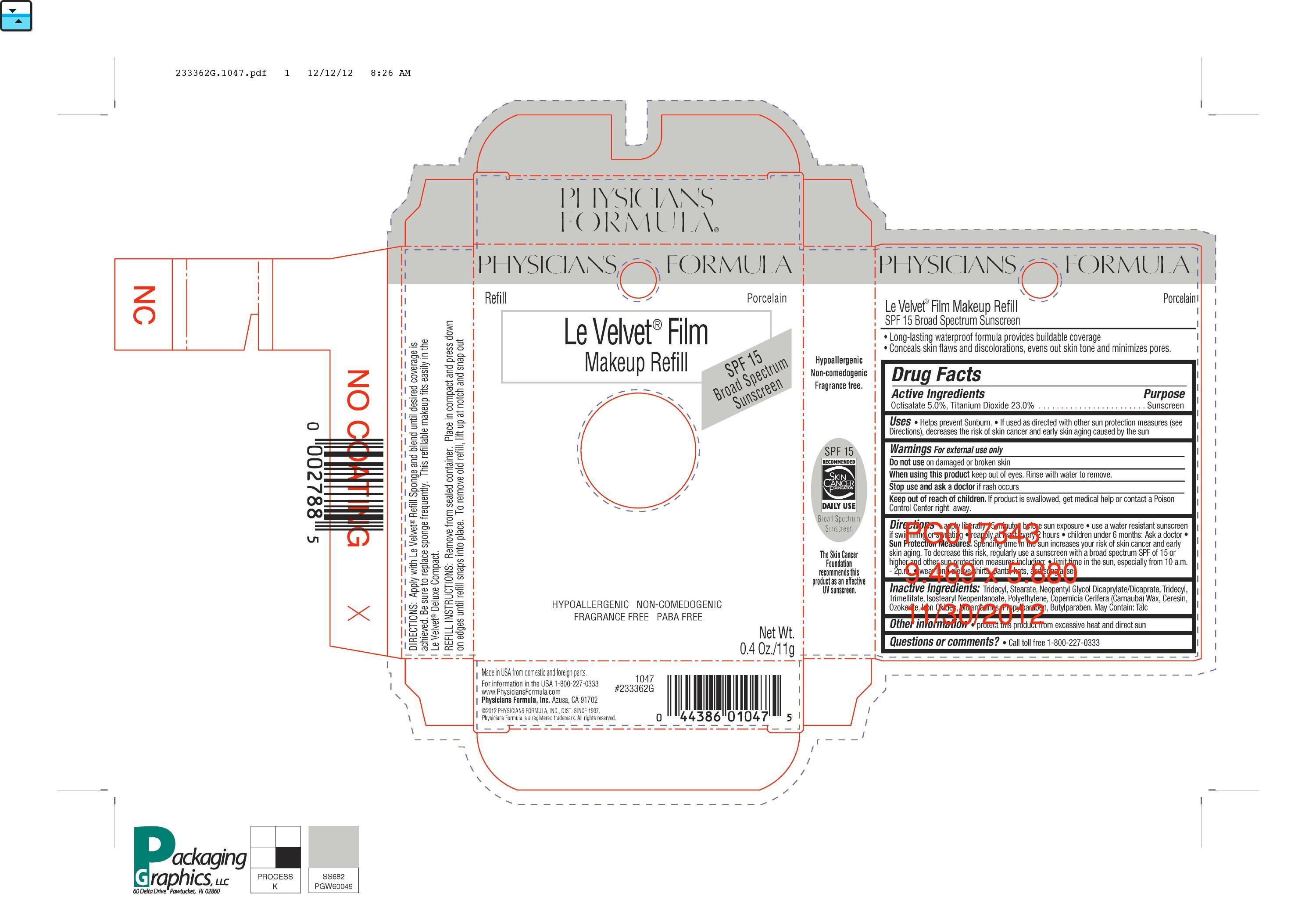
Drug Facts
531e3977-626e-4434-b337-9cdb9f998426
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings : For external use only. Do not use on broken or damaged skin.
Medication Information
Description
Warnings : For external use only. Do not use on broken or damaged skin.
Section 43685-7
Warnings : For external use only. Do not use on broken or damaged skin.
Section 50565-1
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if rash occurs
Section 50567-7
When using this product keep out of eyes. Rinse with water to remove.
Section 51945-4
Section 55106-9
Active Ingredients: Octinoxate 7.5%, Octisalate 4.0%, Oxybenzone 4.0%
Purpose: Sunscreen
Structured Label Content
Section 43685-7 (43685-7)
Warnings : For external use only. Do not use on broken or damaged skin.
Section 50565-1 (50565-1)
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if rash occurs
Section 50567-7 (50567-7)
When using this product keep out of eyes. Rinse with water to remove.
Section 51945-4 (51945-4)
Section 55106-9 (55106-9)
Active Ingredients: Octinoxate 7.5%, Octisalate 4.0%, Oxybenzone 4.0%
Purpose: Sunscreen
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:34.269550 · Updated: 2026-03-14T22:53:21.930902