Neutrogena ®
54707f0f-6025-4dc3-8be8-7a38bb1363b8
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Anti-Dandruff, Anti-Seborrheic Dermatitis, Anti-Psoriasis
Medication Information
Purpose
Anti-Dandruff, Anti-Seborrheic Dermatitis, Anti-Psoriasis
Description
Drug Facts
Use
Controls the symptoms of dandruff and seborrheic dermatitis
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if condition worsens or does not improve after regular use of this product as directed.
Section 50567-7
When using this product avoid contact with the eyes. If contact occurs, rinse eyes thoroughly with water.
Section 50569-3
Ask a doctor before use if you have a condition that covers a large area of the body.
Purposes
Anti-Dandruff, Anti-Seborrheic Dermatitis, Anti-Psoriasis
Warnings
For external use only.
Directions
For best results, use at least twice a week or as directed by a physician .
- Wet hair thoroughly.
- Massage a liberal amount into the scalp.
- Leave lather on scalp for several minutes.
- Rinse and repeat.
Questions?
877-256-4247; Outside US, dial collect 215-273-8755 www.neutrogena.com
Active Ingredient
Salicylic Acid (3%)
Other Information
Store at room temperature.
Inactive Ingredients
Water, Sodium C14-16 Olefin Sulfonate, Cocamidopropyl Betaine, Sodium Chloride, Polyquaternium-22, Sodium Citrate, Hexylene Glycol, Sodium Lauroyl Sarcosinate, Linoleamidopropyl PG-Dimonium Chloride Phosphate, Sodium Hydroxide, Citric Acid
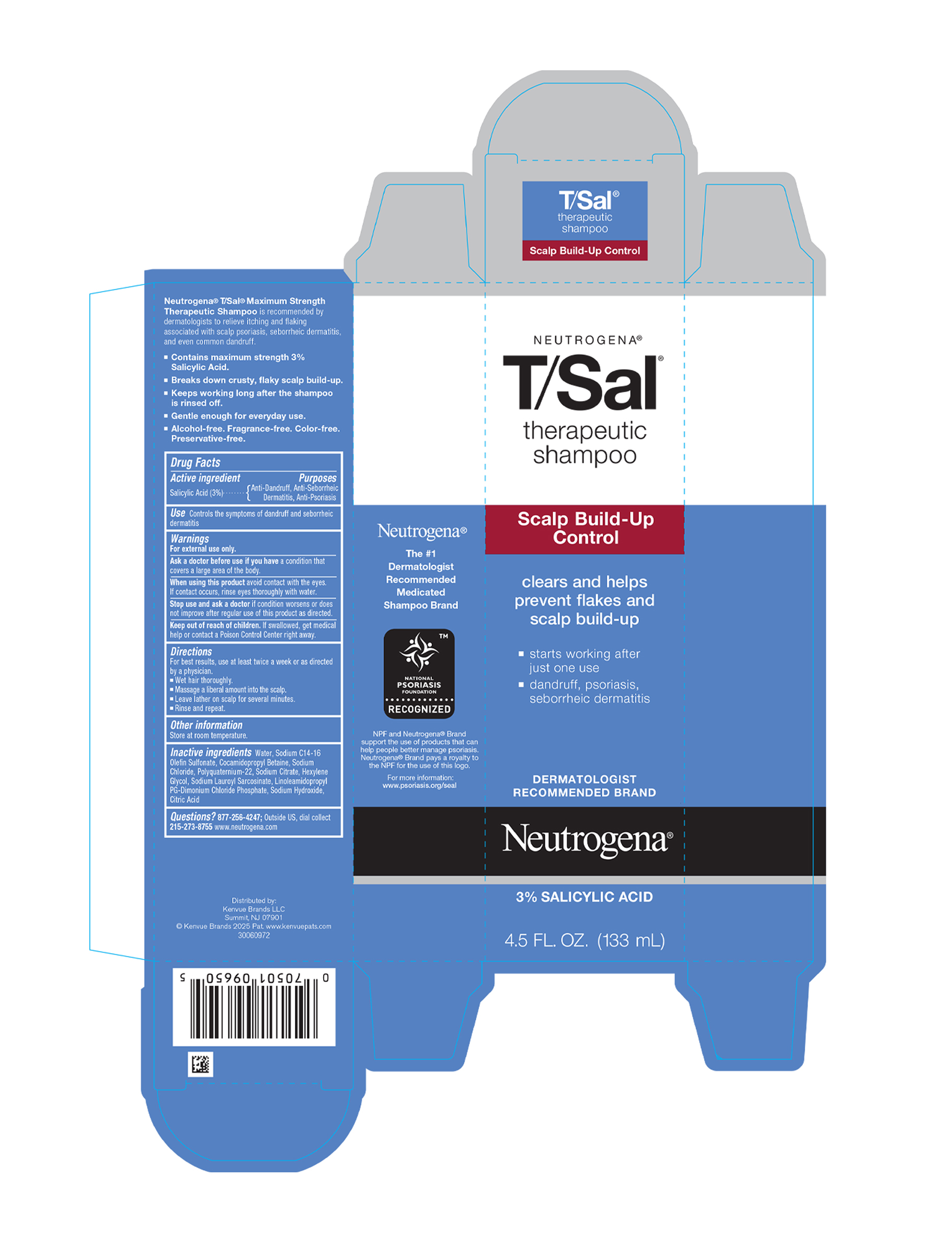
Principal Display Panel 133 Ml Bottle Carton
NEUTROGENA
®
T/Sal
®
therapeutic
shampoo
Scalp Build-Up
Control
clears and helps
prevent flakes and
scalp build-up
- starts working after
just one use - dandruff, psoriasis,
seborrheic dermatitis
DERMATOLOGIST
RECOMMENDED BRAND
Neutrogena
®
3% SALICYLIC ACID
4.5 FL. OZ. (133 mL)
Structured Label Content
Use
Controls the symptoms of dandruff and seborrheic dermatitis
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if condition worsens or does not improve after regular use of this product as directed.
Section 50567-7 (50567-7)
When using this product avoid contact with the eyes. If contact occurs, rinse eyes thoroughly with water.
Section 50569-3 (50569-3)
Ask a doctor before use if you have a condition that covers a large area of the body.
Purposes
Anti-Dandruff, Anti-Seborrheic Dermatitis, Anti-Psoriasis
Warnings
For external use only.
Directions
For best results, use at least twice a week or as directed by a physician .
- Wet hair thoroughly.
- Massage a liberal amount into the scalp.
- Leave lather on scalp for several minutes.
- Rinse and repeat.
Questions?
877-256-4247; Outside US, dial collect 215-273-8755 www.neutrogena.com
Active Ingredient (Active ingredient)
Salicylic Acid (3%)
Other Information (Other information)
Store at room temperature.
Inactive Ingredients (Inactive ingredients)
Water, Sodium C14-16 Olefin Sulfonate, Cocamidopropyl Betaine, Sodium Chloride, Polyquaternium-22, Sodium Citrate, Hexylene Glycol, Sodium Lauroyl Sarcosinate, Linoleamidopropyl PG-Dimonium Chloride Phosphate, Sodium Hydroxide, Citric Acid
Principal Display Panel 133 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 133 mL Bottle Carton)
NEUTROGENA
®
T/Sal
®
therapeutic
shampoo
Scalp Build-Up
Control
clears and helps
prevent flakes and
scalp build-up
- starts working after
just one use - dandruff, psoriasis,
seborrheic dermatitis
DERMATOLOGIST
RECOMMENDED BRAND
Neutrogena
®
3% SALICYLIC ACID
4.5 FL. OZ. (133 mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:17.900719 · Updated: 2026-03-14T23:08:59.288880