saline nasal spray
520e20a3-1e16-4cbd-9da3-948747f4f51d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Moisturizer
Description
Sodium Chloride 0.65%
Medication Information
Warnings
If pregnant or breast-feeding, ask a healthcare professional before use.
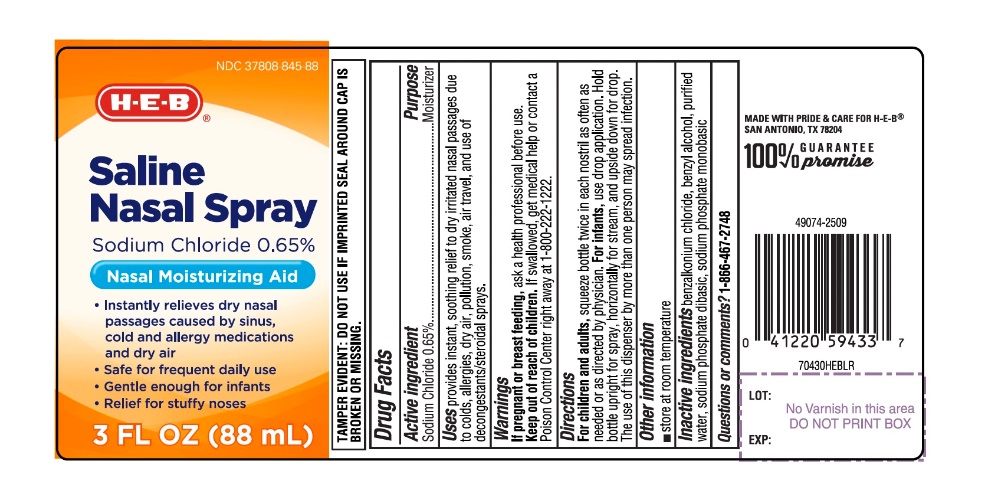
Uses
provides instant, soothing relief dry irritated nasal passages due to colds, allergies, dry air, pollution, smoke, air travel and use of decongestants/steroidal sprays.
Purpose
Moisturizer
Directions
For children and adults,squeeze bottle twice in each nostril as often as needed or as directed by physician. For infants,use drop application. Hold bottle upright for spray, horizontally for stream, and upside down for drop. The use of this dispenser by more than one person may spread infection.
Other Information
- store at room temperature
Inactive Ingredients
benzalkonium chloride, benzyl alcohol, purified water, sodium phosphate dibasic, sodium phosphate monobasic
Description
Sodium Chloride 0.65%
Purpose
Moisturizer
Active Ingredients
Sodium Chloride 0.65%
Saline Nasal Spray
Compare to the active ingredient in Ocean®*
NDC 37808-845-88
Saline Nasal Spray
Sodium Chloride 0.65%
- Instantly relieves dry nasal passages caused by sinus, cold and allergy medications and dry air
- Safe for frequent daily use
- Gentle enough for infants
- Relief for stuffy noses
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SEAL AROUND CAP IS BROKEN OR MISSING.
Distributed by:
Questions Or Comments?
1-866-467-2748
*This product is not manufactured or distributed by Valeant Pharmaceuticals North America LLC, owner of the registered trademark Ocean®.
Package Label for 88 Ml
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away at 1-800-222-1222
Structured Label Content
Warnings
If pregnant or breast-feeding, ask a healthcare professional before use.
Uses
provides instant, soothing relief dry irritated nasal passages due to colds, allergies, dry air, pollution, smoke, air travel and use of decongestants/steroidal sprays.
Directions
For children and adults,squeeze bottle twice in each nostril as often as needed or as directed by physician. For infants,use drop application. Hold bottle upright for spray, horizontally for stream, and upside down for drop. The use of this dispenser by more than one person may spread infection.
Other Information
- store at room temperature
Inactive Ingredients
benzalkonium chloride, benzyl alcohol, purified water, sodium phosphate dibasic, sodium phosphate monobasic
Purpose
Moisturizer
Active Ingredients
Sodium Chloride 0.65%
Saline Nasal Spray
Compare to the active ingredient in Ocean®*
NDC 37808-845-88
Saline Nasal Spray
Sodium Chloride 0.65%
- Instantly relieves dry nasal passages caused by sinus, cold and allergy medications and dry air
- Safe for frequent daily use
- Gentle enough for infants
- Relief for stuffy noses
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SEAL AROUND CAP IS BROKEN OR MISSING.
Distributed by:
Questions Or Comments? (Questions or comments?)
1-866-467-2748
*This product is not manufactured or distributed by Valeant Pharmaceuticals North America LLC, owner of the registered trademark Ocean®.
Package Label for 88 Ml (Package Label for 88 mL)
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away at 1-800-222-1222
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:11.836257 · Updated: 2026-03-14T23:10:24.980101