I.v. Prep Antiseptic Wipe
50f1c74a-bb8e-41c2-a7ee-f2d310a372a7
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Isopropyl Alcohol 70% v/v
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Isopropyl Alcohol 70% v/v
Uses
For preparation of skin prior to injection or venipuncture
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poisin Control Center immediately
Section 51945-4
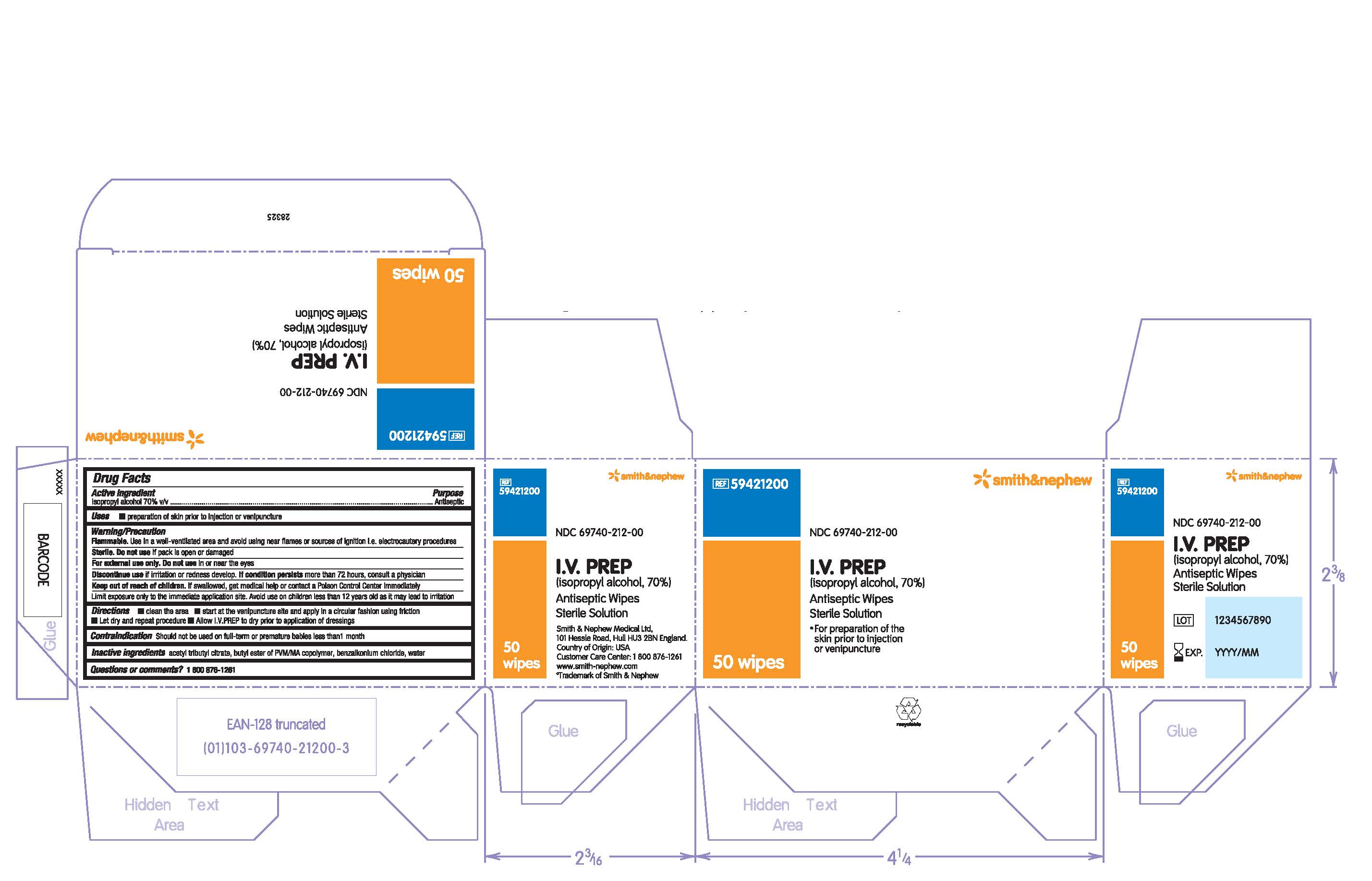
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - BOX OF 50 WIPES (Front)
#59421200
NDC 69740-212-00
I.V. PREP
(isopropyl alcohol, 70%)
Antiseptic Wipes
Sterile Solution
- For preparation of the skin prior to injection or venipuncture
50 Wipes
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - 1 Packet
#59421200
NDC 69740-212-00
I.V. PREP
(isopropyl alcohol, 70%)
Antiseptic Wipe
Sterile Solution
- For preparation of the skin prior to injection or venipuncture
1 Wipe
Made in the USA for Smith & Nephew Medical Ltd
101 Hessle Road, Hull, HU3 2BN, England
Customer Care Center: 1 800 876-1261
www.smith-nephew.com
Section 53413-1
Made in the USA for Smith & Nephew Medical Ltd
101 Hessle Road, Hull, HU3 2BN, England
Customer Care Center: 1 800 876-1261
www.smith-nephew.com
Warnings
- FLAMMABLEUse in a well-ventilated area and avoid using near flames or sources of ignition i.e. electrocautery procedures
- Sterile. Do not useif pack is open or damaged
- For external use only. Do not usein or near the eyes
- Discontinue useif irritation or redness develop. If condition persistsmore than 72 hours, consult a physician
- Limit exposure only to the immediate application site. Avoid use on children less than 12 years old as it may lead to irritation
Directions
- clean the area
- start at the venipuncture site and apply in a circular fashion using friction
- Let dry and repeat procedure
- Allow I.V. PREP to dry prior to application of dressings
Contraindication
Should not be used on full-term or premature babies less than 1 month
Inactive Ingredients
acetyl tributyl citrate, butyl ester of PVM/MA copolymer, benzalkonium chloride, water
Active Ingredients (in Each Packet)
Isopropyl Alcohol 70% v/v
Structured Label Content
Uses (USES)
For preparation of skin prior to injection or venipuncture
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poisin Control Center immediately
Section 51945-4 (51945-4)
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - BOX OF 50 WIPES (Front)
#59421200
NDC 69740-212-00
I.V. PREP
(isopropyl alcohol, 70%)
Antiseptic Wipes
Sterile Solution
- For preparation of the skin prior to injection or venipuncture
50 Wipes
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - 1 Packet
#59421200
NDC 69740-212-00
I.V. PREP
(isopropyl alcohol, 70%)
Antiseptic Wipe
Sterile Solution
- For preparation of the skin prior to injection or venipuncture
1 Wipe
Made in the USA for Smith & Nephew Medical Ltd
101 Hessle Road, Hull, HU3 2BN, England
Customer Care Center: 1 800 876-1261
www.smith-nephew.com
Section 53413-1 (53413-1)
Made in the USA for Smith & Nephew Medical Ltd
101 Hessle Road, Hull, HU3 2BN, England
Customer Care Center: 1 800 876-1261
www.smith-nephew.com
Purpose (PURPOSE)
Antiseptic
Warnings (WARNINGS)
- FLAMMABLEUse in a well-ventilated area and avoid using near flames or sources of ignition i.e. electrocautery procedures
- Sterile. Do not useif pack is open or damaged
- For external use only. Do not usein or near the eyes
- Discontinue useif irritation or redness develop. If condition persistsmore than 72 hours, consult a physician
- Limit exposure only to the immediate application site. Avoid use on children less than 12 years old as it may lead to irritation
Directions (DIRECTIONS)
- clean the area
- start at the venipuncture site and apply in a circular fashion using friction
- Let dry and repeat procedure
- Allow I.V. PREP to dry prior to application of dressings
Contraindication (CONTRAINDICATION)
Should not be used on full-term or premature babies less than 1 month
Inactive Ingredients (INACTIVE INGREDIENTS)
acetyl tributyl citrate, butyl ester of PVM/MA copolymer, benzalkonium chloride, water
Active Ingredients (in Each Packet) (ACTIVE INGREDIENTS (IN EACH PACKET))
Isopropyl Alcohol 70% v/v
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:08.617257 · Updated: 2026-03-14T23:03:01.087200