These Highlights Do Not Include All The Information Needed To Use Fluticasone Propionate Nasal Spray Safely And Effectively. See Full Prescribing Information For Fluticasone Propionate Nasal Spray.
549ff4f2-fa68-3dd8-e054-00144ff8d46c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 01/2015
Indications and Usage
Fluticasone Propionate Nasal Spray, USP 50 mcg per spray is indicated for the management of the nasal symptoms of perennial nonallergic rhinitis in adult and pediatric patients aged 4 years and older.
Dosage and Administration
Administer Fluticasone Propionate Nasal Spray by the intranasal route only. Prime Fluticasone Propionate Nasal Spray before using for the first time or after a period of non-use (1 week or more) by shaking the contents well and releasing 6 sprays into the air away from the face. Shake Fluticasone Propionate Nasal Spray gently before each use. Patients should use Fluticasone Propionate Nasal Spray at regular intervals since its effectiveness depends on its regular use. Maximum effect may take several days and individual patients will experience a variable time to onset and different degree of symptom relief.
Warnings and Precautions
Epistaxis, nasal ulceration, Candida albicans infection, nasal septal perforation, and impaired wound healing. Monitor patients periodically for signs of adverse effects on the nasal mucosa. Avoid use in patients with recent nasal ulcers, nasal surgery, or nasal trauma. ( 5.1 ) Close monitoring for glaucoma and cataracts is warranted. ( 5.2 ) Hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria, contact dermatitis, and rash) have been reported after administration of Fluticasone Propionate Nasal Spray. Discontinue Fluticasone Propionate Nasal Spray if such reactions occur. ( 5.3 ) Potential worsening of infections (e.g., existing tuberculosis; fungal, bacterial, viral, or parasitic infection; ocular herpes simplex). Use with caution in patients with these infections. More serious or even fatal course of chickenpox or measles can occur in susceptible patients. ( 5.4 ) Hypercorticism and adrenal suppression may occur with very high dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue Fluticasone Propionate Nasal Spray slowly. ( 5.5 ) Monitor growth of pediatric patients. ( 5.7 )
Contraindications
Fluticasone Propionate Nasal Spray is contraindicated in patients with hypersensitivity to any of its ingredients [see Warnings and Precautions (5.3) , Description (11) ].
Adverse Reactions
Systemic and local corticosteroid use may result in the following: Epistaxis, nasal ulceration, Candida albicans infection, nasal septal perforation, and impaired wound healing [see Warnings and Precautions (5.1) ] Cataracts and glaucoma [see Warnings and Precautions (5.2) ] Immunosuppression [see Warnings and Precautions (5.4) ] Hypercorticism and adrenal suppression [see Warnings and Precautions (5.5) ] Effect on growth [see Warnings and Precautions (5.7) ]
Drug Interactions
Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir, ketoconazole): Use not recommended. May increase risk of systemic corticosteroid effects. ( 7.1 )
Storage and Handling
Fluticasone Propionate Nasal Spray, 50 mcg is supplied in an amber glass bottle fitted with a white metering atomizing pump, white nasal adapter, and transparent dust cap in a box of 1 (NDC 68071-1638-5) with FDA-approved Patient Labeling (see Patient Instructions for Use for proper actuation of the device). Each bottle contains a net fill weight of 16 g and will provide 120 actuations. Each actuation delivers 50 mcg of fluticasone propionate in 100 mg of formulation through the nasal adapter. The correct amount of medication in each spray cannot be assured after 120 sprays even though the bottle is not completely empty. The bottle should be discarded when the labeled number of actuations has been used.
How Supplied
Fluticasone Propionate Nasal Spray, 50 mcg is supplied in an amber glass bottle fitted with a white metering atomizing pump, white nasal adapter, and transparent dust cap in a box of 1 (NDC 68071-1638-5) with FDA-approved Patient Labeling (see Patient Instructions for Use for proper actuation of the device). Each bottle contains a net fill weight of 16 g and will provide 120 actuations. Each actuation delivers 50 mcg of fluticasone propionate in 100 mg of formulation through the nasal adapter. The correct amount of medication in each spray cannot be assured after 120 sprays even though the bottle is not completely empty. The bottle should be discarded when the labeled number of actuations has been used.
Patient Information
Read the Patient Information that comes with Fluticasone Propionate Nasal Spray before you start using it and each time you get a refill. There may be new information. This Patient Information does not take the place of talking to your healthcare provider about your medical condition or treatment. What is Fluticasone Propionate Nasal Spray? Fluticasone Propionate Nasal Spray is a prescription medicine used to treat non-allergy nasal symptoms such as runny nose, stuffy nose, sneezing, and nasal itching in adults and children aged 4 years and older. It is not known if Fluticasone Propionate Nasal Spray is safe and effective in children younger than 4 years of age. Who should not use Fluticasone Propionate Nasal Spray? Do not use Fluticasone Propionate Nasal Spray if you are allergic to fluticasone propionate or any of the ingredients in Fluticasone Propionate Nasal Spray. See " What are the ingredients in Fluticasone Propionate Nasal Spray? " below for a complete list of ingredients. What should I tell my healthcare provider before using Fluticasone Propionate Nasal Spray? Tell your healthcare provider about all of your health conditions, including if you: have or have had nasal sores, nasal surgery, or nasal injury. have eye problems, such as cataracts or glaucoma. have an immune system problem. are allergic to any of the ingredients in Fluticasone Propionate Nasal Spray, any other medicines, or food products. See " What are the ingredients in Fluticasone Propionate Nasal Spray? " below for a complete list of ingredients. have any type of viral, bacterial, or fungal infection. are exposed to chickenpox or measles. have any other medical conditions. are pregnant or planning to become pregnant. It is not known if Fluticasone Propionate Nasal Spray may harm your unborn baby. are breastfeeding or plan to breastfeed. It is not known if Fluticasone Propionate Nasal Spray passes into your breast milk and if it can harm your baby. Tell your healthcare provider about all the medicines you take , including prescription and over-the-counter medicines, vitamins, and herbal supplements. Fluticasone Propionate Nasal Spray and certain other medicines may interact with each other. This may cause serious side effects. Especially, tell your healthcare provider if you take antifungal or anti-HIV medicines. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. How should I use Fluticasone Propionate Nasal Spray? Read the step-by-step instructions for using Fluticasone Propionate Nasal Spray at the end of this Patient Information. Fluticasone Propionate Nasal Spray is for use in your nose only. Do not spray it in your eyes or mouth. Children should use Fluticasone Propionate Nasal Spray with an adult's help, as instructed by the child's healthcare provider. Use Fluticasone Propionate Nasal Spray exactly as your healthcare provider tells you. Do not use Fluticasone Propionate Nasal Spray more often than prescribed. Fluticasone Propionate Nasal Spray may take several days of regular use for your rhinitis symptoms to get better. If your symptoms do not improve or get worse, call your healthcare provider. You will get the best results if you keep using Fluticasone Propionate Nasal Spray regularly each day without missing a dose. After you begin to feel better, your healthcare provider may decrease your dose. Do not stop using Fluticasone Propionate Nasal Spray unless your healthcare provider tells you to do so. What are the possible side effects of Fluticasone Propionate Nasal Spray? Fluticasone Propionate Nasal Spray may cause serious side effects, including: nose problems . Nose problems may include: nose bleeds. sores (ulcers) in your nose. a certain fungal infection in your nose, mouth, and/or throat (thrush). hole in the cartilage of your nose (nasal septal perforation). Symptoms of nasal septal perforation may include: crusting in the nose nose bleeds runny nose whistling sound when you breathe slow wound healing. You should not use Fluticasone Propionate Nasal Spray until your nose has healed if you have a sore in your nose, have had surgery on your nose, or if your nose has been injured. eye problems including glaucoma and cataracts. You should have regular eye exams while you use Fluticasone Propionate Nasal Spray. serious allergic reactions. Call your healthcare provider or get emergency medical care if you get any of the following signs of a serious allergic reaction: rash hives swelling of your face, mouth, and tongue breathing problems weakened immune system and increased chance of getting infections (immunosuppression). Taking medicines that weaken your immune system makes you more likely to get infections and can make certain infections worse. These infections may include tuberculosis (TB), ocular herpes simplex infections, and infections caused by fungi, bacteria, viruses, and parasites. Avoid contact with people who have a contagious disease such as chickenpox or measles while using Fluticasone Propionate Nasal Spray. If you come in contact with someone who has chickenpox or measles call your healthcare provider right away. Symptoms of an infection may include: fever pain aches chills feeling tired nausea vomiting lowered steroid hormone levels (adrenal insufficiency). Adrenal insufficiency happens when your adrenal glands do not make enough steroid hormones. This can happen when you stop taking oral corticosteroid medicines (such as prednisone) and start taking medicine containing an inhaled steroid (such as Fluticasone Propionate Nasal Spray). Symptoms of adrenal insufficiency may include: feeling tired lack of energy weakness nausea and vomiting low blood pressure slowed growth in children. A child's growth should be checked often. The most common side effects of Fluticasone Propionate Nasal Spray include: headache sore throat nose bleeds nose burning or itching nausea and vomiting trouble breathing cough Tell your healthcare provider about any side effect that bothers you or does not go away. These are not all the side effects with Fluticasone Propionate Nasal Spray. Ask your healthcare provider or pharmacist for more information. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. How do I store Fluticasone Propionate Nasal Spray? Store Fluticasone Propionate between 39°F and 86°F (4°C and 30°C). Keep Fluticasone Propionate Nasal Spray and all medicines out of the reach of children. General information about the safe and effective use of Fluticasone Propionate Nasal Spray. Medicines are sometimes prescribed for purposes not mentioned in a Patient Information leaflet. Do not use Fluticasone Propionate Nasal Spray for a condition for which it was not prescribed. Do not give your Fluticasone Propionate Nasal Spray to other people, even if they have the same condition that you have. It may harm them. This Patient Information leaflet summarizes the most important information about Fluticasone Propionate Nasal Spray. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Fluticasone Propionate Nasal Spray that was written for healthcare professionals. For more information about Fluticasone Propionate Nasal Spray, call 1-800-346-6854. What are the ingredients in Fluticasone Propionate Nasal Spray? Active ingredient: fluticasone propionate. Inactive ingredients: microcrystalline cellulose, carboxymethylcellulose sodium, dextrose, 0.02% w/w benzalkonium chloride, polysorbate 80, and 0.25% w/w phenylethyl alcohol.
Medication Information
Warnings and Precautions
Epistaxis, nasal ulceration, Candida albicans infection, nasal septal perforation, and impaired wound healing. Monitor patients periodically for signs of adverse effects on the nasal mucosa. Avoid use in patients with recent nasal ulcers, nasal surgery, or nasal trauma. ( 5.1 ) Close monitoring for glaucoma and cataracts is warranted. ( 5.2 ) Hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria, contact dermatitis, and rash) have been reported after administration of Fluticasone Propionate Nasal Spray. Discontinue Fluticasone Propionate Nasal Spray if such reactions occur. ( 5.3 ) Potential worsening of infections (e.g., existing tuberculosis; fungal, bacterial, viral, or parasitic infection; ocular herpes simplex). Use with caution in patients with these infections. More serious or even fatal course of chickenpox or measles can occur in susceptible patients. ( 5.4 ) Hypercorticism and adrenal suppression may occur with very high dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue Fluticasone Propionate Nasal Spray slowly. ( 5.5 ) Monitor growth of pediatric patients. ( 5.7 )
Indications and Usage
Fluticasone Propionate Nasal Spray, USP 50 mcg per spray is indicated for the management of the nasal symptoms of perennial nonallergic rhinitis in adult and pediatric patients aged 4 years and older.
Dosage and Administration
Administer Fluticasone Propionate Nasal Spray by the intranasal route only. Prime Fluticasone Propionate Nasal Spray before using for the first time or after a period of non-use (1 week or more) by shaking the contents well and releasing 6 sprays into the air away from the face. Shake Fluticasone Propionate Nasal Spray gently before each use. Patients should use Fluticasone Propionate Nasal Spray at regular intervals since its effectiveness depends on its regular use. Maximum effect may take several days and individual patients will experience a variable time to onset and different degree of symptom relief.
Contraindications
Fluticasone Propionate Nasal Spray is contraindicated in patients with hypersensitivity to any of its ingredients [see Warnings and Precautions (5.3) , Description (11) ].
Adverse Reactions
Systemic and local corticosteroid use may result in the following: Epistaxis, nasal ulceration, Candida albicans infection, nasal septal perforation, and impaired wound healing [see Warnings and Precautions (5.1) ] Cataracts and glaucoma [see Warnings and Precautions (5.2) ] Immunosuppression [see Warnings and Precautions (5.4) ] Hypercorticism and adrenal suppression [see Warnings and Precautions (5.5) ] Effect on growth [see Warnings and Precautions (5.7) ]
Drug Interactions
Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir, ketoconazole): Use not recommended. May increase risk of systemic corticosteroid effects. ( 7.1 )
Storage and Handling
Fluticasone Propionate Nasal Spray, 50 mcg is supplied in an amber glass bottle fitted with a white metering atomizing pump, white nasal adapter, and transparent dust cap in a box of 1 (NDC 68071-1638-5) with FDA-approved Patient Labeling (see Patient Instructions for Use for proper actuation of the device). Each bottle contains a net fill weight of 16 g and will provide 120 actuations. Each actuation delivers 50 mcg of fluticasone propionate in 100 mg of formulation through the nasal adapter. The correct amount of medication in each spray cannot be assured after 120 sprays even though the bottle is not completely empty. The bottle should be discarded when the labeled number of actuations has been used.
How Supplied
Fluticasone Propionate Nasal Spray, 50 mcg is supplied in an amber glass bottle fitted with a white metering atomizing pump, white nasal adapter, and transparent dust cap in a box of 1 (NDC 68071-1638-5) with FDA-approved Patient Labeling (see Patient Instructions for Use for proper actuation of the device). Each bottle contains a net fill weight of 16 g and will provide 120 actuations. Each actuation delivers 50 mcg of fluticasone propionate in 100 mg of formulation through the nasal adapter. The correct amount of medication in each spray cannot be assured after 120 sprays even though the bottle is not completely empty. The bottle should be discarded when the labeled number of actuations has been used.
Patient Information
Read the Patient Information that comes with Fluticasone Propionate Nasal Spray before you start using it and each time you get a refill. There may be new information. This Patient Information does not take the place of talking to your healthcare provider about your medical condition or treatment.
What is Fluticasone Propionate Nasal Spray?
Fluticasone Propionate Nasal Spray is a prescription medicine used to treat non-allergy nasal symptoms such as runny nose, stuffy nose, sneezing, and nasal itching in adults and children aged 4 years and older.
It is not known if Fluticasone Propionate Nasal Spray is safe and effective in children younger than 4 years of age.
Who should not use Fluticasone Propionate Nasal Spray?
Do not use Fluticasone Propionate Nasal Spray if you are allergic to fluticasone propionate or any of the ingredients in Fluticasone Propionate Nasal Spray. See " What are the ingredients in Fluticasone Propionate Nasal Spray?" below for a complete list of ingredients.
What should I tell my healthcare provider before using Fluticasone Propionate Nasal Spray?
Tell your healthcare provider about all of your health conditions, including if you:
- have or have had nasal sores, nasal surgery, or nasal injury.
- have eye problems, such as cataracts or glaucoma.
- have an immune system problem.
- are allergic to any of the ingredients in Fluticasone Propionate Nasal Spray, any other medicines, or food products. See " What are the ingredients in Fluticasone Propionate Nasal Spray?" below for a complete list of ingredients.
- have any type of viral, bacterial, or fungal infection.
- are exposed to chickenpox or measles.
- have any other medical conditions.
- are pregnant or planning to become pregnant. It is not known if Fluticasone Propionate Nasal Spray may harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if Fluticasone Propionate Nasal Spray passes into your breast milk and if it can harm your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Fluticasone Propionate Nasal Spray and certain other medicines may interact with each other. This may cause serious side effects. Especially, tell your healthcare provider if you take antifungal or anti-HIV medicines.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I use Fluticasone Propionate Nasal Spray?
Read the step-by-step instructions for using Fluticasone Propionate Nasal Spray at the end of this Patient Information.
- Fluticasone Propionate Nasal Spray is for use in your nose only. Do not spray it in your eyes or mouth.
- Children should use Fluticasone Propionate Nasal Spray with an adult's help, as instructed by the child's healthcare provider.
- Use Fluticasone Propionate Nasal Spray exactly as your healthcare provider tells you. Do not use Fluticasone Propionate Nasal Spray more often than prescribed.
- Fluticasone Propionate Nasal Spray may take several days of regular use for your rhinitis symptoms to get better. If your symptoms do not improve or get worse, call your healthcare provider.
- You will get the best results if you keep using Fluticasone Propionate Nasal Spray regularly each day without missing a dose. After you begin to feel better, your healthcare provider may decrease your dose. Do not stop using Fluticasone Propionate Nasal Spray unless your healthcare provider tells you to do so.
What are the possible side effects of Fluticasone Propionate Nasal Spray? Fluticasone Propionate Nasal Spray may cause serious side effects, including:
-
nose problems. Nose problems may include:
- nose bleeds.
- sores (ulcers) in your nose.
- a certain fungal infection in your nose, mouth, and/or throat (thrush).
-
hole in the cartilage of your nose (nasal septal perforation).
Symptoms of nasal septal perforation may include:- crusting in the nose
- nose bleeds
- runny nose
- whistling sound when you breathe
- slow wound healing. You should not use Fluticasone Propionate Nasal Spray until your nose has healed if you have a sore in your nose, have had surgery on your nose, or if your nose has been injured.
- eye problems including glaucoma and cataracts. You should have regular eye exams while you use Fluticasone Propionate Nasal Spray.
-
serious allergic reactions. Call your healthcare provider or get emergency medical care if you get any of the following signs of a serious allergic reaction:
- rash
- hives
- swelling of your face, mouth, and tongue
- breathing problems
-
weakened immune system and increased chance of getting infections (immunosuppression). Taking medicines that weaken your immune system makes you more likely to get infections and can make certain infections worse. These infections may include tuberculosis (TB), ocular herpes simplex infections, and infections caused by fungi, bacteria, viruses, and parasites. Avoid contact with people who have a contagious disease such as chickenpox or measles while using Fluticasone Propionate Nasal Spray. If you come in contact with someone who has chickenpox or measles call your healthcare provider right away.
Symptoms of an infection may include:- fever
- pain
- aches
- chills
- feeling tired
- nausea
- vomiting
-
lowered steroid hormone levels (adrenal insufficiency). Adrenal insufficiency happens when your adrenal glands do not make enough steroid hormones. This can happen when you stop taking oral corticosteroid medicines (such as prednisone) and start taking medicine containing an inhaled steroid (such as Fluticasone Propionate Nasal Spray). Symptoms of adrenal insufficiency may include:
- feeling tired
- lack of energy
- weakness
- nausea and vomiting
- low blood pressure
- slowed growth in children. A child's growth should be checked often.
The most common side effects of Fluticasone Propionate Nasal Spray include:
|
|
Tell your healthcare provider about any side effect that bothers you or does not go away.
These are not all the side effects with Fluticasone Propionate Nasal Spray. Ask your healthcare provider or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How do I store Fluticasone Propionate Nasal Spray?
- Store Fluticasone Propionate between 39°F and 86°F (4°C and 30°C).
Keep Fluticasone Propionate Nasal Spray and all medicines out of the reach of children.
General information about the safe and effective use of Fluticasone Propionate Nasal Spray.
Medicines are sometimes prescribed for purposes not mentioned in a Patient Information leaflet. Do not use Fluticasone Propionate Nasal Spray for a condition for which it was not prescribed. Do not give your Fluticasone Propionate Nasal Spray to other people, even if they have the same condition that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about Fluticasone Propionate Nasal Spray. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Fluticasone Propionate Nasal Spray that was written for healthcare professionals.
For more information about Fluticasone Propionate Nasal Spray, call 1-800-346-6854.
What are the ingredients in Fluticasone Propionate Nasal Spray?
Active ingredient: fluticasone propionate.
Inactive ingredients: microcrystalline cellulose, carboxymethylcellulose sodium, dextrose, 0.02% w/w benzalkonium chloride, polysorbate 80, and 0.25% w/w phenylethyl alcohol.
Description
Indications and Usage ( 1 ) 01/2015
Section 34077-8
Teratogenic Effects
Section 34078-6
Nonteratogenic Effects
Hypoadrenalism may occur in infants born of mothers receiving corticosteroids during pregnancy. Such infants should be carefully monitored.
Section 42229-5
Epistaxis
In clinical trials of 2 to 26 weeks' duration, epistaxis was observed more frequently in subjects treated with Fluticasone Propionate Nasal Spray than those who received placebo [see Adverse Reactions (6.1)].
Section 43683-2
| Indications and Usage ( 1) | 01/2015 |
Section 44425-7
Store between 4° and 30°C (39° and 86°F).
2.1 Adults
The recommended starting dosage in adults is 2 sprays (50 mcg of fluticasone propionate each) in each nostril once daily (total daily dose, 200 mcg). The same total daily dose, 1 spray in each nostril administered twice daily (e.g., 8 a.m. and 8 p.m.) is also effective. After the first few days, patients may be able to reduce their dose to 1 spray in each nostril once daily for maintenance therapy.
Maximum total daily doses should not exceed 2 sprays in each nostril (total dose, 200 mcg/day). There is no evidence that exceeding the recommended dose is more effective.
10 Overdosage
Chronic overdosage may result in signs/symptoms of hypercorticism [see Warnings and Precautions (5.5)]. Intranasal administration of 2 mg (10 times the recommended dose) of fluticasone propionate twice daily for 7 days was administered to healthy human volunteers. Adverse events reported with fluticasone propionate were similar to placebo, and no clinically significant abnormalities in laboratory safety tests were observed. Single oral doses up to 16 mg have been studied in human volunteers with no acute toxic effects reported. Repeat oral doses up to 80 mg daily for 10 days in volunteers and repeat oral doses up to 10 mg daily for 14 days in patients were well tolerated. Adverse reactions were of mild or moderate severity, and incidences were similar in active and placebo treatment groups. Acute overdosage with this dosage form is unlikely since 1 bottle of Fluticasone Propionate Nasal Spray contains approximately 8 mg of fluticasone propionate.
11 Description
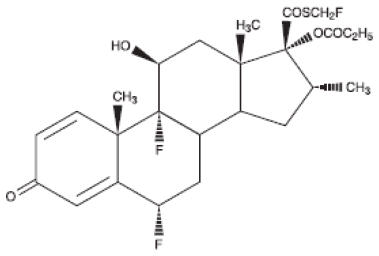
The active component of Fluticasone Propionate Nasal Spray is fluticasone propionate, a corticosteroid having the chemical name S-(fluoromethyl)6α,9-difluoro-11β,17-dihydroxy-16α-methyl-3-oxoandrosta-1,4-diene-17β-carbothioate, 17-propionate and the following chemical structure:
Fluticasone propionate is a white powder with a molecular weight of 500.6, and the empirical formula is C 25H 31F 3O 5S. It is practically insoluble in water, freely soluble in dimethyl sulfoxide and dimethylformamide, and slightly soluble in methanol and 95% ethanol.
Fluticasone Propionate Nasal Spray, 50 mcg is an aqueous suspension of microfine fluticasone propionate for topical administration to the nasal mucosa by means of a metering, atomizing spray pump. Fluticasone Propionate Nasal Spray also contains microcrystalline cellulose and carboxymethylcellulose sodium, dextrose, 0.02% w/w benzalkonium chloride, polysorbate 80, and 0.25% w/w phenylethyl alcohol, and has a pH between 5 and 7.
After initial priming, each actuation delivers 50 mcg of fluticasone propionate in 100 mg of formulation through the nasal adapter.
8.4 Pediatric Use
The safety and effectiveness of Fluticasone Propionate Nasal Spray in children aged 4 years and older have been established [see Adverse Reactions (6.1), Clinical Pharmacology (12.3)] . Six hundred fifty (650) subjects aged 4 to 11 years and 440 subjects aged 12 to 17 years were studied in US clinical trials with fluticasone propionate nasal spray. The safety and effectiveness of Fluticasone Propionate Nasal Spray in children younger than 4 years have not been established.
8.5 Geriatric Use
A limited number of subjects aged 65 years and older (n = 129) or 75 years and older (n = 11) have been treated with Fluticasone Propionate Nasal Spray in clinical trials. While the number of subjects is too small to permit separate analysis of efficacy and safety, the adverse reactions reported in this population were similar to those reported by younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications
Fluticasone Propionate Nasal Spray is contraindicated in patients with hypersensitivity to any of its ingredients [see Warnings and Precautions (5.3), Description (11)].
6 Adverse Reactions
Systemic and local corticosteroid use may result in the following:
- Epistaxis, nasal ulceration, Candida albicans infection, nasal septal perforation, and impaired wound healing [see Warnings and Precautions (5.1)]
- Cataracts and glaucoma [see Warnings and Precautions (5.2)]
- Immunosuppression [see Warnings and Precautions (5.4)]
- Hypercorticism and adrenal suppression [see Warnings and Precautions (5.5)]
- Effect on growth [see Warnings and Precautions (5.7)]
7 Drug Interactions
Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir, ketoconazole): Use not recommended. May increase risk of systemic corticosteroid effects. ( 7.1)
8.3 Nursing Mothers
It is not known whether fluticasone propionate is excreted in human breast milk. However, other corticosteroids have been detected in human milk. Subcutaneous administration to lactating rats of tritiated fluticasone propionate at a dose approximately 0.4 times the MRHDID for adults on a mg/m 2 basis resulted in measurable radioactivity in milk.
Since there are no data from controlled trials on the use of intranasal Fluticasone Propionate Nasal Spray by nursing mothers, caution should be exercised when Fluticasone Propionate Nasal Spray is administered to a nursing woman.
5.7 Effect On Growth
Intranasal corticosteroids may cause a reduction in growth velocity when administered to pediatric patients [see Use in Specific Populations (8.4)]. Monitor the growth routinely of pediatric patients receiving Fluticasone Propionate Nasal Spray. To minimize the systemic effects of intranasal corticosteroids, including Fluticasone Propionate Nasal Spray, titrate each patient's dose to the lowest dosage that effectively controls his/her symptoms [see Dosage and Administration (2), Use in Specific Populations (8.4)].
8.7 Renal Impairment
Formal pharmacokinetic trials using Fluticasone Propionate Nasal Spray have not been conducted in subjects with renal impairment.
Instructions for Use
Fluticasone Propionate Nasal Spray is for use in your nose only.
Read this information before you start using your Fluticasone Propionate Nasal Spray.
|
FIGURE A FIGURE B |
Your Fluticasone Propionate Nasal Spray must be primed before you use it for the first time and when you have not used it for a week or more.
How to prime your Fluticasone Propionate Nasal Spray
|
|
FIGURE C FIGURE D |
Using your Fluticasone Propionate Nasal Spray:
Step 1. Blow your nose to clear your nostrils Step 2. Close 1 nostril. Tilt your head forward slightly and, keeping the bottle upright, carefully insert the nasal applicator into the other nostril (See Figure C). Step 3. Start to breathe in through your nose, and while breathing in press firmly and quickly down 1 time on the applicator to release the spray. To get a full dose, use your forefinger and middle finger to spray while supporting the base of the bottle with your thumb. Avoid spraying in your eyes. Breathe in gently through the nostril. Step 4. Breathe out through your mouth. Step 5. If a second spray is required in that nostril, repeat steps 2 through 4. Step 6. Repeat steps 2 through 5 in the other nostril. Step 7. Wipe the nasal applicator with a clean tissue and replace the dust cover (See Figure D). |
| Do not use this bottle for more than the labeled number of sprays even though the bottle is not completely empty. Before you throw the bottle away, you should talk to your healthcare provider to see if a refill is needed. Do not take extra doses or stop taking Fluticasone Propionate Nasal Spray without talking to your healthcare provider. |
Cleaning your Fluticasone Propionate Nasal Spray:
Your nasal spray should be cleaned at least 1 time each week.
- Remove the dust cover and then gently pull upwards to free the nasal applicator.
- Wash the applicator and dust cover under warm tap water. Allow to dry at room temperature.
- Place the applicator and dust cover back on the bottle.
- If the nasal applicator becomes blocked, it can be removed and left to soak in warm water. Rinse the nasal applicator with cold tap water. Dry the nasal applicator and place it back on the bottle. Do not try to unblock the nasal applicator by inserting a pin or other sharp object.
Storing your Fluticasone Propionate Nasal Spray:
- Store Fluticasone Propionate Nasal Spray between 39°F and 86°F (4°C and 30°C).
- Do not use your Fluticasone Propionate Nasal Spray after the date shown as "EXP" on the label or box.
This Patient Information and Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured For:
Wockhardt USA, LLC
Parsippany, NJ 07054
Manufactured By:
Morton Grove Pharmaceuticals, Inc.
Morton Grove, IL 60053
27264A REV. 03-16
12.3 Pharmacokinetics
The activity of Fluticasone Propionate Nasal Spray is due to the parent drug, fluticasone propionate. Due to the low bioavailability by the intranasal route, the majority of the pharmacokinetic data was obtained via other routes of administration.
5.4 Immunosuppression
Persons who are using drugs that suppress the immune system are more susceptible to infections than healthy individuals. Chickenpox and measles, for example, can have a more serious or even fatal course in susceptible children or adults using corticosteroids. In such children or adults who have not had these diseases or been properly immunized, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If a patient is exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If a patient is exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See the complete prescribing information for VZIG and IG.) If chickenpox develops, treatment with antiviral agents may be considered.
Intranasal corticosteroids should be used with caution, if at all, in patients with active or quiescent tuberculous infections of the respiratory tract; systemic fungal, bacterial, viral, or parasitic infections; or ocular herpes simplex.
8.6 Hepatic Impairment
Formal pharmacokinetic trials using Fluticasone Propionate Nasal Spray have not been conducted in subjects with hepatic impairment. Since fluticasone propionate is predominantly cleared by hepatic metabolism, impairment of liver function may lead to accumulation of fluticasone propionate in plasma. Therefore, patients with hepatic disease should be closely monitored.
1 Indications and Usage
Fluticasone Propionate Nasal Spray, USP 50 mcg per spray is indicated for the management of the nasal symptoms of perennial nonallergic rhinitis in adult and pediatric patients aged 4 years and older.
12.1 Mechanism of Action
Fluticasone propionate is a synthetic trifluorinated corticosteroid with anti-inflammatory activity. Fluticasone propionate has been shown in vitro to exhibit a binding affinity for the human glucocorticoid receptor that is 18 times that of dexamethasone, almost twice that of beclomethasone-17-monopropionate (BMP), the active metabolite of beclomethasone dipropionate, and over 3 times that of budesonide. Data from the McKenzie vasoconstrictor assay in man are consistent with these results. The clinical significance of these findings is unknown.
The precise mechanism through which fluticasone propionate affects rhinitis symptoms is not known. Corticosteroids have been shown to have a wide range of effects on multiple cell types (e.g., mast cells, eosinophils, neutrophils, macrophages, lymphocytes) and mediators (e.g., histamine, eicosanoids, leukotrienes, cytokines) involved in inflammation. In 7 trials in adults, Fluticasone Propionate Nasal Spray has decreased nasal mucosal eosinophils in 66% of patients (35% for placebo) and basophils in 39% of patients (28% for placebo). The direct relationship of these findings to long-term symptom relief is not known.
5 Warnings and Precautions
- Epistaxis, nasal ulceration, Candida albicans infection, nasal septal perforation, and impaired wound healing. Monitor patients periodically for signs of adverse effects on the nasal mucosa. Avoid use in patients with recent nasal ulcers, nasal surgery, or nasal trauma. ( 5.1)
- Close monitoring for glaucoma and cataracts is warranted. ( 5.2)
- Hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria, contact dermatitis, and rash) have been reported after administration of Fluticasone Propionate Nasal Spray. Discontinue Fluticasone Propionate Nasal Spray if such reactions occur. ( 5.3)
- Potential worsening of infections (e.g., existing tuberculosis; fungal, bacterial, viral, or parasitic infection; ocular herpes simplex). Use with caution in patients with these infections. More serious or even fatal course of chickenpox or measles can occur in susceptible patients. ( 5.4)
- Hypercorticism and adrenal suppression may occur with very high dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue Fluticasone Propionate Nasal Spray slowly. ( 5.5)
- Monitor growth of pediatric patients. ( 5.7)
5.2 Glaucoma and Cataracts
Use of intranasal and inhaled corticosteroids may result in the development of glaucoma and/or cataracts. Therefore, close monitoring is warranted in patients with a change in vision or with a history of increased intraocular pressure, glaucoma, and/or cataracts.
2 Dosage and Administration
Administer Fluticasone Propionate Nasal Spray by the intranasal route only. Prime Fluticasone Propionate Nasal Spray before using for the first time or after a period of non-use (1 week or more) by shaking the contents well and releasing 6 sprays into the air away from the face. Shake Fluticasone Propionate Nasal Spray gently before each use.
Patients should use Fluticasone Propionate Nasal Spray at regular intervals since its effectiveness depends on its regular use. Maximum effect may take several days and individual patients will experience a variable time to onset and different degree of symptom relief.
3 Dosage Forms and Strengths
Fluticasone Propionate Nasal Spray is a nasal spray suspension. Each 100-mg spray delivers 50 mcg of fluticasone propionate.
6.2 Postmarketing Experience
In addition to adverse events reported from clinical trials, the following adverse events have been identified during postapproval use of intranasal fluticasone propionate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These events have been chosen for inclusion due to either their seriousness, frequency of reporting, or causal connection to fluticasone propionate or a combination of these factors.
8 Use in Specific Populations
Hepatic impairment: Monitor patients for signs of increased drug exposure. ( 8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In controlled US clinical trials, more than 3,300 subjects with allergic and nonallergic rhinitis received treatment with intranasal fluticasone propionate. In general, adverse reactions in clinical trials have been primarily associated with irritation of the nasal mucous membranes, and the adverse reactions were reported with approximately the same frequency by subjects treated with placebo. Less than 2% of subjects in clinical trials discontinued because of adverse reactions; this rate was similar for vehicle placebo and active comparators.
The safety data described below are based on 7 placebo-controlled clinical trials in subjects with allergic rhinitis. The 7 trials included 536 subjects (57 girls and 108 boys aged 4 to 11 years, 137 female and 234 male adolescents and adults) treated with Fluticasone Propionate 200 mcg once daily over 2 to 4 weeks and 2 placebo-controlled clinical trials which included 246 subjects (119 female and 127 male adolescents and adults) treated with Fluticasone Propionate 200 mcg once daily over 6 months (Table 1). Also included in Table 1 are adverse reactions from 2 trials in which 167 children (45 girls and 122 boys aged 4 to 11 years) were treated with Fluticasone Propionate 100 mcg once daily for 2 to 4 weeks.
| Adverse Reaction | Fluticasone Propionate 100 mcg Once Daily
(n = 167) % |
Fluticasone Propionate 200 mcg Once Daily
(n = 782) % |
Placebo
(n = 758) % |
|---|---|---|---|
| Headache | 6.6 | 16.1 | 14.6 |
| Pharyngitis | 6.0 | 7.8 | 7.2 |
| Epistaxis | 6.0 | 6.9 | 5.4 |
| Nasal burning/nasal irritation | 2.4 | 3.2 | 2.6 |
| Nausea/vomiting | 4.8 | 2.6 | 2.0 |
| Asthma symptoms | 7.2 | 3.3 | 2.9 |
| Cough | 3.6 | 3.8 | 2.8 |
Other adverse reactions with Fluticasone Propionate Nasal Spray observed with an incidence less than or equal to 3% but greater than or equal to 1% and more common than with placebo included: blood in nasal mucus, runny nose, abdominal pain, diarrhea, fever, flu-like symptoms, aches and pains, dizziness, and bronchitis.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied/storage and Handling
Fluticasone Propionate Nasal Spray, 50 mcg is supplied in an amber glass bottle fitted with a white metering atomizing pump, white nasal adapter, and transparent dust cap in a box of 1 (NDC 68071-1638-5) with FDA-approved Patient Labeling (see Patient Instructions for Use for proper actuation of the device). Each bottle contains a net fill weight of 16 g and will provide 120 actuations. Each actuation delivers 50 mcg of fluticasone propionate in 100 mg of formulation through the nasal adapter. The correct amount of medication in each spray cannot be assured after 120 sprays even though the bottle is not completely empty. The bottle should be discarded when the labeled number of actuations has been used.
7.1 Inhibitors of Cytochrome P450 3a4
Fluticasone propionate is a substrate of CYP3A4. The use of strong CYP3A4 inhibitors (e.g., ritonavir, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, ketoconazole, telithromycin, conivaptan, lopinavir, nefazodone, voriconazole) with Fluticasone Propionate Nasal Spray is not recommended because increased systemic corticosteroid adverse effects may occur.
Principal Display Panel 16 G Bottle Box
5.5 Hypercorticism and Adrenal Suppression
When intranasal corticosteroids are used at higher than recommended dosages or in susceptible individuals at recommended dosages, systemic corticosteroid effects such as hypercorticism and adrenal suppression may appear. If such changes occur, the dosage of Fluticasone Propionate Nasal Spray should be discontinued slowly consistent with accepted procedures for discontinuing oral corticosteroid therapy.
The replacement of a systemic corticosteroid with a topical corticosteroid can be accompanied by signs of adrenal insufficiency. In addition, some patients may experience symptoms of corticosteroid withdrawal (e.g., joint and/or muscular pain, lassitude, depression). Patients previously treated for prolonged periods with systemic corticosteroids and transferred to topical corticosteroids should be carefully monitored for acute adrenal insufficiency in response to stress. In patients who have asthma or other clinical conditions requiring long-term systemic corticosteroid treatment, rapid decreases in systemic corticosteroid dosages may cause a severe exacerbation of their symptoms.
5.3 Hypersensitivity Reactions Including Anaphylaxis
Hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria, contact dermatitis, and rash) have been reported after administration of Fluticasone Propionate Nasal Spray. Discontinue Fluticasone Propionate Nasal Spray if such reactions occur [see Contraindications (4)]. Rarely, immediate hypersensitivity reactions may occur after the administration of Fluticasone Propionate Nasal Spray.
2.2 Adolescents and Children (aged 4 Years and Older)
The recommended starting dosage in adolescents and children, aged 4 years and older is 1 spray in each nostril once daily (total daily dose, 100 mcg). Patients not adequately responding to 1 spray in each nostril may use 2 sprays in each nostril once daily (total daily dose, 200 mcg). Once adequate control is achieved, the dosage should be decreased to 1 spray in each nostril once daily.
The maximum total daily dosage should not exceed 2 sprays in each nostril (200 mcg/day). There is no evidence that exceeding the recommended dose is more effective.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Fluticasone propionate demonstrated no tumorigenic potential in mice at oral doses up to 1,000 mcg/kg (approximately 20 times the MRHDID in adults and approximately 10 times the MRHDID in children on a mcg/m 2 basis) for 78 weeks or in rats at inhalation doses up to 57 mcg/kg (approximately 2 times the MRHDID in adults and approximately equivalent to the MRHDID in children on a mcg/m 2 basis) for 104 weeks.
Fluticasone propionate did not induce gene mutation in prokaryotic or eukaryotic cells in vitro. No significant clastogenic effect was seen in cultured human peripheral lymphocytes in vitro or in the mouse micronucleus test.
No evidence of impairment of fertility was observed in male and female rats at subcutaneous doses up to 50 mcg/kg (approximately 2 times the MRHDID in adults on a mcg/m 2 basis). Prostate weight was significantly reduced at a subcutaneous dose of 50 mcg/kg.
5.6 Drug Interactions With Strong Cytochrome P450 3a4 Inhibitors
The use of strong cytochrome P450 3A4 (CYP3A4) inhibitors (e.g., ritonavir, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, ketoconazole, telithromycin, conivaptan, lopinavir, nefazodone, voriconazole) with Fluticasone Propionate Nasal Spray is not recommended because increased systemic corticosteroid adverse effects may occur [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
Structured Label Content
Section 34077-8 (34077-8)
Teratogenic Effects
Section 34078-6 (34078-6)
Nonteratogenic Effects
Hypoadrenalism may occur in infants born of mothers receiving corticosteroids during pregnancy. Such infants should be carefully monitored.
Section 42229-5 (42229-5)
Epistaxis
In clinical trials of 2 to 26 weeks' duration, epistaxis was observed more frequently in subjects treated with Fluticasone Propionate Nasal Spray than those who received placebo [see Adverse Reactions (6.1)].
Section 43683-2 (43683-2)
| Indications and Usage ( 1) | 01/2015 |
Section 44425-7 (44425-7)
Store between 4° and 30°C (39° and 86°F).
2.1 Adults
The recommended starting dosage in adults is 2 sprays (50 mcg of fluticasone propionate each) in each nostril once daily (total daily dose, 200 mcg). The same total daily dose, 1 spray in each nostril administered twice daily (e.g., 8 a.m. and 8 p.m.) is also effective. After the first few days, patients may be able to reduce their dose to 1 spray in each nostril once daily for maintenance therapy.
Maximum total daily doses should not exceed 2 sprays in each nostril (total dose, 200 mcg/day). There is no evidence that exceeding the recommended dose is more effective.
10 Overdosage (10 OVERDOSAGE)
Chronic overdosage may result in signs/symptoms of hypercorticism [see Warnings and Precautions (5.5)]. Intranasal administration of 2 mg (10 times the recommended dose) of fluticasone propionate twice daily for 7 days was administered to healthy human volunteers. Adverse events reported with fluticasone propionate were similar to placebo, and no clinically significant abnormalities in laboratory safety tests were observed. Single oral doses up to 16 mg have been studied in human volunteers with no acute toxic effects reported. Repeat oral doses up to 80 mg daily for 10 days in volunteers and repeat oral doses up to 10 mg daily for 14 days in patients were well tolerated. Adverse reactions were of mild or moderate severity, and incidences were similar in active and placebo treatment groups. Acute overdosage with this dosage form is unlikely since 1 bottle of Fluticasone Propionate Nasal Spray contains approximately 8 mg of fluticasone propionate.
11 Description (11 DESCRIPTION)
The active component of Fluticasone Propionate Nasal Spray is fluticasone propionate, a corticosteroid having the chemical name S-(fluoromethyl)6α,9-difluoro-11β,17-dihydroxy-16α-methyl-3-oxoandrosta-1,4-diene-17β-carbothioate, 17-propionate and the following chemical structure:
Fluticasone propionate is a white powder with a molecular weight of 500.6, and the empirical formula is C 25H 31F 3O 5S. It is practically insoluble in water, freely soluble in dimethyl sulfoxide and dimethylformamide, and slightly soluble in methanol and 95% ethanol.
Fluticasone Propionate Nasal Spray, 50 mcg is an aqueous suspension of microfine fluticasone propionate for topical administration to the nasal mucosa by means of a metering, atomizing spray pump. Fluticasone Propionate Nasal Spray also contains microcrystalline cellulose and carboxymethylcellulose sodium, dextrose, 0.02% w/w benzalkonium chloride, polysorbate 80, and 0.25% w/w phenylethyl alcohol, and has a pH between 5 and 7.
After initial priming, each actuation delivers 50 mcg of fluticasone propionate in 100 mg of formulation through the nasal adapter.
8.4 Pediatric Use
The safety and effectiveness of Fluticasone Propionate Nasal Spray in children aged 4 years and older have been established [see Adverse Reactions (6.1), Clinical Pharmacology (12.3)] . Six hundred fifty (650) subjects aged 4 to 11 years and 440 subjects aged 12 to 17 years were studied in US clinical trials with fluticasone propionate nasal spray. The safety and effectiveness of Fluticasone Propionate Nasal Spray in children younger than 4 years have not been established.
8.5 Geriatric Use
A limited number of subjects aged 65 years and older (n = 129) or 75 years and older (n = 11) have been treated with Fluticasone Propionate Nasal Spray in clinical trials. While the number of subjects is too small to permit separate analysis of efficacy and safety, the adverse reactions reported in this population were similar to those reported by younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications (4 CONTRAINDICATIONS)
Fluticasone Propionate Nasal Spray is contraindicated in patients with hypersensitivity to any of its ingredients [see Warnings and Precautions (5.3), Description (11)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
Systemic and local corticosteroid use may result in the following:
- Epistaxis, nasal ulceration, Candida albicans infection, nasal septal perforation, and impaired wound healing [see Warnings and Precautions (5.1)]
- Cataracts and glaucoma [see Warnings and Precautions (5.2)]
- Immunosuppression [see Warnings and Precautions (5.4)]
- Hypercorticism and adrenal suppression [see Warnings and Precautions (5.5)]
- Effect on growth [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir, ketoconazole): Use not recommended. May increase risk of systemic corticosteroid effects. ( 7.1)
8.3 Nursing Mothers
It is not known whether fluticasone propionate is excreted in human breast milk. However, other corticosteroids have been detected in human milk. Subcutaneous administration to lactating rats of tritiated fluticasone propionate at a dose approximately 0.4 times the MRHDID for adults on a mg/m 2 basis resulted in measurable radioactivity in milk.
Since there are no data from controlled trials on the use of intranasal Fluticasone Propionate Nasal Spray by nursing mothers, caution should be exercised when Fluticasone Propionate Nasal Spray is administered to a nursing woman.
Patient Information
Read the Patient Information that comes with Fluticasone Propionate Nasal Spray before you start using it and each time you get a refill. There may be new information. This Patient Information does not take the place of talking to your healthcare provider about your medical condition or treatment.
What is Fluticasone Propionate Nasal Spray?
Fluticasone Propionate Nasal Spray is a prescription medicine used to treat non-allergy nasal symptoms such as runny nose, stuffy nose, sneezing, and nasal itching in adults and children aged 4 years and older.
It is not known if Fluticasone Propionate Nasal Spray is safe and effective in children younger than 4 years of age.
Who should not use Fluticasone Propionate Nasal Spray?
Do not use Fluticasone Propionate Nasal Spray if you are allergic to fluticasone propionate or any of the ingredients in Fluticasone Propionate Nasal Spray. See " What are the ingredients in Fluticasone Propionate Nasal Spray?" below for a complete list of ingredients.
What should I tell my healthcare provider before using Fluticasone Propionate Nasal Spray?
Tell your healthcare provider about all of your health conditions, including if you:
- have or have had nasal sores, nasal surgery, or nasal injury.
- have eye problems, such as cataracts or glaucoma.
- have an immune system problem.
- are allergic to any of the ingredients in Fluticasone Propionate Nasal Spray, any other medicines, or food products. See " What are the ingredients in Fluticasone Propionate Nasal Spray?" below for a complete list of ingredients.
- have any type of viral, bacterial, or fungal infection.
- are exposed to chickenpox or measles.
- have any other medical conditions.
- are pregnant or planning to become pregnant. It is not known if Fluticasone Propionate Nasal Spray may harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if Fluticasone Propionate Nasal Spray passes into your breast milk and if it can harm your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Fluticasone Propionate Nasal Spray and certain other medicines may interact with each other. This may cause serious side effects. Especially, tell your healthcare provider if you take antifungal or anti-HIV medicines.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I use Fluticasone Propionate Nasal Spray?
Read the step-by-step instructions for using Fluticasone Propionate Nasal Spray at the end of this Patient Information.
- Fluticasone Propionate Nasal Spray is for use in your nose only. Do not spray it in your eyes or mouth.
- Children should use Fluticasone Propionate Nasal Spray with an adult's help, as instructed by the child's healthcare provider.
- Use Fluticasone Propionate Nasal Spray exactly as your healthcare provider tells you. Do not use Fluticasone Propionate Nasal Spray more often than prescribed.
- Fluticasone Propionate Nasal Spray may take several days of regular use for your rhinitis symptoms to get better. If your symptoms do not improve or get worse, call your healthcare provider.
- You will get the best results if you keep using Fluticasone Propionate Nasal Spray regularly each day without missing a dose. After you begin to feel better, your healthcare provider may decrease your dose. Do not stop using Fluticasone Propionate Nasal Spray unless your healthcare provider tells you to do so.
What are the possible side effects of Fluticasone Propionate Nasal Spray? Fluticasone Propionate Nasal Spray may cause serious side effects, including:
-
nose problems. Nose problems may include:
- nose bleeds.
- sores (ulcers) in your nose.
- a certain fungal infection in your nose, mouth, and/or throat (thrush).
-
hole in the cartilage of your nose (nasal septal perforation).
Symptoms of nasal septal perforation may include:- crusting in the nose
- nose bleeds
- runny nose
- whistling sound when you breathe
- slow wound healing. You should not use Fluticasone Propionate Nasal Spray until your nose has healed if you have a sore in your nose, have had surgery on your nose, or if your nose has been injured.
- eye problems including glaucoma and cataracts. You should have regular eye exams while you use Fluticasone Propionate Nasal Spray.
-
serious allergic reactions. Call your healthcare provider or get emergency medical care if you get any of the following signs of a serious allergic reaction:
- rash
- hives
- swelling of your face, mouth, and tongue
- breathing problems
-
weakened immune system and increased chance of getting infections (immunosuppression). Taking medicines that weaken your immune system makes you more likely to get infections and can make certain infections worse. These infections may include tuberculosis (TB), ocular herpes simplex infections, and infections caused by fungi, bacteria, viruses, and parasites. Avoid contact with people who have a contagious disease such as chickenpox or measles while using Fluticasone Propionate Nasal Spray. If you come in contact with someone who has chickenpox or measles call your healthcare provider right away.
Symptoms of an infection may include:- fever
- pain
- aches
- chills
- feeling tired
- nausea
- vomiting
-
lowered steroid hormone levels (adrenal insufficiency). Adrenal insufficiency happens when your adrenal glands do not make enough steroid hormones. This can happen when you stop taking oral corticosteroid medicines (such as prednisone) and start taking medicine containing an inhaled steroid (such as Fluticasone Propionate Nasal Spray). Symptoms of adrenal insufficiency may include:
- feeling tired
- lack of energy
- weakness
- nausea and vomiting
- low blood pressure
- slowed growth in children. A child's growth should be checked often.
The most common side effects of Fluticasone Propionate Nasal Spray include:
|
|
Tell your healthcare provider about any side effect that bothers you or does not go away.
These are not all the side effects with Fluticasone Propionate Nasal Spray. Ask your healthcare provider or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How do I store Fluticasone Propionate Nasal Spray?
- Store Fluticasone Propionate between 39°F and 86°F (4°C and 30°C).
Keep Fluticasone Propionate Nasal Spray and all medicines out of the reach of children.
General information about the safe and effective use of Fluticasone Propionate Nasal Spray.
Medicines are sometimes prescribed for purposes not mentioned in a Patient Information leaflet. Do not use Fluticasone Propionate Nasal Spray for a condition for which it was not prescribed. Do not give your Fluticasone Propionate Nasal Spray to other people, even if they have the same condition that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about Fluticasone Propionate Nasal Spray. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Fluticasone Propionate Nasal Spray that was written for healthcare professionals.
For more information about Fluticasone Propionate Nasal Spray, call 1-800-346-6854.
What are the ingredients in Fluticasone Propionate Nasal Spray?
Active ingredient: fluticasone propionate.
Inactive ingredients: microcrystalline cellulose, carboxymethylcellulose sodium, dextrose, 0.02% w/w benzalkonium chloride, polysorbate 80, and 0.25% w/w phenylethyl alcohol.
5.7 Effect On Growth (5.7 Effect on Growth)
Intranasal corticosteroids may cause a reduction in growth velocity when administered to pediatric patients [see Use in Specific Populations (8.4)]. Monitor the growth routinely of pediatric patients receiving Fluticasone Propionate Nasal Spray. To minimize the systemic effects of intranasal corticosteroids, including Fluticasone Propionate Nasal Spray, titrate each patient's dose to the lowest dosage that effectively controls his/her symptoms [see Dosage and Administration (2), Use in Specific Populations (8.4)].
8.7 Renal Impairment
Formal pharmacokinetic trials using Fluticasone Propionate Nasal Spray have not been conducted in subjects with renal impairment.
Instructions for Use
Fluticasone Propionate Nasal Spray is for use in your nose only.
Read this information before you start using your Fluticasone Propionate Nasal Spray.
|
FIGURE A FIGURE B |
Your Fluticasone Propionate Nasal Spray must be primed before you use it for the first time and when you have not used it for a week or more.
How to prime your Fluticasone Propionate Nasal Spray
|
|
FIGURE C FIGURE D |
Using your Fluticasone Propionate Nasal Spray:
Step 1. Blow your nose to clear your nostrils Step 2. Close 1 nostril. Tilt your head forward slightly and, keeping the bottle upright, carefully insert the nasal applicator into the other nostril (See Figure C). Step 3. Start to breathe in through your nose, and while breathing in press firmly and quickly down 1 time on the applicator to release the spray. To get a full dose, use your forefinger and middle finger to spray while supporting the base of the bottle with your thumb. Avoid spraying in your eyes. Breathe in gently through the nostril. Step 4. Breathe out through your mouth. Step 5. If a second spray is required in that nostril, repeat steps 2 through 4. Step 6. Repeat steps 2 through 5 in the other nostril. Step 7. Wipe the nasal applicator with a clean tissue and replace the dust cover (See Figure D). |
| Do not use this bottle for more than the labeled number of sprays even though the bottle is not completely empty. Before you throw the bottle away, you should talk to your healthcare provider to see if a refill is needed. Do not take extra doses or stop taking Fluticasone Propionate Nasal Spray without talking to your healthcare provider. |
Cleaning your Fluticasone Propionate Nasal Spray:
Your nasal spray should be cleaned at least 1 time each week.
- Remove the dust cover and then gently pull upwards to free the nasal applicator.
- Wash the applicator and dust cover under warm tap water. Allow to dry at room temperature.
- Place the applicator and dust cover back on the bottle.
- If the nasal applicator becomes blocked, it can be removed and left to soak in warm water. Rinse the nasal applicator with cold tap water. Dry the nasal applicator and place it back on the bottle. Do not try to unblock the nasal applicator by inserting a pin or other sharp object.
Storing your Fluticasone Propionate Nasal Spray:
- Store Fluticasone Propionate Nasal Spray between 39°F and 86°F (4°C and 30°C).
- Do not use your Fluticasone Propionate Nasal Spray after the date shown as "EXP" on the label or box.
This Patient Information and Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured For:
Wockhardt USA, LLC
Parsippany, NJ 07054
Manufactured By:
Morton Grove Pharmaceuticals, Inc.
Morton Grove, IL 60053
27264A REV. 03-16
12.3 Pharmacokinetics
The activity of Fluticasone Propionate Nasal Spray is due to the parent drug, fluticasone propionate. Due to the low bioavailability by the intranasal route, the majority of the pharmacokinetic data was obtained via other routes of administration.
5.4 Immunosuppression
Persons who are using drugs that suppress the immune system are more susceptible to infections than healthy individuals. Chickenpox and measles, for example, can have a more serious or even fatal course in susceptible children or adults using corticosteroids. In such children or adults who have not had these diseases or been properly immunized, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If a patient is exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If a patient is exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See the complete prescribing information for VZIG and IG.) If chickenpox develops, treatment with antiviral agents may be considered.
Intranasal corticosteroids should be used with caution, if at all, in patients with active or quiescent tuberculous infections of the respiratory tract; systemic fungal, bacterial, viral, or parasitic infections; or ocular herpes simplex.
8.6 Hepatic Impairment
Formal pharmacokinetic trials using Fluticasone Propionate Nasal Spray have not been conducted in subjects with hepatic impairment. Since fluticasone propionate is predominantly cleared by hepatic metabolism, impairment of liver function may lead to accumulation of fluticasone propionate in plasma. Therefore, patients with hepatic disease should be closely monitored.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Fluticasone Propionate Nasal Spray, USP 50 mcg per spray is indicated for the management of the nasal symptoms of perennial nonallergic rhinitis in adult and pediatric patients aged 4 years and older.
12.1 Mechanism of Action
Fluticasone propionate is a synthetic trifluorinated corticosteroid with anti-inflammatory activity. Fluticasone propionate has been shown in vitro to exhibit a binding affinity for the human glucocorticoid receptor that is 18 times that of dexamethasone, almost twice that of beclomethasone-17-monopropionate (BMP), the active metabolite of beclomethasone dipropionate, and over 3 times that of budesonide. Data from the McKenzie vasoconstrictor assay in man are consistent with these results. The clinical significance of these findings is unknown.
The precise mechanism through which fluticasone propionate affects rhinitis symptoms is not known. Corticosteroids have been shown to have a wide range of effects on multiple cell types (e.g., mast cells, eosinophils, neutrophils, macrophages, lymphocytes) and mediators (e.g., histamine, eicosanoids, leukotrienes, cytokines) involved in inflammation. In 7 trials in adults, Fluticasone Propionate Nasal Spray has decreased nasal mucosal eosinophils in 66% of patients (35% for placebo) and basophils in 39% of patients (28% for placebo). The direct relationship of these findings to long-term symptom relief is not known.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Epistaxis, nasal ulceration, Candida albicans infection, nasal septal perforation, and impaired wound healing. Monitor patients periodically for signs of adverse effects on the nasal mucosa. Avoid use in patients with recent nasal ulcers, nasal surgery, or nasal trauma. ( 5.1)
- Close monitoring for glaucoma and cataracts is warranted. ( 5.2)
- Hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria, contact dermatitis, and rash) have been reported after administration of Fluticasone Propionate Nasal Spray. Discontinue Fluticasone Propionate Nasal Spray if such reactions occur. ( 5.3)
- Potential worsening of infections (e.g., existing tuberculosis; fungal, bacterial, viral, or parasitic infection; ocular herpes simplex). Use with caution in patients with these infections. More serious or even fatal course of chickenpox or measles can occur in susceptible patients. ( 5.4)
- Hypercorticism and adrenal suppression may occur with very high dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue Fluticasone Propionate Nasal Spray slowly. ( 5.5)
- Monitor growth of pediatric patients. ( 5.7)
5.2 Glaucoma and Cataracts
Use of intranasal and inhaled corticosteroids may result in the development of glaucoma and/or cataracts. Therefore, close monitoring is warranted in patients with a change in vision or with a history of increased intraocular pressure, glaucoma, and/or cataracts.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Administer Fluticasone Propionate Nasal Spray by the intranasal route only. Prime Fluticasone Propionate Nasal Spray before using for the first time or after a period of non-use (1 week or more) by shaking the contents well and releasing 6 sprays into the air away from the face. Shake Fluticasone Propionate Nasal Spray gently before each use.
Patients should use Fluticasone Propionate Nasal Spray at regular intervals since its effectiveness depends on its regular use. Maximum effect may take several days and individual patients will experience a variable time to onset and different degree of symptom relief.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Fluticasone Propionate Nasal Spray is a nasal spray suspension. Each 100-mg spray delivers 50 mcg of fluticasone propionate.
6.2 Postmarketing Experience
In addition to adverse events reported from clinical trials, the following adverse events have been identified during postapproval use of intranasal fluticasone propionate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These events have been chosen for inclusion due to either their seriousness, frequency of reporting, or causal connection to fluticasone propionate or a combination of these factors.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Hepatic impairment: Monitor patients for signs of increased drug exposure. ( 8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In controlled US clinical trials, more than 3,300 subjects with allergic and nonallergic rhinitis received treatment with intranasal fluticasone propionate. In general, adverse reactions in clinical trials have been primarily associated with irritation of the nasal mucous membranes, and the adverse reactions were reported with approximately the same frequency by subjects treated with placebo. Less than 2% of subjects in clinical trials discontinued because of adverse reactions; this rate was similar for vehicle placebo and active comparators.
The safety data described below are based on 7 placebo-controlled clinical trials in subjects with allergic rhinitis. The 7 trials included 536 subjects (57 girls and 108 boys aged 4 to 11 years, 137 female and 234 male adolescents and adults) treated with Fluticasone Propionate 200 mcg once daily over 2 to 4 weeks and 2 placebo-controlled clinical trials which included 246 subjects (119 female and 127 male adolescents and adults) treated with Fluticasone Propionate 200 mcg once daily over 6 months (Table 1). Also included in Table 1 are adverse reactions from 2 trials in which 167 children (45 girls and 122 boys aged 4 to 11 years) were treated with Fluticasone Propionate 100 mcg once daily for 2 to 4 weeks.
| Adverse Reaction | Fluticasone Propionate 100 mcg Once Daily
(n = 167) % |
Fluticasone Propionate 200 mcg Once Daily
(n = 782) % |
Placebo
(n = 758) % |
|---|---|---|---|
| Headache | 6.6 | 16.1 | 14.6 |
| Pharyngitis | 6.0 | 7.8 | 7.2 |
| Epistaxis | 6.0 | 6.9 | 5.4 |
| Nasal burning/nasal irritation | 2.4 | 3.2 | 2.6 |
| Nausea/vomiting | 4.8 | 2.6 | 2.0 |
| Asthma symptoms | 7.2 | 3.3 | 2.9 |
| Cough | 3.6 | 3.8 | 2.8 |
Other adverse reactions with Fluticasone Propionate Nasal Spray observed with an incidence less than or equal to 3% but greater than or equal to 1% and more common than with placebo included: blood in nasal mucus, runny nose, abdominal pain, diarrhea, fever, flu-like symptoms, aches and pains, dizziness, and bronchitis.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Fluticasone Propionate Nasal Spray, 50 mcg is supplied in an amber glass bottle fitted with a white metering atomizing pump, white nasal adapter, and transparent dust cap in a box of 1 (NDC 68071-1638-5) with FDA-approved Patient Labeling (see Patient Instructions for Use for proper actuation of the device). Each bottle contains a net fill weight of 16 g and will provide 120 actuations. Each actuation delivers 50 mcg of fluticasone propionate in 100 mg of formulation through the nasal adapter. The correct amount of medication in each spray cannot be assured after 120 sprays even though the bottle is not completely empty. The bottle should be discarded when the labeled number of actuations has been used.
7.1 Inhibitors of Cytochrome P450 3a4 (7.1 Inhibitors of Cytochrome P450 3A4)
Fluticasone propionate is a substrate of CYP3A4. The use of strong CYP3A4 inhibitors (e.g., ritonavir, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, ketoconazole, telithromycin, conivaptan, lopinavir, nefazodone, voriconazole) with Fluticasone Propionate Nasal Spray is not recommended because increased systemic corticosteroid adverse effects may occur.
Principal Display Panel 16 G Bottle Box (PRINCIPAL DISPLAY PANEL - 16 g Bottle Box)
5.5 Hypercorticism and Adrenal Suppression
When intranasal corticosteroids are used at higher than recommended dosages or in susceptible individuals at recommended dosages, systemic corticosteroid effects such as hypercorticism and adrenal suppression may appear. If such changes occur, the dosage of Fluticasone Propionate Nasal Spray should be discontinued slowly consistent with accepted procedures for discontinuing oral corticosteroid therapy.
The replacement of a systemic corticosteroid with a topical corticosteroid can be accompanied by signs of adrenal insufficiency. In addition, some patients may experience symptoms of corticosteroid withdrawal (e.g., joint and/or muscular pain, lassitude, depression). Patients previously treated for prolonged periods with systemic corticosteroids and transferred to topical corticosteroids should be carefully monitored for acute adrenal insufficiency in response to stress. In patients who have asthma or other clinical conditions requiring long-term systemic corticosteroid treatment, rapid decreases in systemic corticosteroid dosages may cause a severe exacerbation of their symptoms.
5.3 Hypersensitivity Reactions Including Anaphylaxis (5.3 Hypersensitivity Reactions including Anaphylaxis)
Hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria, contact dermatitis, and rash) have been reported after administration of Fluticasone Propionate Nasal Spray. Discontinue Fluticasone Propionate Nasal Spray if such reactions occur [see Contraindications (4)]. Rarely, immediate hypersensitivity reactions may occur after the administration of Fluticasone Propionate Nasal Spray.
2.2 Adolescents and Children (aged 4 Years and Older) (2.2 Adolescents and Children (Aged 4 Years and Older))
The recommended starting dosage in adolescents and children, aged 4 years and older is 1 spray in each nostril once daily (total daily dose, 100 mcg). Patients not adequately responding to 1 spray in each nostril may use 2 sprays in each nostril once daily (total daily dose, 200 mcg). Once adequate control is achieved, the dosage should be decreased to 1 spray in each nostril once daily.
The maximum total daily dosage should not exceed 2 sprays in each nostril (200 mcg/day). There is no evidence that exceeding the recommended dose is more effective.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Fluticasone propionate demonstrated no tumorigenic potential in mice at oral doses up to 1,000 mcg/kg (approximately 20 times the MRHDID in adults and approximately 10 times the MRHDID in children on a mcg/m 2 basis) for 78 weeks or in rats at inhalation doses up to 57 mcg/kg (approximately 2 times the MRHDID in adults and approximately equivalent to the MRHDID in children on a mcg/m 2 basis) for 104 weeks.
Fluticasone propionate did not induce gene mutation in prokaryotic or eukaryotic cells in vitro. No significant clastogenic effect was seen in cultured human peripheral lymphocytes in vitro or in the mouse micronucleus test.
No evidence of impairment of fertility was observed in male and female rats at subcutaneous doses up to 50 mcg/kg (approximately 2 times the MRHDID in adults on a mcg/m 2 basis). Prostate weight was significantly reduced at a subcutaneous dose of 50 mcg/kg.
5.6 Drug Interactions With Strong Cytochrome P450 3a4 Inhibitors (5.6 Drug Interactions with Strong Cytochrome P450 3A4 Inhibitors)
The use of strong cytochrome P450 3A4 (CYP3A4) inhibitors (e.g., ritonavir, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, ketoconazole, telithromycin, conivaptan, lopinavir, nefazodone, voriconazole) with Fluticasone Propionate Nasal Spray is not recommended because increased systemic corticosteroid adverse effects may occur [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:42:30.536841 · Updated: 2026-03-14T22:09:35.558921