These Highlights Do Not Include All The Information Needed To Use Tecvayli Safely And Effectively. See Full Prescribing Information For Tecvayli.
54e0f974-ccee-44ea-9254-40e9883cee1e
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Cytokine release syndrome (CRS), including life-threatening or fatal reactions, can occur in patients receiving TECVAYLI. Initiate treatment with TECVAYLI step-up dosing schedule to reduce risk of CRS. Withhold TECVAYLI until CRS resolves or permanently discontinue based on severity [see Dosage and Administration (2.1 , 2.5) and Warnings and Precautions (5.1) ] . Neurologic toxicity, including Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS) and serious, life-threatening, or fatal reactions, can occur in patients receiving TECVAYLI. Monitor patients for signs or symptoms of neurologic toxicity, including ICANS, during treatment. Withhold TECVAYLI until neurologic toxicity resolves or permanently discontinue based on severity [see Dosage and Administration (2.5) and Warnings and Precautions (5.2) ] . Because of the risk of CRS and neurologic toxicity, including ICANS, TECVAYLI is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the TECVAYLI and TALVEY REMS [see Warnings and Precautions (5.3) ] .
Indications and Usage
TECVAYLI is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma in combination with daratumumab and hyaluronidase-fihj in patients who have received at least one prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent. as monotherapy, in patients who have received at least four prior lines of therapy, including a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody.
Dosage and Administration
For subcutaneous injection only. ( 2.1 ) Patients should be hospitalized for 48 hours after administration of both step-up dose 1 and step-up dose 2. Instruct patients to remain within proximity of a healthcare facility and monitored daily for 48 hours after the first treatment dose within the TECVAYLI step-up dosing schedule. ( 2.1 ) See Full Prescribing Information for the recommended dosage for TECVAYLI monotherapy and combination therapy. ( 2.2 , 2.3 ) Administer pretreatment medications as recommended. ( 2.3 ) Refer to Tables 8, 9, 10, and 11 to determine the total dose, injection volume, and number of vials based on the patient's body weight. ( 2.6 ) See Full Prescribing Information for instructions on preparation and administration. ( 2.6 )
Warnings and Precautions
Hepatotoxicity : Can cause hepatotoxicity, including fatalities. Monitor liver enzymes and bilirubin at baseline and during treatment as clinically indicated. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity ( 2.5 , 5.4 ) Infections : Can cause severe, life-threatening, or fatal infections. Monitor patients for signs and symptoms of infection and treat appropriately. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity. ( 2.5 , 5.5 ) Neutropenia : Monitor complete blood cell counts at baseline and periodically during treatment. Withhold TECVAYLI based on severity. ( 2.5 , 5.6 ) Hypersensitivity and Other Administration Reactions : Can cause systemic administration-related reactions and local injection site reactions. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity. ( 2.5 , 5.7 ) Embryo-Fetal Toxicity : May cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception. ( 5.8 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are also described elsewhere in the labeling: Cytokine Release Syndrome [see Warnings and Precautions (5.1) ] Neurologic Toxicity including ICANS [see Warnings and Precautions (5.2) ] Hepatotoxicity [see Warnings and Precautions (5.4) ] Infections [see Warnings and Precautions (5.5) ] Neutropenia [see Warnings and Precautions (5.6) ] Hypersensitivity and Other Administration Reactions [see Warnings and Precautions (5.7) ]
Drug Interactions
TECVAYLI causes release of cytokines [see Clinical Pharmacology (12.2) ] that may suppress activity of certain cytochrome P450 (CYP) enzymes, resulting in increased exposure of CYP substrates, which may increase the risk of adverse reactions of the CYP substrates. The highest risk of drug-drug interaction is expected to occur after initiation of TECVAYLI step-up dosing schedule up to 7 days after the first treatment dose and during and after CRS [see Warnings and Precautions (5.1) ] . Monitor for toxicity and/or concentrations of CYP substrates where minimal increases in concentration may lead to serious adverse reactions. Consider decreasing the dosage of the concomitant CYP substrate as needed.
Storage and Handling
TECVAYLI ® (teclistamab-cqyv) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to light yellow solution supplied as follows: One 30 mg/3 mL (10 mg/mL) single-dose vial in a carton: NDC: 57894-449-01 One 153 mg/1.7 mL (90 mg/mL) single-dose vial in a carton: NDC: 57894-450-01
How Supplied
TECVAYLI ® (teclistamab-cqyv) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to light yellow solution supplied as follows: One 30 mg/3 mL (10 mg/mL) single-dose vial in a carton: NDC: 57894-449-01 One 153 mg/1.7 mL (90 mg/mL) single-dose vial in a carton: NDC: 57894-450-01
Medication Information
Warnings and Precautions
Hepatotoxicity : Can cause hepatotoxicity, including fatalities. Monitor liver enzymes and bilirubin at baseline and during treatment as clinically indicated. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity ( 2.5 , 5.4 ) Infections : Can cause severe, life-threatening, or fatal infections. Monitor patients for signs and symptoms of infection and treat appropriately. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity. ( 2.5 , 5.5 ) Neutropenia : Monitor complete blood cell counts at baseline and periodically during treatment. Withhold TECVAYLI based on severity. ( 2.5 , 5.6 ) Hypersensitivity and Other Administration Reactions : Can cause systemic administration-related reactions and local injection site reactions. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity. ( 2.5 , 5.7 ) Embryo-Fetal Toxicity : May cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception. ( 5.8 , 8.1 , 8.3 )
Indications and Usage
TECVAYLI is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma in combination with daratumumab and hyaluronidase-fihj in patients who have received at least one prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent. as monotherapy, in patients who have received at least four prior lines of therapy, including a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody.
Dosage and Administration
For subcutaneous injection only. ( 2.1 ) Patients should be hospitalized for 48 hours after administration of both step-up dose 1 and step-up dose 2. Instruct patients to remain within proximity of a healthcare facility and monitored daily for 48 hours after the first treatment dose within the TECVAYLI step-up dosing schedule. ( 2.1 ) See Full Prescribing Information for the recommended dosage for TECVAYLI monotherapy and combination therapy. ( 2.2 , 2.3 ) Administer pretreatment medications as recommended. ( 2.3 ) Refer to Tables 8, 9, 10, and 11 to determine the total dose, injection volume, and number of vials based on the patient's body weight. ( 2.6 ) See Full Prescribing Information for instructions on preparation and administration. ( 2.6 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are also described elsewhere in the labeling: Cytokine Release Syndrome [see Warnings and Precautions (5.1) ] Neurologic Toxicity including ICANS [see Warnings and Precautions (5.2) ] Hepatotoxicity [see Warnings and Precautions (5.4) ] Infections [see Warnings and Precautions (5.5) ] Neutropenia [see Warnings and Precautions (5.6) ] Hypersensitivity and Other Administration Reactions [see Warnings and Precautions (5.7) ]
Drug Interactions
TECVAYLI causes release of cytokines [see Clinical Pharmacology (12.2) ] that may suppress activity of certain cytochrome P450 (CYP) enzymes, resulting in increased exposure of CYP substrates, which may increase the risk of adverse reactions of the CYP substrates. The highest risk of drug-drug interaction is expected to occur after initiation of TECVAYLI step-up dosing schedule up to 7 days after the first treatment dose and during and after CRS [see Warnings and Precautions (5.1) ] . Monitor for toxicity and/or concentrations of CYP substrates where minimal increases in concentration may lead to serious adverse reactions. Consider decreasing the dosage of the concomitant CYP substrate as needed.
Storage and Handling
TECVAYLI ® (teclistamab-cqyv) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to light yellow solution supplied as follows: One 30 mg/3 mL (10 mg/mL) single-dose vial in a carton: NDC: 57894-449-01 One 153 mg/1.7 mL (90 mg/mL) single-dose vial in a carton: NDC: 57894-450-01
How Supplied
TECVAYLI ® (teclistamab-cqyv) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to light yellow solution supplied as follows: One 30 mg/3 mL (10 mg/mL) single-dose vial in a carton: NDC: 57894-449-01 One 153 mg/1.7 mL (90 mg/mL) single-dose vial in a carton: NDC: 57894-450-01
Description
Cytokine release syndrome (CRS), including life-threatening or fatal reactions, can occur in patients receiving TECVAYLI. Initiate treatment with TECVAYLI step-up dosing schedule to reduce risk of CRS. Withhold TECVAYLI until CRS resolves or permanently discontinue based on severity [see Dosage and Administration (2.1 , 2.5) and Warnings and Precautions (5.1) ] . Neurologic toxicity, including Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS) and serious, life-threatening, or fatal reactions, can occur in patients receiving TECVAYLI. Monitor patients for signs or symptoms of neurologic toxicity, including ICANS, during treatment. Withhold TECVAYLI until neurologic toxicity resolves or permanently discontinue based on severity [see Dosage and Administration (2.5) and Warnings and Precautions (5.2) ] . Because of the risk of CRS and neurologic toxicity, including ICANS, TECVAYLI is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the TECVAYLI and TALVEY REMS [see Warnings and Precautions (5.3) ] .
Section 42229-5
Manufactured by:
Janssen Biotech, Inc.
Horsham, PA 19044, USA
U.S. License Number 1864
For patent information: www.janssenpatents.com
© Johnson & Johnson and its affiliates 2022, 2026
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: Mar 2026 | ||
|
MEDICATION GUIDE TECVAYLI® [tek vay lee] (teclistamab-cqyv) injection, for subcutaneous use |
|||
|
What is the most important information I should know about TECVAYLI?
TECVAYLI can cause side effects that are serious, life-threatening or cause death, including cytokine release syndrome (CRS) and neurologic problems.
|
|||
|
|
||
Due to the risk of CRS, you will receive TECVAYLI on a "step-up dosing" schedule. You should be hospitalized for 48 hours after the first and second step-up doses and should stay close to a healthcare facility and be monitored by a healthcare provider for 48 hours after you receive your first treatment dose.
|
|||
|
|||
|
|
|
|
Your healthcare provider will monitor you for signs and symptoms of CRS and neurologic problems during treatment with TECVAYLI and treat you as needed. Your healthcare provider may temporarily stop or completely stop your treatment with TECVAYLI if you develop CRS or neurologic problems.
See " What are the possible side effects of TECVAYLI?" for more information about side effects. |
|||
|
What is TECVAYLI?
TECVAYLI is a prescription medicine used to treat adults with multiple myeloma that has come back or did not respond to prior treatment:
|
|||
Before you receive TECVAYLI, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
How will I receive TECVAYLI?
|
|||
|
What should I avoid while receiving TECVAYLI?
Do not drive or operate heavy or dangerous machinery during and for 48 hours after your TECVAYLI step-up dosing schedule is completed, or at any time during treatment with TECVAYLI if you develop new neurologic symptoms such as dizziness, confusion, tremors, sleepiness, or any other symptoms that impair your consciousness, until the symptoms go away. See "What is the most important information I should know about TECVAYLI?" for more information about signs and symptoms of neurologic problems. |
|||
|
What are the possible side effects of TECVAYLI?
TECVAYLI can cause serious side effects, including:
|
|||
|
|
||
Your healthcare provider will monitor you for signs and symptoms of these serious side effects during treatment and may temporarily or permanently stop TECVAYLI if you develop severe side effects. The most common side effects of TECVAYLI when used alone include: |
|||
|
|
|
|
| The most common side effects of TECVAYLI when used with daratumumab and hyaluronidase-fihj include: | |||
|
|
|
|
|
The most common severe abnormal blood test results with TECVAYLI include: decreased white blood cells, red blood cells, and platelets. These are not all the possible side effects of TECVAYLI. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
General Information about the safe and effective use of TECVAYLI.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about TECVAYLI that is written for health professionals. |
|||
|
What are the ingredients in TECVAYLI?
Active ingredient: teclistamab-cqyv Inactive ingredients: edetate disodium, glacial acetic acid, polysorbate 20, sodium acetate, sucrose, Water for Injection Manufactured by: Janssen Biotech, Inc., Horsham, PA 19044, USA U.S. License Number 1864 For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2022, 2026 For more information about TECVAYLI go to www.TECVAYLI.com or call 1-800-526-7736. |
Section 43683-2
Section 44425-7
Store refrigerated at 2 °C to 8 °C (36 °F to 46 °F) in the original carton to protect from light.
Do not freeze.
8.1 Pregnancy
Risk Summary
Based on the mechanism of action, TECVAYLI may cause fetal harm when administered to a pregnant patient [see Clinical Pharmacology (12.1)]. There are no available data on the use of TECVAYLI in pregnant patients to evaluate for a drug associated risk. No animal reproductive or developmental toxicity studies have been conducted with TECVAYLI. Teclistamab-cqyv causes T-cell activation and cytokine release; immune activation may compromise pregnancy maintenance. Human immunoglobulin G (IgG) is known to cross the placenta; therefore, teclistamab-cqyv has the potential to be transmitted from the mother to the developing fetus. Advise women of the potential risk to the fetus.
TECVAYLI is associated with hypogammaglobulinemia, therefore, assessment of immunoglobulin levels in newborns of mothers treated with TECVAYLI should be considered.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data on the presence of teclistamab-cqyv in human milk, the effect on the breastfed child, or the effects on milk production. Maternal IgG is known to be present in human milk. The effects of local gastrointestinal exposure and limited systemic exposure in the breastfed child to TECVAYLI are unknown. Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during treatment with TECVAYLI and for 5 months after the last dose.
11 Description
Teclistamab-cqyv, a bispecific B-cell maturation antigen (BCMA)-directed CD3 T-cell engager, is a humanized immunoglobulin G4-proline, alanine, alanine (IgG4-PAA) antibody. Teclistamab-cqyv is produced in Chinese Hamster Ovary (CHO) cells using recombinant DNA technology. Teclistamab-cqyv consists of an anti-BCMA heavy chain and light chain and an anti-CD3 heavy chain and light chain with two interchain disulfide bonds connecting the two arms. The molecular weight of teclistamab-cqyv is approximately 146 kDa.
TECVAYLI® (teclistamab-cqyv) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to light yellow solution supplied in a single-dose vial for subcutaneous administration.
Each TECVAYLI 3 mL single-dose vial contains 30 mg of teclistamab-cqyv, edetate disodium (0.054 mg), glacial acetic acid (0.72 mg), polysorbate 20 (1.2 mg), sodium acetate (2.7 mg), sucrose (240 mg), and Water for Injection, USP.
Each TECVAYLI 1.7 mL single-dose vial contains 153 mg of teclistamab-cqyv, edetate disodium (0.031 mg), glacial acetic acid (0.41 mg), polysorbate 20 (0.68 mg), sodium acetate (1.5 mg), sucrose (140 mg), and Water for Injection, USP.
5.5 Infections
TECVAYLI can cause severe, life-threatening, or fatal infections.
In MajesTEC-1 (N=165), in patients who received the recommended TECVAYLI dosage, serious infections, including opportunistic infections, occurred in 30% of patients, Grade 3 or 4 infections occurred in 35% of patients, and fatal infections occurred in 4.2% of patients [see Adverse Reactions (6.1)].
In MajesTEC-3 (N=283), in patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj at the recommended dosage, serious infections, including opportunistic infections, occurred in 54% of patients, Grade 3 or Grade 4 infections occurred in 54% of patients and fatal infections occurred in 4.6% of patients [see Adverse Reactions (6.1)].
Monitor patients for signs and symptoms of infection prior to and during treatment with TECVAYLI and treat appropriately. Administer prophylactic antimicrobials according to current practice guidelines [see Dosage and Administration (2.3)].
Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity [see Dosage and Administration (2.5)].
Monitor immunoglobulin levels prior to and during treatment with TECVAYLI and administer subcutaneous or intravenous immunoglobulin (IVIG) to maintain the serum levels >400 mg/dL.
5.6 Neutropenia
TECVAYLI can cause neutropenia and febrile neutropenia. In patients who received TECVAYLI at the recommended dosage in the clinical trials (monotherapy and combination therapy trials; N=448), decreased neutrophils occurred in 88% of patients, with Grade 3 or 4 decreased neutrophils in 70%. Febrile neutropenia occurred in 6% of patients [see Adverse Reactions (6.1)].
Monitor complete blood cell counts at baseline and periodically during treatment and provide supportive care per local institutional guidelines.
Monitor patients with neutropenia for signs of infection.
Withhold TECVAYLI based on severity [see Dosage and Administration (2.5)].
8.4 Pediatric Use
The safety and efficacy of TECVAYLI have not been established in pediatric patients.
8.5 Geriatric Use
- Of the 165 patients with relapsed or refractory multiple myeloma treated with TECVAYLI in MajesTEC-1 at the recommended dosage, 48% were 65 years of age or older, and 15% were 75 years of age or older. No overall differences in safety or effectiveness were observed between patients 65 to 74 years of age compared to younger patients. The study did not include a sufficient number of patients 75 years of age or older to assess whether there are differences in safety or effectiveness.
- Of the 291 patients with relapsed or refractory multiple myeloma treated with TECVAYLI at the recommended dosage in combination with daratumumab and hyaluronidase-fihj in MajesTEC-3, 51% were younger than 65 years of age, 39% were 65 to 74 years of age, and 11% were 75 years of age or older. No overall differences in effectiveness were observed between patients 65 to 74 years of age compared to younger patients. The study did not include a sufficient number of patients 75 years of age or older to assess whether there are differences in effectiveness compared to younger patients. There was a higher incidence of serious adverse reactions in patients 75 years of age or older (83%) and in patients 65 to 74 years of age (77%) as compared to patients younger than 65 years of age (63%).
5.4 Hepatotoxicity
TECVAYLI can cause hepatotoxicity, including fatalities. There was one fatal case of hepatic failure in MajesTEC-1. In patients who received TECVAYLI at the recommended dosage in the clinical trials (monotherapy and combination therapy trials; N=448), elevated aspartate aminotransferase (AST) occurred in 47% of patients, with Grade 3 or 4 elevations in 2.9%. Elevated alanine aminotransferase (ALT) occurred in 48% of patients, with Grade 3 or 4 elevations in 3.8%. Elevated total bilirubin occurred in 10% of patients with Grade 3 or 4 elevations in 0.7%. Liver enzyme elevation can occur with or without concurrent CRS.
Monitor liver enzymes and bilirubin at baseline and during TECVAYLI treatment as clinically indicated. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity [see Dosage and Administration (2.5)].
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of teclistamab-cqyv or of other teclistamab products.
Among patients treated with subcutaneous TECVAYLI at various dosages, anti-teclistamab-cqyv antibodies developed in 1/186 (0.5%) of patients who received monotherapy (up to 27 months) and in 2/379 (0.5%) of patients who received combination therapy with daratumumab and hyaluronidase-fihj (up to 43 months). Because of the low occurrence of anti-teclistamab-cqyv antibodies, the effect of these antibodies on the pharmacokinetics, pharmacodynamics, safety, and/or effectiveness of teclistamab products is unknown.
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are also described elsewhere in the labeling:
- Cytokine Release Syndrome [see Warnings and Precautions (5.1)]
- Neurologic Toxicity including ICANS [see Warnings and Precautions (5.2)]
- Hepatotoxicity [see Warnings and Precautions (5.4)]
- Infections [see Warnings and Precautions (5.5)]
- Neutropenia [see Warnings and Precautions (5.6)]
- Hypersensitivity and Other Administration Reactions [see Warnings and Precautions (5.7)]
7 Drug Interactions
TECVAYLI causes release of cytokines [see Clinical Pharmacology (12.2)] that may suppress activity of certain cytochrome P450 (CYP) enzymes, resulting in increased exposure of CYP substrates, which may increase the risk of adverse reactions of the CYP substrates. The highest risk of drug-drug interaction is expected to occur after initiation of TECVAYLI step-up dosing schedule up to 7 days after the first treatment dose and during and after CRS [see Warnings and Precautions (5.1)].
Monitor for toxicity and/or concentrations of CYP substrates where minimal increases in concentration may lead to serious adverse reactions. Consider decreasing the dosage of the concomitant CYP substrate as needed.
12.2 Pharmacodynamics
Serum concentrations of cytokines (IL-6, IL-10, TNF-α, and IFN-γ) and IL-2R were measured before and after administration of step-up dose 1, step-up dose 2, and the three weekly doses of 1.5 mg/kg of TECVAYLI. Increased concentrations of IL-6, IL-10, and IL-2R were observed during this period.
12.3 Pharmacokinetics
The Cmax and Cavg of teclistamab-cqyv after the first subcutaneous 1.5 mg/kg TECVAYLI dose increase proportionally over a dosage range of 0.08 mg/kg to 3 mg/kg in monotherapy (0.05 to 2 times the approved recommended treatment dosage). Following the recommended dosage of TECVAYLI monotherapy, 90% of steady state exposure was achieved after 12 weekly TECVAYLI doses (1.5 mg/kg once weekly). The mean accumulation ratio between the first and 13th weekly dose of TECVAYLI 1.5 mg/kg was 4.2-fold for Cmax, 4.1-fold for Ctrough, and 5.3-fold for Cavg.
The pharmacokinetic characteristics of teclistamab-cqyv were consistent whether used as monotherapy or in combination with daratumumab and hyaluronidase-fihj. The Cmax, Ctrough, and Cavg of teclistamab-cqyv are presented in Table 16.
| Cmax (µg/mL) | Ctrough (µg/mL) | Cavg (µg/mL) | |
|---|---|---|---|
| Cavg=average serum teclistamab-cqyv concentration over a dosing interval; Cmax=maximum serum teclistamab-cqyv concentration; Ctrough=serum teclistamab-cqyv concentration prior to next dose | |||
| Note: Data are presented as geometric mean (CV %). | |||
| TECVAYLI Monotherapy | |||
| First 1.5 mg/kg dose | 6.34 (60%) | 5.77 (63%) | 4.98 (61%) |
| Steady state of 1.5 mg/kg weekly dosing steady-state exposure for the 13th weekly dose.
|
23.8 (55%) | 21.1 (63%) | 22.8 (57%) |
| TECVAYLI in Combination with Daratumumab and Hyaluronidase-fihj | |||
| First 1.5 mg/kg dose | 6.36 (54%) | 5.79 (59%) | 4.96 (55%) |
| End of 1.5 mg/kg weekly dosing exposure for the 7th weekly dose.
|
19.6 (54%) | 17.3 (62%) | 18.6 (56%) |
| End of 3 mg/kg every two weeks dosing exposure for the 8th every two weeks dose.
|
29.9 (55%) | 19.8 (85%) | 26.2 (62%) |
| Steady state of 3 mg/kg every 4 weeks dosing steady-state exposure for the 5th Q4W dose.
|
20.2 (52%) | 5.82 (152%) | 13.7 (67%) |
Absorption
The mean bioavailability of teclistamab-cqyv was 72% after subcutaneous administration of TECVAYLI. The median (range) Tmax of teclistamab-cqyv after the first and 13th TECVAYLI monotherapy treatment doses were 139 (19 to 168) hours and 72 (24 to 168) hours, respectively.
Distribution
The mean (coefficient of variation [CV]%) volume of distribution of teclistamab-cqyv was 5.63 L (29%).
Elimination
Teclistamab-cqyv clearance decreases over time, with a mean (CV%) maximal reduction from baseline to the 13th weekly TECVAYLI monotherapy treatment dose of 40.8% (56%). The geometric mean (CV%) clearance is 0.472 L/day (64%) at the 13th weekly TECVAYLI monotherapy treatment dose. Patients who discontinue TECVAYLI after the 13th weekly monotherapy treatment dose are expected to have a 50% reduction from Cmax in teclistamab-cqyv concentration at a median (5th to 95th percentile) time of 15 days (7 to 33 days) after Tmax and a 97% reduction from Cmax in teclistamab-cqyv concentration at a median time of 69 days (32 to 163 days) after Tmax.
Specific Populations
There were no clinically significant differences in the exposure of teclistamab-cqyv based on age (24 to 84 years), sex, race (White, Black or African American), ethnicity (Hispanic/Latino, not Hispanic/Latino), mild or moderate renal impairment (estimated glomerular filtration rate [eGFR] by Modification of Diet in Renal Disease [MDRD] method: 30 to 89 mL/minute), or mild hepatic impairment (total bilirubin less than or equal to upper limit of normal [ULN] with AST greater than ULN or total bilirubin greater than 1 to 1.5 times ULN with any AST). The effects of severe renal impairment (eGFR less than 30 mL/minute) or moderate to severe hepatic impairment (total bilirubin greater than 1.5 times ULN with any AST) on the exposures of teclistamab-cqyv are unknown.
Body Weight
The volume of distribution and clearance of teclistamab-cqyv increase with increasing body weight (37.6 kg to 164 kg), supporting a weight-based dose.
Drug Interaction Studies
No clinical studies evaluating the drug interaction potential of teclistamab-cqyv have been conducted.
1 Indications and Usage
TECVAYLI is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma
- in combination with daratumumab and hyaluronidase-fihj in patients who have received at least one prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent.
- as monotherapy, in patients who have received at least four prior lines of therapy, including a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody.
12.1 Mechanism of Action
Teclistamab-cqyv is a bispecific T-cell engaging antibody that binds to the CD3 receptor expressed on the surface of T-cells and B-cell maturation antigen (BCMA) expressed on the surface of multiple myeloma cells and some healthy B-lineage cells.
In vitro, teclistamab-cqyv activated T-cells, caused the release of various proinflammatory cytokines, and resulted in the lysis of multiple myeloma cells.
5.8 Embryo Fetal Toxicity
Based on its mechanism of action, TECVAYLI may cause fetal harm when administered to a pregnant patient. Advise pregnant patients of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with TECVAYLI and for 5 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions
- Hepatotoxicity: Can cause hepatotoxicity, including fatalities. Monitor liver enzymes and bilirubin at baseline and during treatment as clinically indicated. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity (2.5, 5.4)
- Infections: Can cause severe, life-threatening, or fatal infections. Monitor patients for signs and symptoms of infection and treat appropriately. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity. (2.5, 5.5)
- Neutropenia: Monitor complete blood cell counts at baseline and periodically during treatment. Withhold TECVAYLI based on severity. (2.5, 5.6)
- Hypersensitivity and Other Administration Reactions: Can cause systemic administration-related reactions and local injection site reactions. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity. (2.5, 5.7)
- Embryo-Fetal Toxicity: May cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception. (5.8, 8.1, 8.3)
2 Dosage and Administration
- For subcutaneous injection only. (2.1)
- Patients should be hospitalized for 48 hours after administration of both step-up dose 1 and step-up dose 2. Instruct patients to remain within proximity of a healthcare facility and monitored daily for 48 hours after the first treatment dose within the TECVAYLI step-up dosing schedule. (2.1)
- See Full Prescribing Information for the recommended dosage for TECVAYLI monotherapy and combination therapy. (2.2, 2.3)
- Administer pretreatment medications as recommended. (2.3)
- Refer to Tables 8, 9, 10, and 11 to determine the total dose, injection volume, and number of vials based on the patient's body weight. (2.6)
- See Full Prescribing Information for instructions on preparation and administration. (2.6)
3 Dosage Forms and Strengths
Injection
- 30 mg/3 mL (10 mg/mL) clear to slightly opalescent, colorless to light yellow solution in a single-dose vial.
- 153 mg/1.7 mL (90 mg/mL) clear to slightly opalescent, colorless to light yellow solution in a single-dose vial.
5.3 Tecvayli and Talvey Rems
TECVAYLI is available only through a restricted program under a REMS called the "TECVAYLI and TALVEY REMS" because of the risks of CRS and neurologic toxicity, including ICANS [see Warnings and Precautions (5.1, 5.2)].
Notable requirements of the TECVAYLI and TALVEY REMS include the following:
- Prescribers must be certified with the REMS by enrolling and completing training.
- Prescribers must counsel patients receiving TECVAYLI about the risk of CRS and neurologic toxicity, including ICANS, and provide patients with Patient Wallet Card.
- Pharmacies and healthcare settings that dispense TECVAYLI must be certified with this REMS and must verify prescribers are certified through this REMS.
- Wholesalers and distributers must only distribute TECVAYLI to certified pharmacies or healthcare settings.
Further information about the "TECVAYLI and TALVEY REMS" is available at www.TEC-TALREMS.com or by telephone at 1-855-810-8064.
5.1 Cytokine Release Syndrome
TECVAYLI can cause cytokine release syndrome (CRS), including life-threatening or fatal reactions [see Adverse Reactions (6.1)].
In the clinical trials (monotherapy and combination therapy trials; N=448), CRS occurred in 64% of patients who received TECVAYLI at the recommended dosage, with Grade 1 CRS occurring in 46% of patients, Grade 2 in 18%, and Grade 3 in 0.2%. Recurrent CRS occurred in 27% of patients. Most patients experienced CRS during the initial step-up dosing schedule (step-up dose 1 (37%), step-up dose 2 (32%), or the initial treatment dose (20%)). CRS first occurred following subsequent doses of TECVAYLI in 2.5% of patients. The median time to onset of CRS was 2 (range: 1 to 9) days after the most recent dose and the median duration of CRS was 2 (range: 1 to 22) days.
Clinical signs and symptoms of CRS included, but were not limited to, fever, hypoxia, chills, hypotension, sinus tachycardia, headache, and elevated liver enzymes (aspartate aminotransferase and alanine aminotransferase elevation).
Initiate therapy according to TECVAYLI step-up dosing schedule to reduce risk of CRS [see Dosage and Administration (2.1, 2.5)]. Administer pretreatment medications to reduce risk of CRS and monitor patients following administration of TECVAYLI accordingly [see Dosage and Administration (2.3, 2.5)].
At the first sign of CRS, immediately evaluate the patient for hospitalization. Administer supportive care based on severity and consider further management per current practice guidelines. Withhold until CRS resolves or permanently discontinue TECVAYLI based on severity [see Dosage and Administration (2.5)].
TECVAYLI is available only through a restricted program under a REMS [see Warnings and Precautions (5.3)].
8 Use in Specific Populations
Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Relapsed/Refractory Multiple Myeloma
In Combination with Daratumumab and Hyaluronidase-fihj
The safety of TECVAYLI in combination with daratumumab and hyaluronidase-fihj (N=283) compared with either daratumumab and hyaluronidase-fihj, pomalidomide and dexamethasone (DPd) or daratumumab and hyaluronidase-fihj, bortezomib and dexamethasone (DVd) (N=290) was evaluated in patients with relapsed or refractory multiple myeloma in MajesTEC-3 [see Clinical Studies (14.1)]. Patients received step-up doses of 0.06 mg/kg and 0.3 mg/kg of TECVAYLI followed by TECVAYLI 1.5 mg/kg once weekly, followed by TECVAYLI 3 mg/kg every two weeks, followed by TECVAYLI 3 mg/kg every four weeks, subcutaneously.
Among patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj the median exposure was 32 (range 0.03 to 43) months. Among patients who received DPd or DVd the median exposure was 16 (range 0.03 to 45) months.
Serious adverse reactions occurred in 71% of patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj. Serious adverse reactions reported in ≥3% of patients included pneumonia (33%), upper respiratory tract infection (15%), cytokine release syndrome (10%), COVID-19 (7%), sepsis (6%), second primary malignancy (5%), pyrexia (4.9%), febrile neutropenia (4.6%), and gastroenteritis (4.2%).
Fatal adverse reactions occurred in 2.5% of patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj, and included sepsis (0.7%), pneumonia (0.4%), sudden death (0.4%), myocardial infarction (0.4%), enterovirus myocarditis (0.4%) and hemophagocytic lymphohistiocytosis (0.4%).
Permanent discontinuation of TECVAYLI due to adverse reactions occurred in 6% of patients. Adverse reactions leading to discontinuation of TECVAYLI in more than one patient were pneumonia (1.1%), diarrhea (0.7%), fatigue (0.7%), second primary malignancy (0.7%), upper respiratory tract infection (0.7%) and cough (0.7%).
Dosage interruptions of TECVAYLI due to an adverse reaction occurred in 94% of patients. Adverse reactions which required dosage interruption of TECVAYLI in ≥5% of patients included neutropenia (53%), upper respiratory tract infection (48%), COVID-19 (34%), pneumonia (34%), thrombocytopenia (14%), cytokine release syndrome (13%), gastroenteritis (10%), cough (10%), pyrexia (8%), diarrhea (7%), sepsis (6%) and fatigue (5%).
The most common adverse reactions (≥20%) were hypogammaglobulinemia, upper respiratory tract infection, cytokine release syndrome, cough, diarrhea, musculoskeletal pain, COVID-19, pneumonia, injection site reaction, fatigue, pyrexia, headache, nausea, gastroenteritis, and weight decreased.
The most common Grade 3 to 4 laboratory abnormalities (≥20%) were decreased lymphocytes, decreased neutrophils, decreased white blood cells and decreased platelets.
Table 12 summarizes the adverse reactions in MajesTEC-3.
| Adverse Reaction | TECVAYLI with daratumumab and hyaluronidase-fihj (N=283) |
DPd or DVd (N=290) |
||
|---|---|---|---|---|
| Any Grade (%) |
Grade 3 or 4 (%) |
Any Grade (%) |
Grade 3 or 4 (%) |
|
| DPd = daratumumab and hyaluronidase-fihj, pomalidomide, dexamethasone; DVd = daratumumab and hyaluronidase-fihj, bortezomib, dexamethasone. | ||||
| Adverse reactions were graded according to NCI-CTCAE Version 5.0, with the exception of ICANS and CRS, which were graded by ASTCT 2019 consensus grading system; adverse reactions that were considered symptoms of CRS or ICANS were not included. | ||||
| Immune system disorders | ||||
| Hypogammaglobulinemia Hypogammaglobulinemia includes hypogammaglobulinemia, hypoglobulinemia; and/or patients with laboratory IgG levels below 400 mg/dL following treatment with TECVAYLI.
|
84 | 6 | 60 | 1.4 |
| Cytokine release syndrome | 60 | 0 | 0 | 0 |
| Infections | ||||
| Upper respiratory tract infection Upper respiratory tract infection includes bronchitis, bronchiolitis, pharyngitis, rhinitis, sinusitis, sinobronchitis, tracheitis, and tracheobronchitis and other related terms.
|
79 | 17 | 62 | 13 |
| COVID-19 Includes other related terms.
|
45 | 6 | 33 | 2.1 |
| Pneumonia Pneumonia includes atypical pneumonia, bacterial pneumonia, fungal pneumonia, viral pneumonia, and other related terms.
Includes the following fatal adverse reactions: Tec-Dara: Pneumonia (n=1); DPd/DVd: Pneumonia (n=2).
|
42 | 33 | 35 | 28 |
| Gastroenteritis | 20 | 4.9 | 8 | 0.7 |
| Urinary tract infection | 17 | 2.8 | 13 | 1 |
| Herpes virus infection | 11 | 2.1 | 6 | 0.3 |
| Respiratory, thoracic and mediastinal disorders | ||||
| Cough | 53 | 0.7 | 24 | 0 |
| Dyspnea | 13 | 1.8 | 20 | 2.1 |
| Gastrointestinal disorders | ||||
| Diarrhea | 52 | 3.9 | 31 | 2.4 |
| Nausea | 23 | 0 | 12 | 0.3 |
| Vomiting | 17 | 0 | 7 | 0 |
| Abdominal pain | 16 | 0.7 | 13 | 0 |
| Constipation | 14 | 0 | 20 | 0.3 |
| Musculoskeletal and connective tissue disorders | ||||
| Musculoskeletal pain | 50 | 1.4 | 47 | 4.5 |
| General disorders and administration site conditions | ||||
| Injection site reaction
Includes injection site reactions related to teclistamab, daratumumab and hyaluronidase-fihj, or bortezomib.
|
41 | 0 | 4.5 | 0 |
| Fatigue | 39 | 3.9 | 40 | 4.1 |
| Pyrexia | 37 | 1.4 | 19 | 0.3 |
| Edema | 13 | 0.4 | 22 | 0.3 |
| Nervous system disorders | ||||
| Headache | 26 | 1.4 | 12 | 0.3 |
| Sensory neuropathy Sensory neuropathy includes dysaesthesia, hyperaesthesia, hypoaesthesia, paraesthesia, neuralgia, peripheral neuropathy, polyneuropathy, sciatica and other related terms.
|
17 | 0.7 | 25 | 0.3 |
| Motor dysfunction Motor dysfunction includes balance disorder, dysarthria, dysphonia, gait disturbance, muscle contracture, muscle spasms, muscle spasticity, muscle twitching, muscular weakness, myopathy, peripheral motor neuropathy, tremor and other related terms.
|
14 | 0 | 30 | 1 |
| Investigations | ||||
| Weight decreased | 20 | 2.8 | 7 | 1.4 |
| Metabolism and nutrition disorders | ||||
| Decreased appetite | 19 | 1.1 | 7 | 0 |
| Cardiac disorders | ||||
| Cardiac arrhythmia | 11 | 1.1 | 8 | 2.1 |
| Neoplasms | ||||
| Second primary malignancy | 11 | 4.6 | 9 | 4.8 |
| Vascular disorders | ||||
| Hypertension | 11 | 4.6 | 6 | 2.4 |
Clinically relevant adverse reactions in <10% of patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj included sepsis, encephalopathy, CMV infection, febrile neutropenia and ICANS.
Table 13 summarizes laboratory abnormalities in MajesTEC-3.
| Laboratory Abnormality | TECVAYLI with daratumumab and hyaluronidase-fihj (N=283) |
DPd or DVd (N=290) |
||
|---|---|---|---|---|
| All Grades (%) |
Grade 3 or 4 (%) |
All Grades (%) |
Grade 3 or 4 (%) |
|
| DPd = daratumumab and hyaluronidase-fihj, pomalidomide, dexamethasone; DVd = daratumumab and hyaluronidase-fihj, bortezomib, dexamethasone. Percentages calculated with the number of subjects with a baseline and at least one post-treatment value for each lab test as denominator. Laboratory toxicity grades are derived based on NCI CTCAE Version 5.0. |
||||
| The denominator used to calculate the incidence varied from 137 to 282 in Tec-Dara and 135 to 289 in DPd/DVd based on the number of patients with a baseline value and at least one post-treatment value. | ||||
| Hematology | ||||
| Lymphocyte Count Decreased | 99 | 95 | 93 | 64 |
| White Blood Cell Decreased | 94 | 60 | 96 | 74 |
| Neutrophil Count Decreased | 91 | 78 | 94 | 84 |
| Platelet Count Decreased | 73 | 21 | 72 | 25 |
| Hemoglobin Decreased | 64 | 17 | 61 | 19 |
| Chemistry | ||||
| Alanine Aminotransferase Increased | 60 | 5 | 33 | 2.4 |
| Aspartate Aminotransferase Increased | 55 | 3.9 | 22 | 1.7 |
| Potassium Decreased | 52 | 15 | 35 | 8 |
| GGT Increased | 51 | 5 | 24 | 0 |
| Sodium Decreased | 46 | 10 | 37 | 6 |
| Lipase Increased | 49 | 18 | 21 | 4.7 |
| Serum Amylase Increased | 31 | 7 | 13 | 0 |
Monotherapy
The safety of TECVAYLI monotherapy (N=165) in patients with relapsed or refractory multiple myeloma was evaluated in MajesTEC-1 [see Clinical Studies (14.1)]. Patients received step-up doses of 0.06 mg/kg and 0.3 mg/kg of TECVAYLI followed by TECVAYLI 1.5 mg/kg, subcutaneously once weekly. Among patients who received TECVAYLI, 47% were exposed for 6 months or longer and 7% were exposed for one year or longer.
The median age of patients who received TECVAYLI was 64 years (range: 33 to 84 years); 58% were male; 81% were White, 13% were Black or African American, and 2% were Asian.
Serious adverse reactions occurred in 54% of patients who received TECVAYLI. Serious adverse reactions in >2% of patients included pneumonia (15%), cytokine release syndrome (8%), sepsis (6%), general physical health deterioration (6%), COVID-19 (6%), acute kidney injury (4.8%), pyrexia (4.8%), musculoskeletal pain (2.4%), and encephalopathy (2.4%).
Fatal adverse reactions occurred in 5% of patients who received TECVAYLI, including COVID-19 (1.8%), pneumonia (1.8%), septic shock (0.6%), acute renal failure (0.6%), and hemoperitoneum (0.6%).
Permanent discontinuation of TECVAYLI due to adverse reactions occurred in 1.2% of patients. Adverse reactions resulting in permanent discontinuation of TECVAYLI included pneumonia (adenoviral and pneumocystis jirovecii pneumonia in the same patient) and hypercalcemia.
Dosage interruptions of TECVAYLI due to an adverse reaction occurred in 73% of patients. Adverse reactions which required dosage interruption in >5% of patients included neutropenia, pneumonia, pyrexia, cytokine release syndrome, upper respiratory tract infection, and COVID-19.
The most common adverse reactions (≥20%) were pyrexia, CRS, musculoskeletal pain, injection site reaction, fatigue, upper respiratory tract infection, nausea, headache, pneumonia, and diarrhea. The most common Grade 3 to 4 laboratory abnormalities (≥20%) were decreased lymphocytes, decreased neutrophils, decreased white blood cells, decreased hemoglobin, and decreased platelets.
Table 14 summarizes the adverse reactions in MajesTEC-1.
| Adverse Reactions | TECVAYLI (N=165) |
|
|---|---|---|
| Any Grade (%) |
Grade 3 or 4 (%) |
|
| Adverse reactions were graded based on NCI-CTCAE Version 4.03, with the exception of CRS, which was graded per ASTCT 2019 criteria. | ||
| General disorders and administration site conditions | ||
| Pyrexia | 76 | 3 Only grade 3 adverse reactions occurred.
|
| Injection site reaction Includes other related terms.
|
37 | 0.6 |
| Fatigue | 33 | 2.4 |
| Chills | 16 | 0 |
| Pain | 15 | 1.8 |
| Edema | 13 | 0 |
| Immune system disorders | ||
| Cytokine release syndrome | 72 | 0.6 |
| Hypogammaglobulinemia Hypogammaglobulinemia includes hypogammaglobulinemia and hypoglobulinemia.
|
11 | 1.2 |
| Musculoskeletal and connective tissue disorders | ||
| Musculoskeletal pain | 44 | 4.2 |
| Bone pain | 16 | 3 |
| Infections | ||
| Upper respiratory tract infection Upper respiratory tract infection includes bronchitis, nasopharyngitis, pharyngitis, rhinitis, sinusitis, tracheitis and other related terms.
|
26 | 2.4 |
| Pneumonia Pneumonia includes bacterial pneumonia, viral pneumonia, and other related terms.
Includes the following fatal adverse reactions: hemorrhage (n=1), pneumonia (n=3).
|
24 | 15 |
| Urinary tract infection | 11 | 5 |
| Gastrointestinal disorders | ||
| Nausea | 25 | 0.6 |
| Diarrhea | 21 | 2.4 |
| Constipation | 18 | 0 |
| Vomiting | 12 | 0.6 |
| Nervous system disorders | ||
| Headache | 25 | 0.6 |
| Motor dysfunction Motor dysfunction includes cogwheel rigidity, dysgraphia, dysphonia, gait disturbance, hypokinesia, muscle rigidity, muscle spasms, muscular weakness, peroneal nerve palsy, psychomotor hyperactivity, tremor and VIth nerve paralysis.
|
16 | 0 |
| Sensory neuropathy Sensory neuropathy includes dysesthesia, hypoesthesia, hypoesthesia oral, neuralgia, paresthesia, paresthesia oral, peripheral sensory neuropathy, sciatica and vestibular neuronitis.
|
15 | 1.2 |
| Encephalopathy Encephalopathy includes agitation, apathy, aphasia, confusional state, delirium, depressed level of consciousness, disorientation, dyscalculia, hallucination, lethargy, memory impairment, mental status changes and somnolence.
|
13 | 0 |
| Vascular disorders | ||
| Hypotension | 18 | 1.2 |
| Hemorrhage | 12 | 1.8 |
| Hypertension | 12 | 4.8 |
| Respiratory, thoracic, and mediastinal disorders | ||
| Hypoxia | 18 | 1.8 |
| Cough | 15 | 0 |
| Cardiac disorders | ||
| Cardiac arrhythmia | 16 | 1.8 |
| Metabolism and nutrition disorders | ||
| Decreased appetite | 11 | 0.6 |
| Renal and urinary disorders | ||
| Acute kidney injury | 11 | 3.6 |
Clinically relevant adverse reactions in <10% of patients who received TECVAYLI included febrile neutropenia, sepsis, ICANS, seizure, Guillain-Barré syndrome, hepatic failure, and new onset or reactivated viral infections (including adenovirus, hepatitis B virus (HBV), cytomegalovirus (CMV), varicella zoster virus (VZV), herpes simplex virus (HSV), and progressive multifocal leukoencephalopathy (PML).
Table 15 summarizes laboratory abnormalities in MajesTEC-1.
| Laboratory Abnormality | TECVAYLI (N=165 The denominator used to calculate the rate varied from 164 to 165 based on the number of patients with a baseline value and at least one post-treatment value. ) |
|
|---|---|---|
| All Grades (%) | Grade 3 or 4 (%) | |
| Laboratory toxicity grades are derived based on NCI CTCAE Version 4.03. | ||
| Hematology | ||
| Lymphocyte count decreased | 92 | 84 |
| White blood cell decreased | 86 | 41 |
| Neutrophil count decreased | 84 | 56 |
| Platelet count decreased | 71 | 22 |
| Hemoglobin decreased | 67 | 33 |
| Chemistry | ||
| Albumin decreased | 68 | 6 |
| Alkaline phosphatase increased | 42 | 2.4 |
| Phosphorus decreased | 38 | 13 |
| Gamma-glutamyl transferase increased | 37 | 8 |
| Sodium decreased | 35 | 10 |
| Aspartate aminotransferase increased | 34 | 1.2 |
| Calcium (corrected) decreased | 31 | 1.2 |
| Creatinine increased | 30 | 3 |
2.2 Recommended Tecvayli Dosage
In Combination with Daratumumab and Hyaluronidase-fihj
The recommended dosing schedule for TECVAYLI in combination with subcutaneous daratumumab and hyaluronidase-fihj is provided in Table 1. TECVAYLI should be administered until disease progression or unacceptable toxicity.
| Dosing schedule | Week/Day | TECVAYLI Dosage
See Table 3 for recommendations on restarting TECVAYLI after dose delays.
|
Concomitant Therapy |
|---|---|---|---|
| Day 0 | N/A | Daratumumab and hyaluronidase-fihj | |
|
Step-up dosing schedule
The Step-up dosing schedule is a component of the recommended TECVAYLI dosage but is not applicable for the daratumumab and hyaluronidase-fihj dosing.
|
Day 1 | Step-up dose 1 (0.06 mg/kg)
Step-up dose 1 must be administered 20 hours or more after the daratumumab and hyaluronidase-fihj dose.
|
N/A |
| Day 3 | Step-up dose 2 (0.3 mg/kg)
Step-up dose 2 may be given between 2 to 4 days after step-up dose 1 and if adverse reactions occur, step-up dose 2 may be given up to 7 days after step-up dose 1 to allow for resolution of adverse reactions.
|
N/A | |
| Day 7 | First treatment dose (1.5 mg/kg)
First treatment dose (1.5 mg/kg) may be given between 2 to 4 days after step-up dose 2 and if adverse reactions occur, first full treatment dose may be given up to 7 days after step-up dose 2 to allow for resolution of adverse reactions.
,
Administer TECVAYLI at least 3 hours after the daratumumab and hyaluronidase-fihj dose for the first treatment dose. For subsequent doses, administer TECVAYLI at least 15 minutes after the daratumumab and hyaluronidase-fihj dose.
|
Daratumumab and hyaluronidase-fihj | |
| Weekly dosing schedule | Weeks 2 to 8 | 1.5 mg/kg once weekly
,
Maintain a minimum of 5 days between 1.5 mg/kg once weekly doses.
|
Daratumumab and hyaluronidase-fihj once weekly |
| Biweekly (every two weeks) dosing schedule | Weeks 9 to 24 | 3 mg/kg every two weeks
,
Maintain a minimum of 12 days between 3 mg/kg every two weeks doses.
|
Daratumumab and hyaluronidase-fihj every two weeks |
| Every four weeks dosing schedule | Week 25 onwards | 3 mg/kg every four weeks
,
Maintain a minimum of 25 days between 3 mg/kg every four weeks doses.
|
Daratumumab and hyaluronidase-fihj every four weeks |
For dosage and administration instructions for daratumumab and hyaluronidase-fihj, see Clinical Studies (14.1) and refer to daratumumab and hyaluronidase-fihj monotherapy Prescribing Information.
Monotherapy
The recommended dosing schedule for TECVAYLI monotherapy is provided in Table 2. TECVAYLI should be administered until disease progression or unacceptable toxicity.
| Dosing schedule | Day | Dosage | |
|---|---|---|---|
| All Patients | |||
|
Step-up dosing schedule See Table 3 for recommendations on restarting TECVAYLI after dose delays [see Dosage and Administration (2.4)].
|
Day 1 | Step-up dose 1 | 0.06 mg/kg |
| Day 4 Step-up dose 2 may be given between 2 to 4 days after step-up dose 1 and may be given up to 7 days after step-up dose 1 to allow for resolution of adverse reactions.
|
Step-up dose 2 | 0.3 mg/kg | |
| Day 7 First treatment dose may be given between 2 to 4 days after step-up dose 2 and may be given up to 7 days after step-up dose 2 to allow for resolution of adverse reactions.
|
First treatment dose | 1.5 mg/kg | |
| Weekly dosing schedule | One week after first treatment dose and once weekly thereafter
Maintain a minimum of 5 days between 1.5 mg/kg once weekly doses
|
Subsequent treatment doses | 1.5 mg/kg once weekly |
| Patients who have achieved and maintained a complete response or better for a minimum of 6 months | |||
| Biweekly (every two weeks) dosing schedule | The dosing frequency may be decreased to 1.5 mg/kg every two weeks.
Maintain a minimum of 12 days between 1.5 mg/kg every two week doses
|
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Cytokine Release Syndrome (CRS)
Discuss the signs and symptoms associated with CRS, including fever, hypoxia, chills, hypotension, sinus tachycardia, headache, and elevated liver enzymes. Advise patients to immediately contact their healthcare provider if they experience any signs or symptoms of CRS. Advise patients that they will be hospitalized for 48 hours after administration of both step-up dose 1 and step-up dose 2. Advise patients to remain within proximity of a healthcare facility and that they will be monitored daily for 48 hours after the first treatment dose within the TECVAYLI step-up dosing schedule [see Dosage and Administration (2.5) and Warnings and Precautions (5.1)].
Neurologic Toxicity including ICANS
Discuss the signs and symptoms associated with neurologic toxicity, including ICANS, including headache, confusion, dysgraphia, decreased level of consciousness, aphasia, apraxia, somnolence, disorientation, motor dysfunction, neuropathy, or encephalopathy. Advise patients to immediately contact their healthcare provider if they experience any signs or symptoms of neurologic toxicity, including ICANS. Advise patients to refrain from driving or operating heavy or potentially dangerous machinery during and for 48 hours after completion of TECVAYLI step-up dosing schedule and in the event of new onset of any neurologic toxicity, including ICANS symptoms until neurologic toxicity resolves [see Dosage and Administration (2.5) and Warnings and Precautions (5.2)].
TECVAYLI and TALVEY REMS
TECVAYLI is available only through a restricted program called TECVAYLI and TALVEY REMS. Inform patients that they will be given a Patient Wallet Card that they should carry with them at all times and show to all of their healthcare providers. This card describes signs and symptoms of CRS and neurologic toxicity, including ICANS which, if experienced, should prompt the patient to immediately seek medical attention [see Warnings and Precautions (5.3)].
Hepatotoxicity
Advise patients that liver enzyme elevations may occur and that they should report symptoms that may indicate liver toxicity, including fatigue, anorexia, right upper abdominal discomfort, dark urine, or jaundice [see Warnings and Precautions (5.4)].
Infections
Discuss the signs and symptoms of infection [see Dosage and Administration (2.5) and Warnings and Precautions (5.5)].
Neutropenia
Discuss the signs and symptoms associated with neutropenia and febrile neutropenia [see Dosage and Administration (2.5) and Warnings and Precautions (5.6)].
Hypersensitivity and Other Administration Reactions
Advise patients to immediately seek medical attention for any signs and symptoms of systemic administration-related reactions. Advise patients that local injection-site reactions may occur and to report any severe reactions [see Warnings and Precautions (5.7)].
Embryo-Fetal Toxicity
Advise pregnant patients and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to inform their healthcare provider if they are pregnant or become pregnant. Advise females of reproductive potential to use effective contraception during treatment with TECVAYLI and for 5 months after the last dose [see Warnings and Precautions (5.8) and Use in Specific Populations (8.1, 8.3)].
Lactation
Advise women not to breastfeed during treatment with TECVAYLI and for 5 months after the last dose [see Use in Specific Populations (8.2)].
2.6 Preparation and Administration
TECVAYLI is for subcutaneous use by a healthcare provider only.
TECVAYLI should be administered by a healthcare provider with adequate medical personnel and appropriate medical equipment to manage severe reactions, including CRS and ICANS [see Warnings and Precautions (5.1, 5.2)].
TECVAYLI is a clear to slightly opalescent, colorless to light yellow solution. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use if the solution is discolored, or cloudy, or if foreign particles are present.
TECVAYLI 30 mg/3 mL (10 mg/mL) vial and TECVAYLI 153 mg/1.7 mL (90 mg/mL) vial are supplied as ready-to-use solution that do not need dilution prior to administration.
Do not combine TECVAYLI vials of different concentrations to achieve treatment dose.
Use aseptic technique to prepare and administer TECVAYLI.
Preparation of TECVAYLI
Refer to the following reference tables for the preparation of TECVAYLI.
Refer to Tables 8, 9, 10 and 11 below to determine the dosage based on predetermined weight ranges.
Use Table 8 to determine total dose, injection volume and number of vials required based on patient's actual body weight for step-up dose 1 using TECVAYLI 30 mg/3 mL (10 mg/mL) vial.
|
Patient Body Weight (kg) |
Total Dose (mg) |
Volume of Injection (mL) |
Number of Vials (1 vial=3 mL) |
|---|---|---|---|
| 35 to 39.9 | 2.2 | 0.22 | 1 |
| 40 to 44.9 | 2.5 | 0.25 | 1 |
| 45 to 49.9 | 2.8 | 0.28 | 1 |
| 50 to 59.9 | 3.3 | 0.33 | 1 |
| 60 to 69.9 | 3.9 | 0.39 | 1 |
| 70 to 79.9 | 4.5 | 0.45 | 1 |
| 80 to 89.9 | 5.1 | 0.51 | 1 |
| 90 to 99.9 | 5.7 | 0.57 | 1 |
| 100 to 109.9 | 6.3 | 0.63 | 1 |
| 110 to 119.9 | 6.9 | 0.69 | 1 |
| 120 to 129.9 | 7.5 | 0.75 | 1 |
| 130 to 139.9 | 8.1 | 0.81 | 1 |
| 140 to 149.9 | 8.7 | 0.87 | 1 |
| 150 to 160 | 9.3 | 0.93 | 1 |
Use Table 9 to determine total dose, injection volume and number of vials required based on patient's actual body weight for step-up dose 2 using TECVAYLI 30 mg/3 mL (10 mg/mL) vial.
|
Patient Body Weight (kg) |
Total Dose (mg) |
Volume of Injection (mL) |
Number of Vials (1 vial=3 mL) |
|---|---|---|---|
| 35 to 39.9 | 11 | 1.1 | 1 |
| 40 to 44.9 | 13 | 1.3 | 1 |
| 45 to 49.9 | 14 | 1.4 | 1 |
| 50 to 59.9 | 16 | 1.6 | 1 |
| 60 to 69.9 | 19 | 1.9 | 1 |
| 70 to 79.9 | 22 | 2.2 | 1 |
| 80 to 89.9 | 25 | 2.5 | 1 |
| 90 to 99.9 | 28 | 2.8 | 1 |
| 100 to 109.9 | 31 | 3.1 | 2 |
| 110 to 119.9 | 34 | 3.4 | 2 |
| 120 to 129.9 | 37 | 3.7 | 2 |
| 130 to 139.9 | 40 | 4 | 2 |
| 140 to 149.9 | 43 | 4.3 | 2 |
| 150 to 160 | 47 | 4.7 | 2 |
Use Table 10 to determine total dose, injection volume and number of vials required based on patient's actual body weight for the 1.5 mg/kg dose using TECVAYLI 153 mg/1.7 mL (90 mg/mL) vial.
|
Patient Body Weight (kg) |
Total Dose (mg) |
Volume of Injection (mL) |
Number of Vials (1 vial=1.7 mL) |
|---|---|---|---|
| 35 to 39.9 | 56 | 0.62 | 1 |
| 40 to 44.9 | 64 | 0.71 | 1 |
| 45 to 49.9 | 71 | 0.79 | 1 |
| 50 to 59.9 | 83 | 0.92 | 1 |
| 60 to 69.9 | 99 | 1.1 | 1 |
| 70 to 79.9 | 108 | 1.2 | 1 |
| 80 to 89.9 | 126 | 1.4 | 1 |
| 90 to 99.9 | 144 | 1.6 | 1 |
| 100 to 109.9 | 153 | 1.7 | 1 |
| 110 to 119.9 | 171 | 1.9 | 2 |
| 120 to 129.9 | 189 | 2.1 | 2 |
| 130 to 139.9 | 198 | 2.2 | 2 |
| 140 to 149.9 | 216 | 2.4 | 2 |
| 150 to 160 | 234 | 2.6 | 2 |
Use Table 11 to determine total dose, injection volume and number of vials required based on patient's actual body weight for the 3 mg/kg dose using TECVAYLI 153 mg/1.7 mL (90 mg/mL) vial.
|
Patient Body Weight (kg) |
Total Dose (mg) |
Volume of Injection (mL) |
Number of Vials (1 vial=1.7 mL) |
|---|---|---|---|
| 35 to 39.9 | 108 | 1.2 | 1 |
| 40 to 44.9 | 126 | 1.4 | 1 |
| 45 to 49.9 | 144 | 1.6 | 1 |
| 50 to 59.9 | 162 | 1.8 | 2 |
| 60 to 69.9 | 198 | 2.2 | 2 |
| 70 to 79.9 | 225 | 2.5 | 2 |
| 80 to 89.9 | 252 | 2.8 | 2 |
| 90 to 99.9 | 288 | 3.2 | 2 |
| 100 to 109.9 | 315 | 3.5 | 3 |
| 110 to 119.9 | 342 | 3.8 | 3 |
| 120 to 129.9 | 378 | 4.2 | 3 |
| 130 to 139.9 | 405 | 4.5 | 3 |
| 140 to 149.9 | 432 | 4.8 | 3 |
| 150 to 160 | 468 | 5.2 | 4 |
- Remove the appropriate strength TECVAYLI vial(s) from refrigerated storage [2 °C to 8 °C (36 °F to 46 °F)].
- Once removed from refrigerated storage, equilibrate TECVAYLI to ambient temperature [15 °C to 30 °C (59 °F to 86 °F)] for at least 15 minutes. Do not warm TECVAYLI in any other way.
- Gently swirl the vial for approximately 10 seconds to mix. Do not shake.
- Withdraw the required injection volume of TECVAYLI from the vial(s) into an appropriately sized syringe using a transfer needle.
- Each injection volume should not exceed 2 mL. Divide doses that require greater than 2 mL equally into multiple syringes.
- Use TECVAYLI with stainless steel injection needles and polypropylene or polycarbonate syringe material.
- Replace the transfer needle with an appropriately sized needle for injection.
Administration of TECVAYLI
Inject the required volume of TECVAYLI into the subcutaneous tissue of the abdomen (preferred injection site). Alternatively, TECVAYLI may be injected into the subcutaneous tissue at other sites (e.g., thigh). If multiple injections are needed, administer injections at least 2 cm apart.
Do not inject into tattoos or scars or areas where the skin is red, bruised, tender, hard or not intact.
Storage and Disposal
If the prepared dosing syringe(s) of TECVAYLI is not used immediately, store syringe(s) at 2 °C to 8 °C (36 °F to 46 °F) or at ambient temperature 15 °C to 30 °C (59 °F to 86 °F) for a maximum of 20 hours. Discard syringe(s) after 20 hours, if not used.
Dispose of any unused product or waste material in accordance with local requirements.
16 How Supplied/storage and Handling
TECVAYLI® (teclistamab-cqyv) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to light yellow solution supplied as follows:
- One 30 mg/3 mL (10 mg/mL) single-dose vial in a carton: NDC: 57894-449-01
- One 153 mg/1.7 mL (90 mg/mL) single-dose vial in a carton: NDC: 57894-450-01
2.3 Recommended Pretreatment Medications
Administer the following pretreatment medications 1 to 3 hours before each dose of the TECVAYLI step-up dosing schedule, which includes step-up dose 1, step-up dose 2, and the first treatment dose (see Tables 1 and 2), to reduce the risk of CRS [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
- Corticosteroid (oral or intravenous dexamethasone 16 mg)
- Histamine-1 (H1) receptor antagonist (oral or intravenous diphenhydramine 50 mg or equivalent)
- Antipyretics (oral or intravenous acetaminophen 650 mg to 1,000 mg)
Administration of pretreatment medications may be required prior to administration of subsequent doses of TECVAYLI in patients who:
- Repeat doses within the TECVAYLI step-up dosing schedule following a dose delay [see Dosage and Administration (2.4)].
- Experienced CRS following the prior dose of TECVAYLI [see Dosage and Administration (2.5)].
Prophylaxis for Herpes Zoster Reactivation
Prior to starting treatment with TECVAYLI, consider initiation of antiviral prophylaxis to prevent herpes zoster reactivation per guidelines.
2.4 Restarting Tecvayli After Dosage Delay
If a dose of TECVAYLI is delayed, restart therapy based on the recommendations in Table 3 and resume the treatment schedule accordingly [see Dosage and Administration (2.2)]. Administer pretreatment medications as indicated in Table 3 [see Dosage and Administration (2.3)].
| Last dose administered | Time since the last dose administered | Action |
|---|---|---|
| Step-up dose 1 | More than 7 days | Restart TECVAYLI step-up dosing schedule at step-up dose 1 (0.06 mg/kg). Administer pretreatment medications prior to TECVAYLI dose and monitor patients accordingly [see Dosage and Administration (2.3, 2.5)].
|
| Step-up dose 2 | 8 days to 28 days | Repeat step-up dose 2 (0.3 mg/kg) and continue TECVAYLI step-up dosing schedule. |
|
More than 28 days Consider benefit-risk of restarting TECVAYLI in patients who require a dose delay of more than 28 days due to an adverse reaction.
|
Restart TECVAYLI step-up dosing schedule at step-up dose 1 (0.06 mg/kg). | |
| Weekly treatment dose | 28 days or less | Continue TECVAYLI 1.5 mg/kg once weekly. |
| 29 days to 56 days | Restart TECVAYLI step-up dosing schedule at step-up dose 2 (0.3 mg/kg). | |
| More than 56 days | Restart TECVAYLI step-up dosing schedule at step-up dose 1 (0.06 mg/kg). | |
| Any biweekly (every two weeks) or every four weeks treatment dose | 63 days or less | Continue TECVAYLI at last dose given every two weeks or every four weeks schedule. |
| 64 days to 112 days | Restart TECVAYLI step-up dosing schedule at step-up dose 2 (0.3 mg/kg). | |
| More than 112 days | Restart TECVAYLI step-up dosing schedule at step-up dose 1 (0.06 mg/kg). |
14.1 Relapsed Or Refractory Multiple Myeloma
In Combination with Daratumumab and Hyaluronidase-fihj
The efficacy of TECVAYLI in combination with subcutaneous daratumumab and hyaluronidase-fihj compared with investigator's choice of either daratumumab and hyaluronidase-fihj, pomalidomide and dexamethasone (DPd) or daratumumab and hyaluronidase-fihj, bortezomib and dexamethasone (DVd) was evaluated in adult patients with relapsed or refractory multiple myeloma in a randomized, open-label, multi-center study (MajesTEC-3) (NCT05083169). The study included patients who had previously received one to three prior lines of therapy including a proteasome inhibitor and lenalidomide. Patients who had received only one prior line of therapy must have been refractory to lenalidomide. Patients who had disease refractory to a prior anti-CD38 monoclonal antibody therapy, or who had received any prior BCMA-directed therapy were excluded.
Patients received TECVAYLI in combination with daratumumab and hyaluronidase-fihj as follows:
- Subcutaneous TECVAYLI step-up doses of 0.06 mg/kg and 0.3 mg/kg, followed by TECVAYLI 1.5 mg/kg once weekly from Weeks 2 to 8, then 3 mg/kg every two weeks from Weeks 9 to 24, then 3 mg/kg every four weeks starting on Week 25 until disease progression or unacceptable toxicity,
AND - Subcutaneous daratumumab and hyaluronidase-fihj 1,800 mg/30,000 units (1,800 mg of daratumumab and 30,000 units of hyaluronidase) starting one day prior to TECVAYLI and continuing once weekly from Weeks 1 to 8, then every two weeks from Weeks 9 to 24, then every four weeks starting on Week 25 until disease progression or unacceptable toxicity [see Dosage and Administration (2.2)].
The major efficacy outcome measure was progression-free survival (PFS) by Independent Review Committee (IRC) assessment based on International Myeloma Working Group (IMWG) 2016 criteria.
A total of 587 patients were randomized in MajesTEC-3: 291 to the TECVAYLI in combination with daratumumab and hyaluronidase-fihj group and 296 to the control group (DPd or DVd). The median age was 64 years (range: 25 to 88) with 38% aged 65 to 74 years and 10% aged 75 years or older; 55% were male; 65% were White, 6% were Black or African American, and 22% were Asian. The International Staging System (ISS) at screening was stage I in 63%, stage II in 29%, and stage III in 8%. High-risk cytogenetics (presence of del(17p), t(4;14) or t(14;16)) were present in 35% of patients. Extramedullary disease was present in 5% of patients.
The median number of prior lines of therapy was 2 (range: 1 to 3), with 38% who received one prior line of therapy. All patients received prior lenalidomide and 99.8% received a prior proteasome inhibitor; 84% were refractory to lenalidomide. Seventy-four percent of patients previously received autologous stem cell transplantation. Five percent of patients received a prior anti-CD38 monoclonal antibody; there is limited data with TECVAYLI in combination with daratumumab and hyaluronidase-fihj in patients who have received or are refractory to prior anti-CD38 monoclonal antibody therapy.
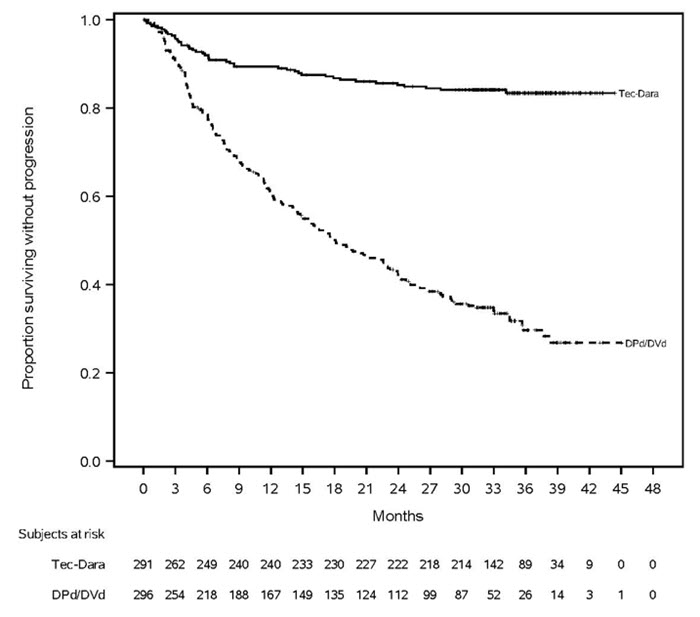
The trial demonstrated a statistically significant improvement in the TECVAYLI in combination with daratumumab and hyaluronidase-fihj group compared with the control group in PFS and overall survival (OS). The efficacy results are shown in Table 17 and Figures 1 and 2.
| TECVAYLI with daratumumab and hyaluronidase-fihj (N=291) |
DPd or DVd (N=296) |
|
|---|---|---|
| DPd = daratumumab and hyaluronidase-fihj, pomalidomide, dexamethasone; DVd = daratumumab and hyaluronidase-fihj, bortezomib, dexamethasone; CI=confidence interval; NE=not estimable; NR=not reached; MRD = minimal residual disease. | ||
|
Progression-free survival (PFS) Based on intent-to-treat analysis set
,
Median duration of follow-up = 34.5 months
|
||
| Number of events, n (%) | 44 (15) | 187 (63) |
| Median, months (95% CI) | NR (NE, NE) | 18.1 (14.6, 22.8) |
| Hazard ratio (95% CI) Stratified Cox proportional hazard model. For all stratified analyses, stratification was based on ISS staging (I vs. II or III) and number of prior lines of therapy (1 vs. 2 or 3), as randomized ; p-valueStratified log-rank test
|
0.17 (0.12, 0.23); <0.0001 | |
| Overall survival (OS) , | ||
| Number of events, n (%) | 46 (16) | 98 (33) |
| Median, months (95% CI) | NR (NE, NE) | NR (41.4, NE) |
| Hazard ratio (95% CI); p-value | 0.46 (0.32, 0.65); <0.0001 | |
| Overall response (sCR+CR+VGPR+PR), n (%) | 259 (89.0) | 223 (75.3) |
| 95% CI (%) | (84.8, 92.4) | (70.0, 80.1) |
| p-value Stratified Cochran Mantel-Haenszel Chi-Squared test
|
<0.0001 | |
| Stringent complete response (sCR), n (%) | 225 (77.3) | 69 (23.3) |
| Complete response (CR), n (%) | 13 (4.5) | 26 (8.8) |
| Very good partial response (VGPR), n (%) | 14 (4.8) | 74 (25.0) |
| Partial response (PR), n (%) | 7 (2.4) | 54 (18.2) |
| Complete response or better (sCR+CR), n (%) | 238 (81.8) | 95 (32.1) |
| 95% CI (%) | (76.9, 86.0) | (26.8, 37.7) |
| p-value | <0.0001 | |
|
MRD negativity rate Based on MRD next-generation sequencing (NGS) primary analysis set, defined as all randomized patients enrolled at sites where next generation sequencing assay was available.
,
Based on a threshold of 10-5 using a next generation sequencing assay (clonoSEQ). , N
|
262 | 269 |
| n (%) | 153 (58.4) | 46 (17.1) |
| 95% CI (%) | (52.2, 64.4) | (12.8, 22.1) |
| p-value Fisher's exact test
|
<0.0001 | |
| MRD negativity rate in patients with CR or better | ||
| Number of patients with CR or better, n | 216 | 86 |
| MRD negativity rate, n (%) | 153 (70.8) | 46 (53.5) |
| 95% CI (%) | (64.3, 76.8) | (42.4, 64.3) |
| Figure 1: Kaplan-Meier Curve of PFS (MajesTEC-3) |
|---|
| Tec-Dara = TECVAYLI in combination with daratumumab and hyaluronidase-fihj; DPd = daratumumab and hyaluronidase-fihj, pomalidomide, dexamethasone; DVd = daratumumab and hyaluronidase-fihj, bortezomib, dexamethasone. |
| Figure 2: Kaplan-Meier Curve of OS (MajesTEC-3) |
|---|
| Tec-Dara = TECVAYLI in combination with daratumumab and hyaluronidase-fihj; DPd = daratumumab and hyaluronidase-fihj, pomalidomide, dexamethasone; DVd = daratumumab and hyaluronidase-fihj, bortezomib, dexamethasone. |
In the 15 patients in MajesTEC-3 who had received a prior anti-CD38 monoclonal antibody, the ORR was 93.3% (95% CI: 68.1, 99.8).
Monotherapy
The efficacy of TECVAYLI was evaluated in patients with relapsed or refractory multiple myeloma in a single-arm, open-label, multi-center study (MajesTEC-1, NCT03145181 [Phase 1] and NCT04557098 [Phase 2]). The study included patients who had previously received at least three prior therapies, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody. The study excluded patients who had stroke, seizure, allogeneic stem cell transplantation within the past 6 months, Eastern Cooperative Oncology Group (ECOG) performance score of 2 or higher, known active CNS involvement or clinical signs of meningeal involvement of multiple myeloma, or active or documented history of autoimmune disease, with the exception of vitiligo, Type 1 diabetes, and prior autoimmune thyroiditis.
Patients received step-up doses of 0.06 mg/kg and 0.3 mg/kg of TECVAYLI followed by TECVAYLI 1.5 mg/kg subcutaneously once weekly thereafter until disease progression or unacceptable toxicity [see Dosage and Administration (2.2)].
The efficacy population included 110 patients. The median age was 66 (range: 33 to 82) years with 16% of patients 75 years of age or older; 56% were male; 91% were White, 5% were Black or African American, 3% were Asian. The International Staging System (ISS) at study entry was Stage I in 50%, Stage II in 38%, and Stage III in 12% of patients. High-risk cytogenetics (presence of del(17p), t(4;14) or t(14;16)) were present in 25% of patients. Seventeen percent of patients had extramedullary plasmacytomas. Patients with prior BCMA-targeted therapy were not included in the efficacy population.
The median number of prior lines of therapy was 5 (range: 2 to 14); 78% of patients had received at least 4 prior lines of therapy. Eighty-one percent of patients received prior stem cell transplantation. All patients had received prior therapy with a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody, and 76% were triple-class refractory (refractory to a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody).
Efficacy was established based on overall response rate (ORR) as determined by the Independent Review Committee (IRC) assessment using International Myeloma Working Group (IMWG) 2016 criteria (see Table 18).
The median time to first response was 1.2 months (range: 0.2 to 5.5 months). With a median follow-up of 7.4 months among responders, the estimated duration of response (DOR) rate was 90.6% (95% CI: 80.3%, 95.7%) at 6 months and 66.5% (95% CI: 38.8%, 83.9%) at 9 months.
| N=110 | |
|---|---|
| NE=not estimable | |
| Overall response rate (ORR: sCR+CR+VGPR+PR) n (%) | 68 (61.8) |
| 95% CI (%) | (52.1, 70.9) |
| Complete response (CR) or better Complete response or better = Stringent complete response (sCR) + complete response (CR).
|
31 (28.2) |
| Very good partial response (VGPR) | 32 (29.1) |
| Partial response (PR) | 5 (4.5) |
| Duration of Response (DOR) (months) | |
| DOR (Months): Median (95% CI) | NE (9.0, NE) |
2.5 Dosage Modifications for Adverse Reactions
Dosage reductions of TECVAYLI are not recommended.
Refer to daratumumab and hyaluronidase-fihj Prescribing Information for information about dosage modifications for daratumumab and hyaluronidase-fihj.
Dosage delays may be required to manage toxicities related to TECVAYLI [see Warnings and Precautions (5)].
See Tables 4, 5 and 6 for recommended actions for adverse reactions of CRS, neurologic toxicity, and ICANS. See Table 7 for recommended actions for other adverse reactions following administration of TECVAYLI.
Management of CRS, Neurologic Toxicity, and ICANS
Cytokine Release Syndrome
Management recommendations for cytokine release syndrome (CRS) are summarized in Table 4.
Identify CRS based on clinical presentation [see Warnings and Precautions (5.1)]. Evaluate and treat other causes of fever, hypoxia, and hypotension.
If CRS is suspected, withhold TECVAYLI until CRS resolves. Manage according to the recommendations in Table 4 and consider further management per current practice guidelines. Administer supportive therapy for CRS, which may include intensive care for severe or life-threatening CRS. Consider laboratory testing to monitor for disseminated intravascular coagulation (DIC), hematology parameters, as well as pulmonary, cardiac, renal, and hepatic function.
| Grade Based on American Society for Transplantation and Cellular Therapy (ASTCT) 2019 grading for CRS.
|
Presenting Symptoms | Actions |
|---|---|---|
| Grade 1 | Temperature ≥100.4 °F (38 °C) Attributed to CRS. Fever may not always be present concurrently with hypotension or hypoxia as it may be masked by interventions such as antipyretics or anticytokine therapy.
|
|
| Grade 2 | Temperature ≥100.4 °F (38 °C) with: Hypotension responsive to fluids and not requiring vasopressors, and/or, Oxygen requirement of low-flow nasal cannula Low-flow nasal cannula is ≤6 L/minute, and high-flow nasal cannula is >6 L/minute. or blow-by. |
|
| Grade 3 | Temperature ≥100.4 °F (38 °C) with: Hypotension requiring one vasopressor with or without vasopressin, and/or, Oxygen requirement of high-flow nasal cannula, facemask, non-rebreather mask, or Venturi mask. |
First Occurrence of Grade 3 CRS with Duration <48 Hours:
|
Recurrent Grade 3 CRS or Grade 3 CRS with Duration 48 Hours or Longer:
|
||
| Grade 4 | Temperature ≥100.4 °F (38 °C) with: Hypotension requiring multiple vasopressors (excluding vasopressin), and/or, Oxygen requirement of positive pressure (e.g., continuous positive airway pressure (CPAP), bilevel positive airway pressure (BiPAP), intubation, and mechanical ventilation). |
|
Neurologic Toxicity and Immune Effector Cell-Associated Neurotoxicity Syndrome
Management recommendations for neurologic toxicity and immune effector cell-associated neurotoxicity syndrome (ICANS) are summarized in Tables 5 and 6.
At the first sign of neurologic toxicity, including ICANS, withhold TECVAYLI and consider neurology evaluation. Rule out other causes of neurologic symptoms. Provide supportive therapy, which may include intensive care, for severe or life-threatening neurologic toxicities, including ICANS [see Warnings and Precautions (5.2)]. Manage ICANS according to the recommendations in Table 6 and consider further management per current practice guidelines.
| Severity Based on National Cancer Institute Common Terminology Criteria for Adverse Events (NCI- CTCAE), version 4.03.
|
Actions |
|---|---|
| Grade 1 | Withhold TECVAYLI until neurologic toxicity symptoms resolve or stabilize. See Table 3 for recommendations on restarting TECVAYLI after dose delays [see Dosage and Administration (2.4)].
|
| Grade 2 Grade 3 (First occurrence) |
|
| Grade 3 (Recurrent) Grade 4 |
|
| Grade Based on American Society for Transplantation and Cellular Therapy (ASTCT) 2019 grading for ICANS.
|
Presenting Symptoms Management is determined by the most severe reaction, not attributable to any other cause.
|
Actions |
|---|---|---|
| Grade 1 | ICE score 7–9 If patient is arousable and able to perform Immune Effector Cell-Associated Encephalopathy (ICE) Assessment, assess: Orientation (oriented to year, month, city, hospital = 4 points); Naming (name 3 objects, e.g., point to clock, pen, button = 3 points); Following Commands (e.g., "show me 2 fingers" or "close your eyes and stick out your tongue" = 1 point); Writing (ability to write a standard sentence = 1 point; and Attention (count backwards from 100 by ten = 1 point). If patient is unarousable and unable to perform ICE Assessment (Grade 4 ICANS) = 0 points. , or depressed level of consciousness Not attributable to any other cause. : awakens spontaneously. |
|
| Grade 2 | ICE score 3–6, or depressed level of consciousness: awakens to voice. |
|
| Grade 3 | ICE score 0–2, or depressed level of consciousness: awakens only to tactile stimulus, or seizures, either:
|
First Occurrence of Grade 3 ICANS:
|
Recurrent Grade 3 ICANS:
|
||
| Grade 4 | ICE score 0, or depressed level of consciousness: either:
|
|
| Adverse Reactions | Severity | Actions |
|---|---|---|
| Infections Based on National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE), version 4.03.
[see Warnings and Precautions (5.5)]
|
All Grades | Withhold TECVAYLI in patients with active infection during the step-up dosing schedule. See Table 3 for recommendations on restarting TECVAYLI after dose delays [see Dosage and Administration (2.4)].
|
| Grade 3 | Withhold subsequent TECVAYLI doses administered after TECVAYLI step-up dosing schedule until infection improves to Grade 1 or less. | |
| Grade 4 |
|
|
| Hematologic Toxicities [see Warnings and Precautions (5.6) and Adverse Reactions (6.1)] | Absolute neutrophil count (ANC) < 0.5 × 109/L |
|
| Febrile neutropenia |
|
|
| Hemoglobin < 8 g/dL |
|
|
|
Monotherapy: Platelet count < 25,000/mcL or platelet count between 25,000/mcL and 50,000/mcL with bleeding Combination therapy: Platelet count < 50,000/mcL |
Monotherapy:
|
|
| Other Non-Hematologic Adverse Reactions [see Warnings and Precautions (5.4) and Adverse Reactions (6.1)] | Grade 3 | Withhold TECVAYLI until adverse reaction improves to Grade 1 or less. |
| Grade 4 |
|
8.3 Females and Males of Reproductive Potential
TECVAYLI may cause fetal harm when administered to a pregnant patient [see Use in Specific Populations (8.1)].
Pregnancy Testing
Verify pregnancy status of females of reproductive potential prior to initiating TECVAYLI.
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment and for 5 months after the last dose of TECVAYLI.
Principal Display Panel 30 Mg/3 Ml Vial Carton
Rx only
One Vial
NDC 57894-449-01
TECVAYLI®
(teclistamab-cqyv)
Injection
30 mg/3 mL
(10 mg/mL)
TECVAYLI®vials of different
concentrations should not be
combined to achieve treatment dose.
For subcutaneous injection
by a healthcare provider only.
Attention: Dispense the enclosed
Medication Guide to each patient.
Single-dose vial
Discard unused portion.
janssen
2.1 Important Dosage and Administration Information
TECVAYLI is for subcutaneous injection only.
Administer pretreatment medications prior to each dose of the TECVAYLI step-up dosing schedule, which includes step-up dose 1, step-up dose 2, and the first treatment dose as described in Tables 1 and 2 [see Dosage and Administration (2.3)].
Administer TECVAYLI subcutaneously according to the step-up dosing schedule in Tables 1 and 2 to reduce the incidence and severity of cytokine release syndrome (CRS). Due to the risk of CRS and neurologic toxicity, including ICANS, patients should be hospitalized for 48 hours after administration of both step-up dose 1 and step-up dose 2. Instruct patients to remain within proximity of a healthcare facility and monitor them daily for 48 hours after the first treatment dose within the TECVAYLI step-up dosing schedule [see Dosage and Administration (2.5) and Warnings and Precautions (5.1, 5.2)].
Refer to Tables 8, 9, 10, and 11 to determine the dosage based on predetermined weight ranges [see Dosage and Administration (2.6)].
Principal Display Panel 153 Mg/1.7 Ml Vial Carton
Rx only
One Vial
NDC 57894-450-01
TECVAYLI®
(teclistamab-cqyv)
Injection
153 mg/1.7 mL
(90 mg/mL)
TECVAYLI®vials of different
concentrations should not be
combined to achieve treatment dose.
For subcutaneous injection
by a healthcare provider only.
Attention: Dispense the enclosed
Medication Guide to each patient.
Single-dose vial
Discard unused portion.
janssen
5.7 Hypersensitivity and Other Administration Reactions
TECVAYLI can cause both systemic administration-related reactions and local injection-site reactions.
Systemic Reactions
In patients who received the recommended TECVAYLI dosage in the clinical trials (monotherapy and combination therapy trials; N=448), 2.5% of patients experienced systemic-administration reactions, which included recurrent pyrexia and rash.
Local Reactions
In patients who received TECVAYLI at the recommended dosage in the clinical trials (monotherapy and combination therapy trials; N=448), injection-site reactions occurred in 37% of patients with Grade 1 injection-site reactions in 29% and Grade 2 in 9%.
Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity [see Dosage and Administration (2.5)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity or genotoxicity studies have been conducted with teclistamab-cqyv.
No studies have been conducted to evaluate the effects of teclistamab-cqyv on fertility.
5.2 Neurologic Toxicity Including Immune Effector Cell Associated Neurotoxicity Syndrome
TECVAYLI can cause serious, life-threatening or fatal neurologic toxicity, including immune effector cell-associated neurotoxicity syndrome (ICANS) [see Adverse Reactions (6.1)].
In the clinical trials (monotherapy and combination therapy trials; N=448), neurologic toxicity occurred in 60% of patients who received TECVAYLI at the recommended dosage, with Grade 3 or 4 neurologic toxicity in 6%. Neurologic toxicities reported in ≥5% of patients included headache (27%), sensory neuropathy (16%), motor dysfunction (15%), insomnia (12%), encephalopathy (11%), and dizziness (8%). Fatal neurologic toxicity occurred in 0.4% of patients, including Guillain-Barré syndrome and status epilepticus (one patient each).
In MajesTEC-1, ICANS was reported in 6% of patients who received TECVAYLI as monotherapy at the recommended dosage [see Adverse Reactions (6.1)]. Recurrent ICANS occurred in 1.8% of patients. Most patients experienced ICANS following step-up dose 1 (1.2%), step-up dose 2 (0.6%), or the initial treatment dose (1.8%). Less than 3% of patients developed first occurrence of ICANS following subsequent TECVAYLI doses. The median time to onset of ICANS was 4 days (range: 2 to 8 days) after the most recent TECVAYLI dose with a median duration of 3 days (range: 1 to 20 days). The most frequent clinical manifestations of ICANS reported were confusional state and dysgraphia.
In MajesTEC-3, ICANS was reported in 1.1% of patients who received the recommended TECVAYLI dosage in combination with daratumumab and hyaluronidase-fihj, including Grade 4 ICANS in 1 patient. All events of ICANS occurred during the step-up dosing schedule [see Adverse Reactions (6.1)]. The median time to onset of ICANS was 2 days (range: 1 to 3 days) after the most recent dose and the median duration of ICANS was 2 days (range: 1 to 2 days). The clinical manifestations of ICANS reported were amnesia, encephalopathy and delirium.
The onset of ICANS can be concurrent with CRS, following resolution of CRS, or in the absence of CRS.
Monitor patients for signs and symptoms of neurologic toxicity, including ICANS during TECVAYLI treatment. At the first sign of neurologic toxicity, including ICANS, immediately evaluate patient and provide supportive therapy based on severity. Withhold until neurologic toxicity resolves or permanently discontinue TECVAYLI based on severity per recommendations and consider further management per current practice guidelines [see Dosage and Administration (2.5)].
Due to the potential for neurologic toxicity, patients receiving TECVAYLI are at risk of depressed level of consciousness [see Adverse Reactions (6.1)]. Advise patients to refrain from driving or operating heavy or potentially dangerous machinery during and for 48 hours after completion of TECVAYLI step-up dosing schedule and in the event of new onset of any neurologic toxicity symptoms until neurologic toxicity resolves [see Dosage and Administration (2.1)].
TECVAYLI is available only through a restricted program under a REMS [see Warnings and Precautions (5.3)].
Warning: Cytokine Release Syndrome and Neurologic Toxicity, Including Immune Effector Cell Associated Neurotoxicity Syndrome
Cytokine release syndrome (CRS), including life-threatening or fatal reactions, can occur in patients receiving TECVAYLI. Initiate treatment with TECVAYLI step-up dosing schedule to reduce risk of CRS. Withhold TECVAYLI until CRS resolves or permanently discontinue based on severity [see Dosage and Administration (2.1 , 2.5) and Warnings and Precautions (5.1)].
Neurologic toxicity, including Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS) and serious, life-threatening, or fatal reactions, can occur in patients receiving TECVAYLI. Monitor patients for signs or symptoms of neurologic toxicity, including ICANS, during treatment. Withhold TECVAYLI until neurologic toxicity resolves or permanently discontinue based on severity [see Dosage and Administration (2.5) and Warnings and Precautions (5.2)].
Because of the risk of CRS and neurologic toxicity, including ICANS, TECVAYLI is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the TECVAYLI and TALVEY REMS [see Warnings and Precautions (5.3)].
Structured Label Content
Section 42229-5 (42229-5)
Manufactured by:
Janssen Biotech, Inc.
Horsham, PA 19044, USA
U.S. License Number 1864
For patent information: www.janssenpatents.com
© Johnson & Johnson and its affiliates 2022, 2026
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: Mar 2026 | ||
|
MEDICATION GUIDE TECVAYLI® [tek vay lee] (teclistamab-cqyv) injection, for subcutaneous use |
|||
|
What is the most important information I should know about TECVAYLI?
TECVAYLI can cause side effects that are serious, life-threatening or cause death, including cytokine release syndrome (CRS) and neurologic problems.
|
|||
|
|
||
Due to the risk of CRS, you will receive TECVAYLI on a "step-up dosing" schedule. You should be hospitalized for 48 hours after the first and second step-up doses and should stay close to a healthcare facility and be monitored by a healthcare provider for 48 hours after you receive your first treatment dose.
|
|||
|
|||
|
|
|
|
Your healthcare provider will monitor you for signs and symptoms of CRS and neurologic problems during treatment with TECVAYLI and treat you as needed. Your healthcare provider may temporarily stop or completely stop your treatment with TECVAYLI if you develop CRS or neurologic problems.
See " What are the possible side effects of TECVAYLI?" for more information about side effects. |
|||
|
What is TECVAYLI?
TECVAYLI is a prescription medicine used to treat adults with multiple myeloma that has come back or did not respond to prior treatment:
|
|||
Before you receive TECVAYLI, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
How will I receive TECVAYLI?
|
|||
|
What should I avoid while receiving TECVAYLI?
Do not drive or operate heavy or dangerous machinery during and for 48 hours after your TECVAYLI step-up dosing schedule is completed, or at any time during treatment with TECVAYLI if you develop new neurologic symptoms such as dizziness, confusion, tremors, sleepiness, or any other symptoms that impair your consciousness, until the symptoms go away. See "What is the most important information I should know about TECVAYLI?" for more information about signs and symptoms of neurologic problems. |
|||
|
What are the possible side effects of TECVAYLI?
TECVAYLI can cause serious side effects, including:
|
|||
|
|
||
Your healthcare provider will monitor you for signs and symptoms of these serious side effects during treatment and may temporarily or permanently stop TECVAYLI if you develop severe side effects. The most common side effects of TECVAYLI when used alone include: |
|||
|
|
|
|
| The most common side effects of TECVAYLI when used with daratumumab and hyaluronidase-fihj include: | |||
|
|
|
|
|
The most common severe abnormal blood test results with TECVAYLI include: decreased white blood cells, red blood cells, and platelets. These are not all the possible side effects of TECVAYLI. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
General Information about the safe and effective use of TECVAYLI.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about TECVAYLI that is written for health professionals. |
|||
|
What are the ingredients in TECVAYLI?
Active ingredient: teclistamab-cqyv Inactive ingredients: edetate disodium, glacial acetic acid, polysorbate 20, sodium acetate, sucrose, Water for Injection Manufactured by: Janssen Biotech, Inc., Horsham, PA 19044, USA U.S. License Number 1864 For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2022, 2026 For more information about TECVAYLI go to www.TECVAYLI.com or call 1-800-526-7736. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store refrigerated at 2 °C to 8 °C (36 °F to 46 °F) in the original carton to protect from light.
Do not freeze.
8.1 Pregnancy
Risk Summary
Based on the mechanism of action, TECVAYLI may cause fetal harm when administered to a pregnant patient [see Clinical Pharmacology (12.1)]. There are no available data on the use of TECVAYLI in pregnant patients to evaluate for a drug associated risk. No animal reproductive or developmental toxicity studies have been conducted with TECVAYLI. Teclistamab-cqyv causes T-cell activation and cytokine release; immune activation may compromise pregnancy maintenance. Human immunoglobulin G (IgG) is known to cross the placenta; therefore, teclistamab-cqyv has the potential to be transmitted from the mother to the developing fetus. Advise women of the potential risk to the fetus.
TECVAYLI is associated with hypogammaglobulinemia, therefore, assessment of immunoglobulin levels in newborns of mothers treated with TECVAYLI should be considered.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data on the presence of teclistamab-cqyv in human milk, the effect on the breastfed child, or the effects on milk production. Maternal IgG is known to be present in human milk. The effects of local gastrointestinal exposure and limited systemic exposure in the breastfed child to TECVAYLI are unknown. Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during treatment with TECVAYLI and for 5 months after the last dose.
11 Description (11 DESCRIPTION)
Teclistamab-cqyv, a bispecific B-cell maturation antigen (BCMA)-directed CD3 T-cell engager, is a humanized immunoglobulin G4-proline, alanine, alanine (IgG4-PAA) antibody. Teclistamab-cqyv is produced in Chinese Hamster Ovary (CHO) cells using recombinant DNA technology. Teclistamab-cqyv consists of an anti-BCMA heavy chain and light chain and an anti-CD3 heavy chain and light chain with two interchain disulfide bonds connecting the two arms. The molecular weight of teclistamab-cqyv is approximately 146 kDa.
TECVAYLI® (teclistamab-cqyv) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to light yellow solution supplied in a single-dose vial for subcutaneous administration.
Each TECVAYLI 3 mL single-dose vial contains 30 mg of teclistamab-cqyv, edetate disodium (0.054 mg), glacial acetic acid (0.72 mg), polysorbate 20 (1.2 mg), sodium acetate (2.7 mg), sucrose (240 mg), and Water for Injection, USP.
Each TECVAYLI 1.7 mL single-dose vial contains 153 mg of teclistamab-cqyv, edetate disodium (0.031 mg), glacial acetic acid (0.41 mg), polysorbate 20 (0.68 mg), sodium acetate (1.5 mg), sucrose (140 mg), and Water for Injection, USP.
5.5 Infections
TECVAYLI can cause severe, life-threatening, or fatal infections.
In MajesTEC-1 (N=165), in patients who received the recommended TECVAYLI dosage, serious infections, including opportunistic infections, occurred in 30% of patients, Grade 3 or 4 infections occurred in 35% of patients, and fatal infections occurred in 4.2% of patients [see Adverse Reactions (6.1)].
In MajesTEC-3 (N=283), in patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj at the recommended dosage, serious infections, including opportunistic infections, occurred in 54% of patients, Grade 3 or Grade 4 infections occurred in 54% of patients and fatal infections occurred in 4.6% of patients [see Adverse Reactions (6.1)].
Monitor patients for signs and symptoms of infection prior to and during treatment with TECVAYLI and treat appropriately. Administer prophylactic antimicrobials according to current practice guidelines [see Dosage and Administration (2.3)].
Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity [see Dosage and Administration (2.5)].
Monitor immunoglobulin levels prior to and during treatment with TECVAYLI and administer subcutaneous or intravenous immunoglobulin (IVIG) to maintain the serum levels >400 mg/dL.
5.6 Neutropenia
TECVAYLI can cause neutropenia and febrile neutropenia. In patients who received TECVAYLI at the recommended dosage in the clinical trials (monotherapy and combination therapy trials; N=448), decreased neutrophils occurred in 88% of patients, with Grade 3 or 4 decreased neutrophils in 70%. Febrile neutropenia occurred in 6% of patients [see Adverse Reactions (6.1)].
Monitor complete blood cell counts at baseline and periodically during treatment and provide supportive care per local institutional guidelines.
Monitor patients with neutropenia for signs of infection.
Withhold TECVAYLI based on severity [see Dosage and Administration (2.5)].
8.4 Pediatric Use
The safety and efficacy of TECVAYLI have not been established in pediatric patients.
8.5 Geriatric Use
- Of the 165 patients with relapsed or refractory multiple myeloma treated with TECVAYLI in MajesTEC-1 at the recommended dosage, 48% were 65 years of age or older, and 15% were 75 years of age or older. No overall differences in safety or effectiveness were observed between patients 65 to 74 years of age compared to younger patients. The study did not include a sufficient number of patients 75 years of age or older to assess whether there are differences in safety or effectiveness.
- Of the 291 patients with relapsed or refractory multiple myeloma treated with TECVAYLI at the recommended dosage in combination with daratumumab and hyaluronidase-fihj in MajesTEC-3, 51% were younger than 65 years of age, 39% were 65 to 74 years of age, and 11% were 75 years of age or older. No overall differences in effectiveness were observed between patients 65 to 74 years of age compared to younger patients. The study did not include a sufficient number of patients 75 years of age or older to assess whether there are differences in effectiveness compared to younger patients. There was a higher incidence of serious adverse reactions in patients 75 years of age or older (83%) and in patients 65 to 74 years of age (77%) as compared to patients younger than 65 years of age (63%).
5.4 Hepatotoxicity
TECVAYLI can cause hepatotoxicity, including fatalities. There was one fatal case of hepatic failure in MajesTEC-1. In patients who received TECVAYLI at the recommended dosage in the clinical trials (monotherapy and combination therapy trials; N=448), elevated aspartate aminotransferase (AST) occurred in 47% of patients, with Grade 3 or 4 elevations in 2.9%. Elevated alanine aminotransferase (ALT) occurred in 48% of patients, with Grade 3 or 4 elevations in 3.8%. Elevated total bilirubin occurred in 10% of patients with Grade 3 or 4 elevations in 0.7%. Liver enzyme elevation can occur with or without concurrent CRS.
Monitor liver enzymes and bilirubin at baseline and during TECVAYLI treatment as clinically indicated. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity [see Dosage and Administration (2.5)].
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of teclistamab-cqyv or of other teclistamab products.
Among patients treated with subcutaneous TECVAYLI at various dosages, anti-teclistamab-cqyv antibodies developed in 1/186 (0.5%) of patients who received monotherapy (up to 27 months) and in 2/379 (0.5%) of patients who received combination therapy with daratumumab and hyaluronidase-fihj (up to 43 months). Because of the low occurrence of anti-teclistamab-cqyv antibodies, the effect of these antibodies on the pharmacokinetics, pharmacodynamics, safety, and/or effectiveness of teclistamab products is unknown.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are also described elsewhere in the labeling:
- Cytokine Release Syndrome [see Warnings and Precautions (5.1)]
- Neurologic Toxicity including ICANS [see Warnings and Precautions (5.2)]
- Hepatotoxicity [see Warnings and Precautions (5.4)]
- Infections [see Warnings and Precautions (5.5)]
- Neutropenia [see Warnings and Precautions (5.6)]
- Hypersensitivity and Other Administration Reactions [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
TECVAYLI causes release of cytokines [see Clinical Pharmacology (12.2)] that may suppress activity of certain cytochrome P450 (CYP) enzymes, resulting in increased exposure of CYP substrates, which may increase the risk of adverse reactions of the CYP substrates. The highest risk of drug-drug interaction is expected to occur after initiation of TECVAYLI step-up dosing schedule up to 7 days after the first treatment dose and during and after CRS [see Warnings and Precautions (5.1)].
Monitor for toxicity and/or concentrations of CYP substrates where minimal increases in concentration may lead to serious adverse reactions. Consider decreasing the dosage of the concomitant CYP substrate as needed.
12.2 Pharmacodynamics
Serum concentrations of cytokines (IL-6, IL-10, TNF-α, and IFN-γ) and IL-2R were measured before and after administration of step-up dose 1, step-up dose 2, and the three weekly doses of 1.5 mg/kg of TECVAYLI. Increased concentrations of IL-6, IL-10, and IL-2R were observed during this period.
12.3 Pharmacokinetics
The Cmax and Cavg of teclistamab-cqyv after the first subcutaneous 1.5 mg/kg TECVAYLI dose increase proportionally over a dosage range of 0.08 mg/kg to 3 mg/kg in monotherapy (0.05 to 2 times the approved recommended treatment dosage). Following the recommended dosage of TECVAYLI monotherapy, 90% of steady state exposure was achieved after 12 weekly TECVAYLI doses (1.5 mg/kg once weekly). The mean accumulation ratio between the first and 13th weekly dose of TECVAYLI 1.5 mg/kg was 4.2-fold for Cmax, 4.1-fold for Ctrough, and 5.3-fold for Cavg.
The pharmacokinetic characteristics of teclistamab-cqyv were consistent whether used as monotherapy or in combination with daratumumab and hyaluronidase-fihj. The Cmax, Ctrough, and Cavg of teclistamab-cqyv are presented in Table 16.
| Cmax (µg/mL) | Ctrough (µg/mL) | Cavg (µg/mL) | |
|---|---|---|---|
| Cavg=average serum teclistamab-cqyv concentration over a dosing interval; Cmax=maximum serum teclistamab-cqyv concentration; Ctrough=serum teclistamab-cqyv concentration prior to next dose | |||
| Note: Data are presented as geometric mean (CV %). | |||
| TECVAYLI Monotherapy | |||
| First 1.5 mg/kg dose | 6.34 (60%) | 5.77 (63%) | 4.98 (61%) |
| Steady state of 1.5 mg/kg weekly dosing steady-state exposure for the 13th weekly dose.
|
23.8 (55%) | 21.1 (63%) | 22.8 (57%) |
| TECVAYLI in Combination with Daratumumab and Hyaluronidase-fihj | |||
| First 1.5 mg/kg dose | 6.36 (54%) | 5.79 (59%) | 4.96 (55%) |
| End of 1.5 mg/kg weekly dosing exposure for the 7th weekly dose.
|
19.6 (54%) | 17.3 (62%) | 18.6 (56%) |
| End of 3 mg/kg every two weeks dosing exposure for the 8th every two weeks dose.
|
29.9 (55%) | 19.8 (85%) | 26.2 (62%) |
| Steady state of 3 mg/kg every 4 weeks dosing steady-state exposure for the 5th Q4W dose.
|
20.2 (52%) | 5.82 (152%) | 13.7 (67%) |
Absorption
The mean bioavailability of teclistamab-cqyv was 72% after subcutaneous administration of TECVAYLI. The median (range) Tmax of teclistamab-cqyv after the first and 13th TECVAYLI monotherapy treatment doses were 139 (19 to 168) hours and 72 (24 to 168) hours, respectively.
Distribution
The mean (coefficient of variation [CV]%) volume of distribution of teclistamab-cqyv was 5.63 L (29%).
Elimination
Teclistamab-cqyv clearance decreases over time, with a mean (CV%) maximal reduction from baseline to the 13th weekly TECVAYLI monotherapy treatment dose of 40.8% (56%). The geometric mean (CV%) clearance is 0.472 L/day (64%) at the 13th weekly TECVAYLI monotherapy treatment dose. Patients who discontinue TECVAYLI after the 13th weekly monotherapy treatment dose are expected to have a 50% reduction from Cmax in teclistamab-cqyv concentration at a median (5th to 95th percentile) time of 15 days (7 to 33 days) after Tmax and a 97% reduction from Cmax in teclistamab-cqyv concentration at a median time of 69 days (32 to 163 days) after Tmax.
Specific Populations
There were no clinically significant differences in the exposure of teclistamab-cqyv based on age (24 to 84 years), sex, race (White, Black or African American), ethnicity (Hispanic/Latino, not Hispanic/Latino), mild or moderate renal impairment (estimated glomerular filtration rate [eGFR] by Modification of Diet in Renal Disease [MDRD] method: 30 to 89 mL/minute), or mild hepatic impairment (total bilirubin less than or equal to upper limit of normal [ULN] with AST greater than ULN or total bilirubin greater than 1 to 1.5 times ULN with any AST). The effects of severe renal impairment (eGFR less than 30 mL/minute) or moderate to severe hepatic impairment (total bilirubin greater than 1.5 times ULN with any AST) on the exposures of teclistamab-cqyv are unknown.
Body Weight
The volume of distribution and clearance of teclistamab-cqyv increase with increasing body weight (37.6 kg to 164 kg), supporting a weight-based dose.
Drug Interaction Studies
No clinical studies evaluating the drug interaction potential of teclistamab-cqyv have been conducted.
1 Indications and Usage (1 INDICATIONS AND USAGE)
TECVAYLI is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma
- in combination with daratumumab and hyaluronidase-fihj in patients who have received at least one prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent.
- as monotherapy, in patients who have received at least four prior lines of therapy, including a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody.
12.1 Mechanism of Action
Teclistamab-cqyv is a bispecific T-cell engaging antibody that binds to the CD3 receptor expressed on the surface of T-cells and B-cell maturation antigen (BCMA) expressed on the surface of multiple myeloma cells and some healthy B-lineage cells.
In vitro, teclistamab-cqyv activated T-cells, caused the release of various proinflammatory cytokines, and resulted in the lysis of multiple myeloma cells.
5.8 Embryo Fetal Toxicity (5.8 Embryo-Fetal Toxicity)
Based on its mechanism of action, TECVAYLI may cause fetal harm when administered to a pregnant patient. Advise pregnant patients of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with TECVAYLI and for 5 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hepatotoxicity: Can cause hepatotoxicity, including fatalities. Monitor liver enzymes and bilirubin at baseline and during treatment as clinically indicated. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity (2.5, 5.4)
- Infections: Can cause severe, life-threatening, or fatal infections. Monitor patients for signs and symptoms of infection and treat appropriately. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity. (2.5, 5.5)
- Neutropenia: Monitor complete blood cell counts at baseline and periodically during treatment. Withhold TECVAYLI based on severity. (2.5, 5.6)
- Hypersensitivity and Other Administration Reactions: Can cause systemic administration-related reactions and local injection site reactions. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity. (2.5, 5.7)
- Embryo-Fetal Toxicity: May cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception. (5.8, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For subcutaneous injection only. (2.1)
- Patients should be hospitalized for 48 hours after administration of both step-up dose 1 and step-up dose 2. Instruct patients to remain within proximity of a healthcare facility and monitored daily for 48 hours after the first treatment dose within the TECVAYLI step-up dosing schedule. (2.1)
- See Full Prescribing Information for the recommended dosage for TECVAYLI monotherapy and combination therapy. (2.2, 2.3)
- Administer pretreatment medications as recommended. (2.3)
- Refer to Tables 8, 9, 10, and 11 to determine the total dose, injection volume, and number of vials based on the patient's body weight. (2.6)
- See Full Prescribing Information for instructions on preparation and administration. (2.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection
- 30 mg/3 mL (10 mg/mL) clear to slightly opalescent, colorless to light yellow solution in a single-dose vial.
- 153 mg/1.7 mL (90 mg/mL) clear to slightly opalescent, colorless to light yellow solution in a single-dose vial.
5.3 Tecvayli and Talvey Rems (5.3 TECVAYLI and TALVEY REMS)
TECVAYLI is available only through a restricted program under a REMS called the "TECVAYLI and TALVEY REMS" because of the risks of CRS and neurologic toxicity, including ICANS [see Warnings and Precautions (5.1, 5.2)].
Notable requirements of the TECVAYLI and TALVEY REMS include the following:
- Prescribers must be certified with the REMS by enrolling and completing training.
- Prescribers must counsel patients receiving TECVAYLI about the risk of CRS and neurologic toxicity, including ICANS, and provide patients with Patient Wallet Card.
- Pharmacies and healthcare settings that dispense TECVAYLI must be certified with this REMS and must verify prescribers are certified through this REMS.
- Wholesalers and distributers must only distribute TECVAYLI to certified pharmacies or healthcare settings.
Further information about the "TECVAYLI and TALVEY REMS" is available at www.TEC-TALREMS.com or by telephone at 1-855-810-8064.
5.1 Cytokine Release Syndrome
TECVAYLI can cause cytokine release syndrome (CRS), including life-threatening or fatal reactions [see Adverse Reactions (6.1)].
In the clinical trials (monotherapy and combination therapy trials; N=448), CRS occurred in 64% of patients who received TECVAYLI at the recommended dosage, with Grade 1 CRS occurring in 46% of patients, Grade 2 in 18%, and Grade 3 in 0.2%. Recurrent CRS occurred in 27% of patients. Most patients experienced CRS during the initial step-up dosing schedule (step-up dose 1 (37%), step-up dose 2 (32%), or the initial treatment dose (20%)). CRS first occurred following subsequent doses of TECVAYLI in 2.5% of patients. The median time to onset of CRS was 2 (range: 1 to 9) days after the most recent dose and the median duration of CRS was 2 (range: 1 to 22) days.
Clinical signs and symptoms of CRS included, but were not limited to, fever, hypoxia, chills, hypotension, sinus tachycardia, headache, and elevated liver enzymes (aspartate aminotransferase and alanine aminotransferase elevation).
Initiate therapy according to TECVAYLI step-up dosing schedule to reduce risk of CRS [see Dosage and Administration (2.1, 2.5)]. Administer pretreatment medications to reduce risk of CRS and monitor patients following administration of TECVAYLI accordingly [see Dosage and Administration (2.3, 2.5)].
At the first sign of CRS, immediately evaluate the patient for hospitalization. Administer supportive care based on severity and consider further management per current practice guidelines. Withhold until CRS resolves or permanently discontinue TECVAYLI based on severity [see Dosage and Administration (2.5)].
TECVAYLI is available only through a restricted program under a REMS [see Warnings and Precautions (5.3)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Relapsed/Refractory Multiple Myeloma
In Combination with Daratumumab and Hyaluronidase-fihj
The safety of TECVAYLI in combination with daratumumab and hyaluronidase-fihj (N=283) compared with either daratumumab and hyaluronidase-fihj, pomalidomide and dexamethasone (DPd) or daratumumab and hyaluronidase-fihj, bortezomib and dexamethasone (DVd) (N=290) was evaluated in patients with relapsed or refractory multiple myeloma in MajesTEC-3 [see Clinical Studies (14.1)]. Patients received step-up doses of 0.06 mg/kg and 0.3 mg/kg of TECVAYLI followed by TECVAYLI 1.5 mg/kg once weekly, followed by TECVAYLI 3 mg/kg every two weeks, followed by TECVAYLI 3 mg/kg every four weeks, subcutaneously.
Among patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj the median exposure was 32 (range 0.03 to 43) months. Among patients who received DPd or DVd the median exposure was 16 (range 0.03 to 45) months.
Serious adverse reactions occurred in 71% of patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj. Serious adverse reactions reported in ≥3% of patients included pneumonia (33%), upper respiratory tract infection (15%), cytokine release syndrome (10%), COVID-19 (7%), sepsis (6%), second primary malignancy (5%), pyrexia (4.9%), febrile neutropenia (4.6%), and gastroenteritis (4.2%).
Fatal adverse reactions occurred in 2.5% of patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj, and included sepsis (0.7%), pneumonia (0.4%), sudden death (0.4%), myocardial infarction (0.4%), enterovirus myocarditis (0.4%) and hemophagocytic lymphohistiocytosis (0.4%).
Permanent discontinuation of TECVAYLI due to adverse reactions occurred in 6% of patients. Adverse reactions leading to discontinuation of TECVAYLI in more than one patient were pneumonia (1.1%), diarrhea (0.7%), fatigue (0.7%), second primary malignancy (0.7%), upper respiratory tract infection (0.7%) and cough (0.7%).
Dosage interruptions of TECVAYLI due to an adverse reaction occurred in 94% of patients. Adverse reactions which required dosage interruption of TECVAYLI in ≥5% of patients included neutropenia (53%), upper respiratory tract infection (48%), COVID-19 (34%), pneumonia (34%), thrombocytopenia (14%), cytokine release syndrome (13%), gastroenteritis (10%), cough (10%), pyrexia (8%), diarrhea (7%), sepsis (6%) and fatigue (5%).
The most common adverse reactions (≥20%) were hypogammaglobulinemia, upper respiratory tract infection, cytokine release syndrome, cough, diarrhea, musculoskeletal pain, COVID-19, pneumonia, injection site reaction, fatigue, pyrexia, headache, nausea, gastroenteritis, and weight decreased.
The most common Grade 3 to 4 laboratory abnormalities (≥20%) were decreased lymphocytes, decreased neutrophils, decreased white blood cells and decreased platelets.
Table 12 summarizes the adverse reactions in MajesTEC-3.
| Adverse Reaction | TECVAYLI with daratumumab and hyaluronidase-fihj (N=283) |
DPd or DVd (N=290) |
||
|---|---|---|---|---|
| Any Grade (%) |
Grade 3 or 4 (%) |
Any Grade (%) |
Grade 3 or 4 (%) |
|
| DPd = daratumumab and hyaluronidase-fihj, pomalidomide, dexamethasone; DVd = daratumumab and hyaluronidase-fihj, bortezomib, dexamethasone. | ||||
| Adverse reactions were graded according to NCI-CTCAE Version 5.0, with the exception of ICANS and CRS, which were graded by ASTCT 2019 consensus grading system; adverse reactions that were considered symptoms of CRS or ICANS were not included. | ||||
| Immune system disorders | ||||
| Hypogammaglobulinemia Hypogammaglobulinemia includes hypogammaglobulinemia, hypoglobulinemia; and/or patients with laboratory IgG levels below 400 mg/dL following treatment with TECVAYLI.
|
84 | 6 | 60 | 1.4 |
| Cytokine release syndrome | 60 | 0 | 0 | 0 |
| Infections | ||||
| Upper respiratory tract infection Upper respiratory tract infection includes bronchitis, bronchiolitis, pharyngitis, rhinitis, sinusitis, sinobronchitis, tracheitis, and tracheobronchitis and other related terms.
|
79 | 17 | 62 | 13 |
| COVID-19 Includes other related terms.
|
45 | 6 | 33 | 2.1 |
| Pneumonia Pneumonia includes atypical pneumonia, bacterial pneumonia, fungal pneumonia, viral pneumonia, and other related terms.
Includes the following fatal adverse reactions: Tec-Dara: Pneumonia (n=1); DPd/DVd: Pneumonia (n=2).
|
42 | 33 | 35 | 28 |
| Gastroenteritis | 20 | 4.9 | 8 | 0.7 |
| Urinary tract infection | 17 | 2.8 | 13 | 1 |
| Herpes virus infection | 11 | 2.1 | 6 | 0.3 |
| Respiratory, thoracic and mediastinal disorders | ||||
| Cough | 53 | 0.7 | 24 | 0 |
| Dyspnea | 13 | 1.8 | 20 | 2.1 |
| Gastrointestinal disorders | ||||
| Diarrhea | 52 | 3.9 | 31 | 2.4 |
| Nausea | 23 | 0 | 12 | 0.3 |
| Vomiting | 17 | 0 | 7 | 0 |
| Abdominal pain | 16 | 0.7 | 13 | 0 |
| Constipation | 14 | 0 | 20 | 0.3 |
| Musculoskeletal and connective tissue disorders | ||||
| Musculoskeletal pain | 50 | 1.4 | 47 | 4.5 |
| General disorders and administration site conditions | ||||
| Injection site reaction
Includes injection site reactions related to teclistamab, daratumumab and hyaluronidase-fihj, or bortezomib.
|
41 | 0 | 4.5 | 0 |
| Fatigue | 39 | 3.9 | 40 | 4.1 |
| Pyrexia | 37 | 1.4 | 19 | 0.3 |
| Edema | 13 | 0.4 | 22 | 0.3 |
| Nervous system disorders | ||||
| Headache | 26 | 1.4 | 12 | 0.3 |
| Sensory neuropathy Sensory neuropathy includes dysaesthesia, hyperaesthesia, hypoaesthesia, paraesthesia, neuralgia, peripheral neuropathy, polyneuropathy, sciatica and other related terms.
|
17 | 0.7 | 25 | 0.3 |
| Motor dysfunction Motor dysfunction includes balance disorder, dysarthria, dysphonia, gait disturbance, muscle contracture, muscle spasms, muscle spasticity, muscle twitching, muscular weakness, myopathy, peripheral motor neuropathy, tremor and other related terms.
|
14 | 0 | 30 | 1 |
| Investigations | ||||
| Weight decreased | 20 | 2.8 | 7 | 1.4 |
| Metabolism and nutrition disorders | ||||
| Decreased appetite | 19 | 1.1 | 7 | 0 |
| Cardiac disorders | ||||
| Cardiac arrhythmia | 11 | 1.1 | 8 | 2.1 |
| Neoplasms | ||||
| Second primary malignancy | 11 | 4.6 | 9 | 4.8 |
| Vascular disorders | ||||
| Hypertension | 11 | 4.6 | 6 | 2.4 |
Clinically relevant adverse reactions in <10% of patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj included sepsis, encephalopathy, CMV infection, febrile neutropenia and ICANS.
Table 13 summarizes laboratory abnormalities in MajesTEC-3.
| Laboratory Abnormality | TECVAYLI with daratumumab and hyaluronidase-fihj (N=283) |
DPd or DVd (N=290) |
||
|---|---|---|---|---|
| All Grades (%) |
Grade 3 or 4 (%) |
All Grades (%) |
Grade 3 or 4 (%) |
|
| DPd = daratumumab and hyaluronidase-fihj, pomalidomide, dexamethasone; DVd = daratumumab and hyaluronidase-fihj, bortezomib, dexamethasone. Percentages calculated with the number of subjects with a baseline and at least one post-treatment value for each lab test as denominator. Laboratory toxicity grades are derived based on NCI CTCAE Version 5.0. |
||||
| The denominator used to calculate the incidence varied from 137 to 282 in Tec-Dara and 135 to 289 in DPd/DVd based on the number of patients with a baseline value and at least one post-treatment value. | ||||
| Hematology | ||||
| Lymphocyte Count Decreased | 99 | 95 | 93 | 64 |
| White Blood Cell Decreased | 94 | 60 | 96 | 74 |
| Neutrophil Count Decreased | 91 | 78 | 94 | 84 |
| Platelet Count Decreased | 73 | 21 | 72 | 25 |
| Hemoglobin Decreased | 64 | 17 | 61 | 19 |
| Chemistry | ||||
| Alanine Aminotransferase Increased | 60 | 5 | 33 | 2.4 |
| Aspartate Aminotransferase Increased | 55 | 3.9 | 22 | 1.7 |
| Potassium Decreased | 52 | 15 | 35 | 8 |
| GGT Increased | 51 | 5 | 24 | 0 |
| Sodium Decreased | 46 | 10 | 37 | 6 |
| Lipase Increased | 49 | 18 | 21 | 4.7 |
| Serum Amylase Increased | 31 | 7 | 13 | 0 |
Monotherapy
The safety of TECVAYLI monotherapy (N=165) in patients with relapsed or refractory multiple myeloma was evaluated in MajesTEC-1 [see Clinical Studies (14.1)]. Patients received step-up doses of 0.06 mg/kg and 0.3 mg/kg of TECVAYLI followed by TECVAYLI 1.5 mg/kg, subcutaneously once weekly. Among patients who received TECVAYLI, 47% were exposed for 6 months or longer and 7% were exposed for one year or longer.
The median age of patients who received TECVAYLI was 64 years (range: 33 to 84 years); 58% were male; 81% were White, 13% were Black or African American, and 2% were Asian.
Serious adverse reactions occurred in 54% of patients who received TECVAYLI. Serious adverse reactions in >2% of patients included pneumonia (15%), cytokine release syndrome (8%), sepsis (6%), general physical health deterioration (6%), COVID-19 (6%), acute kidney injury (4.8%), pyrexia (4.8%), musculoskeletal pain (2.4%), and encephalopathy (2.4%).
Fatal adverse reactions occurred in 5% of patients who received TECVAYLI, including COVID-19 (1.8%), pneumonia (1.8%), septic shock (0.6%), acute renal failure (0.6%), and hemoperitoneum (0.6%).
Permanent discontinuation of TECVAYLI due to adverse reactions occurred in 1.2% of patients. Adverse reactions resulting in permanent discontinuation of TECVAYLI included pneumonia (adenoviral and pneumocystis jirovecii pneumonia in the same patient) and hypercalcemia.
Dosage interruptions of TECVAYLI due to an adverse reaction occurred in 73% of patients. Adverse reactions which required dosage interruption in >5% of patients included neutropenia, pneumonia, pyrexia, cytokine release syndrome, upper respiratory tract infection, and COVID-19.
The most common adverse reactions (≥20%) were pyrexia, CRS, musculoskeletal pain, injection site reaction, fatigue, upper respiratory tract infection, nausea, headache, pneumonia, and diarrhea. The most common Grade 3 to 4 laboratory abnormalities (≥20%) were decreased lymphocytes, decreased neutrophils, decreased white blood cells, decreased hemoglobin, and decreased platelets.
Table 14 summarizes the adverse reactions in MajesTEC-1.
| Adverse Reactions | TECVAYLI (N=165) |
|
|---|---|---|
| Any Grade (%) |
Grade 3 or 4 (%) |
|
| Adverse reactions were graded based on NCI-CTCAE Version 4.03, with the exception of CRS, which was graded per ASTCT 2019 criteria. | ||
| General disorders and administration site conditions | ||
| Pyrexia | 76 | 3 Only grade 3 adverse reactions occurred.
|
| Injection site reaction Includes other related terms.
|
37 | 0.6 |
| Fatigue | 33 | 2.4 |
| Chills | 16 | 0 |
| Pain | 15 | 1.8 |
| Edema | 13 | 0 |
| Immune system disorders | ||
| Cytokine release syndrome | 72 | 0.6 |
| Hypogammaglobulinemia Hypogammaglobulinemia includes hypogammaglobulinemia and hypoglobulinemia.
|
11 | 1.2 |
| Musculoskeletal and connective tissue disorders | ||
| Musculoskeletal pain | 44 | 4.2 |
| Bone pain | 16 | 3 |
| Infections | ||
| Upper respiratory tract infection Upper respiratory tract infection includes bronchitis, nasopharyngitis, pharyngitis, rhinitis, sinusitis, tracheitis and other related terms.
|
26 | 2.4 |
| Pneumonia Pneumonia includes bacterial pneumonia, viral pneumonia, and other related terms.
Includes the following fatal adverse reactions: hemorrhage (n=1), pneumonia (n=3).
|
24 | 15 |
| Urinary tract infection | 11 | 5 |
| Gastrointestinal disorders | ||
| Nausea | 25 | 0.6 |
| Diarrhea | 21 | 2.4 |
| Constipation | 18 | 0 |
| Vomiting | 12 | 0.6 |
| Nervous system disorders | ||
| Headache | 25 | 0.6 |
| Motor dysfunction Motor dysfunction includes cogwheel rigidity, dysgraphia, dysphonia, gait disturbance, hypokinesia, muscle rigidity, muscle spasms, muscular weakness, peroneal nerve palsy, psychomotor hyperactivity, tremor and VIth nerve paralysis.
|
16 | 0 |
| Sensory neuropathy Sensory neuropathy includes dysesthesia, hypoesthesia, hypoesthesia oral, neuralgia, paresthesia, paresthesia oral, peripheral sensory neuropathy, sciatica and vestibular neuronitis.
|
15 | 1.2 |
| Encephalopathy Encephalopathy includes agitation, apathy, aphasia, confusional state, delirium, depressed level of consciousness, disorientation, dyscalculia, hallucination, lethargy, memory impairment, mental status changes and somnolence.
|
13 | 0 |
| Vascular disorders | ||
| Hypotension | 18 | 1.2 |
| Hemorrhage | 12 | 1.8 |
| Hypertension | 12 | 4.8 |
| Respiratory, thoracic, and mediastinal disorders | ||
| Hypoxia | 18 | 1.8 |
| Cough | 15 | 0 |
| Cardiac disorders | ||
| Cardiac arrhythmia | 16 | 1.8 |
| Metabolism and nutrition disorders | ||
| Decreased appetite | 11 | 0.6 |
| Renal and urinary disorders | ||
| Acute kidney injury | 11 | 3.6 |
Clinically relevant adverse reactions in <10% of patients who received TECVAYLI included febrile neutropenia, sepsis, ICANS, seizure, Guillain-Barré syndrome, hepatic failure, and new onset or reactivated viral infections (including adenovirus, hepatitis B virus (HBV), cytomegalovirus (CMV), varicella zoster virus (VZV), herpes simplex virus (HSV), and progressive multifocal leukoencephalopathy (PML).
Table 15 summarizes laboratory abnormalities in MajesTEC-1.
| Laboratory Abnormality | TECVAYLI (N=165 The denominator used to calculate the rate varied from 164 to 165 based on the number of patients with a baseline value and at least one post-treatment value. ) |
|
|---|---|---|
| All Grades (%) | Grade 3 or 4 (%) | |
| Laboratory toxicity grades are derived based on NCI CTCAE Version 4.03. | ||
| Hematology | ||
| Lymphocyte count decreased | 92 | 84 |
| White blood cell decreased | 86 | 41 |
| Neutrophil count decreased | 84 | 56 |
| Platelet count decreased | 71 | 22 |
| Hemoglobin decreased | 67 | 33 |
| Chemistry | ||
| Albumin decreased | 68 | 6 |
| Alkaline phosphatase increased | 42 | 2.4 |
| Phosphorus decreased | 38 | 13 |
| Gamma-glutamyl transferase increased | 37 | 8 |
| Sodium decreased | 35 | 10 |
| Aspartate aminotransferase increased | 34 | 1.2 |
| Calcium (corrected) decreased | 31 | 1.2 |
| Creatinine increased | 30 | 3 |
2.2 Recommended Tecvayli Dosage (2.2 Recommended TECVAYLI Dosage)
In Combination with Daratumumab and Hyaluronidase-fihj
The recommended dosing schedule for TECVAYLI in combination with subcutaneous daratumumab and hyaluronidase-fihj is provided in Table 1. TECVAYLI should be administered until disease progression or unacceptable toxicity.
| Dosing schedule | Week/Day | TECVAYLI Dosage
See Table 3 for recommendations on restarting TECVAYLI after dose delays.
|
Concomitant Therapy |
|---|---|---|---|
| Day 0 | N/A | Daratumumab and hyaluronidase-fihj | |
|
Step-up dosing schedule
The Step-up dosing schedule is a component of the recommended TECVAYLI dosage but is not applicable for the daratumumab and hyaluronidase-fihj dosing.
|
Day 1 | Step-up dose 1 (0.06 mg/kg)
Step-up dose 1 must be administered 20 hours or more after the daratumumab and hyaluronidase-fihj dose.
|
N/A |
| Day 3 | Step-up dose 2 (0.3 mg/kg)
Step-up dose 2 may be given between 2 to 4 days after step-up dose 1 and if adverse reactions occur, step-up dose 2 may be given up to 7 days after step-up dose 1 to allow for resolution of adverse reactions.
|
N/A | |
| Day 7 | First treatment dose (1.5 mg/kg)
First treatment dose (1.5 mg/kg) may be given between 2 to 4 days after step-up dose 2 and if adverse reactions occur, first full treatment dose may be given up to 7 days after step-up dose 2 to allow for resolution of adverse reactions.
,
Administer TECVAYLI at least 3 hours after the daratumumab and hyaluronidase-fihj dose for the first treatment dose. For subsequent doses, administer TECVAYLI at least 15 minutes after the daratumumab and hyaluronidase-fihj dose.
|
Daratumumab and hyaluronidase-fihj | |
| Weekly dosing schedule | Weeks 2 to 8 | 1.5 mg/kg once weekly
,
Maintain a minimum of 5 days between 1.5 mg/kg once weekly doses.
|
Daratumumab and hyaluronidase-fihj once weekly |
| Biweekly (every two weeks) dosing schedule | Weeks 9 to 24 | 3 mg/kg every two weeks
,
Maintain a minimum of 12 days between 3 mg/kg every two weeks doses.
|
Daratumumab and hyaluronidase-fihj every two weeks |
| Every four weeks dosing schedule | Week 25 onwards | 3 mg/kg every four weeks
,
Maintain a minimum of 25 days between 3 mg/kg every four weeks doses.
|
Daratumumab and hyaluronidase-fihj every four weeks |
For dosage and administration instructions for daratumumab and hyaluronidase-fihj, see Clinical Studies (14.1) and refer to daratumumab and hyaluronidase-fihj monotherapy Prescribing Information.
Monotherapy
The recommended dosing schedule for TECVAYLI monotherapy is provided in Table 2. TECVAYLI should be administered until disease progression or unacceptable toxicity.
| Dosing schedule | Day | Dosage | |
|---|---|---|---|
| All Patients | |||
|
Step-up dosing schedule See Table 3 for recommendations on restarting TECVAYLI after dose delays [see Dosage and Administration (2.4)].
|
Day 1 | Step-up dose 1 | 0.06 mg/kg |
| Day 4 Step-up dose 2 may be given between 2 to 4 days after step-up dose 1 and may be given up to 7 days after step-up dose 1 to allow for resolution of adverse reactions.
|
Step-up dose 2 | 0.3 mg/kg | |
| Day 7 First treatment dose may be given between 2 to 4 days after step-up dose 2 and may be given up to 7 days after step-up dose 2 to allow for resolution of adverse reactions.
|
First treatment dose | 1.5 mg/kg | |
| Weekly dosing schedule | One week after first treatment dose and once weekly thereafter
Maintain a minimum of 5 days between 1.5 mg/kg once weekly doses
|
Subsequent treatment doses | 1.5 mg/kg once weekly |
| Patients who have achieved and maintained a complete response or better for a minimum of 6 months | |||
| Biweekly (every two weeks) dosing schedule | The dosing frequency may be decreased to 1.5 mg/kg every two weeks.
Maintain a minimum of 12 days between 1.5 mg/kg every two week doses
|
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Cytokine Release Syndrome (CRS)
Discuss the signs and symptoms associated with CRS, including fever, hypoxia, chills, hypotension, sinus tachycardia, headache, and elevated liver enzymes. Advise patients to immediately contact their healthcare provider if they experience any signs or symptoms of CRS. Advise patients that they will be hospitalized for 48 hours after administration of both step-up dose 1 and step-up dose 2. Advise patients to remain within proximity of a healthcare facility and that they will be monitored daily for 48 hours after the first treatment dose within the TECVAYLI step-up dosing schedule [see Dosage and Administration (2.5) and Warnings and Precautions (5.1)].
Neurologic Toxicity including ICANS
Discuss the signs and symptoms associated with neurologic toxicity, including ICANS, including headache, confusion, dysgraphia, decreased level of consciousness, aphasia, apraxia, somnolence, disorientation, motor dysfunction, neuropathy, or encephalopathy. Advise patients to immediately contact their healthcare provider if they experience any signs or symptoms of neurologic toxicity, including ICANS. Advise patients to refrain from driving or operating heavy or potentially dangerous machinery during and for 48 hours after completion of TECVAYLI step-up dosing schedule and in the event of new onset of any neurologic toxicity, including ICANS symptoms until neurologic toxicity resolves [see Dosage and Administration (2.5) and Warnings and Precautions (5.2)].
TECVAYLI and TALVEY REMS
TECVAYLI is available only through a restricted program called TECVAYLI and TALVEY REMS. Inform patients that they will be given a Patient Wallet Card that they should carry with them at all times and show to all of their healthcare providers. This card describes signs and symptoms of CRS and neurologic toxicity, including ICANS which, if experienced, should prompt the patient to immediately seek medical attention [see Warnings and Precautions (5.3)].
Hepatotoxicity
Advise patients that liver enzyme elevations may occur and that they should report symptoms that may indicate liver toxicity, including fatigue, anorexia, right upper abdominal discomfort, dark urine, or jaundice [see Warnings and Precautions (5.4)].
Infections
Discuss the signs and symptoms of infection [see Dosage and Administration (2.5) and Warnings and Precautions (5.5)].
Neutropenia
Discuss the signs and symptoms associated with neutropenia and febrile neutropenia [see Dosage and Administration (2.5) and Warnings and Precautions (5.6)].
Hypersensitivity and Other Administration Reactions
Advise patients to immediately seek medical attention for any signs and symptoms of systemic administration-related reactions. Advise patients that local injection-site reactions may occur and to report any severe reactions [see Warnings and Precautions (5.7)].
Embryo-Fetal Toxicity
Advise pregnant patients and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to inform their healthcare provider if they are pregnant or become pregnant. Advise females of reproductive potential to use effective contraception during treatment with TECVAYLI and for 5 months after the last dose [see Warnings and Precautions (5.8) and Use in Specific Populations (8.1, 8.3)].
Lactation
Advise women not to breastfeed during treatment with TECVAYLI and for 5 months after the last dose [see Use in Specific Populations (8.2)].
2.6 Preparation and Administration
TECVAYLI is for subcutaneous use by a healthcare provider only.
TECVAYLI should be administered by a healthcare provider with adequate medical personnel and appropriate medical equipment to manage severe reactions, including CRS and ICANS [see Warnings and Precautions (5.1, 5.2)].
TECVAYLI is a clear to slightly opalescent, colorless to light yellow solution. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use if the solution is discolored, or cloudy, or if foreign particles are present.
TECVAYLI 30 mg/3 mL (10 mg/mL) vial and TECVAYLI 153 mg/1.7 mL (90 mg/mL) vial are supplied as ready-to-use solution that do not need dilution prior to administration.
Do not combine TECVAYLI vials of different concentrations to achieve treatment dose.
Use aseptic technique to prepare and administer TECVAYLI.
Preparation of TECVAYLI
Refer to the following reference tables for the preparation of TECVAYLI.
Refer to Tables 8, 9, 10 and 11 below to determine the dosage based on predetermined weight ranges.
Use Table 8 to determine total dose, injection volume and number of vials required based on patient's actual body weight for step-up dose 1 using TECVAYLI 30 mg/3 mL (10 mg/mL) vial.
|
Patient Body Weight (kg) |
Total Dose (mg) |
Volume of Injection (mL) |
Number of Vials (1 vial=3 mL) |
|---|---|---|---|
| 35 to 39.9 | 2.2 | 0.22 | 1 |
| 40 to 44.9 | 2.5 | 0.25 | 1 |
| 45 to 49.9 | 2.8 | 0.28 | 1 |
| 50 to 59.9 | 3.3 | 0.33 | 1 |
| 60 to 69.9 | 3.9 | 0.39 | 1 |
| 70 to 79.9 | 4.5 | 0.45 | 1 |
| 80 to 89.9 | 5.1 | 0.51 | 1 |
| 90 to 99.9 | 5.7 | 0.57 | 1 |
| 100 to 109.9 | 6.3 | 0.63 | 1 |
| 110 to 119.9 | 6.9 | 0.69 | 1 |
| 120 to 129.9 | 7.5 | 0.75 | 1 |
| 130 to 139.9 | 8.1 | 0.81 | 1 |
| 140 to 149.9 | 8.7 | 0.87 | 1 |
| 150 to 160 | 9.3 | 0.93 | 1 |
Use Table 9 to determine total dose, injection volume and number of vials required based on patient's actual body weight for step-up dose 2 using TECVAYLI 30 mg/3 mL (10 mg/mL) vial.
|
Patient Body Weight (kg) |
Total Dose (mg) |
Volume of Injection (mL) |
Number of Vials (1 vial=3 mL) |
|---|---|---|---|
| 35 to 39.9 | 11 | 1.1 | 1 |
| 40 to 44.9 | 13 | 1.3 | 1 |
| 45 to 49.9 | 14 | 1.4 | 1 |
| 50 to 59.9 | 16 | 1.6 | 1 |
| 60 to 69.9 | 19 | 1.9 | 1 |
| 70 to 79.9 | 22 | 2.2 | 1 |
| 80 to 89.9 | 25 | 2.5 | 1 |
| 90 to 99.9 | 28 | 2.8 | 1 |
| 100 to 109.9 | 31 | 3.1 | 2 |
| 110 to 119.9 | 34 | 3.4 | 2 |
| 120 to 129.9 | 37 | 3.7 | 2 |
| 130 to 139.9 | 40 | 4 | 2 |
| 140 to 149.9 | 43 | 4.3 | 2 |
| 150 to 160 | 47 | 4.7 | 2 |
Use Table 10 to determine total dose, injection volume and number of vials required based on patient's actual body weight for the 1.5 mg/kg dose using TECVAYLI 153 mg/1.7 mL (90 mg/mL) vial.
|
Patient Body Weight (kg) |
Total Dose (mg) |
Volume of Injection (mL) |
Number of Vials (1 vial=1.7 mL) |
|---|---|---|---|
| 35 to 39.9 | 56 | 0.62 | 1 |
| 40 to 44.9 | 64 | 0.71 | 1 |
| 45 to 49.9 | 71 | 0.79 | 1 |
| 50 to 59.9 | 83 | 0.92 | 1 |
| 60 to 69.9 | 99 | 1.1 | 1 |
| 70 to 79.9 | 108 | 1.2 | 1 |
| 80 to 89.9 | 126 | 1.4 | 1 |
| 90 to 99.9 | 144 | 1.6 | 1 |
| 100 to 109.9 | 153 | 1.7 | 1 |
| 110 to 119.9 | 171 | 1.9 | 2 |
| 120 to 129.9 | 189 | 2.1 | 2 |
| 130 to 139.9 | 198 | 2.2 | 2 |
| 140 to 149.9 | 216 | 2.4 | 2 |
| 150 to 160 | 234 | 2.6 | 2 |
Use Table 11 to determine total dose, injection volume and number of vials required based on patient's actual body weight for the 3 mg/kg dose using TECVAYLI 153 mg/1.7 mL (90 mg/mL) vial.
|
Patient Body Weight (kg) |
Total Dose (mg) |
Volume of Injection (mL) |
Number of Vials (1 vial=1.7 mL) |
|---|---|---|---|
| 35 to 39.9 | 108 | 1.2 | 1 |
| 40 to 44.9 | 126 | 1.4 | 1 |
| 45 to 49.9 | 144 | 1.6 | 1 |
| 50 to 59.9 | 162 | 1.8 | 2 |
| 60 to 69.9 | 198 | 2.2 | 2 |
| 70 to 79.9 | 225 | 2.5 | 2 |
| 80 to 89.9 | 252 | 2.8 | 2 |
| 90 to 99.9 | 288 | 3.2 | 2 |
| 100 to 109.9 | 315 | 3.5 | 3 |
| 110 to 119.9 | 342 | 3.8 | 3 |
| 120 to 129.9 | 378 | 4.2 | 3 |
| 130 to 139.9 | 405 | 4.5 | 3 |
| 140 to 149.9 | 432 | 4.8 | 3 |
| 150 to 160 | 468 | 5.2 | 4 |
- Remove the appropriate strength TECVAYLI vial(s) from refrigerated storage [2 °C to 8 °C (36 °F to 46 °F)].
- Once removed from refrigerated storage, equilibrate TECVAYLI to ambient temperature [15 °C to 30 °C (59 °F to 86 °F)] for at least 15 minutes. Do not warm TECVAYLI in any other way.
- Gently swirl the vial for approximately 10 seconds to mix. Do not shake.
- Withdraw the required injection volume of TECVAYLI from the vial(s) into an appropriately sized syringe using a transfer needle.
- Each injection volume should not exceed 2 mL. Divide doses that require greater than 2 mL equally into multiple syringes.
- Use TECVAYLI with stainless steel injection needles and polypropylene or polycarbonate syringe material.
- Replace the transfer needle with an appropriately sized needle for injection.
Administration of TECVAYLI
Inject the required volume of TECVAYLI into the subcutaneous tissue of the abdomen (preferred injection site). Alternatively, TECVAYLI may be injected into the subcutaneous tissue at other sites (e.g., thigh). If multiple injections are needed, administer injections at least 2 cm apart.
Do not inject into tattoos or scars or areas where the skin is red, bruised, tender, hard or not intact.
Storage and Disposal
If the prepared dosing syringe(s) of TECVAYLI is not used immediately, store syringe(s) at 2 °C to 8 °C (36 °F to 46 °F) or at ambient temperature 15 °C to 30 °C (59 °F to 86 °F) for a maximum of 20 hours. Discard syringe(s) after 20 hours, if not used.
Dispose of any unused product or waste material in accordance with local requirements.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
TECVAYLI® (teclistamab-cqyv) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to light yellow solution supplied as follows:
- One 30 mg/3 mL (10 mg/mL) single-dose vial in a carton: NDC: 57894-449-01
- One 153 mg/1.7 mL (90 mg/mL) single-dose vial in a carton: NDC: 57894-450-01
2.3 Recommended Pretreatment Medications
Administer the following pretreatment medications 1 to 3 hours before each dose of the TECVAYLI step-up dosing schedule, which includes step-up dose 1, step-up dose 2, and the first treatment dose (see Tables 1 and 2), to reduce the risk of CRS [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
- Corticosteroid (oral or intravenous dexamethasone 16 mg)
- Histamine-1 (H1) receptor antagonist (oral or intravenous diphenhydramine 50 mg or equivalent)
- Antipyretics (oral or intravenous acetaminophen 650 mg to 1,000 mg)
Administration of pretreatment medications may be required prior to administration of subsequent doses of TECVAYLI in patients who:
- Repeat doses within the TECVAYLI step-up dosing schedule following a dose delay [see Dosage and Administration (2.4)].
- Experienced CRS following the prior dose of TECVAYLI [see Dosage and Administration (2.5)].
Prophylaxis for Herpes Zoster Reactivation
Prior to starting treatment with TECVAYLI, consider initiation of antiviral prophylaxis to prevent herpes zoster reactivation per guidelines.
2.4 Restarting Tecvayli After Dosage Delay (2.4 Restarting TECVAYLI after Dosage Delay)
If a dose of TECVAYLI is delayed, restart therapy based on the recommendations in Table 3 and resume the treatment schedule accordingly [see Dosage and Administration (2.2)]. Administer pretreatment medications as indicated in Table 3 [see Dosage and Administration (2.3)].
| Last dose administered | Time since the last dose administered | Action |
|---|---|---|
| Step-up dose 1 | More than 7 days | Restart TECVAYLI step-up dosing schedule at step-up dose 1 (0.06 mg/kg). Administer pretreatment medications prior to TECVAYLI dose and monitor patients accordingly [see Dosage and Administration (2.3, 2.5)].
|
| Step-up dose 2 | 8 days to 28 days | Repeat step-up dose 2 (0.3 mg/kg) and continue TECVAYLI step-up dosing schedule. |
|
More than 28 days Consider benefit-risk of restarting TECVAYLI in patients who require a dose delay of more than 28 days due to an adverse reaction.
|
Restart TECVAYLI step-up dosing schedule at step-up dose 1 (0.06 mg/kg). | |
| Weekly treatment dose | 28 days or less | Continue TECVAYLI 1.5 mg/kg once weekly. |
| 29 days to 56 days | Restart TECVAYLI step-up dosing schedule at step-up dose 2 (0.3 mg/kg). | |
| More than 56 days | Restart TECVAYLI step-up dosing schedule at step-up dose 1 (0.06 mg/kg). | |
| Any biweekly (every two weeks) or every four weeks treatment dose | 63 days or less | Continue TECVAYLI at last dose given every two weeks or every four weeks schedule. |
| 64 days to 112 days | Restart TECVAYLI step-up dosing schedule at step-up dose 2 (0.3 mg/kg). | |
| More than 112 days | Restart TECVAYLI step-up dosing schedule at step-up dose 1 (0.06 mg/kg). |
14.1 Relapsed Or Refractory Multiple Myeloma (14.1 Relapsed or Refractory Multiple Myeloma)
In Combination with Daratumumab and Hyaluronidase-fihj
The efficacy of TECVAYLI in combination with subcutaneous daratumumab and hyaluronidase-fihj compared with investigator's choice of either daratumumab and hyaluronidase-fihj, pomalidomide and dexamethasone (DPd) or daratumumab and hyaluronidase-fihj, bortezomib and dexamethasone (DVd) was evaluated in adult patients with relapsed or refractory multiple myeloma in a randomized, open-label, multi-center study (MajesTEC-3) (NCT05083169). The study included patients who had previously received one to three prior lines of therapy including a proteasome inhibitor and lenalidomide. Patients who had received only one prior line of therapy must have been refractory to lenalidomide. Patients who had disease refractory to a prior anti-CD38 monoclonal antibody therapy, or who had received any prior BCMA-directed therapy were excluded.
Patients received TECVAYLI in combination with daratumumab and hyaluronidase-fihj as follows:
- Subcutaneous TECVAYLI step-up doses of 0.06 mg/kg and 0.3 mg/kg, followed by TECVAYLI 1.5 mg/kg once weekly from Weeks 2 to 8, then 3 mg/kg every two weeks from Weeks 9 to 24, then 3 mg/kg every four weeks starting on Week 25 until disease progression or unacceptable toxicity,
AND - Subcutaneous daratumumab and hyaluronidase-fihj 1,800 mg/30,000 units (1,800 mg of daratumumab and 30,000 units of hyaluronidase) starting one day prior to TECVAYLI and continuing once weekly from Weeks 1 to 8, then every two weeks from Weeks 9 to 24, then every four weeks starting on Week 25 until disease progression or unacceptable toxicity [see Dosage and Administration (2.2)].
The major efficacy outcome measure was progression-free survival (PFS) by Independent Review Committee (IRC) assessment based on International Myeloma Working Group (IMWG) 2016 criteria.
A total of 587 patients were randomized in MajesTEC-3: 291 to the TECVAYLI in combination with daratumumab and hyaluronidase-fihj group and 296 to the control group (DPd or DVd). The median age was 64 years (range: 25 to 88) with 38% aged 65 to 74 years and 10% aged 75 years or older; 55% were male; 65% were White, 6% were Black or African American, and 22% were Asian. The International Staging System (ISS) at screening was stage I in 63%, stage II in 29%, and stage III in 8%. High-risk cytogenetics (presence of del(17p), t(4;14) or t(14;16)) were present in 35% of patients. Extramedullary disease was present in 5% of patients.
The median number of prior lines of therapy was 2 (range: 1 to 3), with 38% who received one prior line of therapy. All patients received prior lenalidomide and 99.8% received a prior proteasome inhibitor; 84% were refractory to lenalidomide. Seventy-four percent of patients previously received autologous stem cell transplantation. Five percent of patients received a prior anti-CD38 monoclonal antibody; there is limited data with TECVAYLI in combination with daratumumab and hyaluronidase-fihj in patients who have received or are refractory to prior anti-CD38 monoclonal antibody therapy.
The trial demonstrated a statistically significant improvement in the TECVAYLI in combination with daratumumab and hyaluronidase-fihj group compared with the control group in PFS and overall survival (OS). The efficacy results are shown in Table 17 and Figures 1 and 2.
| TECVAYLI with daratumumab and hyaluronidase-fihj (N=291) |
DPd or DVd (N=296) |
|
|---|---|---|
| DPd = daratumumab and hyaluronidase-fihj, pomalidomide, dexamethasone; DVd = daratumumab and hyaluronidase-fihj, bortezomib, dexamethasone; CI=confidence interval; NE=not estimable; NR=not reached; MRD = minimal residual disease. | ||
|
Progression-free survival (PFS) Based on intent-to-treat analysis set
,
Median duration of follow-up = 34.5 months
|
||
| Number of events, n (%) | 44 (15) | 187 (63) |
| Median, months (95% CI) | NR (NE, NE) | 18.1 (14.6, 22.8) |
| Hazard ratio (95% CI) Stratified Cox proportional hazard model. For all stratified analyses, stratification was based on ISS staging (I vs. II or III) and number of prior lines of therapy (1 vs. 2 or 3), as randomized ; p-valueStratified log-rank test
|
0.17 (0.12, 0.23); <0.0001 | |
| Overall survival (OS) , | ||
| Number of events, n (%) | 46 (16) | 98 (33) |
| Median, months (95% CI) | NR (NE, NE) | NR (41.4, NE) |
| Hazard ratio (95% CI); p-value | 0.46 (0.32, 0.65); <0.0001 | |
| Overall response (sCR+CR+VGPR+PR), n (%) | 259 (89.0) | 223 (75.3) |
| 95% CI (%) | (84.8, 92.4) | (70.0, 80.1) |
| p-value Stratified Cochran Mantel-Haenszel Chi-Squared test
|
<0.0001 | |
| Stringent complete response (sCR), n (%) | 225 (77.3) | 69 (23.3) |
| Complete response (CR), n (%) | 13 (4.5) | 26 (8.8) |
| Very good partial response (VGPR), n (%) | 14 (4.8) | 74 (25.0) |
| Partial response (PR), n (%) | 7 (2.4) | 54 (18.2) |
| Complete response or better (sCR+CR), n (%) | 238 (81.8) | 95 (32.1) |
| 95% CI (%) | (76.9, 86.0) | (26.8, 37.7) |
| p-value | <0.0001 | |
|
MRD negativity rate Based on MRD next-generation sequencing (NGS) primary analysis set, defined as all randomized patients enrolled at sites where next generation sequencing assay was available.
,
Based on a threshold of 10-5 using a next generation sequencing assay (clonoSEQ). , N
|
262 | 269 |
| n (%) | 153 (58.4) | 46 (17.1) |
| 95% CI (%) | (52.2, 64.4) | (12.8, 22.1) |
| p-value Fisher's exact test
|
<0.0001 | |
| MRD negativity rate in patients with CR or better | ||
| Number of patients with CR or better, n | 216 | 86 |
| MRD negativity rate, n (%) | 153 (70.8) | 46 (53.5) |
| 95% CI (%) | (64.3, 76.8) | (42.4, 64.3) |
| Figure 1: Kaplan-Meier Curve of PFS (MajesTEC-3) |
|---|
| Tec-Dara = TECVAYLI in combination with daratumumab and hyaluronidase-fihj; DPd = daratumumab and hyaluronidase-fihj, pomalidomide, dexamethasone; DVd = daratumumab and hyaluronidase-fihj, bortezomib, dexamethasone. |
| Figure 2: Kaplan-Meier Curve of OS (MajesTEC-3) |
|---|
| Tec-Dara = TECVAYLI in combination with daratumumab and hyaluronidase-fihj; DPd = daratumumab and hyaluronidase-fihj, pomalidomide, dexamethasone; DVd = daratumumab and hyaluronidase-fihj, bortezomib, dexamethasone. |
In the 15 patients in MajesTEC-3 who had received a prior anti-CD38 monoclonal antibody, the ORR was 93.3% (95% CI: 68.1, 99.8).
Monotherapy
The efficacy of TECVAYLI was evaluated in patients with relapsed or refractory multiple myeloma in a single-arm, open-label, multi-center study (MajesTEC-1, NCT03145181 [Phase 1] and NCT04557098 [Phase 2]). The study included patients who had previously received at least three prior therapies, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody. The study excluded patients who had stroke, seizure, allogeneic stem cell transplantation within the past 6 months, Eastern Cooperative Oncology Group (ECOG) performance score of 2 or higher, known active CNS involvement or clinical signs of meningeal involvement of multiple myeloma, or active or documented history of autoimmune disease, with the exception of vitiligo, Type 1 diabetes, and prior autoimmune thyroiditis.
Patients received step-up doses of 0.06 mg/kg and 0.3 mg/kg of TECVAYLI followed by TECVAYLI 1.5 mg/kg subcutaneously once weekly thereafter until disease progression or unacceptable toxicity [see Dosage and Administration (2.2)].
The efficacy population included 110 patients. The median age was 66 (range: 33 to 82) years with 16% of patients 75 years of age or older; 56% were male; 91% were White, 5% were Black or African American, 3% were Asian. The International Staging System (ISS) at study entry was Stage I in 50%, Stage II in 38%, and Stage III in 12% of patients. High-risk cytogenetics (presence of del(17p), t(4;14) or t(14;16)) were present in 25% of patients. Seventeen percent of patients had extramedullary plasmacytomas. Patients with prior BCMA-targeted therapy were not included in the efficacy population.
The median number of prior lines of therapy was 5 (range: 2 to 14); 78% of patients had received at least 4 prior lines of therapy. Eighty-one percent of patients received prior stem cell transplantation. All patients had received prior therapy with a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody, and 76% were triple-class refractory (refractory to a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody).
Efficacy was established based on overall response rate (ORR) as determined by the Independent Review Committee (IRC) assessment using International Myeloma Working Group (IMWG) 2016 criteria (see Table 18).
The median time to first response was 1.2 months (range: 0.2 to 5.5 months). With a median follow-up of 7.4 months among responders, the estimated duration of response (DOR) rate was 90.6% (95% CI: 80.3%, 95.7%) at 6 months and 66.5% (95% CI: 38.8%, 83.9%) at 9 months.
| N=110 | |
|---|---|
| NE=not estimable | |
| Overall response rate (ORR: sCR+CR+VGPR+PR) n (%) | 68 (61.8) |
| 95% CI (%) | (52.1, 70.9) |
| Complete response (CR) or better Complete response or better = Stringent complete response (sCR) + complete response (CR).
|
31 (28.2) |
| Very good partial response (VGPR) | 32 (29.1) |
| Partial response (PR) | 5 (4.5) |
| Duration of Response (DOR) (months) | |
| DOR (Months): Median (95% CI) | NE (9.0, NE) |
2.5 Dosage Modifications for Adverse Reactions
Dosage reductions of TECVAYLI are not recommended.
Refer to daratumumab and hyaluronidase-fihj Prescribing Information for information about dosage modifications for daratumumab and hyaluronidase-fihj.
Dosage delays may be required to manage toxicities related to TECVAYLI [see Warnings and Precautions (5)].
See Tables 4, 5 and 6 for recommended actions for adverse reactions of CRS, neurologic toxicity, and ICANS. See Table 7 for recommended actions for other adverse reactions following administration of TECVAYLI.
Management of CRS, Neurologic Toxicity, and ICANS
Cytokine Release Syndrome
Management recommendations for cytokine release syndrome (CRS) are summarized in Table 4.
Identify CRS based on clinical presentation [see Warnings and Precautions (5.1)]. Evaluate and treat other causes of fever, hypoxia, and hypotension.
If CRS is suspected, withhold TECVAYLI until CRS resolves. Manage according to the recommendations in Table 4 and consider further management per current practice guidelines. Administer supportive therapy for CRS, which may include intensive care for severe or life-threatening CRS. Consider laboratory testing to monitor for disseminated intravascular coagulation (DIC), hematology parameters, as well as pulmonary, cardiac, renal, and hepatic function.
| Grade Based on American Society for Transplantation and Cellular Therapy (ASTCT) 2019 grading for CRS.
|
Presenting Symptoms | Actions |
|---|---|---|
| Grade 1 | Temperature ≥100.4 °F (38 °C) Attributed to CRS. Fever may not always be present concurrently with hypotension or hypoxia as it may be masked by interventions such as antipyretics or anticytokine therapy.
|
|
| Grade 2 | Temperature ≥100.4 °F (38 °C) with: Hypotension responsive to fluids and not requiring vasopressors, and/or, Oxygen requirement of low-flow nasal cannula Low-flow nasal cannula is ≤6 L/minute, and high-flow nasal cannula is >6 L/minute. or blow-by. |
|
| Grade 3 | Temperature ≥100.4 °F (38 °C) with: Hypotension requiring one vasopressor with or without vasopressin, and/or, Oxygen requirement of high-flow nasal cannula, facemask, non-rebreather mask, or Venturi mask. |
First Occurrence of Grade 3 CRS with Duration <48 Hours:
|
Recurrent Grade 3 CRS or Grade 3 CRS with Duration 48 Hours or Longer:
|
||
| Grade 4 | Temperature ≥100.4 °F (38 °C) with: Hypotension requiring multiple vasopressors (excluding vasopressin), and/or, Oxygen requirement of positive pressure (e.g., continuous positive airway pressure (CPAP), bilevel positive airway pressure (BiPAP), intubation, and mechanical ventilation). |
|
Neurologic Toxicity and Immune Effector Cell-Associated Neurotoxicity Syndrome
Management recommendations for neurologic toxicity and immune effector cell-associated neurotoxicity syndrome (ICANS) are summarized in Tables 5 and 6.
At the first sign of neurologic toxicity, including ICANS, withhold TECVAYLI and consider neurology evaluation. Rule out other causes of neurologic symptoms. Provide supportive therapy, which may include intensive care, for severe or life-threatening neurologic toxicities, including ICANS [see Warnings and Precautions (5.2)]. Manage ICANS according to the recommendations in Table 6 and consider further management per current practice guidelines.
| Severity Based on National Cancer Institute Common Terminology Criteria for Adverse Events (NCI- CTCAE), version 4.03.
|
Actions |
|---|---|
| Grade 1 | Withhold TECVAYLI until neurologic toxicity symptoms resolve or stabilize. See Table 3 for recommendations on restarting TECVAYLI after dose delays [see Dosage and Administration (2.4)].
|
| Grade 2 Grade 3 (First occurrence) |
|
| Grade 3 (Recurrent) Grade 4 |
|
| Grade Based on American Society for Transplantation and Cellular Therapy (ASTCT) 2019 grading for ICANS.
|
Presenting Symptoms Management is determined by the most severe reaction, not attributable to any other cause.
|
Actions |
|---|---|---|
| Grade 1 | ICE score 7–9 If patient is arousable and able to perform Immune Effector Cell-Associated Encephalopathy (ICE) Assessment, assess: Orientation (oriented to year, month, city, hospital = 4 points); Naming (name 3 objects, e.g., point to clock, pen, button = 3 points); Following Commands (e.g., "show me 2 fingers" or "close your eyes and stick out your tongue" = 1 point); Writing (ability to write a standard sentence = 1 point; and Attention (count backwards from 100 by ten = 1 point). If patient is unarousable and unable to perform ICE Assessment (Grade 4 ICANS) = 0 points. , or depressed level of consciousness Not attributable to any other cause. : awakens spontaneously. |
|
| Grade 2 | ICE score 3–6, or depressed level of consciousness: awakens to voice. |
|
| Grade 3 | ICE score 0–2, or depressed level of consciousness: awakens only to tactile stimulus, or seizures, either:
|
First Occurrence of Grade 3 ICANS:
|
Recurrent Grade 3 ICANS:
|
||
| Grade 4 | ICE score 0, or depressed level of consciousness: either:
|
|
| Adverse Reactions | Severity | Actions |
|---|---|---|
| Infections Based on National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE), version 4.03.
[see Warnings and Precautions (5.5)]
|
All Grades | Withhold TECVAYLI in patients with active infection during the step-up dosing schedule. See Table 3 for recommendations on restarting TECVAYLI after dose delays [see Dosage and Administration (2.4)].
|
| Grade 3 | Withhold subsequent TECVAYLI doses administered after TECVAYLI step-up dosing schedule until infection improves to Grade 1 or less. | |
| Grade 4 |
|
|
| Hematologic Toxicities [see Warnings and Precautions (5.6) and Adverse Reactions (6.1)] | Absolute neutrophil count (ANC) < 0.5 × 109/L |
|
| Febrile neutropenia |
|
|
| Hemoglobin < 8 g/dL |
|
|
|
Monotherapy: Platelet count < 25,000/mcL or platelet count between 25,000/mcL and 50,000/mcL with bleeding Combination therapy: Platelet count < 50,000/mcL |
Monotherapy:
|
|
| Other Non-Hematologic Adverse Reactions [see Warnings and Precautions (5.4) and Adverse Reactions (6.1)] | Grade 3 | Withhold TECVAYLI until adverse reaction improves to Grade 1 or less. |
| Grade 4 |
|
8.3 Females and Males of Reproductive Potential
TECVAYLI may cause fetal harm when administered to a pregnant patient [see Use in Specific Populations (8.1)].
Pregnancy Testing
Verify pregnancy status of females of reproductive potential prior to initiating TECVAYLI.
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment and for 5 months after the last dose of TECVAYLI.
Principal Display Panel 30 Mg/3 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 30 mg/3 mL Vial Carton)
Rx only
One Vial
NDC 57894-449-01
TECVAYLI®
(teclistamab-cqyv)
Injection
30 mg/3 mL
(10 mg/mL)
TECVAYLI®vials of different
concentrations should not be
combined to achieve treatment dose.
For subcutaneous injection
by a healthcare provider only.
Attention: Dispense the enclosed
Medication Guide to each patient.
Single-dose vial
Discard unused portion.
janssen
2.1 Important Dosage and Administration Information
TECVAYLI is for subcutaneous injection only.
Administer pretreatment medications prior to each dose of the TECVAYLI step-up dosing schedule, which includes step-up dose 1, step-up dose 2, and the first treatment dose as described in Tables 1 and 2 [see Dosage and Administration (2.3)].
Administer TECVAYLI subcutaneously according to the step-up dosing schedule in Tables 1 and 2 to reduce the incidence and severity of cytokine release syndrome (CRS). Due to the risk of CRS and neurologic toxicity, including ICANS, patients should be hospitalized for 48 hours after administration of both step-up dose 1 and step-up dose 2. Instruct patients to remain within proximity of a healthcare facility and monitor them daily for 48 hours after the first treatment dose within the TECVAYLI step-up dosing schedule [see Dosage and Administration (2.5) and Warnings and Precautions (5.1, 5.2)].
Refer to Tables 8, 9, 10, and 11 to determine the dosage based on predetermined weight ranges [see Dosage and Administration (2.6)].
Principal Display Panel 153 Mg/1.7 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 153 mg/1.7 mL Vial Carton)
Rx only
One Vial
NDC 57894-450-01
TECVAYLI®
(teclistamab-cqyv)
Injection
153 mg/1.7 mL
(90 mg/mL)
TECVAYLI®vials of different
concentrations should not be
combined to achieve treatment dose.
For subcutaneous injection
by a healthcare provider only.
Attention: Dispense the enclosed
Medication Guide to each patient.
Single-dose vial
Discard unused portion.
janssen
5.7 Hypersensitivity and Other Administration Reactions
TECVAYLI can cause both systemic administration-related reactions and local injection-site reactions.
Systemic Reactions
In patients who received the recommended TECVAYLI dosage in the clinical trials (monotherapy and combination therapy trials; N=448), 2.5% of patients experienced systemic-administration reactions, which included recurrent pyrexia and rash.
Local Reactions
In patients who received TECVAYLI at the recommended dosage in the clinical trials (monotherapy and combination therapy trials; N=448), injection-site reactions occurred in 37% of patients with Grade 1 injection-site reactions in 29% and Grade 2 in 9%.
Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity [see Dosage and Administration (2.5)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity or genotoxicity studies have been conducted with teclistamab-cqyv.
No studies have been conducted to evaluate the effects of teclistamab-cqyv on fertility.
5.2 Neurologic Toxicity Including Immune Effector Cell Associated Neurotoxicity Syndrome (5.2 Neurologic Toxicity including Immune Effector Cell-Associated Neurotoxicity Syndrome)
TECVAYLI can cause serious, life-threatening or fatal neurologic toxicity, including immune effector cell-associated neurotoxicity syndrome (ICANS) [see Adverse Reactions (6.1)].
In the clinical trials (monotherapy and combination therapy trials; N=448), neurologic toxicity occurred in 60% of patients who received TECVAYLI at the recommended dosage, with Grade 3 or 4 neurologic toxicity in 6%. Neurologic toxicities reported in ≥5% of patients included headache (27%), sensory neuropathy (16%), motor dysfunction (15%), insomnia (12%), encephalopathy (11%), and dizziness (8%). Fatal neurologic toxicity occurred in 0.4% of patients, including Guillain-Barré syndrome and status epilepticus (one patient each).
In MajesTEC-1, ICANS was reported in 6% of patients who received TECVAYLI as monotherapy at the recommended dosage [see Adverse Reactions (6.1)]. Recurrent ICANS occurred in 1.8% of patients. Most patients experienced ICANS following step-up dose 1 (1.2%), step-up dose 2 (0.6%), or the initial treatment dose (1.8%). Less than 3% of patients developed first occurrence of ICANS following subsequent TECVAYLI doses. The median time to onset of ICANS was 4 days (range: 2 to 8 days) after the most recent TECVAYLI dose with a median duration of 3 days (range: 1 to 20 days). The most frequent clinical manifestations of ICANS reported were confusional state and dysgraphia.
In MajesTEC-3, ICANS was reported in 1.1% of patients who received the recommended TECVAYLI dosage in combination with daratumumab and hyaluronidase-fihj, including Grade 4 ICANS in 1 patient. All events of ICANS occurred during the step-up dosing schedule [see Adverse Reactions (6.1)]. The median time to onset of ICANS was 2 days (range: 1 to 3 days) after the most recent dose and the median duration of ICANS was 2 days (range: 1 to 2 days). The clinical manifestations of ICANS reported were amnesia, encephalopathy and delirium.
The onset of ICANS can be concurrent with CRS, following resolution of CRS, or in the absence of CRS.
Monitor patients for signs and symptoms of neurologic toxicity, including ICANS during TECVAYLI treatment. At the first sign of neurologic toxicity, including ICANS, immediately evaluate patient and provide supportive therapy based on severity. Withhold until neurologic toxicity resolves or permanently discontinue TECVAYLI based on severity per recommendations and consider further management per current practice guidelines [see Dosage and Administration (2.5)].
Due to the potential for neurologic toxicity, patients receiving TECVAYLI are at risk of depressed level of consciousness [see Adverse Reactions (6.1)]. Advise patients to refrain from driving or operating heavy or potentially dangerous machinery during and for 48 hours after completion of TECVAYLI step-up dosing schedule and in the event of new onset of any neurologic toxicity symptoms until neurologic toxicity resolves [see Dosage and Administration (2.1)].
TECVAYLI is available only through a restricted program under a REMS [see Warnings and Precautions (5.3)].
Warning: Cytokine Release Syndrome and Neurologic Toxicity, Including Immune Effector Cell Associated Neurotoxicity Syndrome (WARNING: CYTOKINE RELEASE SYNDROME and NEUROLOGIC TOXICITY, including IMMUNE EFFECTOR CELL-ASSOCIATED NEUROTOXICITY SYNDROME)
Cytokine release syndrome (CRS), including life-threatening or fatal reactions, can occur in patients receiving TECVAYLI. Initiate treatment with TECVAYLI step-up dosing schedule to reduce risk of CRS. Withhold TECVAYLI until CRS resolves or permanently discontinue based on severity [see Dosage and Administration (2.1 , 2.5) and Warnings and Precautions (5.1)].
Neurologic toxicity, including Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS) and serious, life-threatening, or fatal reactions, can occur in patients receiving TECVAYLI. Monitor patients for signs or symptoms of neurologic toxicity, including ICANS, during treatment. Withhold TECVAYLI until neurologic toxicity resolves or permanently discontinue based on severity [see Dosage and Administration (2.5) and Warnings and Precautions (5.2)].
Because of the risk of CRS and neurologic toxicity, including ICANS, TECVAYLI is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the TECVAYLI and TALVEY REMS [see Warnings and Precautions (5.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:06.209849 · Updated: 2026-03-14T22:51:41.355943