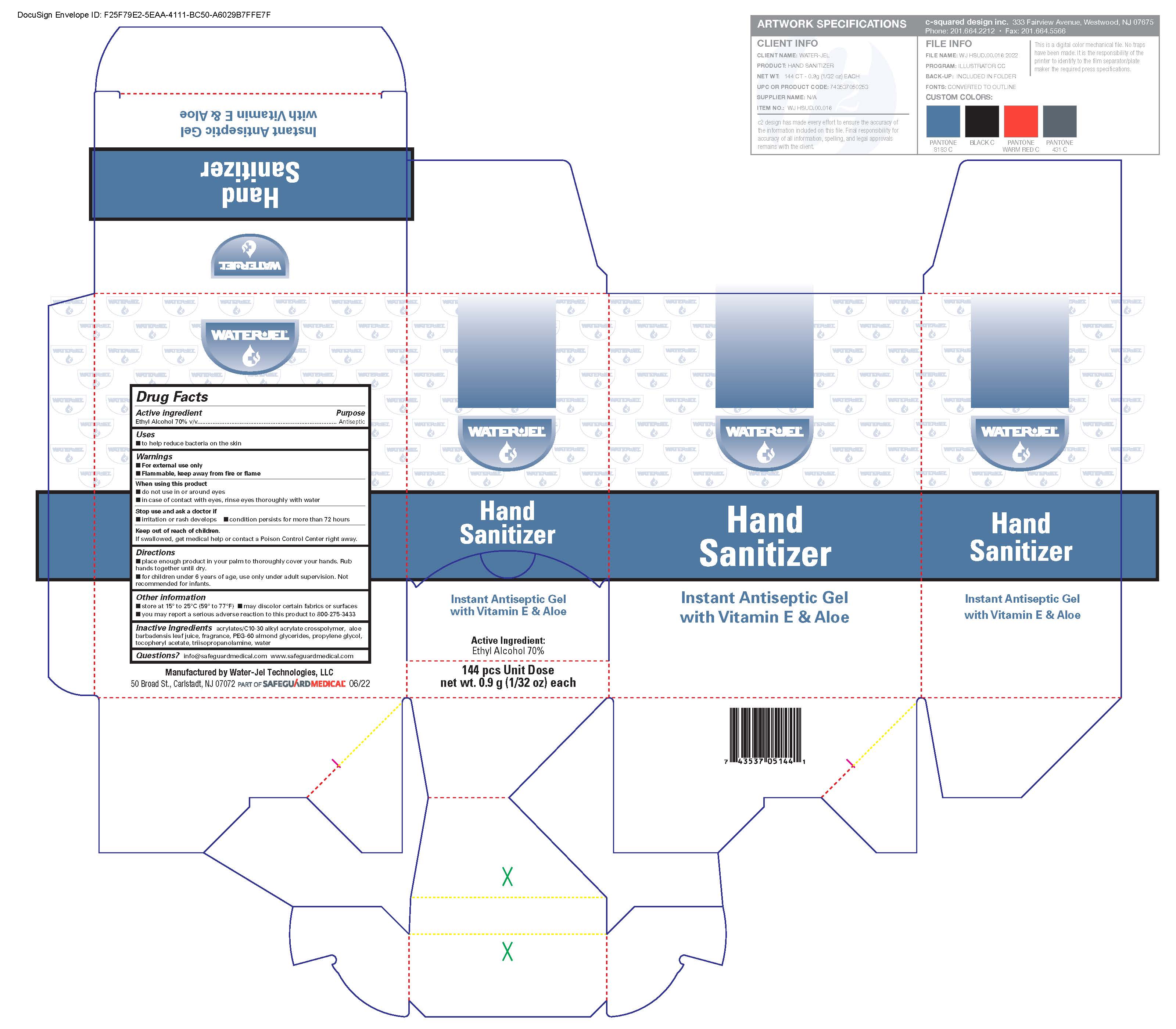
Instant Hand Sanitizer
54e62104-8076-41db-afe5-6dc5c3563acc
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyl alcohol 70% v/v
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Ethyl alcohol 70% v/v
Uses
- to help reduce bacteria on the skin
Section 42229-5
- store at 15 0 to 25 0 C (59 0 to 77 0 F)
- may discolor certain fabrics or surfaces
- you may report a serious adverse reaction to this product to 800-275-3433
Warnings
For external use only
Flammable, keep away from fire or flame
Questions
[email protected] www.safeguardmedical.com
Directions
- place enough product in your palm to thoroughly cover your hands. Rub hands together until dry.
- for children under 6 years of age, use only under adult supervision. Not recommended for infants.
Active Ingredient
Ethyl alcohol 70% v/v
Inactive Ingredients
acrylates/C10-30 alkyl acrylate crosspolymer, aloe barbadensis leaf juice, dl-alpha tocopheryl acetate, fragrance, PEG-60 almond glycerides, propylene glycol, tocopheryl acetate, purified water, triisopropanolamine
Principal Display Panel
Enter section text here
When Using This Product
- do not use in the eyes
- discontinue use if irritation and redness develops. If condition persists for more than 72 hours consult a doctor.
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
- to help reduce bacteria on the skin
Section 42229-5 (42229-5)
- store at 15 0 to 25 0 C (59 0 to 77 0 F)
- may discolor certain fabrics or surfaces
- you may report a serious adverse reaction to this product to 800-275-3433
Purpose
Antiseptic
Warnings
For external use only
Flammable, keep away from fire or flame
Questions
[email protected] www.safeguardmedical.com
Directions
- place enough product in your palm to thoroughly cover your hands. Rub hands together until dry.
- for children under 6 years of age, use only under adult supervision. Not recommended for infants.
Active Ingredient (Active ingredient)
Ethyl alcohol 70% v/v
Inactive Ingredients (Inactive ingredients)
acrylates/C10-30 alkyl acrylate crosspolymer, aloe barbadensis leaf juice, dl-alpha tocopheryl acetate, fragrance, PEG-60 almond glycerides, propylene glycol, tocopheryl acetate, purified water, triisopropanolamine
Principal Display Panel
Enter section text here
When Using This Product (When using this product)
- do not use in the eyes
- discontinue use if irritation and redness develops. If condition persists for more than 72 hours consult a doctor.
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:13.642746 · Updated: 2026-03-14T23:06:44.003626