher than 6 mg.
Anticholinergic Effects:constipation and urinary retention.
Central Nervous System:disorientation, hallucinations, numbness, paresthesias, extrapyramidal symptoms, seizures, tardive dyskinesia.
Cardiovascular:hypotension.
Gastrointestinal:aphthous stomatitis, indigestion.
Endocrine:raised libido, testicular swelling, gynecomastia in males, enlargement of breasts and galactorrhea in the female, raising or lowering of blood sugar levels, and syndrome of inappropriate antidiuretic hormone secretion.
Other:tinnitus, weight gain, sweating, flushing, jaundice, alopecia, exacerbation of asthma, and hyperpyrexia (in association with chlorpromazine).
5.1 Need to Evaluate for Comorbid Diagnoses
Because sleep disturbances may be the presenting manifestation of a physical and/or psychiatric disorder, symptomatic treatment of insomnia should be initiated only after careful evaluation of the patient. The failure of insomnia to remit after 7 to 10 days of treatment may indicate the presence of a primary psychiatric and/or medical illness that should be evaluated.Exacerbation of insomnia or the emergence of new cognitive or behavioral abnormalities may be the consequence of an unrecognized psychiatric or physical disorder. Such findings have emerged during the course of treatment with hypnotic drugs.
8.6 Use in Patients With Hepatic Impairment (8.6 Use in Patients with Hepatic Impairment)
Patients with hepatic impairment may display higher doxepin concentrations than healthy individuals. Initiate doxepin treatment with 3 mg in patients with hepatic impairment and monitor closely for adverse daytime effects. [see Clinical Pharmacology (12.3)]
10.2 Signs and Symptoms of Critical Overdose
Manifestations of doxepin critical overdose include: cardiac dysrhythmias, severe hypotension, convulsions, and CNS depression including coma. Electrocardiogram changes, particularly in QRS axis or width, are clinically significant indicators of tricyclic compound toxicity. Other signs of overdose may include, but are not limited to: confusion, disturbed concentration, transient visual hallucinations, dilated pupils, agitation, hyperactive reflexes, stupor, drowsiness, muscle rigidity, vomiting, hypothermia, hyperpyrexia.
5.2 Abnormal Thinking and Behavioral Changes
Complex behaviors such as "sleep-driving" (i.e., driving while not fully awake after ingestion of a hypnotic, with amnesia for the event) have been reported with hypnotics. These events can occur in hypnotic-naive as well as in hypnotic-experienced persons. Although behaviors such as "sleep-driving" may occur with hypnotics alone at therapeutic doses, the use of alcohol and other CNS depressants with hypnotics appears to increase the risk of such behaviors, as does the use of hypnotics at doses exceeding the maximum recommended dose. Due to the risk to the patient and the community, discontinuation of doxepin should be strongly considered for patients who report a "sleep-driving" episode. Other complex behaviors (e.g., preparing and eating food, making phone calls, or having sex) have been reported in patients who are not fully awake after taking a hypnotic. As with "sleep-driving", patients usually do not remember these events. Amnesia, anxiety and other neuro-psychiatric symptoms may occur unpredictably.
5.3 Suicide Risk and Worsening of Depression
In primarily depressed patients, worsening of depression, including suicidal thoughts and actions (including completed suicides), has been reported in association with the use of hypnotics.
Doxepin, the active ingredient in doxepin tablets, is an antidepressant at doses 10-to 100-fold higher than in doxepin tablets. Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Risk from the lower dose of doxepin in doxepin tablets cannot be excluded.
It can rarely be determined with certainty whether a particular instance of the abnormal behaviors listed above is drug induced, spontaneous in origin, or a result of an underlying psychiatric or physical disorder. Nonetheless, the emergence of any new behavioral sign or symptom of concern requires careful and immediate evaluation.
7.4 Cns Depressants and Sedating Antihistamines (7.4 CNS Depressants and Sedating Antihistamines)
When taken with doxepin, the sedative effects of sedating antihistamines and CNS depressants may be potentiated [see Warnings and Precautions (5.2, 5.4)].
Principal Display Panel 3 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL 3 mg Tablet Bottle Label)
NDC 51672-4246-2
30 Tablets
Doxepin
Tablets
3 mg*
Rx only
Principal Display Panel 6 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL 6 mg Tablet Bottle Label)
NDC 51672-4247-2
30 Tablets
Doxepin
Tablets
6 mg*
Rx only
4.2 Co Administration With Monoamine Oxidase Inhibitors (maois) (4.2 Co-administration with Monoamine Oxidase Inhibitors (MAOIs))
Serious side effects and even death have been reported following the concomitant use of certain drugs with MAO inhibitors. Do not administer doxepin if patient is currently on MAOIs or has MAOIs within the past two weeks. The exact length of time may vary depending on the particular MAOI dosage and duration of treatment.
6.3 Other Reactions Observed During the Pre Marketing Evaluation of Doxepin (6.3 Other Reactions Observed During the Pre-marketing Evaluation of Doxepin)
Doxepin was administered to 1017 subjects in clinical trials in the United States. Treatment-emergent adverse reactions recorded by clinical investigators were standardized using a modified MedDRA dictionary of preferred terms. The following is a list of MedDRA terms that reflect treatment-emergent adverse reactions reported by subjects treated with doxepin.
Adverse reactions are further categorized by body system and listed in order of decreasing frequency according to the following definitions: Frequent adverse reactions are those that occurred on one or more occasions in at least 1/100 subjects; Infrequent adverse reactions are those that occurred in fewer than 1/100 subjects and more than 1/1000 subjects. Rare adverse reactions are those that occurred in fewer than 1/1000 subjects. Adverse reactions that are listed in Table 1 are not included in the following listing of frequent, infrequent, and rare AEs.
Blood and Lymphatic System Disorders:Infrequent: anemia; Rare: thrombocythemia.
Cardiac Disorders:Rare: atrioventricular block, palpitations, tachycardia, ventricular extrasystoles.
Ear and Labyrinth Disorders:Rare: ear pain, hypoacusis, motion sickness, tinnitus, tympanic membrane perforation.
Eye Disorders:Infrequent: eye redness, vision blurred; Rare: blepharospasm, diplopia, eye pain, lacrimation decreased.
Gastrointestinal Disorders:Infrequent: abdominal pain, dry mouth, gastroesophageal reflux disease, vomiting; Rare: dyspepsia, constipation, gingival recession, haematochezia, lip blister.
General Disorders and Administration Site Conditions: Infrequent: asthenia, chest pain, fatigue; Rare: chills, gait abnormal, edema peripheral.
Hepatobiliary Disorders:Rare: hyperbilirubinemia.
Immune System Disorders:Rare: hypersensitivity.
Infections and Infestations: Infrequent: bronchitis, fungal infection, laryngitis, sinusitis, tooth infection, urinary tract infection, viral infection; Rare: cellulitis staphylococcal, eye infection, folliculitis, gastroenteritis viral, herpes zoster, infective tenosynovitis, influenza, lower respiratory tract infection, onychomycosis, pharyngitis, pneumonia.
Injury, Poisoning and Procedural Complications:Infrequent: back injury, fall, joint sprain; Rare: bone fracture, skin laceration.
Investigations:Infrequent: blood glucose increased; Rare: alanine aminotransferase increased, blood pressure decreased, blood pressure increased, electrocardiogram ST-T segment abnormal, electrocardiogram QRS complex abnormal, heart rate decreased, neutrophil count decreased, QRS axis abnormal, transaminases increased.
Metabolism and Nutrition Disorders:Infrequent: anorexia, decreased appetite, hyperkalemia, hypermagnesemia, increased appetite; Rare: hypokalemia.
Musculoskeletal and Connective Tissue Disorders:Infrequent: arthralgia, back pain, myalgia, neck pain, pain in extremity; Rare: joint range of motion decreased, muscle cramp, sensation of heaviness.
Neoplasms Benign, Malignant and Unspecified (Including Cysts and Polyps): Rare: lung adenocarcinoma stage I, malignant melanoma.
Nervous System Disorders: Frequent: dizziness; Infrequent: dysgeusia, lethargy, parasthesia, syncope; Rare: ageusia, ataxia, cerebrovascular accident, disturbance in attention, migraine, sleep paralysis, syncope vasovagal, tremor.
Psychiatric Disorders: Infrequent: abnormal dreams, adjustment disorder, anxiety, depression; Rare: confusional state, elevated mood, insomnia, libido decreased, nightmare.
Reproductive System and Breast Disorders: Rare: breast cyst, dysmenorrhea.
Renal and Urinary Disorders: Rare: dysuria, enuresis, hemoglobinuria, nocturia.
Respiratory, Thoracic and Mediastinal Disorders: Infrequent: nasal congestion, pharyngolaryngeal pain, sinus congestion, wheezing; Rare: cough, crackles lung, nasopharyngeal disorder, rhinorrhea, dyspnea.
Skin and Subcutaneous Tissue Disorders: Infrequent: skin irritation; Rare: cold sweat, dermatitis, erythema, hyperhidrosis, pruritis, rash, rosacea.
Surgical and Medical Procedures: Rare: arthrodesis.
Vascular Disorders: Infrequent: pallor; Rare: blood pressure inadequately controlled, hematoma, hot flush.
In addition, the reactions below have been reported for other tricyclics and may be idiosyncratic (not related to dose).
Allergic:photosensitization, skin rash.
Hematologic:agranulocytosis, eosinophilia, leukopenia, purpura, thrombocytopenia
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:41.879701 · Updated: 2026-03-14T22:30:36.203052