Desmopressin Acetate Tablets
52133714-37d1-458d-9839-f0bd206711ea
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
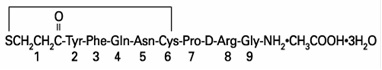
Desmopressin Acetate Tablets (desmopressin acetate) are a synthetic analogue of the natural pituitary hormone 8-arginine vasopressin (ADH), an antidiuretic hormone affecting renal water conservation. It is chemically defined as follows: Mol. Wt. 1183.34 Empirical Formula: C 46 H 64 N 14 O 12 S 2 •C 2 H 4 O 2 •3H 2 O 1-(3-mercaptopropionic acid)-8-D-arginine vasopressin monoacetate (salt) trihydrate. Desmopressin Acetate Tablets contain either 0.1 or 0.2 mg desmopressin acetate. Inactive ingredients include: lactose, potato starch, magnesium stearate and povidone.
Contraindications
Desmopressin Acetate Tablets are contraindicated in individuals with known hypersensitivity to desmopressin acetate or to any of the components of Desmopressin Acetate Tablets . Desmopressin acetate is contraindicated in patients with moderate to severe renal impairment (defined as a creatinine clearance below 50 mL/min). Desmopressin acetate is contraindicated in patients with hyponatremia or a history of hyponatremia.
Adverse Reactions
Infrequently, large doses of the intranasal formulations of desmopressin acetate and Desmopressin Acetate Injection have produced transient headache, nausea, flushing and mild abdominal cramps. These symptoms have disappeared with reduction in dosage.
How Supplied
Desmopressin Acetate 0.2 mg Tablet NDC 71335-2342-1: 90 TABLETs in a BOTTLE Store at Controlled Room Temperature 20 to 25°C (68 to 77°F) [see USP]. Avoid exposure to excessive heat or light. This product should be dispensed in a container with a child-resistant cap. Keep out of the reach of children. Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
Medication Information
Contraindications
Desmopressin Acetate Tablets are contraindicated in individuals with known hypersensitivity to desmopressin acetate or to any of the components of Desmopressin Acetate Tablets.
Desmopressin acetate is contraindicated in patients with moderate to severe renal impairment (defined as a creatinine clearance below 50 mL/min).
Desmopressin acetate is contraindicated in patients with hyponatremia or a history of hyponatremia.
Adverse Reactions
Infrequently, large doses of the intranasal formulations of desmopressin acetate and Desmopressin Acetate Injection have produced transient headache, nausea, flushing and mild abdominal cramps. These symptoms have disappeared with reduction in dosage.
How Supplied
Desmopressin Acetate 0.2 mg Tablet
- NDC 71335-2342-1: 90 TABLETs in a BOTTLE
Store at Controlled Room Temperature 20 to 25°C (68 to 77°F) [see USP]. Avoid exposure to excessive heat or light.
This product should be dispensed in a container with a child-resistant cap.
Keep out of the reach of children.
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
Description
Desmopressin Acetate Tablets (desmopressin acetate) are a synthetic analogue of the natural pituitary hormone 8-arginine vasopressin (ADH), an antidiuretic hormone affecting renal water conservation. It is chemically defined as follows:
Mol. Wt. 1183.34 Empirical Formula: C46H64N14O12S2•C2H4O2•3H2O
1-(3-mercaptopropionic acid)-8-D-arginine vasopressin monoacetate (salt) trihydrate.
Desmopressin Acetate Tablets contain either 0.1 or 0.2 mg desmopressin acetate. Inactive ingredients include: lactose, potato starch, magnesium stearate and povidone.
Section 34072-9
General: Intranasal formulations of desmopressin acetate at high doses and Desmopressin Acetate Injection have infrequently produced a slight elevation of blood pressure which disappears with a reduction of dosage. Although this effect has not been observed when single oral doses up to 0.6 mg have been administered, the drug should be used with caution in patients with coronary artery insufficiency and/or hypertensive cardiovascular disease, because of a possible rise in blood pressure.
Desmopressin acetate should be used with caution in patients with conditions associated with fluid and electrolyte imbalance, such as cystic fibrosis, heart failure and renal disorders because these patients are prone to hyponatremia.
Rare severe allergic reactions have been reported with desmopressin acetate. Anaphylaxis has been reported rarely with intravenous and intranasal administration of desmopressin acetate but not with Desmopressin Acetate Tablets.
Section 34073-7
Drug Interactions: Although the pressor activity of desmopressin acetate is very low compared to its antidiuretic activity, large doses of Desmopressin Acetate Tablets should be used with other pressor agents only with careful patient monitoring. The concomitant administration of drugs that may increase the risk of water intoxication with hyponatremia, (e.g. tricyclic antidepressants, selective serotonin re-uptake inhibitors, chlorpromazine, opiate analgesics, NSAIDs, lamotrigine and carbamazepine) should be performed with caution.
Section 34075-2
Laboratory Tests: Central Diabetes Insipidus: Laboratory tests for monitoring the patient with central diabetes insipidus or post-surgical or head trauma-related polyuria and polydipsia include urine volume and osmolality. In some cases, measurements of plasma osmolality may be useful.
Section 34080-2
Nursing Mothers: There have been no controlled studies in nursing mothers. A single study in postpartum women demonstrated a marked change in plasma, but little if any change in assayable desmopressin acetate in breast milk following an intranasal dose of 0.01 mg.
It is not known whether the drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when desmopressin acetate is administered to nursing mothers.
Section 34081-0
Pediatric Use: Central Diabetes Insipidus: Desmopressin Acetate Tablets have been used safely in pediatric patients, age 4 years and older, with diabetes insipidus for periods up to 44 months. In younger pediatric patients the dose must be individually adjusted in order to prevent an excessive decrease in plasma osmolality leading to hyponatremia and possible convulsions; dosing should start at 0.05 mg (1/2 of the 0.1 mg tablet). Use of Desmopressin Acetate Tablets in pediatric patients requires careful fluid intake restrictions to prevent possible hyponatremia and water intoxication. Fluid restriction should be discussed with the patient and/or guardian. (See WARNINGS.)
Section 34082-8
Geriatric Use: Clinical studies of Desmopressin Acetate Tablets did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function. Desmopressin acetate is contraindicated in patients with moderate to severe renal impairment (defined as a creatinine clearance below 50 mL/min). (See CLINICAL PHARMACOLOGY, Human Pharmacokinetics and CONTRAINDICATIONS .)
Use of Desmopressin Acetate Tablets in geriatric patients requires careful fluid intake restrictions to prevent possible hyponatremia and water intoxication. Fluid restriction should be discussed with the patient. (See WARNINGS .)
Section 34083-6
Carcinogenicity, Mutagenicity, Impairment of Fertility: Studies with desmopressin acetate have not been performed to evaluate carcinogenic potential, mutagenic potential or effects on fertility.
Section 42228-7
Pregnancy: Category B: Fertility studies have not been done. Teratology studies in rats and rabbits at doses from 0.05 to 10 mcg/ kg/day (approximately 0.1 times the maximum systemic human exposure in rats and up to 38 times the maximum systemic human exposure in rabbits based on surface area, mg/m2) revealed no harm to the fetus due to desmopressin acetate. There are, however, no adequate and well-controlled studies in pregnant women. Because animal studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Several publications where desmopressin acetate was used in the management of diabetes insipidus during pregnancy are available; these include a few anecdotal reports of congenital anomalies and low birth weight babies. However, no causal connection between these events and desmopressin acetate has been established. A fifteen year Swedish epidemiologic study of the use of desmopressin acetate in pregnant women with diabetes insipidus found the rate of birth defects to be no greater than that in the general population; however, the statistical power of this study is low. As opposed to preparations containing natural hormones, desmopressin acetate in antidiuretic doses has no uterotonic action and the physician will have to weigh the possible therapeutic advantages against the possible risks in each case.
Section 42229-5
Central Diabetes Insipidus: Dose response studies in patients with diabetes insipidus have demonstrated that oral doses of 0.025 mg to 0.4 mg produced clinically significant antidiuretic effects. In most patients, doses of 0.1 mg to 0.2 mg produced optimal antidiuretic effects lasting up to eight hours. With doses of 0.4 mg, antidiuretic effects were observed for up to 12 hours; measurements beyond 12 hours were not recorded. Increasing oral doses produced dose dependent increases in the plasma levels of desmopressin acetate.
The plasma half-life of desmopressin acetate followed a monoexponential time course with t1/2 values of 1.5 to 2.5 hours which was independent of dose.
The bioavailability of Desmopressin Acetate oral tablets is about 5% compared to intranasal desmopressin acetate, and about 0.16% compared to intravenous desmopressin acetate. The time to reach maximum plasma desmopressin acetate levels ranged from 0.9 to 1.5 hours following oral or intranasal administration, respectively. Following administration of Desmopressin Acetate Tablets, the onset of antidiuretic effect occurs at around 1 hour, and it reaches a maximum at about 4 to 7 hours based on the measurement of increased urine osmolality.
The use of Desmopressin Acetate Tablets in patients with an established diagnosis will result in a reduction in urinary output with an accompanying increase in urine osmolality. These effects usually will allow resumption of a more normal life style, with a decrease in urinary frequency and nocturia.
There are reports of an occasional change in response to the intranasal formulations of desmopressin acetate (Desmopressin Acetate Nasal Spray and Desmopressin Acetate Rhinal Tube). Usually, the change occurred over a period of time greater than six months. This change may be due to decreased responsiveness, or to shortened duration of effect. There is no evidence that this effect is due to the development of binding antibodies, but may be due to a local inactivation of the peptide. No lessening of effect was observed in the 46 patients who were treated with Desmopressin Acetate Tablets for 12 to 44 months and no serum antibodies to desmopressin were detected.
The change in structure of arginine vasopressin to desmopressin acetate resulted in less vasopressor activity and decreased action on visceral smooth muscle relative to enhanced antidiuretic activity. Consequently, clinically effective antidiuretic doses are usually below the threshold for effects on vascular or visceral smooth muscle. In the four long-term studies of Desmopressin Acetate Tablets, no increases in blood pressure in 46 patients receiving Desmopressin Acetate Tablets for periods of 12 to 44 months were reported. In one study, the pharmacodynamic characteristics of Desmopressin Acetate Tablets and intranasal formulation were compared during an 8-hour dosing interval at steady state. The doses administered to 36 hydrated (water loaded) healthy male adult volunteers every 8 hours were 0.1, 0.2, 0.4 mg orally and 0.01 mg intranasally by rhinal tube. The results are shown in the following table:
| Treatment | Total Urine Volume in mL | Maximum Urine Osmolality in mOsm/kg |
|---|---|---|
| (SE) = Standard error of the mean | ||
| 0.1 mg PO q8h | -3689.3 (149.6) | 514.8 (21.9) |
| 0.2 mg PO q8h | -4429.9 (149.6) | 686.3 (21.9) |
| 0.4 mg PO q8h | -4998.8 (149.6) | 769.3 (21.9) |
| 0.01 mg IN q8h | -4844.9 (149.6) | 754.1 (21.9) |
With respect to the mean values of total urine volume decrease and maximum urine osmolality increase from baseline, the 90% confidence limits estimated that the 0.4 mg and 0.2 mg oral dose produced between 95% and 110% and 84% to 99% of pharmacodynamic activity, respectively, when compared to the 0.01 mg intranasal dose.
While both the 0.2 mg and 0.4 mg oral doses are considered pharmacodynamically similar to the 0.01 mg intranasal dose, the pharmacodynamic data on an inter-subject basis was highly variable and, therefore, individual dosing is recommended.
In another study in diabetes insipidus patients, the pharmacodynamic characteristics of Desmopressin Acetate Tablets and intranasal formulations were compared over a 12-hour period. Ten fluid-controlled patients under age 18 were administered tablet doses of 0.2 mg and 0.4 mg, and intranasal doses of 0.01 mg and 0.02 mg.
| Treatment | Urine Volume in mL/min | Maximum Urine Osmolality in mOsm/kg |
|---|---|---|
| (SD) = Standard Deviation | ||
| 0.01 mg IN | 0.3 (0.15) | 717.0 (224.63) |
| 0.02 mg IN | 0.3 (0.25) | 761.8 (298.82) |
| 0.2 mg PO | 0.3 (0.12) | 678.3 (147.91) |
| 0.4 mg PO | 0.2 (0.15) | 787.2 (73.34) |
All four dose formulations (0.01 mg IN, 0.02 mg IN, 0.2 mg PO and 0.4 mg PO) have a similar, pronounced pharmacodynamic effect on urine volume and urine osmolality. At two hours after study drug administration, mean urine volume was 4 mL/min and urine osmolality was >500 mOsm/kg. Mean plasma osmolality remained relatively constant over the time course recorded (0 to 12 hours). A statistical separation from baseline did not occur at any dose or time point. In these patients, the 0.2 mg tablets and the 0.01 mg intranasal spray exhibited similar pharmacodynamic profiles as did the 0.4 mg tablets and the 0.02 mg intranasal spray formulation. In another study of adult diabetes insipidus patients previously controlled on Desmopressin Acetate Intranasal Spray, after one week of self-titration from spray to tablets, patients’ diuresis was controlled with 0.1 mg Desmopressin Acetate Tablets three times a day.
Section 43682-4
Human Pharmacokinetics: Desmopressin acetate is mainly excreted in the urine. A pharmacokinetic study conducted in healthy volunteers and patients with mild, moderate, and severe renal impairment (n=24, 6 subjects in each group) receiving single dose desmopressin acetate (2 mcg) injection demonstrated a difference in desmopressin acetate terminal half-life. Terminal half-life significantly increased from 3 hours in normal healthy patients to 9 hours in patients with severe renal impairment. (See CONTRAINDICATIONS .)
Section 51945-4
Desmopressin Acetate 0.2 mg Tablet
Warnings
1. Very rare cases of hyponatremia have been reported from world-wide postmarketing experience in patients treated with desmopressin acetate. Desmopressin acetate is a potent antidiuretic which, when administered, may lead to water intoxication and/or hyponatremia. Unless properly diagnosed and treated hyponatremia can be fatal. Therefore, fluid restriction is recommended and should be discussed with the patient and/or guardian. Careful medical supervision is required.
2. When Desmopressin Acetate Tablets are administered, in particular in pediatric and geriatric patients, fluid intake should be adjusted downward to decrease the potential occurrence of water intoxication and hyponatremia. (See PRECAUTIONS , Pediatric Use and Geriatric Use.) All patients receiving desmopressin acetate therapy should be observed for the following signs of symptoms associated with hyponatremia: headache, nausea/vomiting, decreased serum sodium, weight gain, restlessness, fatigue, lethargy, disorientation, depressed reflexes, loss of appetite, irritability, muscle weakness, muscle spasms or cramps and abnormal mental status such as hallucinations, decreased consciousness and confusion. Severe symptoms may include one or a combination of the following: seizure, coma and/or respiratory arrest. Particular attention should be paid to the possibility of the rare occurrence of an extreme decrease in plasma osmolality that may result in seizures which could lead to coma.
3. Desmopressin acetate should be used with caution in patients with habitual or psychogenic polydipsia who may be more likely to drink excessive amounts of water, putting them at greater risk of hyponatremia.
Overdosage
Signs of overdose may include confusion, drowsiness, continuing headache, problems with passing urine and rapid weight gain due to fluid retention. (See WARNINGS .) In case of overdose, the dose should be reduced, frequency of administration decreased, or the drug withdrawn according to the severity of the condition. There is no known specific antidote for desmopressin acetate. The patient should be observed and treated with appropriate symptomatic therapy. An oral LD50 has not been established. Oral doses up to 0.2 mg/kg/day have been administered to dogs and rats for 6 months without any significant drug-related toxicities reported. An intravenous dose of 2 mg/kg in mice demonstrated no effect.
Clinical Pharmacology
Desmopressin Acetate Tablets contain as active substance, desmopressin acetate, a synthetic analogue of the natural hormone arginine vasopressin.
Structured Label Content
Section 34072-9 (34072-9)
General: Intranasal formulations of desmopressin acetate at high doses and Desmopressin Acetate Injection have infrequently produced a slight elevation of blood pressure which disappears with a reduction of dosage. Although this effect has not been observed when single oral doses up to 0.6 mg have been administered, the drug should be used with caution in patients with coronary artery insufficiency and/or hypertensive cardiovascular disease, because of a possible rise in blood pressure.
Desmopressin acetate should be used with caution in patients with conditions associated with fluid and electrolyte imbalance, such as cystic fibrosis, heart failure and renal disorders because these patients are prone to hyponatremia.
Rare severe allergic reactions have been reported with desmopressin acetate. Anaphylaxis has been reported rarely with intravenous and intranasal administration of desmopressin acetate but not with Desmopressin Acetate Tablets.
Section 34073-7 (34073-7)
Drug Interactions: Although the pressor activity of desmopressin acetate is very low compared to its antidiuretic activity, large doses of Desmopressin Acetate Tablets should be used with other pressor agents only with careful patient monitoring. The concomitant administration of drugs that may increase the risk of water intoxication with hyponatremia, (e.g. tricyclic antidepressants, selective serotonin re-uptake inhibitors, chlorpromazine, opiate analgesics, NSAIDs, lamotrigine and carbamazepine) should be performed with caution.
Section 34075-2 (34075-2)
Laboratory Tests: Central Diabetes Insipidus: Laboratory tests for monitoring the patient with central diabetes insipidus or post-surgical or head trauma-related polyuria and polydipsia include urine volume and osmolality. In some cases, measurements of plasma osmolality may be useful.
Section 34080-2 (34080-2)
Nursing Mothers: There have been no controlled studies in nursing mothers. A single study in postpartum women demonstrated a marked change in plasma, but little if any change in assayable desmopressin acetate in breast milk following an intranasal dose of 0.01 mg.
It is not known whether the drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when desmopressin acetate is administered to nursing mothers.
Section 34081-0 (34081-0)
Pediatric Use: Central Diabetes Insipidus: Desmopressin Acetate Tablets have been used safely in pediatric patients, age 4 years and older, with diabetes insipidus for periods up to 44 months. In younger pediatric patients the dose must be individually adjusted in order to prevent an excessive decrease in plasma osmolality leading to hyponatremia and possible convulsions; dosing should start at 0.05 mg (1/2 of the 0.1 mg tablet). Use of Desmopressin Acetate Tablets in pediatric patients requires careful fluid intake restrictions to prevent possible hyponatremia and water intoxication. Fluid restriction should be discussed with the patient and/or guardian. (See WARNINGS.)
Section 34082-8 (34082-8)
Geriatric Use: Clinical studies of Desmopressin Acetate Tablets did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function. Desmopressin acetate is contraindicated in patients with moderate to severe renal impairment (defined as a creatinine clearance below 50 mL/min). (See CLINICAL PHARMACOLOGY, Human Pharmacokinetics and CONTRAINDICATIONS .)
Use of Desmopressin Acetate Tablets in geriatric patients requires careful fluid intake restrictions to prevent possible hyponatremia and water intoxication. Fluid restriction should be discussed with the patient. (See WARNINGS .)
Section 34083-6 (34083-6)
Carcinogenicity, Mutagenicity, Impairment of Fertility: Studies with desmopressin acetate have not been performed to evaluate carcinogenic potential, mutagenic potential or effects on fertility.
Section 42228-7 (42228-7)
Pregnancy: Category B: Fertility studies have not been done. Teratology studies in rats and rabbits at doses from 0.05 to 10 mcg/ kg/day (approximately 0.1 times the maximum systemic human exposure in rats and up to 38 times the maximum systemic human exposure in rabbits based on surface area, mg/m2) revealed no harm to the fetus due to desmopressin acetate. There are, however, no adequate and well-controlled studies in pregnant women. Because animal studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Several publications where desmopressin acetate was used in the management of diabetes insipidus during pregnancy are available; these include a few anecdotal reports of congenital anomalies and low birth weight babies. However, no causal connection between these events and desmopressin acetate has been established. A fifteen year Swedish epidemiologic study of the use of desmopressin acetate in pregnant women with diabetes insipidus found the rate of birth defects to be no greater than that in the general population; however, the statistical power of this study is low. As opposed to preparations containing natural hormones, desmopressin acetate in antidiuretic doses has no uterotonic action and the physician will have to weigh the possible therapeutic advantages against the possible risks in each case.
Section 42229-5 (42229-5)
Central Diabetes Insipidus: Dose response studies in patients with diabetes insipidus have demonstrated that oral doses of 0.025 mg to 0.4 mg produced clinically significant antidiuretic effects. In most patients, doses of 0.1 mg to 0.2 mg produced optimal antidiuretic effects lasting up to eight hours. With doses of 0.4 mg, antidiuretic effects were observed for up to 12 hours; measurements beyond 12 hours were not recorded. Increasing oral doses produced dose dependent increases in the plasma levels of desmopressin acetate.
The plasma half-life of desmopressin acetate followed a monoexponential time course with t1/2 values of 1.5 to 2.5 hours which was independent of dose.
The bioavailability of Desmopressin Acetate oral tablets is about 5% compared to intranasal desmopressin acetate, and about 0.16% compared to intravenous desmopressin acetate. The time to reach maximum plasma desmopressin acetate levels ranged from 0.9 to 1.5 hours following oral or intranasal administration, respectively. Following administration of Desmopressin Acetate Tablets, the onset of antidiuretic effect occurs at around 1 hour, and it reaches a maximum at about 4 to 7 hours based on the measurement of increased urine osmolality.
The use of Desmopressin Acetate Tablets in patients with an established diagnosis will result in a reduction in urinary output with an accompanying increase in urine osmolality. These effects usually will allow resumption of a more normal life style, with a decrease in urinary frequency and nocturia.
There are reports of an occasional change in response to the intranasal formulations of desmopressin acetate (Desmopressin Acetate Nasal Spray and Desmopressin Acetate Rhinal Tube). Usually, the change occurred over a period of time greater than six months. This change may be due to decreased responsiveness, or to shortened duration of effect. There is no evidence that this effect is due to the development of binding antibodies, but may be due to a local inactivation of the peptide. No lessening of effect was observed in the 46 patients who were treated with Desmopressin Acetate Tablets for 12 to 44 months and no serum antibodies to desmopressin were detected.
The change in structure of arginine vasopressin to desmopressin acetate resulted in less vasopressor activity and decreased action on visceral smooth muscle relative to enhanced antidiuretic activity. Consequently, clinically effective antidiuretic doses are usually below the threshold for effects on vascular or visceral smooth muscle. In the four long-term studies of Desmopressin Acetate Tablets, no increases in blood pressure in 46 patients receiving Desmopressin Acetate Tablets for periods of 12 to 44 months were reported. In one study, the pharmacodynamic characteristics of Desmopressin Acetate Tablets and intranasal formulation were compared during an 8-hour dosing interval at steady state. The doses administered to 36 hydrated (water loaded) healthy male adult volunteers every 8 hours were 0.1, 0.2, 0.4 mg orally and 0.01 mg intranasally by rhinal tube. The results are shown in the following table:
| Treatment | Total Urine Volume in mL | Maximum Urine Osmolality in mOsm/kg |
|---|---|---|
| (SE) = Standard error of the mean | ||
| 0.1 mg PO q8h | -3689.3 (149.6) | 514.8 (21.9) |
| 0.2 mg PO q8h | -4429.9 (149.6) | 686.3 (21.9) |
| 0.4 mg PO q8h | -4998.8 (149.6) | 769.3 (21.9) |
| 0.01 mg IN q8h | -4844.9 (149.6) | 754.1 (21.9) |
With respect to the mean values of total urine volume decrease and maximum urine osmolality increase from baseline, the 90% confidence limits estimated that the 0.4 mg and 0.2 mg oral dose produced between 95% and 110% and 84% to 99% of pharmacodynamic activity, respectively, when compared to the 0.01 mg intranasal dose.
While both the 0.2 mg and 0.4 mg oral doses are considered pharmacodynamically similar to the 0.01 mg intranasal dose, the pharmacodynamic data on an inter-subject basis was highly variable and, therefore, individual dosing is recommended.
In another study in diabetes insipidus patients, the pharmacodynamic characteristics of Desmopressin Acetate Tablets and intranasal formulations were compared over a 12-hour period. Ten fluid-controlled patients under age 18 were administered tablet doses of 0.2 mg and 0.4 mg, and intranasal doses of 0.01 mg and 0.02 mg.
| Treatment | Urine Volume in mL/min | Maximum Urine Osmolality in mOsm/kg |
|---|---|---|
| (SD) = Standard Deviation | ||
| 0.01 mg IN | 0.3 (0.15) | 717.0 (224.63) |
| 0.02 mg IN | 0.3 (0.25) | 761.8 (298.82) |
| 0.2 mg PO | 0.3 (0.12) | 678.3 (147.91) |
| 0.4 mg PO | 0.2 (0.15) | 787.2 (73.34) |
All four dose formulations (0.01 mg IN, 0.02 mg IN, 0.2 mg PO and 0.4 mg PO) have a similar, pronounced pharmacodynamic effect on urine volume and urine osmolality. At two hours after study drug administration, mean urine volume was 4 mL/min and urine osmolality was >500 mOsm/kg. Mean plasma osmolality remained relatively constant over the time course recorded (0 to 12 hours). A statistical separation from baseline did not occur at any dose or time point. In these patients, the 0.2 mg tablets and the 0.01 mg intranasal spray exhibited similar pharmacodynamic profiles as did the 0.4 mg tablets and the 0.02 mg intranasal spray formulation. In another study of adult diabetes insipidus patients previously controlled on Desmopressin Acetate Intranasal Spray, after one week of self-titration from spray to tablets, patients’ diuresis was controlled with 0.1 mg Desmopressin Acetate Tablets three times a day.
Section 43682-4 (43682-4)
Human Pharmacokinetics: Desmopressin acetate is mainly excreted in the urine. A pharmacokinetic study conducted in healthy volunteers and patients with mild, moderate, and severe renal impairment (n=24, 6 subjects in each group) receiving single dose desmopressin acetate (2 mcg) injection demonstrated a difference in desmopressin acetate terminal half-life. Terminal half-life significantly increased from 3 hours in normal healthy patients to 9 hours in patients with severe renal impairment. (See CONTRAINDICATIONS .)
Section 51945-4 (51945-4)
Desmopressin Acetate 0.2 mg Tablet
Warnings (WARNINGS)
1. Very rare cases of hyponatremia have been reported from world-wide postmarketing experience in patients treated with desmopressin acetate. Desmopressin acetate is a potent antidiuretic which, when administered, may lead to water intoxication and/or hyponatremia. Unless properly diagnosed and treated hyponatremia can be fatal. Therefore, fluid restriction is recommended and should be discussed with the patient and/or guardian. Careful medical supervision is required.
2. When Desmopressin Acetate Tablets are administered, in particular in pediatric and geriatric patients, fluid intake should be adjusted downward to decrease the potential occurrence of water intoxication and hyponatremia. (See PRECAUTIONS , Pediatric Use and Geriatric Use.) All patients receiving desmopressin acetate therapy should be observed for the following signs of symptoms associated with hyponatremia: headache, nausea/vomiting, decreased serum sodium, weight gain, restlessness, fatigue, lethargy, disorientation, depressed reflexes, loss of appetite, irritability, muscle weakness, muscle spasms or cramps and abnormal mental status such as hallucinations, decreased consciousness and confusion. Severe symptoms may include one or a combination of the following: seizure, coma and/or respiratory arrest. Particular attention should be paid to the possibility of the rare occurrence of an extreme decrease in plasma osmolality that may result in seizures which could lead to coma.
3. Desmopressin acetate should be used with caution in patients with habitual or psychogenic polydipsia who may be more likely to drink excessive amounts of water, putting them at greater risk of hyponatremia.
Overdosage (OVERDOSAGE)
Signs of overdose may include confusion, drowsiness, continuing headache, problems with passing urine and rapid weight gain due to fluid retention. (See WARNINGS .) In case of overdose, the dose should be reduced, frequency of administration decreased, or the drug withdrawn according to the severity of the condition. There is no known specific antidote for desmopressin acetate. The patient should be observed and treated with appropriate symptomatic therapy. An oral LD50 has not been established. Oral doses up to 0.2 mg/kg/day have been administered to dogs and rats for 6 months without any significant drug-related toxicities reported. An intravenous dose of 2 mg/kg in mice demonstrated no effect.
Description (DESCRIPTION)
Desmopressin Acetate Tablets (desmopressin acetate) are a synthetic analogue of the natural pituitary hormone 8-arginine vasopressin (ADH), an antidiuretic hormone affecting renal water conservation. It is chemically defined as follows:
Mol. Wt. 1183.34 Empirical Formula: C46H64N14O12S2•C2H4O2•3H2O
1-(3-mercaptopropionic acid)-8-D-arginine vasopressin monoacetate (salt) trihydrate.
Desmopressin Acetate Tablets contain either 0.1 or 0.2 mg desmopressin acetate. Inactive ingredients include: lactose, potato starch, magnesium stearate and povidone.
How Supplied (HOW SUPPLIED)
Desmopressin Acetate 0.2 mg Tablet
- NDC 71335-2342-1: 90 TABLETs in a BOTTLE
Store at Controlled Room Temperature 20 to 25°C (68 to 77°F) [see USP]. Avoid exposure to excessive heat or light.
This product should be dispensed in a container with a child-resistant cap.
Keep out of the reach of children.
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
Adverse Reactions (ADVERSE REACTIONS)
Infrequently, large doses of the intranasal formulations of desmopressin acetate and Desmopressin Acetate Injection have produced transient headache, nausea, flushing and mild abdominal cramps. These symptoms have disappeared with reduction in dosage.
Contraindications (CONTRAINDICATIONS)
Desmopressin Acetate Tablets are contraindicated in individuals with known hypersensitivity to desmopressin acetate or to any of the components of Desmopressin Acetate Tablets.
Desmopressin acetate is contraindicated in patients with moderate to severe renal impairment (defined as a creatinine clearance below 50 mL/min).
Desmopressin acetate is contraindicated in patients with hyponatremia or a history of hyponatremia.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Desmopressin Acetate Tablets contain as active substance, desmopressin acetate, a synthetic analogue of the natural hormone arginine vasopressin.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:56.303346 · Updated: 2026-03-14T22:23:37.577322