Uloric
54de10ef-fe5f-4930-b91d-6bbb04c664bd
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
ULORIC is a xanthine oxidase (XO) inhibitor indicated for the chronic management of hyperuricemia in adult patients with gout who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable.
Dosage and Administration
Recommended dosage is 40 mg or 80 mg once daily. The recommended starting dosage is 40 mg once daily. For patients who do not achieve a serum uric acid (sUA) less than 6 mg/dL after 2 weeks, the recommended dosage is 80 mg once daily. ( 2.1 ) Patients with severe renal impairment: Limit the dosage to 40 mg once daily. ( 2.2 , 8.6 ) Flare prophylaxis is recommended upon initiation of ULORIC. ( 2.4 ) Can be administered without regard to food or antacid use. ( 2.1 )
Contraindications
ULORIC is contraindicated in patients being treated with azathioprine or mercaptopurine [see Drug Interactions (7) ] .
Warnings and Precautions
Gout Flares : An increase in gout flares is frequently observed after initiation of ULORIC. If a gout flare occurs during treatment, ULORIC need not be discontinued. Prophylactic therapy (i.e., non-steroidal anti-inflammatory drug or colchicine) upon initiation of treatment may be beneficial for up to six months. ( 2.4 , 5.2 ) Hepatic Effects : Cases of hepatic failure, some fatal, have been reported. If liver injury is detected, promptly interrupt ULORIC and treat cause, if possible, to resolution or stabilization. Permanently discontinue ULORIC if liver injury is confirmed, and no alternate etiology can be found. ( 5.3 ) Serious Skin Reactions : Serious skin and hypersensitivity reactions, including Stevens-Johnson Syndrome, drug reaction with eosinophilia and systemic symptoms and toxic epidermal necrolysis have been reported in patients taking ULORIC. Discontinue ULORIC if serious skin reactions are suspected. ( 5.4 )
Adverse Reactions
The following serious adverse reactions are described elsewhere in the prescribing information: Cardiovascular Death [see Warnings and Precautions (5.1) ] Hepatic Effects [see Warnings and Precautions (5.3) ] Serious Skin Reactions [see Warnings and Precautions (5.4) ]
Drug Interactions
Concomitant administration of ULORIC with XO substrate drugs, azathioprine or mercaptopurine could increase plasma concentrations of these drugs resulting in severe toxicity. ( 7 )
How Supplied
ULORIC 40 mg tablets are light green to green in color, round, debossed with "TAP" on one side and "40" on the other side and supplied as: NDC Number Size 64764-918-11 Hospital Unit Dose Pack of 100 Tablets 64764-918-30 Bottle of 30 Tablets 64764-918-90 Bottle of 90 Tablets 64764-918-18 Bottle of 500 Tablets ULORIC 80 mg tablets are light green to green in color, teardrop shaped, debossed with "TAP" on one side and "80" on the other side and supplied as: NDC Number Size 64764-677-11 Hospital Unit Dose Pack of 100 Tablets 64764-677-30 Bottle of 30 Tablets 64764-677-13 Bottle of 100 Tablets 64764-677-19 Bottle of 1000 Tablets
Storage and Handling
ULORIC 40 mg tablets are light green to green in color, round, debossed with "TAP" on one side and "40" on the other side and supplied as: NDC Number Size 64764-918-11 Hospital Unit Dose Pack of 100 Tablets 64764-918-30 Bottle of 30 Tablets 64764-918-90 Bottle of 90 Tablets 64764-918-18 Bottle of 500 Tablets ULORIC 80 mg tablets are light green to green in color, teardrop shaped, debossed with "TAP" on one side and "80" on the other side and supplied as: NDC Number Size 64764-677-11 Hospital Unit Dose Pack of 100 Tablets 64764-677-30 Bottle of 30 Tablets 64764-677-13 Bottle of 100 Tablets 64764-677-19 Bottle of 1000 Tablets
Description
Gout patients with established cardiovascular (CV) disease treated with ULORIC had a higher rate of CV death compared to those treated with allopurinol in a CV outcomes study [see Warnings and Precautions (5.1) ] . Consider the risks and benefits of ULORIC when deciding to prescribe or continue patients on ULORIC. ULORIC should only be used in patients who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable [see Indications and Usage (1) ] .
Medication Information
Warnings and Precautions
Gout Flares : An increase in gout flares is frequently observed after initiation of ULORIC. If a gout flare occurs during treatment, ULORIC need not be discontinued. Prophylactic therapy (i.e., non-steroidal anti-inflammatory drug or colchicine) upon initiation of treatment may be beneficial for up to six months. ( 2.4 , 5.2 ) Hepatic Effects : Cases of hepatic failure, some fatal, have been reported. If liver injury is detected, promptly interrupt ULORIC and treat cause, if possible, to resolution or stabilization. Permanently discontinue ULORIC if liver injury is confirmed, and no alternate etiology can be found. ( 5.3 ) Serious Skin Reactions : Serious skin and hypersensitivity reactions, including Stevens-Johnson Syndrome, drug reaction with eosinophilia and systemic symptoms and toxic epidermal necrolysis have been reported in patients taking ULORIC. Discontinue ULORIC if serious skin reactions are suspected. ( 5.4 )
Indications and Usage
ULORIC is a xanthine oxidase (XO) inhibitor indicated for the chronic management of hyperuricemia in adult patients with gout who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable.
Dosage and Administration
Recommended dosage is 40 mg or 80 mg once daily. The recommended starting dosage is 40 mg once daily. For patients who do not achieve a serum uric acid (sUA) less than 6 mg/dL after 2 weeks, the recommended dosage is 80 mg once daily. ( 2.1 ) Patients with severe renal impairment: Limit the dosage to 40 mg once daily. ( 2.2 , 8.6 ) Flare prophylaxis is recommended upon initiation of ULORIC. ( 2.4 ) Can be administered without regard to food or antacid use. ( 2.1 )
Contraindications
ULORIC is contraindicated in patients being treated with azathioprine or mercaptopurine [see Drug Interactions (7) ] .
Adverse Reactions
The following serious adverse reactions are described elsewhere in the prescribing information: Cardiovascular Death [see Warnings and Precautions (5.1) ] Hepatic Effects [see Warnings and Precautions (5.3) ] Serious Skin Reactions [see Warnings and Precautions (5.4) ]
Drug Interactions
Concomitant administration of ULORIC with XO substrate drugs, azathioprine or mercaptopurine could increase plasma concentrations of these drugs resulting in severe toxicity. ( 7 )
Storage and Handling
ULORIC 40 mg tablets are light green to green in color, round, debossed with "TAP" on one side and "40" on the other side and supplied as: NDC Number Size 64764-918-11 Hospital Unit Dose Pack of 100 Tablets 64764-918-30 Bottle of 30 Tablets 64764-918-90 Bottle of 90 Tablets 64764-918-18 Bottle of 500 Tablets ULORIC 80 mg tablets are light green to green in color, teardrop shaped, debossed with "TAP" on one side and "80" on the other side and supplied as: NDC Number Size 64764-677-11 Hospital Unit Dose Pack of 100 Tablets 64764-677-30 Bottle of 30 Tablets 64764-677-13 Bottle of 100 Tablets 64764-677-19 Bottle of 1000 Tablets
How Supplied
ULORIC 40 mg tablets are light green to green in color, round, debossed with "TAP" on one side and "40" on the other side and supplied as: NDC Number Size 64764-918-11 Hospital Unit Dose Pack of 100 Tablets 64764-918-30 Bottle of 30 Tablets 64764-918-90 Bottle of 90 Tablets 64764-918-18 Bottle of 500 Tablets ULORIC 80 mg tablets are light green to green in color, teardrop shaped, debossed with "TAP" on one side and "80" on the other side and supplied as: NDC Number Size 64764-677-11 Hospital Unit Dose Pack of 100 Tablets 64764-677-30 Bottle of 30 Tablets 64764-677-13 Bottle of 100 Tablets 64764-677-19 Bottle of 1000 Tablets
Description
Gout patients with established cardiovascular (CV) disease treated with ULORIC had a higher rate of CV death compared to those treated with allopurinol in a CV outcomes study [see Warnings and Precautions (5.1) ] . Consider the risks and benefits of ULORIC when deciding to prescribe or continue patients on ULORIC. ULORIC should only be used in patients who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable [see Indications and Usage (1) ] .
Section 42229-5
Limitations of Use:
ULORIC is not recommended for the treatment of asymptomatic hyperuricemia.
Section 42230-3
| This Medication Guide has been approved by the U.S. Food and Drug Administration | ULR015 R10 | Revised: April 2023 |
|
MEDICATION GUIDE ULORIC (Ū–'lor–ik) (febuxostat) tablets, for oral use |
||
| Read the Medication Guide that comes with ULORIC before you start taking it and each time you get a refill. There may be new information. The Medication Guide does not take the place of talking with your doctor about your medical condition or your treatment. | ||
|
What is the most important information that I should know about ULORIC? ULORIC may cause serious side effects, including: Heart-related deaths. Call your doctor or get emergency medical help right away if you have any of the following symptoms, especially if they are new, worse, or worry you: |
||
|
|
|
|
What is ULORIC?
ULORIC is a prescription medicine called a xanthine oxidase (XO) inhibitor used to lower blood uric acid levels in adult patients with gout when allopurinol has not worked well enough or when allopurinol is not right for you. ULORIC is not for use in people who do not have symptoms of high blood uric acid levels. It is not known if ULORIC is safe and effective in children. |
||
|
Who should not take ULORIC? Do not take ULORIC if you:
|
||
|
What should I tell my doctor before taking ULORIC? Before taking ULORIC tell your doctor about all of your medical conditions, including if you:
Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine. |
||
How should I take ULORIC?
|
||
|
What are the possible side effects of ULORIC? ULORIC may cause serious side effects, including:
|
||
|
|
|
|
||
|
|
|
| The most common side effects of ULORIC include: | ||
|
|
|
| These are not all of the possible side effects of ULORIC. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store ULORIC?
|
||
|
General information about the safe and effective use of ULORIC.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ULORIC for a condition for which it was not prescribed. Do not give ULORIC to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about ULORIC that is written for health professionals. |
||
|
What are the ingredients in ULORIC? Active ingredient: febuxostat Inactive ingredients: lactose monohydrate, microcrystalline cellulose, hydroxypropyl cellulose, sodium croscarmellose, silicon dioxide, magnesium stearate, and Opadry II, green Distributed by: Takeda Pharmaceuticals America, Inc. Lexington, MA 02421 ULORIC is a registered trademark of Teijin Limited registered in the U.S. Patent and Trademark Office and used under license by Takeda Pharmaceuticals America, Inc. All other trademarks are the property of their respective owners. ©2009-2023 Takeda Pharmaceuticals America, Inc. For more information, go to www.ULORIC.com or call 1-877-TAKEDA (1-877-825-3327). |
541887-04
Section 44425-7
Protect from light. Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
7.3 in Vivo
Based on drug interaction studies in healthy patients, ULORIC does not have clinically significant interactions with colchicine, naproxen, indomethacin, hydrochlorothiazide, warfarin or desipramine [see Clinical Pharmacology (12.3)]. Therefore, ULORIC may be used concomitantly with these medications.
10 Overdosage
ULORIC was studied in healthy patients in doses up to 300 mg daily for seven days without evidence of dose-limiting toxicities. No overdose of ULORIC was reported in clinical studies. Patients should be managed by symptomatic and supportive care should there be an overdose.
11 Description
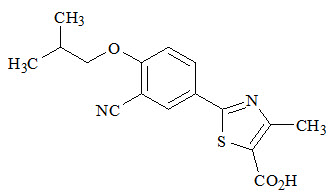
ULORIC (febuxostat) is a xanthine oxidase inhibitor. The active ingredient in ULORIC is 2-[3-cyano-4-(2-methylpropoxy) phenyl]-4-methylthiazole-5-carboxylic acid, with a molecular weight of 316.38. The empirical formula is C16H16N2O3S.
The chemical structure is:
Febuxostat is a non-hygroscopic, white crystalline powder that is freely soluble in dimethylformamide; soluble in dimethylsulfoxide; sparingly soluble in ethanol; slightly soluble in methanol and acetonitrile; and practically insoluble in water. The melting range is 205°C to 208°C.
ULORIC tablets for oral use contain the active ingredient, febuxostat, and are available in two dosage strengths, 40 mg and 80 mg. Inactive ingredients include hydroxypropyl cellulose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, silicon dioxide, and sodium croscarmellose. ULORIC tablets are coated with Opadry II, green.
5.2 Gout Flares
After initiation of ULORIC, an increase in gout flares is frequently observed. This increase is due to reduction in serum uric acid levels, resulting in mobilization of urate from tissue deposits.
In order to prevent gout flares when ULORIC is initiated, concurrent prophylactic treatment with an NSAID or colchicine is recommended [see Dosage and Administration (2.4)].
8.4 Pediatric Use
Safety and effectiveness of ULORIC in pediatric patients have not been established.
8.5 Geriatric Use
No dose adjustment is necessary in elderly patients. Of the total number of patients in Studies 1, 2, and 3 (clinical studies of ULORIC in the treatment of gout) [see Clinical Studies (14.1)], 16% were 65 and over, while 4% were 75 and over. Comparing patients in different age groups, no clinically significant differences in safety or effectiveness were observed but greater sensitivity of some older individuals cannot be ruled out. The Cmax and AUC24 of febuxostat following multiple oral doses of ULORIC in geriatric patients (≥65 years) were similar to those in younger patients (18 to 40 years) [see Clinical Pharmacology (12.3)].
14 Clinical Studies
A serum uric acid level of less than 6 mg/dL is the goal of antihyperuricemic therapy and has been established as appropriate for the treatment of gout.
4 Contraindications
ULORIC is contraindicated in patients being treated with azathioprine or mercaptopurine [see Drug Interactions (7)].
5.3 Hepatic Effects
Cases of fatal and nonfatal hepatic failure in patients taking ULORIC have been reported. During randomized controlled studies, transaminase elevations greater than three times the upper limit of normal (ULN) were observed (AST: 2%, 2%, and ALT: 3%, 2% in ULORIC and allopurinol-treated patients, respectively). No dose-effect relationship for these transaminase elevations was noted [see Clinical Pharmacology (12.3)].
Obtain a liver test panel (serum alanine aminotransferase [ALT], aspartate aminotransferase [AST], alkaline phosphatase, and total bilirubin) as a baseline before initiating ULORIC.
Measure liver tests promptly in patients who report symptoms that may indicate liver injury, including fatigue, anorexia, right upper abdominal discomfort, dark urine or jaundice. In this clinical context, if the patient presents abnormal liver tests (ALT or AST greater than three times the upper limit of the reference range), interrupt ULORIC treatment while investigating the probable cause. Permanently discontinue ULORIC if liver injury is confirmed, and no alternate etiology can be found.
Permanently discontinue ULORIC in patients who have serum ALT or AST greater than three times the reference range with serum total bilirubin greater than two times the reference range without alternative etiologies because they are at risk for severe drug-induced liver injury. For patients with lesser elevations of serum ALT or bilirubin and with an alternate probable cause, treatment with ULORIC can be used with close monitoring.
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the prescribing information:
- Cardiovascular Death [see Warnings and Precautions (5.1)]
- Hepatic Effects [see Warnings and Precautions (5.3)]
- Serious Skin Reactions [see Warnings and Precautions (5.4)]
7 Drug Interactions
Concomitant administration of ULORIC with XO substrate drugs, azathioprine or mercaptopurine could increase plasma concentrations of these drugs resulting in severe toxicity. (7)
8.6 Renal Impairment
No dose adjustment is necessary in patients with mild to moderate renal impairment (Clcr 30 to 89 mL/min). For patients with severe renal impairment (Clcr 15 to 29 mL/min), the recommended dosage of ULORIC is limited to 40 mg once daily [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
In healthy patients, maximum plasma concentrations (Cmax) and AUC of febuxostat increased in a dose proportional manner following single and multiple doses of 10 mg (0.25 times the lowest recommended dosage) to 120 mg (1.5 times the maximum recommended dosage). There is no accumulation when therapeutic doses are administered every 24 hours. Febuxostat has an apparent mean terminal elimination half-life (t1/2) of approximately 5 to 8 hours. Febuxostat pharmacokinetic parameters for patients with hyperuricemia and gout estimated by population pharmacokinetic analyses were similar to those estimated in healthy patients.
13.2 Animal Toxicology
A 12-month toxicity study in beagle dogs showed deposition of xanthine crystals and calculi in kidneys at 15 mg/kg (approximately 4 times the MRHD on an AUC basis). A similar effect of calculus formation was noted in rats in a 6-month study due to deposition of xanthine crystals at 48 mg/kg (approximately 31 and 40 times the MRHD on an AUC basis in males and females respectively).
2.1 Recommended Dosage
The recommended ULORIC dosage is 40 mg or 80 mg once daily.
The recommended starting dosage of ULORIC is 40 mg once daily. For patients who do not achieve a serum uric acid (sUA) less than 6 mg/dL after two weeks, the recommended ULORIC dosage is 80 mg once daily.
ULORIC can be taken without regard to food or antacid use [see Clinical Pharmacology (12.3)].
Concurrent prophylactic treatment with a non-steroidal anti-inflammatory drug (NSAID) or colchicine is recommended [see Dosage and Administration (2.4) and Warnings and Precautions (5.2)].
8.7 Hepatic Impairment
No dose adjustment is necessary in patients with mild or moderate hepatic impairment (Child-Pugh Class A or B). No studies have been conducted in patients with severe hepatic impairment (Child-Pugh Class C); therefore, caution should be exercised in these patients [see Clinical Pharmacology (12.3)].
1 Indications and Usage
ULORIC is a xanthine oxidase (XO) inhibitor indicated for the chronic management of hyperuricemia in adult patients with gout who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable.
12.1 Mechanism of Action
ULORIC, a xanthine oxidase inhibitor, achieves its therapeutic effect by decreasing serum uric acid. ULORIC is not expected to inhibit other enzymes involved in purine and pyrimidine synthesis and metabolism at therapeutic concentrations.
5.1 Cardiovascular Death
In a cardiovascular (CV) outcome study, gout patients with established CV disease treated with ULORIC had a higher rate of CV death compared to those treated with allopurinol. Sudden cardiac death was the most common cause of adjudicated CV deaths, 2.7% in the ULORIC group (83 of 3,098) as compared to 1.8% in the allopurinol group (56 of 3,092). ULORIC was similar to allopurinol for nonfatal myocardial infarction (MI), nonfatal stroke and unstable angina with urgent coronary revascularization [see Clinical Studies (14.2)].
Because of the increased risk of CV death, ULORIC should only be used in patients who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable [see Indications and Usage(1)].
Consider the risks and benefits of ULORIC when deciding to prescribe or continue patients on ULORIC. Consider use of prophylactic low-dose aspirin therapy in patients with a history of CV disease. Monitor patients for the development of CV events. Inform patients about the symptoms of serious CV events and the steps to take if they occur.
5 Warnings and Precautions
- Gout Flares: An increase in gout flares is frequently observed after initiation of ULORIC. If a gout flare occurs during treatment, ULORIC need not be discontinued. Prophylactic therapy (i.e., non-steroidal anti-inflammatory drug or colchicine) upon initiation of treatment may be beneficial for up to six months. (2.4, 5.2)
- Hepatic Effects: Cases of hepatic failure, some fatal, have been reported. If liver injury is detected, promptly interrupt ULORIC and treat cause, if possible, to resolution or stabilization. Permanently discontinue ULORIC if liver injury is confirmed, and no alternate etiology can be found. (5.3)
- Serious Skin Reactions: Serious skin and hypersensitivity reactions, including Stevens-Johnson Syndrome, drug reaction with eosinophilia and systemic symptoms and toxic epidermal necrolysis have been reported in patients taking ULORIC. Discontinue ULORIC if serious skin reactions are suspected. (5.4)
5.4 Serious Skin Reactions
Serious skin and hypersensitivity reactions, including Stevens-Johnson Syndrome, drug reaction with eosinophilia and systemic symptoms (DRESS) and toxic epidermal necrolysis (TEN) have been reported postmarketing in patients taking ULORIC. Discontinue ULORIC if serious skin reactions are suspected [see Patient Counseling Information (17)]. Many of these patients had reported previous similar skin reactions to allopurinol. ULORIC should be used with close monitoring in these patients.
2 Dosage and Administration
- Recommended dosage is 40 mg or 80 mg once daily. The recommended starting dosage is 40 mg once daily. For patients who do not achieve a serum uric acid (sUA) less than 6 mg/dL after 2 weeks, the recommended dosage is 80 mg once daily. (2.1)
- Patients with severe renal impairment: Limit the dosage to 40 mg once daily. (2.2, 8.6)
- Flare prophylaxis is recommended upon initiation of ULORIC. (2.4)
- Can be administered without regard to food or antacid use. (2.1)
8.8 Secondary Hyperuricemia
No studies have been conducted in patients with secondary hyperuricemia (including organ transplant recipients); ULORIC is not recommended for use in patients whom the rate of urate formation is greatly increased (e.g., malignant disease and its treatment, Lesch-Nyhan syndrome). The concentration of xanthine in urine could, in rare cases, rise sufficiently to allow deposition in the urinary tract.
3 Dosage Forms and Strengths
- 40 mg tablets, light green to green, round, debossed with "TAP" and "40"
- 80 mg tablets, light green to green, teardrop shaped, debossed with "TAP" and "80"
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of ULORIC. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: agranulocytosis, eosinophilia.
Hepatobiliary Disorders: hepatic failure (some fatal), jaundice, serious cases of abnormal liver function test results, liver disorder.
Immune System Disorders: anaphylaxis, anaphylactic reaction.
Musculoskeletal and Connective Tissue Disorders: rhabdomyolysis.
Psychiatric Disorders: psychotic behavior including aggressive thoughts.
Renal and Urinary Disorders: tubulointerstitial nephritis.
Skin and Subcutaneous Tissue Disorders: generalized rash, Stevens-Johnson Syndrome, hypersensitivity skin reactions, erythema multiforme, drug reaction with eosinophilia and systemic symptoms, toxic epidermal necrolysis.
8 Use in Specific Populations
- Patients with severe hepatic impairment: No studies have been conducted in this patient population. Caution should be exercised in these patients. (8.7)
- Patients with secondary hyperuricemia (including patients being treated for Lesch-Nyhan syndrome or malignant disease, or in organ transplant recipients): ULORIC is not recommended for use as no studies have been conducted in this patient population. (8.8)
Warning: Cardiovascular Death
Gout patients with established cardiovascular (CV) disease treated with ULORIC had a higher rate of CV death compared to those treated with allopurinol in a CV outcomes study [see Warnings and Precautions (5.1)].
Consider the risks and benefits of ULORIC when deciding to prescribe or continue patients on ULORIC. ULORIC should only be used in patients who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable [see Indications and Usage (1)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In Phase 2 and 3 clinical studies, a total of 2757 patients with hyperuricemia and gout were treated with ULORIC 40 mg or 80 mg daily. For ULORIC 40 mg, 559 patients were treated for ≥6 months. For ULORIC 80 mg, 1377 patients were treated for ≥6 months, 674 patients were treated for ≥1 year and 515 patients were treated for ≥2 years. In the CARES study, a total of 3098 patients were treated with ULORIC 40 mg or 80 mg daily; of these, 2155 patients were treated for ≥1 year and 1539 were treated for ≥2 years [see Clinical Studies (14.2)].
14.2 Cardiovascular Safety Study
A randomized, double-blind, allopurinol-controlled CV outcomes study (CARES) was conducted to evaluate the CV risk of ULORIC (NCT01101035). The study compared the risk of MACE between patients treated with ULORIC (N=3098) and allopurinol-treated patients (N=3092). The primary endpoint was the time to first occurrence of a MACE defined as the composite of CV death, nonfatal MI, nonfatal stroke, or unstable angina with urgent coronary revascularization. The study was designed to exclude a prespecified risk margin of 1.3 for the hazard ratio of MACE. An independent committee conducted a blinded evaluation of serious CV adverse events according to predefined criteria (adjudication) for determination of MACE. The study was event driven and patients were followed until a sufficient number of primary outcome events accrued. The median on-study follow-up time was 2.6 years.
Patients randomized to ULORIC initially received 40 mg once daily which was increased to 80 mg once daily, if their sUA was ≥6mg/dL at Week 2. For patients randomized to allopurinol, those who had normal renal function or mild renal impairment (estimated creatinine clearance (eClcr) ≥60 to ˂90 mL/minute) initially received 300 mg once daily with 100 mg/day dose increments monthly until either sUA ˂6mg/dL or an allopurinol dosage of 600 mg once daily was achieved; those who had moderate renal impairment (eClcr ≥30 to ˂60 mL/minute) initially received 200 mg once daily with 100 mg/day dose increments monthly until either a sUA ˂6 mg/dL or an allopurinol dosage of 400 mg once daily was achieved.
The mean age of the population was 65 years (range: 44 to 93 years). Most patients were male (84%) and Caucasian (69%). Patients had a diagnosis of gout for approximately 12 years, a mean baseline sUA of 8.7 mg/dL, and 90% had experienced at least one gout flare in the past year. CV history included MI (39%), hospitalization for unstable angina (28%), cardiac revascularization (37%), and stroke (14%). The most prevalent comorbid conditions were hypertension (92%), hyperlipidemia (87%), diabetes mellitus (55%), diabetes mellitus with micro- or macrovascular disease (39%), and renal impairment [92% with an eClcr 30 to 89 mL/minute]. The use of CV disease medication was balanced across treatment groups. Baseline CV disease medications included: ACE inhibitors or ARBs (70%), lipid modifying agents (74%), aspirin (62%), beta-blockers (59%), calcium channel blockers (26%), and nonaspirin antiplatelet medications (31%).
Table 5 shows the study results for the primary MACE composite endpoint and its individual components. For the composite primary endpoint, the ULORIC group was non-inferior compared with the allopurinol group. The rates of nonfatal MI, stroke, and unstable angina with urgent coronary revascularization were similar. There was a higher rate of CV deaths in patients treated with ULORIC (134 CV deaths; 1.5 per 100 PY) than in allopurinol-treated patients (100 CV deaths; 1.1 per 100 PY). Sudden cardiac death was the most common cause of adjudicated CV deaths in the ULORIC group (83 of 3,098; 2.7%) as compared to the allopurinol group (56 of 3,092; 1.8%). The biological plausibility of CV death associated with ULORIC is unclear.
All-cause mortality was higher in the ULORIC group (243 deaths [7.8%]; 2.6 per 100 PY) than the allopurinol group (199 deaths [6.4%]; 2.2 per 100 PY) [Hazard Ratio: 1.22, 95% CI: 1.01, 1.47], due to a higher rate of CV deaths.
| ULORIC N=3098 |
Allopurinol N=3092 |
Hazard Ratio | |||
|---|---|---|---|---|---|
| Number of Patients with Event (%) | Rate per 100 PY Patient Years (PY)
|
Number of Patients with Event (%) | Rate per 100 PY | 95% CI | |
| Composite of primary endpoint MACE | 335 (10.8) | 3.8 | 321 (10.4) | 3.7 | 1.03 (0.89, 1.21) |
| Cardiovascular Death | 134 (4.3) | 1.5 | 100 (3.2) | 1.1 | 1.34 (1.03, 1.73) |
| Nonfatal MI | 111 (3.6) | 1.2 | 118 (3.8) | 1.3 | 0.93 (0.72, 1.21) |
| Nonfatal stroke | 71 (2.3) | 0.8 | 70 (2.3) | 0.8 | 1.01 (0.73, 1.41) |
| Unstable angina with urgent coronary revascularization | 49 (1.6) | 0.5 | 56 (1.8) | 0.6 | 0.86 (0.59, 1.26) |
7.2 Cytotoxic Chemotherapy Drugs
Drug interaction studies of ULORIC with cytotoxic chemotherapy have not been conducted. No data are available regarding the safety of ULORIC during cytotoxic chemotherapy.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
ULORIC 40 mg tablets are light green to green in color, round, debossed with "TAP" on one side and "40" on the other side and supplied as:
| NDC Number | Size |
|---|---|
| 64764-918-11 | Hospital Unit Dose Pack of 100 Tablets |
| 64764-918-30 | Bottle of 30 Tablets |
| 64764-918-90 | Bottle of 90 Tablets |
| 64764-918-18 | Bottle of 500 Tablets |
ULORIC 80 mg tablets are light green to green in color, teardrop shaped, debossed with "TAP" on one side and "80" on the other side and supplied as:
| NDC Number | Size |
|---|---|
| 64764-677-11 | Hospital Unit Dose Pack of 100 Tablets |
| 64764-677-30 | Bottle of 30 Tablets |
| 64764-677-13 | Bottle of 100 Tablets |
| 64764-677-19 | Bottle of 1000 Tablets |
2.3 Serum Uric Acid Level Monitoring
Testing for the target serum uric acid level of less than 6 mg/dL may be performed as early as two weeks after initiating ULORIC therapy.
7.1 Xanthine Oxidase Substrate Drugs
ULORIC is an XO inhibitor. Based on a drug interaction study in healthy patients, febuxostat altered the metabolism of theophylline (a substrate of XO) in humans [see Clinical Pharmacology (12.3)]. Therefore, use with caution when coadministering ULORIC with theophylline.
A drug interaction study of ULORIC and azathioprine, also metabolized by XO, showed an increase in exposure of 6-mercaptopurine which may lead to toxicity [see Clinical Pharmacology (12.3)]. Drug interaction studies of ULORIC with other drugs that are metabolized by XO (e.g., mercaptopurine) have not been conducted. ULORIC is contraindicated in patients being treated with azathioprine or mercaptopurine [see Contraindications (4)].
14.1 Management of Hyperuricemia in Gout
The efficacy of ULORIC was demonstrated in three randomized, double-blind, controlled trials in patients with hyperuricemia and gout. Hyperuricemia was defined as a baseline serum uric acid level ≥8 mg/dL.
Study 1 (NCT00430248) randomized patients to: ULORIC 40 mg daily, ULORIC 80 mg daily, or allopurinol (300 mg daily for patients with estimated creatinine clearance (Clcr) ≥60 mL/min or 200 mg daily for patients with estimated Clcr ≥30 mL/min and ≤59 mL/min). The duration of Study 1 was six months.
Study 2 (NCT00174915) randomized patients to: placebo, ULORIC 80 mg daily, ULORIC 120 mg daily, ULORIC 240 mg daily or allopurinol (300 mg daily for patients with a baseline serum creatinine ≤1.5 mg/dL or 100 mg daily for patients with a baseline serum creatinine greater than 1.5 mg/dL and ≤2 mg/dL). The duration of Study 2 was six months.
Study 3 (NCT00102440), a year study, randomized patients to: ULORIC 80 mg daily, ULORIC 120 mg daily, or allopurinol 300 mg daily. Patients who completed Study 2 and Study 3 were eligible to enroll in a Phase 3 long-term extension study in which patients received treatment with ULORIC for over three years.
In all three studies, patients received naproxen 250 mg twice daily or colchicine 0.6 mg once or twice daily for gout flare prophylaxis. In Study 1 the duration of prophylaxis was six months; in Study 2 and Study 3 the duration of prophylaxis was eight weeks.
The efficacy of ULORIC was also evaluated in a 4-week dose ranging study which randomized patients to: placebo, ULORIC 40 mg daily, ULORIC 80 mg daily, or ULORIC 120 mg daily. Patients who completed this study were eligible to enroll in a long-term extension study in which patients received treatment with ULORIC for up to five years.
Patients in these studies were representative of the patient population for which ULORIC use is intended. Table 2 summarizes the demographics and baseline characteristics for the patients enrolled in the studies.
| Male | 95% | |
| Race: | Caucasian | 80% |
| African American | 10% | |
| Ethnicity: Hispanic or Latino | 7% | |
| Alcohol User | 67% | |
| Mild to Moderate Renal Insufficiency (percent with estimated Clcr less than 90 mL/min) |
59% | |
| History of Hypertension | 49% | |
| History of Hyperlipidemia | 38% | |
| BMI ≥30 kg/m2 | 63% | |
| Mean BMI | 33 kg/m2 | |
| Baseline sUA ≥10 mg/dL | 36% | |
| Mean baseline sUA | 9.7 mg/dL | |
| Experienced a gout flare in previous year | 85% |
2.4 Recommended Prophylaxis for Gout Flares
Gout flares may occur after initiation of ULORIC due to changing serum uric acid levels resulting in mobilization of urate from tissue deposits. Flare prophylaxis with a non-steroidal anti-inflammatory drug (NSAID) or colchicine is recommended upon initiation of ULORIC. Prophylactic therapy may be beneficial for up to six months [see Clinical Studies (14.1)].
If a gout flare occurs during ULORIC treatment, ULORIC need not be discontinued. The gout flare should be managed concurrently, as appropriate for the individual patient [see Warnings and Precautions (5.2)].
Principal Display Panel 40 Mg Tablet Bottle Label
NDC 64764-918-30
30 Tablets
Uloric
(febuxostat)
tablets
40 mg
Dispense the accompanying
Medication Guide to each patient.
Principal Display Panel 80 Mg Tablet Bottle Label
NDC 64764-677-30
30 Tablets
Uloric
(febuxostat)
tablets
80 mg
Dispense the accompanying
Medication Guide to each patient.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year carcinogenicity studies were conducted in F344 rats and B6C3F1 mice. Increased transitional cell papilloma and carcinoma of the urinary bladder was observed at 24 mg/kg (25 times the MRHD on an AUC basis and 18.75 mg/kg (12.5 times the MRHD on an AUC basis) in male rats and female mice, respectively. The urinary bladder neoplasms were secondary to calculus formation in the kidney and urinary bladder.
Febuxostat showed a positive clastogenic response in a chromosomal aberration assay in a Chinese hamster lung fibroblast cell line with and without metabolic activation in vitro. Febuxostat was negative in the following genotoxicity assays: the in vitro Ames assay, in vitro chromosomal aberration assay in human peripheral lymphocytes, the L5178Y mouse lymphoma cell line assay, the in vivo mouse micronucleus assay, and the rat unscheduled DNA synthesis assay.
Fertility and reproductive performance were unaffected in male or female rats that received febuxostat at oral doses up to 48 mg/kg/day (approximately 31 and 40 times the MRHD on an AUC basis in males and females respectively).
2.2 Dosage Recommendations in Patients With Renal Impairment and Hepatic Impairment
The recommended dosage of ULORIC is limited to 40 mg once daily in patients with severe renal impairment. No dose modification is necessary when administering ULORIC in patients with mild or moderate renal impairment [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
No dosage modification is necessary in patients with mild to moderate hepatic impairment [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use:
ULORIC is not recommended for the treatment of asymptomatic hyperuricemia.
Section 42230-3 (42230-3)
| This Medication Guide has been approved by the U.S. Food and Drug Administration | ULR015 R10 | Revised: April 2023 |
|
MEDICATION GUIDE ULORIC (Ū–'lor–ik) (febuxostat) tablets, for oral use |
||
| Read the Medication Guide that comes with ULORIC before you start taking it and each time you get a refill. There may be new information. The Medication Guide does not take the place of talking with your doctor about your medical condition or your treatment. | ||
|
What is the most important information that I should know about ULORIC? ULORIC may cause serious side effects, including: Heart-related deaths. Call your doctor or get emergency medical help right away if you have any of the following symptoms, especially if they are new, worse, or worry you: |
||
|
|
|
|
What is ULORIC?
ULORIC is a prescription medicine called a xanthine oxidase (XO) inhibitor used to lower blood uric acid levels in adult patients with gout when allopurinol has not worked well enough or when allopurinol is not right for you. ULORIC is not for use in people who do not have symptoms of high blood uric acid levels. It is not known if ULORIC is safe and effective in children. |
||
|
Who should not take ULORIC? Do not take ULORIC if you:
|
||
|
What should I tell my doctor before taking ULORIC? Before taking ULORIC tell your doctor about all of your medical conditions, including if you:
Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine. |
||
How should I take ULORIC?
|
||
|
What are the possible side effects of ULORIC? ULORIC may cause serious side effects, including:
|
||
|
|
|
|
||
|
|
|
| The most common side effects of ULORIC include: | ||
|
|
|
| These are not all of the possible side effects of ULORIC. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store ULORIC?
|
||
|
General information about the safe and effective use of ULORIC.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ULORIC for a condition for which it was not prescribed. Do not give ULORIC to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about ULORIC that is written for health professionals. |
||
|
What are the ingredients in ULORIC? Active ingredient: febuxostat Inactive ingredients: lactose monohydrate, microcrystalline cellulose, hydroxypropyl cellulose, sodium croscarmellose, silicon dioxide, magnesium stearate, and Opadry II, green Distributed by: Takeda Pharmaceuticals America, Inc. Lexington, MA 02421 ULORIC is a registered trademark of Teijin Limited registered in the U.S. Patent and Trademark Office and used under license by Takeda Pharmaceuticals America, Inc. All other trademarks are the property of their respective owners. ©2009-2023 Takeda Pharmaceuticals America, Inc. For more information, go to www.ULORIC.com or call 1-877-TAKEDA (1-877-825-3327). |
541887-04
Section 44425-7 (44425-7)
Protect from light. Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
7.3 in Vivo (7.3 In Vivo)
Based on drug interaction studies in healthy patients, ULORIC does not have clinically significant interactions with colchicine, naproxen, indomethacin, hydrochlorothiazide, warfarin or desipramine [see Clinical Pharmacology (12.3)]. Therefore, ULORIC may be used concomitantly with these medications.
10 Overdosage (10 OVERDOSAGE)
ULORIC was studied in healthy patients in doses up to 300 mg daily for seven days without evidence of dose-limiting toxicities. No overdose of ULORIC was reported in clinical studies. Patients should be managed by symptomatic and supportive care should there be an overdose.
11 Description (11 DESCRIPTION)
ULORIC (febuxostat) is a xanthine oxidase inhibitor. The active ingredient in ULORIC is 2-[3-cyano-4-(2-methylpropoxy) phenyl]-4-methylthiazole-5-carboxylic acid, with a molecular weight of 316.38. The empirical formula is C16H16N2O3S.
The chemical structure is:
Febuxostat is a non-hygroscopic, white crystalline powder that is freely soluble in dimethylformamide; soluble in dimethylsulfoxide; sparingly soluble in ethanol; slightly soluble in methanol and acetonitrile; and practically insoluble in water. The melting range is 205°C to 208°C.
ULORIC tablets for oral use contain the active ingredient, febuxostat, and are available in two dosage strengths, 40 mg and 80 mg. Inactive ingredients include hydroxypropyl cellulose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, silicon dioxide, and sodium croscarmellose. ULORIC tablets are coated with Opadry II, green.
5.2 Gout Flares
After initiation of ULORIC, an increase in gout flares is frequently observed. This increase is due to reduction in serum uric acid levels, resulting in mobilization of urate from tissue deposits.
In order to prevent gout flares when ULORIC is initiated, concurrent prophylactic treatment with an NSAID or colchicine is recommended [see Dosage and Administration (2.4)].
8.4 Pediatric Use
Safety and effectiveness of ULORIC in pediatric patients have not been established.
8.5 Geriatric Use
No dose adjustment is necessary in elderly patients. Of the total number of patients in Studies 1, 2, and 3 (clinical studies of ULORIC in the treatment of gout) [see Clinical Studies (14.1)], 16% were 65 and over, while 4% were 75 and over. Comparing patients in different age groups, no clinically significant differences in safety or effectiveness were observed but greater sensitivity of some older individuals cannot be ruled out. The Cmax and AUC24 of febuxostat following multiple oral doses of ULORIC in geriatric patients (≥65 years) were similar to those in younger patients (18 to 40 years) [see Clinical Pharmacology (12.3)].
14 Clinical Studies (14 CLINICAL STUDIES)
A serum uric acid level of less than 6 mg/dL is the goal of antihyperuricemic therapy and has been established as appropriate for the treatment of gout.
4 Contraindications (4 CONTRAINDICATIONS)
ULORIC is contraindicated in patients being treated with azathioprine or mercaptopurine [see Drug Interactions (7)].
5.3 Hepatic Effects
Cases of fatal and nonfatal hepatic failure in patients taking ULORIC have been reported. During randomized controlled studies, transaminase elevations greater than three times the upper limit of normal (ULN) were observed (AST: 2%, 2%, and ALT: 3%, 2% in ULORIC and allopurinol-treated patients, respectively). No dose-effect relationship for these transaminase elevations was noted [see Clinical Pharmacology (12.3)].
Obtain a liver test panel (serum alanine aminotransferase [ALT], aspartate aminotransferase [AST], alkaline phosphatase, and total bilirubin) as a baseline before initiating ULORIC.
Measure liver tests promptly in patients who report symptoms that may indicate liver injury, including fatigue, anorexia, right upper abdominal discomfort, dark urine or jaundice. In this clinical context, if the patient presents abnormal liver tests (ALT or AST greater than three times the upper limit of the reference range), interrupt ULORIC treatment while investigating the probable cause. Permanently discontinue ULORIC if liver injury is confirmed, and no alternate etiology can be found.
Permanently discontinue ULORIC in patients who have serum ALT or AST greater than three times the reference range with serum total bilirubin greater than two times the reference range without alternative etiologies because they are at risk for severe drug-induced liver injury. For patients with lesser elevations of serum ALT or bilirubin and with an alternate probable cause, treatment with ULORIC can be used with close monitoring.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the prescribing information:
- Cardiovascular Death [see Warnings and Precautions (5.1)]
- Hepatic Effects [see Warnings and Precautions (5.3)]
- Serious Skin Reactions [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Concomitant administration of ULORIC with XO substrate drugs, azathioprine or mercaptopurine could increase plasma concentrations of these drugs resulting in severe toxicity. (7)
8.6 Renal Impairment
No dose adjustment is necessary in patients with mild to moderate renal impairment (Clcr 30 to 89 mL/min). For patients with severe renal impairment (Clcr 15 to 29 mL/min), the recommended dosage of ULORIC is limited to 40 mg once daily [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
In healthy patients, maximum plasma concentrations (Cmax) and AUC of febuxostat increased in a dose proportional manner following single and multiple doses of 10 mg (0.25 times the lowest recommended dosage) to 120 mg (1.5 times the maximum recommended dosage). There is no accumulation when therapeutic doses are administered every 24 hours. Febuxostat has an apparent mean terminal elimination half-life (t1/2) of approximately 5 to 8 hours. Febuxostat pharmacokinetic parameters for patients with hyperuricemia and gout estimated by population pharmacokinetic analyses were similar to those estimated in healthy patients.
13.2 Animal Toxicology
A 12-month toxicity study in beagle dogs showed deposition of xanthine crystals and calculi in kidneys at 15 mg/kg (approximately 4 times the MRHD on an AUC basis). A similar effect of calculus formation was noted in rats in a 6-month study due to deposition of xanthine crystals at 48 mg/kg (approximately 31 and 40 times the MRHD on an AUC basis in males and females respectively).
2.1 Recommended Dosage
The recommended ULORIC dosage is 40 mg or 80 mg once daily.
The recommended starting dosage of ULORIC is 40 mg once daily. For patients who do not achieve a serum uric acid (sUA) less than 6 mg/dL after two weeks, the recommended ULORIC dosage is 80 mg once daily.
ULORIC can be taken without regard to food or antacid use [see Clinical Pharmacology (12.3)].
Concurrent prophylactic treatment with a non-steroidal anti-inflammatory drug (NSAID) or colchicine is recommended [see Dosage and Administration (2.4) and Warnings and Precautions (5.2)].
8.7 Hepatic Impairment
No dose adjustment is necessary in patients with mild or moderate hepatic impairment (Child-Pugh Class A or B). No studies have been conducted in patients with severe hepatic impairment (Child-Pugh Class C); therefore, caution should be exercised in these patients [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
ULORIC is a xanthine oxidase (XO) inhibitor indicated for the chronic management of hyperuricemia in adult patients with gout who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable.
12.1 Mechanism of Action
ULORIC, a xanthine oxidase inhibitor, achieves its therapeutic effect by decreasing serum uric acid. ULORIC is not expected to inhibit other enzymes involved in purine and pyrimidine synthesis and metabolism at therapeutic concentrations.
5.1 Cardiovascular Death
In a cardiovascular (CV) outcome study, gout patients with established CV disease treated with ULORIC had a higher rate of CV death compared to those treated with allopurinol. Sudden cardiac death was the most common cause of adjudicated CV deaths, 2.7% in the ULORIC group (83 of 3,098) as compared to 1.8% in the allopurinol group (56 of 3,092). ULORIC was similar to allopurinol for nonfatal myocardial infarction (MI), nonfatal stroke and unstable angina with urgent coronary revascularization [see Clinical Studies (14.2)].
Because of the increased risk of CV death, ULORIC should only be used in patients who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable [see Indications and Usage(1)].
Consider the risks and benefits of ULORIC when deciding to prescribe or continue patients on ULORIC. Consider use of prophylactic low-dose aspirin therapy in patients with a history of CV disease. Monitor patients for the development of CV events. Inform patients about the symptoms of serious CV events and the steps to take if they occur.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Gout Flares: An increase in gout flares is frequently observed after initiation of ULORIC. If a gout flare occurs during treatment, ULORIC need not be discontinued. Prophylactic therapy (i.e., non-steroidal anti-inflammatory drug or colchicine) upon initiation of treatment may be beneficial for up to six months. (2.4, 5.2)
- Hepatic Effects: Cases of hepatic failure, some fatal, have been reported. If liver injury is detected, promptly interrupt ULORIC and treat cause, if possible, to resolution or stabilization. Permanently discontinue ULORIC if liver injury is confirmed, and no alternate etiology can be found. (5.3)
- Serious Skin Reactions: Serious skin and hypersensitivity reactions, including Stevens-Johnson Syndrome, drug reaction with eosinophilia and systemic symptoms and toxic epidermal necrolysis have been reported in patients taking ULORIC. Discontinue ULORIC if serious skin reactions are suspected. (5.4)
5.4 Serious Skin Reactions
Serious skin and hypersensitivity reactions, including Stevens-Johnson Syndrome, drug reaction with eosinophilia and systemic symptoms (DRESS) and toxic epidermal necrolysis (TEN) have been reported postmarketing in patients taking ULORIC. Discontinue ULORIC if serious skin reactions are suspected [see Patient Counseling Information (17)]. Many of these patients had reported previous similar skin reactions to allopurinol. ULORIC should be used with close monitoring in these patients.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended dosage is 40 mg or 80 mg once daily. The recommended starting dosage is 40 mg once daily. For patients who do not achieve a serum uric acid (sUA) less than 6 mg/dL after 2 weeks, the recommended dosage is 80 mg once daily. (2.1)
- Patients with severe renal impairment: Limit the dosage to 40 mg once daily. (2.2, 8.6)
- Flare prophylaxis is recommended upon initiation of ULORIC. (2.4)
- Can be administered without regard to food or antacid use. (2.1)
8.8 Secondary Hyperuricemia
No studies have been conducted in patients with secondary hyperuricemia (including organ transplant recipients); ULORIC is not recommended for use in patients whom the rate of urate formation is greatly increased (e.g., malignant disease and its treatment, Lesch-Nyhan syndrome). The concentration of xanthine in urine could, in rare cases, rise sufficiently to allow deposition in the urinary tract.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 40 mg tablets, light green to green, round, debossed with "TAP" and "40"
- 80 mg tablets, light green to green, teardrop shaped, debossed with "TAP" and "80"
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of ULORIC. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: agranulocytosis, eosinophilia.
Hepatobiliary Disorders: hepatic failure (some fatal), jaundice, serious cases of abnormal liver function test results, liver disorder.
Immune System Disorders: anaphylaxis, anaphylactic reaction.
Musculoskeletal and Connective Tissue Disorders: rhabdomyolysis.
Psychiatric Disorders: psychotic behavior including aggressive thoughts.
Renal and Urinary Disorders: tubulointerstitial nephritis.
Skin and Subcutaneous Tissue Disorders: generalized rash, Stevens-Johnson Syndrome, hypersensitivity skin reactions, erythema multiforme, drug reaction with eosinophilia and systemic symptoms, toxic epidermal necrolysis.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Patients with severe hepatic impairment: No studies have been conducted in this patient population. Caution should be exercised in these patients. (8.7)
- Patients with secondary hyperuricemia (including patients being treated for Lesch-Nyhan syndrome or malignant disease, or in organ transplant recipients): ULORIC is not recommended for use as no studies have been conducted in this patient population. (8.8)
Warning: Cardiovascular Death (WARNING: CARDIOVASCULAR DEATH)
Gout patients with established cardiovascular (CV) disease treated with ULORIC had a higher rate of CV death compared to those treated with allopurinol in a CV outcomes study [see Warnings and Precautions (5.1)].
Consider the risks and benefits of ULORIC when deciding to prescribe or continue patients on ULORIC. ULORIC should only be used in patients who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable [see Indications and Usage (1)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In Phase 2 and 3 clinical studies, a total of 2757 patients with hyperuricemia and gout were treated with ULORIC 40 mg or 80 mg daily. For ULORIC 40 mg, 559 patients were treated for ≥6 months. For ULORIC 80 mg, 1377 patients were treated for ≥6 months, 674 patients were treated for ≥1 year and 515 patients were treated for ≥2 years. In the CARES study, a total of 3098 patients were treated with ULORIC 40 mg or 80 mg daily; of these, 2155 patients were treated for ≥1 year and 1539 were treated for ≥2 years [see Clinical Studies (14.2)].
14.2 Cardiovascular Safety Study
A randomized, double-blind, allopurinol-controlled CV outcomes study (CARES) was conducted to evaluate the CV risk of ULORIC (NCT01101035). The study compared the risk of MACE between patients treated with ULORIC (N=3098) and allopurinol-treated patients (N=3092). The primary endpoint was the time to first occurrence of a MACE defined as the composite of CV death, nonfatal MI, nonfatal stroke, or unstable angina with urgent coronary revascularization. The study was designed to exclude a prespecified risk margin of 1.3 for the hazard ratio of MACE. An independent committee conducted a blinded evaluation of serious CV adverse events according to predefined criteria (adjudication) for determination of MACE. The study was event driven and patients were followed until a sufficient number of primary outcome events accrued. The median on-study follow-up time was 2.6 years.
Patients randomized to ULORIC initially received 40 mg once daily which was increased to 80 mg once daily, if their sUA was ≥6mg/dL at Week 2. For patients randomized to allopurinol, those who had normal renal function or mild renal impairment (estimated creatinine clearance (eClcr) ≥60 to ˂90 mL/minute) initially received 300 mg once daily with 100 mg/day dose increments monthly until either sUA ˂6mg/dL or an allopurinol dosage of 600 mg once daily was achieved; those who had moderate renal impairment (eClcr ≥30 to ˂60 mL/minute) initially received 200 mg once daily with 100 mg/day dose increments monthly until either a sUA ˂6 mg/dL or an allopurinol dosage of 400 mg once daily was achieved.
The mean age of the population was 65 years (range: 44 to 93 years). Most patients were male (84%) and Caucasian (69%). Patients had a diagnosis of gout for approximately 12 years, a mean baseline sUA of 8.7 mg/dL, and 90% had experienced at least one gout flare in the past year. CV history included MI (39%), hospitalization for unstable angina (28%), cardiac revascularization (37%), and stroke (14%). The most prevalent comorbid conditions were hypertension (92%), hyperlipidemia (87%), diabetes mellitus (55%), diabetes mellitus with micro- or macrovascular disease (39%), and renal impairment [92% with an eClcr 30 to 89 mL/minute]. The use of CV disease medication was balanced across treatment groups. Baseline CV disease medications included: ACE inhibitors or ARBs (70%), lipid modifying agents (74%), aspirin (62%), beta-blockers (59%), calcium channel blockers (26%), and nonaspirin antiplatelet medications (31%).
Table 5 shows the study results for the primary MACE composite endpoint and its individual components. For the composite primary endpoint, the ULORIC group was non-inferior compared with the allopurinol group. The rates of nonfatal MI, stroke, and unstable angina with urgent coronary revascularization were similar. There was a higher rate of CV deaths in patients treated with ULORIC (134 CV deaths; 1.5 per 100 PY) than in allopurinol-treated patients (100 CV deaths; 1.1 per 100 PY). Sudden cardiac death was the most common cause of adjudicated CV deaths in the ULORIC group (83 of 3,098; 2.7%) as compared to the allopurinol group (56 of 3,092; 1.8%). The biological plausibility of CV death associated with ULORIC is unclear.
All-cause mortality was higher in the ULORIC group (243 deaths [7.8%]; 2.6 per 100 PY) than the allopurinol group (199 deaths [6.4%]; 2.2 per 100 PY) [Hazard Ratio: 1.22, 95% CI: 1.01, 1.47], due to a higher rate of CV deaths.
| ULORIC N=3098 |
Allopurinol N=3092 |
Hazard Ratio | |||
|---|---|---|---|---|---|
| Number of Patients with Event (%) | Rate per 100 PY Patient Years (PY)
|
Number of Patients with Event (%) | Rate per 100 PY | 95% CI | |
| Composite of primary endpoint MACE | 335 (10.8) | 3.8 | 321 (10.4) | 3.7 | 1.03 (0.89, 1.21) |
| Cardiovascular Death | 134 (4.3) | 1.5 | 100 (3.2) | 1.1 | 1.34 (1.03, 1.73) |
| Nonfatal MI | 111 (3.6) | 1.2 | 118 (3.8) | 1.3 | 0.93 (0.72, 1.21) |
| Nonfatal stroke | 71 (2.3) | 0.8 | 70 (2.3) | 0.8 | 1.01 (0.73, 1.41) |
| Unstable angina with urgent coronary revascularization | 49 (1.6) | 0.5 | 56 (1.8) | 0.6 | 0.86 (0.59, 1.26) |
7.2 Cytotoxic Chemotherapy Drugs
Drug interaction studies of ULORIC with cytotoxic chemotherapy have not been conducted. No data are available regarding the safety of ULORIC during cytotoxic chemotherapy.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ULORIC 40 mg tablets are light green to green in color, round, debossed with "TAP" on one side and "40" on the other side and supplied as:
| NDC Number | Size |
|---|---|
| 64764-918-11 | Hospital Unit Dose Pack of 100 Tablets |
| 64764-918-30 | Bottle of 30 Tablets |
| 64764-918-90 | Bottle of 90 Tablets |
| 64764-918-18 | Bottle of 500 Tablets |
ULORIC 80 mg tablets are light green to green in color, teardrop shaped, debossed with "TAP" on one side and "80" on the other side and supplied as:
| NDC Number | Size |
|---|---|
| 64764-677-11 | Hospital Unit Dose Pack of 100 Tablets |
| 64764-677-30 | Bottle of 30 Tablets |
| 64764-677-13 | Bottle of 100 Tablets |
| 64764-677-19 | Bottle of 1000 Tablets |
2.3 Serum Uric Acid Level Monitoring
Testing for the target serum uric acid level of less than 6 mg/dL may be performed as early as two weeks after initiating ULORIC therapy.
7.1 Xanthine Oxidase Substrate Drugs
ULORIC is an XO inhibitor. Based on a drug interaction study in healthy patients, febuxostat altered the metabolism of theophylline (a substrate of XO) in humans [see Clinical Pharmacology (12.3)]. Therefore, use with caution when coadministering ULORIC with theophylline.
A drug interaction study of ULORIC and azathioprine, also metabolized by XO, showed an increase in exposure of 6-mercaptopurine which may lead to toxicity [see Clinical Pharmacology (12.3)]. Drug interaction studies of ULORIC with other drugs that are metabolized by XO (e.g., mercaptopurine) have not been conducted. ULORIC is contraindicated in patients being treated with azathioprine or mercaptopurine [see Contraindications (4)].
14.1 Management of Hyperuricemia in Gout
The efficacy of ULORIC was demonstrated in three randomized, double-blind, controlled trials in patients with hyperuricemia and gout. Hyperuricemia was defined as a baseline serum uric acid level ≥8 mg/dL.
Study 1 (NCT00430248) randomized patients to: ULORIC 40 mg daily, ULORIC 80 mg daily, or allopurinol (300 mg daily for patients with estimated creatinine clearance (Clcr) ≥60 mL/min or 200 mg daily for patients with estimated Clcr ≥30 mL/min and ≤59 mL/min). The duration of Study 1 was six months.
Study 2 (NCT00174915) randomized patients to: placebo, ULORIC 80 mg daily, ULORIC 120 mg daily, ULORIC 240 mg daily or allopurinol (300 mg daily for patients with a baseline serum creatinine ≤1.5 mg/dL or 100 mg daily for patients with a baseline serum creatinine greater than 1.5 mg/dL and ≤2 mg/dL). The duration of Study 2 was six months.
Study 3 (NCT00102440), a year study, randomized patients to: ULORIC 80 mg daily, ULORIC 120 mg daily, or allopurinol 300 mg daily. Patients who completed Study 2 and Study 3 were eligible to enroll in a Phase 3 long-term extension study in which patients received treatment with ULORIC for over three years.
In all three studies, patients received naproxen 250 mg twice daily or colchicine 0.6 mg once or twice daily for gout flare prophylaxis. In Study 1 the duration of prophylaxis was six months; in Study 2 and Study 3 the duration of prophylaxis was eight weeks.
The efficacy of ULORIC was also evaluated in a 4-week dose ranging study which randomized patients to: placebo, ULORIC 40 mg daily, ULORIC 80 mg daily, or ULORIC 120 mg daily. Patients who completed this study were eligible to enroll in a long-term extension study in which patients received treatment with ULORIC for up to five years.
Patients in these studies were representative of the patient population for which ULORIC use is intended. Table 2 summarizes the demographics and baseline characteristics for the patients enrolled in the studies.
| Male | 95% | |
| Race: | Caucasian | 80% |
| African American | 10% | |
| Ethnicity: Hispanic or Latino | 7% | |
| Alcohol User | 67% | |
| Mild to Moderate Renal Insufficiency (percent with estimated Clcr less than 90 mL/min) |
59% | |
| History of Hypertension | 49% | |
| History of Hyperlipidemia | 38% | |
| BMI ≥30 kg/m2 | 63% | |
| Mean BMI | 33 kg/m2 | |
| Baseline sUA ≥10 mg/dL | 36% | |
| Mean baseline sUA | 9.7 mg/dL | |
| Experienced a gout flare in previous year | 85% |
2.4 Recommended Prophylaxis for Gout Flares
Gout flares may occur after initiation of ULORIC due to changing serum uric acid levels resulting in mobilization of urate from tissue deposits. Flare prophylaxis with a non-steroidal anti-inflammatory drug (NSAID) or colchicine is recommended upon initiation of ULORIC. Prophylactic therapy may be beneficial for up to six months [see Clinical Studies (14.1)].
If a gout flare occurs during ULORIC treatment, ULORIC need not be discontinued. The gout flare should be managed concurrently, as appropriate for the individual patient [see Warnings and Precautions (5.2)].
Principal Display Panel 40 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 40 mg Tablet Bottle Label)
NDC 64764-918-30
30 Tablets
Uloric
(febuxostat)
tablets
40 mg
Dispense the accompanying
Medication Guide to each patient.
Principal Display Panel 80 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 80 mg Tablet Bottle Label)
NDC 64764-677-30
30 Tablets
Uloric
(febuxostat)
tablets
80 mg
Dispense the accompanying
Medication Guide to each patient.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year carcinogenicity studies were conducted in F344 rats and B6C3F1 mice. Increased transitional cell papilloma and carcinoma of the urinary bladder was observed at 24 mg/kg (25 times the MRHD on an AUC basis and 18.75 mg/kg (12.5 times the MRHD on an AUC basis) in male rats and female mice, respectively. The urinary bladder neoplasms were secondary to calculus formation in the kidney and urinary bladder.
Febuxostat showed a positive clastogenic response in a chromosomal aberration assay in a Chinese hamster lung fibroblast cell line with and without metabolic activation in vitro. Febuxostat was negative in the following genotoxicity assays: the in vitro Ames assay, in vitro chromosomal aberration assay in human peripheral lymphocytes, the L5178Y mouse lymphoma cell line assay, the in vivo mouse micronucleus assay, and the rat unscheduled DNA synthesis assay.
Fertility and reproductive performance were unaffected in male or female rats that received febuxostat at oral doses up to 48 mg/kg/day (approximately 31 and 40 times the MRHD on an AUC basis in males and females respectively).
2.2 Dosage Recommendations in Patients With Renal Impairment and Hepatic Impairment (2.2 Dosage Recommendations in Patients with Renal Impairment and Hepatic Impairment)
The recommended dosage of ULORIC is limited to 40 mg once daily in patients with severe renal impairment. No dose modification is necessary when administering ULORIC in patients with mild or moderate renal impairment [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
No dosage modification is necessary in patients with mild to moderate hepatic impairment [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:36.771482 · Updated: 2026-03-14T22:41:00.111917