These Highlights Do Not Include All The Information Needed To Use Etravirine Safely And Effectively. See Full Prescribing Information For Etravirine.
50ca19cb-0f43-4d7b-8c9e-8b3aaded0546
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Etravirine, in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-experienced adult patients and pediatric patients 2 years of age and older [see Microbiology (12.4) and Clinical Studies (14) ] .
Indications and Usage
Etravirine, in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-experienced adult patients and pediatric patients 2 years of age and older [see Microbiology (12.4) and Clinical Studies (14) ] .
Dosage and Administration
Adult patients: 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. ( 2.1 , 2.2 , 2.4 ) Pregnant patients: 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. ( 2.2 ) Pediatric patients (2 years to less than 18 years of age and weighing at least 10 kg): dosage of Etravirine is based on body weight and should not exceed the recommended adult dose. Etravirine tablets should be taken following a meal. ( 2.3 )
Warnings and Precautions
Severe, potentially life threatening and fatal skin reactions have been reported. This includes cases of Stevens-Johnson syndrome, hypersensitivity reaction, toxic epidermal necrolysis and erythema multiforme. Immediately discontinue treatment if severe hypersensitivity, severe rash or rash with systemic symptoms or liver transaminase elevations develops and monitor clinical status, including liver transaminases closely. ( 5.1 ) Monitor for immune reconstitution syndrome and fat redistribution. ( 5.3 , 5.4 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are described in greater detail in other sections: Severe skin and hypersensitivity reactions [see Warnings and Precautions (5.1) ] . Immune reconstitution syndrome [see Warnings and Precautions (5.3) ] .
Drug Interactions
Co-administration of Etravirine with other drugs can alter the concentrations of other drugs and other drugs may alter the concentrations of etravirine. The potential drug-drug interactions must be considered prior to and during therapy. ( 7 , 12.3 )
Storage and Handling
Etravirine 100 mg tablets are supplied as white to off-white, oval tablets containing 100 mg of etravirine. Each tablet is debossed with "TMC125" on one side and "100" on the other side. Etravirine 200 mg tablets are supplied as white to off-white, biconvex, oblong tablets containing 200 mg of etravirine. Each tablet is debossed with "T200" on one side. Etravirine tablets are packaged in bottles in the following configuration: 100 mg tablets—bottles of 120 (NDC 10147-0570-1). Each bottle contains 3 desiccant pouches. 200 mg tablets—bottles of 60 (NDC 10147-0571-1). Each bottle contains 3 desiccant pouches.
How Supplied
Etravirine 100 mg tablets are supplied as white to off-white, oval tablets containing 100 mg of etravirine. Each tablet is debossed with "TMC125" on one side and "100" on the other side. Etravirine 200 mg tablets are supplied as white to off-white, biconvex, oblong tablets containing 200 mg of etravirine. Each tablet is debossed with "T200" on one side. Etravirine tablets are packaged in bottles in the following configuration: 100 mg tablets—bottles of 120 (NDC 10147-0570-1). Each bottle contains 3 desiccant pouches. 200 mg tablets—bottles of 60 (NDC 10147-0571-1). Each bottle contains 3 desiccant pouches.
Medication Information
Warnings and Precautions
Severe, potentially life threatening and fatal skin reactions have been reported. This includes cases of Stevens-Johnson syndrome, hypersensitivity reaction, toxic epidermal necrolysis and erythema multiforme. Immediately discontinue treatment if severe hypersensitivity, severe rash or rash with systemic symptoms or liver transaminase elevations develops and monitor clinical status, including liver transaminases closely. ( 5.1 ) Monitor for immune reconstitution syndrome and fat redistribution. ( 5.3 , 5.4 )
Indications and Usage
Etravirine, in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-experienced adult patients and pediatric patients 2 years of age and older [see Microbiology (12.4) and Clinical Studies (14) ] .
Dosage and Administration
Adult patients: 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. ( 2.1 , 2.2 , 2.4 ) Pregnant patients: 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. ( 2.2 ) Pediatric patients (2 years to less than 18 years of age and weighing at least 10 kg): dosage of Etravirine is based on body weight and should not exceed the recommended adult dose. Etravirine tablets should be taken following a meal. ( 2.3 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are described in greater detail in other sections: Severe skin and hypersensitivity reactions [see Warnings and Precautions (5.1) ] . Immune reconstitution syndrome [see Warnings and Precautions (5.3) ] .
Drug Interactions
Co-administration of Etravirine with other drugs can alter the concentrations of other drugs and other drugs may alter the concentrations of etravirine. The potential drug-drug interactions must be considered prior to and during therapy. ( 7 , 12.3 )
Storage and Handling
Etravirine 100 mg tablets are supplied as white to off-white, oval tablets containing 100 mg of etravirine. Each tablet is debossed with "TMC125" on one side and "100" on the other side. Etravirine 200 mg tablets are supplied as white to off-white, biconvex, oblong tablets containing 200 mg of etravirine. Each tablet is debossed with "T200" on one side. Etravirine tablets are packaged in bottles in the following configuration: 100 mg tablets—bottles of 120 (NDC 10147-0570-1). Each bottle contains 3 desiccant pouches. 200 mg tablets—bottles of 60 (NDC 10147-0571-1). Each bottle contains 3 desiccant pouches.
How Supplied
Etravirine 100 mg tablets are supplied as white to off-white, oval tablets containing 100 mg of etravirine. Each tablet is debossed with "TMC125" on one side and "100" on the other side. Etravirine 200 mg tablets are supplied as white to off-white, biconvex, oblong tablets containing 200 mg of etravirine. Each tablet is debossed with "T200" on one side. Etravirine tablets are packaged in bottles in the following configuration: 100 mg tablets—bottles of 120 (NDC 10147-0570-1). Each bottle contains 3 desiccant pouches. 200 mg tablets—bottles of 60 (NDC 10147-0571-1). Each bottle contains 3 desiccant pouches.
Description
Etravirine, in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-experienced adult patients and pediatric patients 2 years of age and older [see Microbiology (12.4) and Clinical Studies (14) ] .
Section 42229-5
Clinical Trials Experience in Adults
The safety assessment is based on all data from 1203 subjects in the Phase 3 placebo-controlled trials, TMC125-C206 and TMC125-C216, conducted in antiretroviral treatment-experienced HIV-1-infected adult subjects, 599 of whom received Etravirine (200 mg twice daily). In these pooled trials, the median exposure for subjects in the Etravirine arm and placebo arm was 52.3 and 51.0 weeks, respectively. Discontinuations due to adverse drug reactions (ADRs) were 5.2% in the Etravirine arm and 2.6% in the placebo arm.
The most frequently reported ADR at least Grade 2 in severity was rash (10.0%). Stevens-Johnson syndrome, drug hypersensitivity reaction and erythema multiforme were reported in less than 0.1% of subjects during clinical development with Etravirine [see Warnings and Precautions (5.1)] . A total of 2.2% of HIV-1-infected subjects in Phase 3 trials receiving Etravirine discontinued due to rash. In general, in clinical trials, rash was mild to moderate, occurred primarily in the second week of therapy, and was infrequent after Week 4. Rash generally resolved within 1 to 2 weeks on continued therapy. The incidence of rash was higher in women compared to men in the Etravirine arm in the Phase 3 trials (rash ≥ Grade 2 was reported in 9/60 [15.0%] women versus 51/539 [9.5%] men; discontinuations due to rash were reported in 3/60 [5.0%] women versus 10/539 [1.9%] men) [see Warnings and Precautions (5.1)] . Patients with a history of NNRTI-related rash did not appear to be at increased risk for the development of Etravirine-related rash compared to patients without a history of NNRTI-related rash.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Issued: April 2025 | |
|
PATIENT INFORMATION
ETRAVIRINE tablets |
||
| Important: Ask your healthcare provider or pharmacist about medicines that should not be taken with Etravirine. For more information, see the section "What should I tell my healthcare provider before taking Etravirine?" | ||
|
What is Etravirine? Etravirine is a prescription medicine that is used to treat human immunodeficiency virus-1 (HIV-1) infection in combination with other HIV-1 medicines, in adults and children 2 years of age and older who have taken HIV-1 medicines in the past. HIV-1 is the virus that causes AIDS (Acquired Immune Deficiency Syndrome). Etravirine is not recommended for use in children less than 2 years of age. |
||
|
What should I tell my healthcare provider before taking Etravirine? Before taking Etravirine tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some medicines may interact with Etravirine. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine.
|
||
|
How should I take Etravirine?
|
||
|
What are the possible side effects of Etravirine? Etravirine can cause serious side effects including:
If you get a rash with any of the following symptoms, stop taking Etravirine and call your healthcare provider or get medical help right away: |
||
|
|
|
|
Sometimes allergic reactions can affect body organs, such as your liver. Call your healthcare provider right away if you have any of the following signs or symptoms of liver problems: |
||
|
|
|
The most common side effects of Etravirine in adults include rash as well as numbness, tingling or pain in the hands or feet. The most common side effects of Etravirine in children include rash and diarrhea. These are not all the possible side effects of Etravirine. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
How should I store Etravirine?
Keep Etravirine and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of Etravirine Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Etravirine for a condition for which it was not prescribed. Do not give Etravirine to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about Etravirine that is written for health professionals. |
||
|
What are the ingredients in Etravirine? Active ingredient:etravirine. 100 mg Etravirine tablets contain the following inactive ingredients:colloidal silicon dioxide, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, and microcrystalline cellulose. 200 mg Etravirine tablets contain the following inactive ingredients:colloidal silicon dioxide, croscarmellose sodium, hypromellose, magnesium stearate, microcrystalline cellulose, and silicified microcrystalline cellulose. Manufactured for:
For more information, call Patriot Pharmaceuticals, LLC at 1-800-667-8570. |
Section 44425-7
Store Etravirine tablets at 25°C (77°F); with excursions permitted to 15° to 30°C (59° to 86°F) [see USP controlled room temperature]. Store in the original bottle. Keep the bottle tightly closed in order to protect from moisture. Do not remove the desiccant pouches.
Keep Etravirine and all medicines out of the reach of children.
10 Overdosage
There is no specific antidote for overdose with Etravirine. Human experience of overdose with Etravirine is limited. The highest dose studied in healthy volunteers was 400 mg once daily. Treatment of overdose with Etravirine consists of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. Because etravirine is highly protein bound, dialysis is unlikely to result in significant removal of the active substance.
11 Description
Etravirine is a non-nucleoside reverse transcriptase inhibitor (NNRTI) of human immunodeficiency virus type 1 (HIV-1).
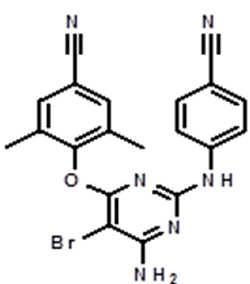
The chemical name for etravirine is 4-[[6-amino-5-bromo-2-[(4-cyanophenyl)amino]-4- pyrimidinyl]oxy]-3,5-dimethylbenzonitrile. Its molecular formula is C 20H 15BrN 6O and its molecular weight is 435.28. Etravirine has the following structural formula:
Etravirine is a white to slightly yellowish-brown powder. Etravirine is practically insoluble in water over a wide pH range. It is very slightly soluble in propylene glycol and slightly soluble in ethanol. Etravirine is soluble in polyethylene glycol (PEG)400 and freely soluble in some organic solvents (e.g., N,N-dimethylformamide and tetrahydrofuran).
Etravirine 100 mg tablets are available as white to off-white, oval tablets for oral administration. Each 100 mg tablet contains 100 mg of etravirine and the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate and microcrystalline cellulose.
Etravirine 200 mg tablets are available as white to off-white, biconvex, oblong tablets for oral administration. Each 200 mg tablet contains 200 mg of etravirine and the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, hypromellose, magnesium stearate, microcrystalline cellulose and silicified microcrystalline cellulose.
8.4 Pediatric Use
The safety and effectiveness of Etravirine have been established for the treatment of HIV-infected pediatric patients from 2 years of age to less than 18 years [see Indications and Usage (1)and Dosage and Administration (2.3)] . Use of Etravirine in pediatric patients 2 years to less than 18 years of age is supported by evidence from adequate and well-controlled studies of Etravirine in adults with additional data from two Phase 2 trials in treatment-experienced pediatric subjects, TMC125-C213, 6 years to less than 18 years of age (N=101) and TMC125-C234/IMPAACT P1090, 2 years to less than 6 years of age (N=20). Both studies were open-label, single arm trials of etravirine plus an optimized background regimen. In clinical trials, the safety, pharmacokinetics, and efficacy were comparable to that observed in adults except for rash (greater than or equal to Grade 2) which was observed more frequently in pediatric subjects [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)] . Postmarketing reports of Stevens-Johnson syndrome in pediatric patients receiving Etravirine have been reported [see Warnings and Precautions (5.1), and Adverse Reactions (6.2)] .
Treatment with Etravirine is not recommended in pediatric patients less than 2 years of age [see Clinical Pharmacology (12.3)] . Five HIV-infected subjects from 1 year to < 2 years of age were enrolled in TMC125-C234/IMPAACT P1090. Etravirine exposure was lower than reported in HIV-infected adults (AUC 12hgeometric mean ratio [90% CI] was 0.59 [0.34, 1.01] for pediatric subjects from 1 year to < 2 years of age compared to adults). Virologic failure at Week 24 (confirmed HIV-RNA greater than or equal to 400 copies/mL) occurred in 3 of 4 evaluable subjects who discontinued before or had reached Week 24. Genotypic and phenotypic resistance to etravirine developed in 1 of the 3 subjects who experienced virologic failure.
8.5 Geriatric Use
Clinical studies of Etravirine did not include sufficient numbers of subjects aged 65 years of age and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)] .
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are described in greater detail in other sections:
- Severe skin and hypersensitivity reactions [see Warnings and Precautions (5.1)] .
- Immune reconstitution syndrome [see Warnings and Precautions (5.3)] .
7 Drug Interactions
8.7 Renal Impairment
Since the renal clearance of etravirine is negligible (less than 1.2%), a decrease in total body clearance is not expected in patients with renal impairment. No dose adjustments are required in patients with renal impairment. As etravirine is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis [see Clinical Pharmacology (12.3)] .
12.3 Pharmacokinetics
The pharmacokinetic properties of Etravirine were determined in healthy adult subjects and in treatment-experienced HIV-1-infected adult and pediatric subjects. The systemic exposures (AUC) to etravirine were lower in HIV-1-infected subjects (Table 5) than in healthy subjects.
| Parameter | Etravirine
N=575 |
|---|---|
| AUC 12h(ng∙h/mL) | |
| Geometric mean ± standard deviation | 4522 ± 4710 |
| Median (range) | 4380 (458–59084) |
| C 0h(ng/mL) | |
| Geometric mean ± standard deviation | 297 ± 391 |
| Median (range) | 298 (2–4852) |
Note: The median protein binding adjusted EC 50for MT4 cells infected with HIV-1/IIIB in vitroequals 4 ng/mL.
5.4 Fat Redistribution
Redistribution/accumulation of body fat, including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
8.6 Hepatic Impairment
No dose adjustment of Etravirine is required in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. The pharmacokinetics of Etravirine have not been evaluated in patients with severe hepatic impairment (Child-Pugh Class C) [see Clinical Pharmacology (12.3)] .
1 Indications and Usage
Etravirine, in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-experienced adult patients and pediatric patients 2 years of age and older [see Microbiology (12.4)and Clinical Studies (14)] .
12.1 Mechanism of Action
Etravirine is an antiretroviral drug [see Microbiology (12.4)] .
5 Warnings and Precautions
- Severe, potentially life threatening and fatal skin reactions have been reported. This includes cases of Stevens-Johnson syndrome, hypersensitivity reaction, toxic epidermal necrolysis and erythema multiforme. Immediately discontinue treatment if severe hypersensitivity, severe rash or rash with systemic symptoms or liver transaminase elevations develops and monitor clinical status, including liver transaminases closely. ( 5.1)
- Monitor for immune reconstitution syndrome and fat redistribution. ( 5.3, 5.4)
2 Dosage and Administration
- Adult patients: 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. ( 2.1, 2.2, 2.4)
- Pregnant patients: 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. ( 2.2)
- Pediatric patients (2 years to less than 18 years of age and weighing at least 10 kg): dosage of Etravirine is based on body weight and should not exceed the recommended adult dose. Etravirine tablets should be taken following a meal. ( 2.3)
2.4 Method of Administration
Instruct patients to swallow the Etravirine tablet(s) whole with liquid such as water. Patients who are unable to swallow the Etravirine tablet(s) whole may disperse the tablet(s) in water. Instruct the patient to do the following:
- place the tablet(s) in 5 mL (1 teaspoon) of water, or at least enough liquid to cover the medication,
- stir well until the water looks milky,
- add approximately 15 mL (1 tablespoon) of liquid. Water may be used but other liquids, such as orange juice or milk, may improve taste. Patients should not place the tablets in orange juice or milk without first adding water. The use of warm (temperature greater than 104°F [greater than 40°C]) or carbonated beverages should be avoided.
- drink the mixture immediately,
- rinse the glass several times with orange juice, milk or water and completely swallow the rinse each time to make sure the patient takes the entire dose.
3 Dosage Forms and Strengths
- 100 mg white to off-white oval tablets debossed with "TMC125" on one side and "100" on the other side.
- 200 mg white to off-white, biconvex, oblong tablets debossed with "T200" on one side.
6.2 Postmarketing Experience
The following events have been identified during postmarketing use of Etravirine. Because these events are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Severe hypersensitivity reactions including DRESS and cases of hepatic failure have been reported [see Warnings and Precautions (5.1)] .
Musculoskeletal and Connective Tissue Disorders: rhabdomyolysis
Skin and Subcutaneous Tissue Disorders: Fatal cases of toxic epidermal necrolysis and Stevens-Johnson syndrome have been reported [see Warnings and Precautions (5.1)] .
8 Use in Specific Populations
- Lactation: Breastfeeding is not recommended due to the potential for HIV-1 transmission. ( 8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
7.3 Significant Drug Interactions
Table 4 shows significant drug interactions based on which, alterations in dose or regimen of Etravirine and/or co-administered drug may be recommended. Drugs that are not recommended for co-administration with Etravirine are also included in Table 4 [see Clinical Pharmacology (12.3)] .
| Concomitant Drug Class:
Drug Name |
Effect on Concentration of Etravirine or Concomitant Drug | Clinical Comment |
|---|---|---|
| ↑ = increase; ↓ = decrease; ↔ = no change | ||
| HIV-antiviral agents: integrase strand inhibitors | ||
| dolutegravir
The interaction between Etravirine and the drug was evaluated in a clinical study. All other drug interactions shown are predicted.
|
↓ dolutegravir
↔ etravirine |
Etravirine significantly reduced plasma concentrations of dolutegravir. Using cross -study comparisons to historical pharmacokinetic data for etravirine, dolutegravir did not appear to affect the pharmacokinetics of etravirine. |
| dolutegravir/darunavir
/ritonavir |
↓ dolutegravir
↔ etravirine |
The effect of etravirine on dolutegravir plasma concentrations was mitigated by co-administration of darunavir/ritonavir or lopinavir/ritonavir, and is expected to be mitigated by atazanavir/ritonavir. Dolutegravir should only be used with Etravirine when co-administered with atazanavir/ritonavir, darunavir/ritonavir, or lopinavir/ritonavir. |
| dolutegravir/lopinavir
/ritonavir |
↔ dolutegravir
↔ etravirine |
|
| HIV-antiviral agents: non-nucleoside reverse transcriptase inhibitors (NNRTIs) | ||
| efavirenz
nevirapine |
↓ etravirine | Combining two NNRTIs has not been shown to be beneficial. Concomitant use of Etravirine with efavirenz or nevirapine may cause a significant decrease in the plasma concentrations of etravirine and loss of therapeutic effect of Etravirine. Co-administration of Etravirine and other NNRTIs is not recommended. |
| delavirdine | ↑ etravirine | Combining two NNRTIs has not been shown to be beneficial. Etravirine and delavirdine should not be co-administered. |
| rilpivirine | ↓ rilpivirine
↔ etravirine |
Combining two NNRTIs has not been shown to be beneficial. Co-administration of Etravirine and rilpivirine is not recommended. |
| HIV-antiviral agents: protease inhibitors (PIs) | ||
| atazanavir
(without ritonavir) |
↓ atazanavir | Co-administration of Etravirine and atazanavir without low-dose ritonavir is not recommended. |
| atazanavir/ritonavir | ↓ atazanavir
↔ etravirine |
Concomitant use of Etravirine with atazanavir/ritonavir decreased atazanavir C minbut it is not considered clinically relevant. The mean systemic exposure (AUC) of etravirine after co-administration of Etravirine with atazanavir/ritonavir in HIV-infected subjects was similar to the mean systemic exposure of etravirine observed in the Phase 3 trials after co-administration of Etravirine and darunavir/ritonavir (as part of the background regimen). Etravirine and atazanavir/ritonavir can be co-administered without dose adjustments. |
| atazanavir/cobicistat | ↓ atazanavir
↓ cobicistat |
Co-administration of Etravirine with atazanavir/cobicistat is not recommended because it may result in loss of therapeutic effect and development of resistance to atazanavir. |
| darunavir/ritonavir | ↓ etravirine | The mean systemic exposure (AUC) of etravirine was reduced when Etravirine was co-administered with darunavir/ritonavir. Because all subjects in the Phase 3 trials received darunavir/ritonavir as part of the background regimen and etravirine exposures from these trials were determined to be safe and effective, Etravirine and darunavir/ritonavir can be co-administered without dose adjustments. |
| darunavir/cobicistat | ↓ cobicistat
darunavir: effect unknown |
Co-administration of Etravirine with darunavir/cobicistat is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. |
| fosamprenavir
(without ritonavir) |
↑ amprenavir | Concomitant use of Etravirine with fosamprenavir without low-dose ritonavir may cause a significant alteration in the plasma concentration of amprenavir. Co-administration of Etravirine and fosamprenavir without low-dose ritonavir is not recommended. |
| fosamprenavir/ritonavir | ↑ amprenavir | Due to a significant increase in the systemic exposure of amprenavir, the appropriate doses of the combination of Etravirine and fosamprenavir/ritonavir have not been established. Co-administration of Etravirine and fosamprenavir/ritonavir is not recommended. |
| indinavir
(without ritonavir) |
↓ indinavir | Concomitant use of Etravirine with indinavir without low-dose ritonavir may cause a significant alteration in the plasma concentration of indinavir. Co-administration of Etravirine and indinavir without low-dose ritonavir is not recommended. |
| lopinavir/ritonavir | ↓ etravirine | The mean systemic exposure (AUC) of etravirine was reduced after co-administration of Etravirine with lopinavir/ritonavir (tablet). Because the reduction in the mean systemic exposures of etravirine in the presence of lopinavir/ritonavir is similar to the reduction in mean systemic exposures of etravirine in the presence of darunavir/ritonavir, Etravirine and lopinavir/ritonavir can be co-administered without dose adjustments. |
| nelfinavir
(without ritonavir) |
↑ nelfinavir | Concomitant use of Etravirine with nelfinavir without low-dose ritonavir may cause a significant alteration in the plasma concentration of nelfinavir. Co-administration of Etravirine and nelfinavir without low-dose ritonavir is not recommended. |
| ritonavir | ↓ etravirine | Concomitant use of Etravirine with ritonavir 600 mg twice daily may cause a significant decrease in the plasma concentration of etravirine and loss of therapeutic effect of Etravirine. Co-administration of Etravirine and ritonavir 600 mg twice daily is not recommended. |
| saquinavir/ritonavir | ↓ etravirine | The mean systemic exposure (AUC) of etravirine was reduced when Etravirine was co-administered with saquinavir/ritonavir. Because the reduction in the mean systemic exposures of etravirine in the presence of saquinavir/ritonavir is similar to the reduction in mean systemic exposures of etravirine in the presence of darunavir/ritonavir, Etravirine and saquinavir/ritonavir can be co-administered without dose adjustments. |
| tipranavir/ritonavir | ↓ etravirine | Concomitant use of Etravirine with tipranavir/ritonavir may cause a significant decrease in the plasma concentrations of etravirine and loss of therapeutic effect of Etravirine. Co-administration of Etravirine and tipranavir/ritonavir is not recommended. |
| CCR5 antagonists | ||
| maraviroc | ↔ etravirine
↓ maraviroc |
When Etravirine is co-administered with maraviroc in the absence of a potent CYP3A inhibitor (e.g., ritonavir boosted protease inhibitor), the recommended dose of maraviroc is 600 mg twice daily. No dose adjustment of Etravirine is needed. |
| maraviroc/darunavir/
ritonavir The reference for etravirine exposure is the pharmacokinetic parameters of etravirine in the presence of darunavir/ritonavir.
|
↔ etravirine
↑ maraviroc |
When Etravirine is co-administered with maraviroc in the presence of a potent CYP3A inhibitor (e.g., ritonavir boosted protease inhibitor), the recommended dose of maraviroc is 150 mg twice daily. No dose adjustment of Etravirine is needed. |
| Other agents | ||
|
Antiarrhythmics:
digoxin |
↔ etravirine
↑ digoxin |
For patients who are initiating a combination of Etravirine and digoxin, the lowest dose of digoxin should initially be prescribed. For patients on a stable digoxin regimen and initiating Etravirine, no dose adjustment of either Etravirine or digoxin is needed. The serum digoxin concentrations should be monitored and used for titration of the digoxin dose to obtain the desired clinical effect. |
| amiodarone
bepridil disopyramide flecainide lidocaine (systemic) mexiletine propafenone quinidine |
↓ antiarrhythmics | Concentrations of these antiarrhythmics may be decreased when co-administered with Etravirine. Etravirine and antiarrhythmics should be co-administered with caution. Drug concentration monitoring is recommended, if available. |
|
Anticoagulant:
warfarin |
↑ anticoagulants | Warfarin concentrations may be increased when co-administered with Etravirine. The international normalized ratio (INR) should be monitored when warfarin is combined with Etravirine. |
|
Anticonvulsants:
carbamazepine phenobarbital phenytoin |
↓ etravirine | Carbamazepine, phenobarbital and phenytoin are inducers of CYP450 enzymes. Etravirine should not be used in combination with carbamazepine, phenobarbital, or phenytoin as co-administration may cause significant decreases in etravirine plasma concentrations and loss of therapeutic effect of Etravirine. |
|
Antifungals:
fluconazole |
↑ etravirine
↔ fluconazole |
Co-administration of etravirine and fluconazole significantly increased etravirine exposures. The amount of safety data at these increased etravirine exposures is limited, therefore, etravirine and fluconazole should be co-administered with caution. No dose adjustment of Etravirine or fluconazole is needed. |
| voriconazole | ↑ voriconazole | Co-administration of etravirine and voriconazole significantly increased etravirine exposures. The amount of safety data at these increased etravirine exposures is limited, therefore, etravirine and voriconazole should be co-administered with caution. No dose adjustment of Etravirine or voriconazole is needed. |
|
Antifungals:
itraconazole ketoconazole posaconazole |
↑ etravirine
↓ itraconazole ↓ ketoconazole ↔ posaconazole |
Posaconazole, a potent inhibitor of CYP3A4, may increase plasma concentrations of etravirine. Itraconazole and ketoconazole are potent inhibitors as well as substrates of CYP3A4. Concomitant systemic use of itraconazole or ketoconazole and Etravirine may increase plasma concentrations of etravirine. Simultaneously, plasma concentrations of itraconazole or ketoconazole may be decreased by Etravirine. Dose adjustments for itraconazole, ketoconazole or posaconazole may be necessary depending on the other co-administered drugs. |
|
Antiinfective:
clarithromycin |
↑ etravirine
↓ clarithromycin ↑ 14-OH-clarithromycin |
Clarithromycin exposure was decreased by Etravirine; however, concentrations of the active metabolite, 14-hydroxy-clarithromycin, were increased. Because 14-hydroxy-clarithromycin has reduced activity against Mycobacterium aviumcomplex (MAC), overall activity against this pathogen may be altered. Alternatives to clarithromycin, such as azithromycin, should be considered for the treatment of MAC. |
|
Antimalarial:
artemether/lumefantrine |
↔ etravirine
↓ artemether ↓ dihydroartemisinin ↓ lumefantrine |
Caution is warranted when co-administering Etravirine and artemether/lumefantrine as it is unknown whether the decrease in exposure of artemether or its active metabolite, dihydroartemisinin, could result in decreased antimalarial efficacy. No dose adjustment is needed for Etravirine. |
|
Antimycobacterials:
rifampin rifapentine |
↓ etravirine | Rifampin and rifapentine are potent inducers of CYP450 enzymes. Etravirine should not be used with rifampin or rifapentine as co-administration may cause significant decreases in etravirine plasma concentrations and loss of therapeutic effect of Etravirine. |
|
Antimycobacterial:
rifabutin |
↓ etravirine
↓ rifabutin ↓ 25- O-desacetylrifabutin |
If Etravirine is NOT co-administered with a protease inhibitor/ritonavir, then rifabutin at a dose of 300 mg once daily is recommended. |
| If Etravirine is co-administered with darunavir/ritonavir, lopinavir/ritonavir or saquinavir/ritonavir, then rifabutin should not be co-administered due to the potential for significant reductions in etravirine exposure. | ||
|
Benzodiazepine:
diazepam |
↑ diazepam | Concomitant use of Etravirine with diazepam may increase plasma concentrations of diazepam. A decrease in diazepam dose may be needed. |
|
Corticosteroid:
dexamethasone (systemic) |
↓ etravirine | Systemic dexamethasone induces CYP3A and can decrease etravirine plasma concentrations. This may result in loss of therapeutic effect of Etravirine. Systemic dexamethasone should be used with caution or alternatives should be considered, particularly for long-term use. |
|
Herbal products:
St. John's wort ( Hypericum perforatum) |
↓ etravirine | Concomitant use of Etravirine with products containing St. John's wort may cause significant decreases in etravirine plasma concentrations and loss of therapeutic effect of Etravirine. Etravirine and products containing St. John's wort should not be co-administered. |
|
Hepatitis C virus (HCV) direct-acting antivirals:
daclatasvir |
↓ daclatasvir | Co-administration of Etravirine with daclatasvir may decrease daclatasvir concentrations. Increase the daclatasvir dose to 90 mg once daily. |
| elbasvir/grazoprevir | ↓ elbasvir
↓ grazoprevir |
Co-administration of Etravirine with elbasvir/grazoprevir may decrease elbasvir and grazoprevir concentrations, leading to reduced therapeutic effect of elbasvir/grazoprevir. Co-administration is not recommended. |
|
HMG-CoA reductase inhibitors:
atorvastatin |
↔ etravirine
↓ atorvastatin ↑ 2-OH-atorvastatin |
The combination of Etravirine and atorvastatin can be given without dose adjustments, however, the dose of atorvastatin may need to be altered based on clinical response. |
| pravastatin
rosuvastatin |
↔ etravirine
↔ pravastatin ↔ rosuvastatin |
No interaction between pravastatin, rosuvastatin and Etravirine is expected. |
| lovastatin
simvastatin |
↓ lovastatin
↓ simvastatin |
Lovastatin and simvastatin are CYP3A substrates and co-administration with Etravirine may result in lower plasma concentrations of the HMG-CoA reductase inhibitor. |
| fluvastatin
pitavastatin |
↑ fluvastatin
↑ pitavastatin |
Fluvastatin and pitavastatin are metabolized by CYP2C9 and co-administration with Etravirine may result in higher plasma concentrations of the HMG-CoA reductase inhibitor. Dose adjustments for these HMG-CoA reductase inhibitors may be necessary. |
|
Immunosuppressants:
cyclosporine sirolimus tacrolimus |
↓ immunosuppressant | Etravirine and systemic immunosuppressants should be co-administered with caution because plasma concentrations of cyclosporine, sirolimus, or tacrolimus may be affected. |
|
Narcotic analgesics/treatment of opioid dependence:
buprenorphine buprenorphine/naloxone methadone |
↔ etravirine
↓ buprenorphine ↔ norbuprenorphine ↔ methadone |
Etravirine and buprenorphine (or buprenorphine/naloxone) can be co-administered without dose adjustments, however, clinical monitoring for withdrawal symptoms is recommended as buprenorphine (or buprenorphine/naloxone) maintenance therapy may need to be adjusted in some patients. |
| Etravirine and methadone can be co-administered without dose adjustments, however, clinical monitoring for withdrawal symptoms is recommended as methadone maintenance therapy may need to be adjusted in some patients. | ||
|
Phosphodiesterase type 5 (PDE-5) inhibitors:
sildenafil tadalafil vardenafil |
↓ sildenafil
↓ N-desmethyl-sildenafil |
Etravirine and sildenafil can be co-administered without dose adjustments, however, the dose of sildenafil may need to be altered based on clinical effect. |
|
Platelet aggregation inhibitors:
clopidogrel |
↓ clopidogrel (active) metabolite | Activation of clopidogrel to its active metabolite may be decreased when clopidogrel is co-administered with Etravirine. Alternatives to clopidogrel should be considered. |
5.3 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including Etravirine. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium aviuminfection, cytomegalovirus, Pneumocystis jirovecipneumonia (PCP) or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
16 How Supplied/storage and Handling
Etravirine 100 mg tablets are supplied as white to off-white, oval tablets containing 100 mg of etravirine. Each tablet is debossed with "TMC125" on one side and "100" on the other side.
Etravirine 200 mg tablets are supplied as white to off-white, biconvex, oblong tablets containing 200 mg of etravirine. Each tablet is debossed with "T200" on one side.
Etravirine tablets are packaged in bottles in the following configuration:
- 100 mg tablets—bottles of 120 (NDC 10147-0570-1). Each bottle contains 3 desiccant pouches.
- 200 mg tablets—bottles of 60 (NDC 10147-0571-1). Each bottle contains 3 desiccant pouches.
2.2 Recommended Dosage During Pregnancy
The recommended oral dosage of Etravirine for pregnant individuals is 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal [see Use in Specific Populations (8.1)] .
2.1 Recommended Dosage in Adult Patients
The recommended oral dosage of Etravirine for adult patients is 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. The type of food does not affect the exposure to Etravirine [see Clinical Pharmacology (12.3)] .
14.1 Treatment Experienced Adult Subjects
The clinical efficacy of Etravirine is derived from the analyses of 48-week data from 2 ongoing, randomized, double-blinded, placebo-controlled, Phase 3 trials, TMC125-C206 and TMC125-C216 (DUET-1 and DUET-2) in subjects with 1 or more NNRTI resistance-associated substitutions. These trials are identical in design and the results below are pooled data from the two trials.
TMC125-C206 and TMC125-C216 are Phase 3 studies designed to evaluate the safety and antiretroviral activity of Etravirine in combination with a background regimen (BR) as compared to placebo in combination with a BR. Eligible subjects were treatment-experienced HIV-1-infected subjects with plasma HIV-1 RNA greater than 5000 copies/mL while on an antiretroviral regimen for at least 8 weeks. In addition, subjects had 1 or more NNRTI resistance-associated substitutions at screening or from prior genotypic analysis, and 3 or more of the following primary PI substitutions at screening: D30N, V32I, L33F, M46I/L, I47A/V, G48V, I50L/V, V82A/F/L/S/T, I84V, N88S, or L90M. Randomization was stratified by the intended use of ENF in the BR, previous use of darunavir/ritonavir, and screening viral load. Virologic response was defined as HIV-1 RNA less than 50 copies/mL at Week 48.
All study subjects received darunavir/ritonavir as part of their BR, and at least 2 other investigator-selected antiretroviral drugs (N[t]RTIs with or without ENF). Of Etravirine-treated subjects, 25.5% used ENF for the first time ( de novo) and 20.0% re-used ENF. Of placebo-treated subjects, 26.5% used de novoENF and 20.4% re-used ENF.
In the pooled analysis for TMC125-C206 and TMC125-C216, demographics and baseline characteristics were balanced between the Etravirine arm and the placebo arm (Table 13). Table 13 displays selected demographic and baseline disease characteristics of the subjects in the Etravirine and placebo arms.
| Etravirine + BR
N=599 |
Placebo + BR
N=604 |
|
|---|---|---|
| RASs = Resistance-Associated Substitutions, BR=background regimen, FC = fold change in EC 50 | ||
| Demographic characteristics | ||
| Median age, years (range) | 46
(18–77) |
45
(18–72) |
| Sex | ||
| Male | 90.0% | 88.6% |
| Female | 10.0% | 11.4% |
| Race | ||
| White | 70.1% | 69.8% |
| Black | 13.2% | 13.0% |
| Hispanic | 11.3% | 12.2% |
| Asian | 1.3% | 0.6% |
| Other | 4.1% | 4.5% |
| Baseline disease characteristics | ||
| Median baseline plasma HIV-1 RNA (range), log 10copies/mL | 4.8
(2.7–6.8) |
4.8
(2.2–6.5) |
| Percentage of subjects with baseline viral load: | ||
| < 30,000 copies/mL | 27.5% | 28.8% |
| ≥ 30,000 copies/mL and < 100,000 copies/mL | 34.4% | 35.3% |
| ≥ 100,000 copies/mL | 38.1% | 35.9% |
| Median baseline CD4+ cell count (range), cells/mm 3 | 99
(1–789) |
109
(0–912) |
| Percentage of subjects with baseline CD4+ cell count: | ||
| < 50 cells/mm 3 | 35.6% | 34.7% |
| ≥ 50 cells/mm 3and < 200 cells/mm 3 | 34.8% | 34.5% |
| ≥ 200 cells/mm 3 | 29.6% | 30.8% |
| Median (range) number of primary PI substitutions
IAS-USA primary PI substitutions [August/September 2007]: D30N, V32I, L33F, M46I/L, I47A/V, G48V, I50L/V, I54L/M, L76V, V82A/F/L/S/T, I84V, N88S, L90M
|
4
(0–7) |
4
(0–8) |
| Percentage of subjects with previous use of NNRTIs: | ||
| 0 | 8.2% | 7.9% |
| 1 | 46.9% | 46.7% |
| > 1 | 44.9% | 45.4% |
| Percentage of subjects with previous use of the following NNRTIs: | 70.3% | 72.5% |
| Efavirenz | 57.1% | 58.6% |
| Nevirapine | 13.7% | 12.6% |
| Delavirdine | ||
| Median (range) number of NNRTI RASs
Tibotec NNRTI RASs [June 2008]: A98G, V90I, L100I, K101E/H/P/Q, K103H/N/S/T, V106A/M/I, V108I, E138A/G/K/Q, V179D/E/F/G/I/T, Y181C/I/V, Y188C/H/L, V189I, G190A/C/E/Q/S, H221Y, P255H, F227C/L, M230I/L, P236L, K238N/T, Y318F
|
2
(0–8) |
2
(0–7) |
| Median fold change of the virus for the following NNRTIs: | ||
| Delavirdine | 27.3 | 26.1 |
| Efavirenz | 63.9 | 45.4 |
| Etravirine | 1.6 | 1.5 |
| Nevirapine | 74.3 | 74.0 |
| Percentage of subjects with previous use of a fusion inhibitor | 39.6% | 42.2% |
| Percentage of subjects with a Phenotypic Sensitivity Score (PSS) for the background therapy
The PSS was calculated for the background therapy (as determined on Day 7). Percentages are based on the number of subjects with available phenotype data. For fusion inhibitors (enfuvirtide), subjects were considered resistant if the drug was used in previous therapy up to baseline. Etravirine is not included in this calculation. of:
|
||
| 0 | 17.0% | 16.2% |
| 1 | 36.5% | 38.7% |
| 2 | 26.9% | 27.8% |
| ≥ 3 | 19.7% | 17.3% |
Efficacy at Week 48 for subjects in the Etravirine and placebo arms for the pooled TMC125-C206 and TMC125-C216 study populations are shown in Table 14.
| Etravirine + BR
N=599 |
Placebo + BR
N=604 |
|
|---|---|---|
| BR=background regimen | ||
| Virologic responders at Week 48
Viral Load < 50 HIV-1 RNA copies/mL |
359 (60%) | 232 (38%) |
| Virologic failures at Week 48
Viral Load ≥ 50 HIV-1 RNA copies/mL |
123 (21%) | 201 (33%) |
| Death | 11 (2%) | 19 (3%) |
| Discontinuations before Week 48: | ||
| due to virologic failures | 58 (10%) | 110 (18%) |
| due to adverse events | 31 (5%) | 14 (2%) |
| due to other reasons | 17 (3%) | 28 (5%) |
At Week 48, 70.8% of Etravirine-treated subjects achieved HIV-1 RNA less than 400 copies/mL as compared to 46.4% of placebo-treated subjects. The mean decrease in plasma HIV-1 RNA from baseline to Week 48 was -2.23 log 10copies/mL for Etravirine-treated subjects and -1.46 log 10copies/mL for placebo-treated subjects. The mean CD4+ cell count increase from baseline for Etravirine-treated subjects was 96 cells/mm 3and 68 cells/mm 3for placebo-treated subjects.
Of the study population who either re-used or did not use ENF, 57.4% of Etravirine-treated subjects and 31.7% of placebo-treated subjects achieved HIV-1 RNA less than 50 copies/mL. Of the study population using ENF de novo,67.3% of Etravirine-treated subjects and 57.2% of placebo-treated subjects achieved HIV-1 RNA less than 50 copies/mL.
Treatment-emergent CDC category C events occurred in 4% of Etravirine-treated subjects and 8.4% of placebo-treated subjects.
Study TMC125-C227 was a randomized, exploratory, active-controlled, open-label, Phase 2b trial. Eligible subjects were treatment-experienced, PI-naïve HIV-1-infected subjects with genotypic evidence of NNRTI resistance at screening or from prior genotypic analysis. The virologic response was evaluated in 116 subjects who were randomized to Etravirine (59 subjects) or an investigator-selected PI (57 subjects), each given with 2 investigator-selected N(t)RTIs. Etravirine-treated subjects had lower antiviral responses associated with reduced susceptibility to the N(t)RTIs and to Etravirine as compared to the control PI-treated subjects.
5.1 Severe Skin and Hypersensitivity Reactions
Severe, potentially life-threatening and fatal skin reactions have been reported. In clinical trials, these include cases of Stevens-Johnson syndrome, toxic epidermal necrolysis and erythema multiforme. Hypersensitivity reactions including Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) have also been reported and were characterized by rash, constitutional findings, and sometimes organ dysfunction, including hepatic failure. In Phase 3 clinical trials, Grade 3 and 4 rashes were reported in 1.3% of subjects receiving Etravirine compared to 0.2% of placebo subjects. A total of 2.2% of HIV-1-infected subjects receiving Etravirine discontinued from Phase 3 trials due to rash [see Adverse Reactions (6.1)] . Rash occurred most commonly during the first 6 weeks of therapy. The incidence of rash was higher in females [see Adverse Reactions (6.1)] . Stevens-Johnson syndrome was reported in 1.1% (2/177) of pediatric patients less than 18 years of age receiving Etravirine in combination with other HIV-1 antiretroviral agents in an observational study.
Discontinue Etravirine immediately if signs or symptoms of severe skin reactions or hypersensitivity reactions develop (including, but not limited to, severe rash or rash accompanied by fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, hepatitis, eosinophilia, angioedema). Clinical status including liver transaminases should be monitored and appropriate therapy initiated. Delay in stopping Etravirine treatment after the onset of severe rash may result in a life-threatening reaction.
7.1 Potential for Other Drugs to Affect Etravirine
Etravirine is a substrate of CYP3A, CYP2C9, and CYP2C19. Therefore, co-administration of Etravirine with drugs that induce or inhibit CYP3A, CYP2C9, and CYP2C19 may alter the therapeutic effect or adverse reaction profile of Etravirine (see Table 4) [see Clinical Pharmacology (12.3)].
7.2 Potential for Etravirine to Affect Other Drugs
Etravirine is an inducer of CYP3A and inhibitor of CYP2C9, CYP2C19 and P-glycoprotein (P-gp). Therefore, co-administration of drugs that are substrates of CYP3A, CYP2C9 and CYP2C19 or are transported by P-gp with Etravirine may alter the therapeutic effect or adverse reaction profile of the co-administered drug(s) (see Table 4) [see Clinical Pharmacology (12.3)].
Principal Display Panel 100 Mg Tablet Bottle Label
120 Tablets
NDC 10147-0570-1
Etravirine
tablets
100 mg
Each tablet contains
100 mg of etravirine.
Rx only
PATRIOT
PHARMACEUTICALS LLC
ALERT: Find out about medicines that
should NOT be taken with Etravirine tablets
from your healthcare provider.
Principal Display Panel 200 Mg Tablet Bottle Label
60 Tablets
NDC 10147-0571-1
Etravirine
tablets
200 mg
Each tablet contains
200 mg of etravirine.
Rx only
PATRIOT
PHARMACEUTICALS LLC
ALERT: Find out about medicines that
should NOT be taken with Etravirine tablets
from your healthcare provider.
7.4 Drugs Without Clinically Significant Interactions With Etravirine
In addition to the drugs included in Table 4, the interaction between Etravirine and the following drugs were evaluated in clinical studies and no dose adjustment is needed for either drug [see Clinical Pharmacology (12.3)] : didanosine, enfuvirtide (ENF), ethinylestradiol/norethindrone, omeprazole, paroxetine, raltegravir, ranitidine, and tenofovir disoproxil fumarate.
2.3 Recommended Dosage in Pediatric Patients (2 Years to Less Than 18 Years of Age)
The recommended dosage of Etravirine for pediatric patients 2 years to less than 18 years of age and weighing at least 10 kg is based on body weight (see Table 1) not exceeding the recommended adult dosage. Etravirine should be taken orally, following a meal. The type of food does not affect the exposure to Etravirine [see Clinical Pharmacology (12.3)] .
| Body Weight kilograms (kg) | Dose |
|---|---|
| greater than or equal to 10 kg to less than 20 kg | 100 mg twice daily |
| greater than or equal to 20 kg to less than 25 kg | 125 mg twice daily |
| greater than or equal to 25 kg to less than 30 kg | 150 mg twice daily |
| greater than or equal to 30 kg | 200 mg twice daily |
14.2 Treatment Experienced Pediatric Subjects (2 Years to Less Than 18 Years of Age)
The efficacy of Etravirine for treatment-experienced pediatric subjects is based on two Phase 2 trials, TMC125-C213 and TMC125-C234/IMPAACT P1090.
5.2 Risk of Adverse Reactions Or Loss of Virologic Response Due to Drug Interactions
The concomitant use of Etravirine and other drugs may result in potentially significant drug interactions, some of which may lead to [see Drug Interactions (7.3)] :
- Loss of therapeutic effect of concomitant drug or Etravirine and possible development of resistance.
- Possible clinically significant adverse reactions from greater exposures of Etravirine or other concomitant drugs.
See Table 4for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during Etravirine therapy and review concomitant medications during Etravirine therapy.
Structured Label Content
Section 42229-5 (42229-5)
Clinical Trials Experience in Adults
The safety assessment is based on all data from 1203 subjects in the Phase 3 placebo-controlled trials, TMC125-C206 and TMC125-C216, conducted in antiretroviral treatment-experienced HIV-1-infected adult subjects, 599 of whom received Etravirine (200 mg twice daily). In these pooled trials, the median exposure for subjects in the Etravirine arm and placebo arm was 52.3 and 51.0 weeks, respectively. Discontinuations due to adverse drug reactions (ADRs) were 5.2% in the Etravirine arm and 2.6% in the placebo arm.
The most frequently reported ADR at least Grade 2 in severity was rash (10.0%). Stevens-Johnson syndrome, drug hypersensitivity reaction and erythema multiforme were reported in less than 0.1% of subjects during clinical development with Etravirine [see Warnings and Precautions (5.1)] . A total of 2.2% of HIV-1-infected subjects in Phase 3 trials receiving Etravirine discontinued due to rash. In general, in clinical trials, rash was mild to moderate, occurred primarily in the second week of therapy, and was infrequent after Week 4. Rash generally resolved within 1 to 2 weeks on continued therapy. The incidence of rash was higher in women compared to men in the Etravirine arm in the Phase 3 trials (rash ≥ Grade 2 was reported in 9/60 [15.0%] women versus 51/539 [9.5%] men; discontinuations due to rash were reported in 3/60 [5.0%] women versus 10/539 [1.9%] men) [see Warnings and Precautions (5.1)] . Patients with a history of NNRTI-related rash did not appear to be at increased risk for the development of Etravirine-related rash compared to patients without a history of NNRTI-related rash.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Issued: April 2025 | |
|
PATIENT INFORMATION
ETRAVIRINE tablets |
||
| Important: Ask your healthcare provider or pharmacist about medicines that should not be taken with Etravirine. For more information, see the section "What should I tell my healthcare provider before taking Etravirine?" | ||
|
What is Etravirine? Etravirine is a prescription medicine that is used to treat human immunodeficiency virus-1 (HIV-1) infection in combination with other HIV-1 medicines, in adults and children 2 years of age and older who have taken HIV-1 medicines in the past. HIV-1 is the virus that causes AIDS (Acquired Immune Deficiency Syndrome). Etravirine is not recommended for use in children less than 2 years of age. |
||
|
What should I tell my healthcare provider before taking Etravirine? Before taking Etravirine tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some medicines may interact with Etravirine. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine.
|
||
|
How should I take Etravirine?
|
||
|
What are the possible side effects of Etravirine? Etravirine can cause serious side effects including:
If you get a rash with any of the following symptoms, stop taking Etravirine and call your healthcare provider or get medical help right away: |
||
|
|
|
|
Sometimes allergic reactions can affect body organs, such as your liver. Call your healthcare provider right away if you have any of the following signs or symptoms of liver problems: |
||
|
|
|
The most common side effects of Etravirine in adults include rash as well as numbness, tingling or pain in the hands or feet. The most common side effects of Etravirine in children include rash and diarrhea. These are not all the possible side effects of Etravirine. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
How should I store Etravirine?
Keep Etravirine and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of Etravirine Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Etravirine for a condition for which it was not prescribed. Do not give Etravirine to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about Etravirine that is written for health professionals. |
||
|
What are the ingredients in Etravirine? Active ingredient:etravirine. 100 mg Etravirine tablets contain the following inactive ingredients:colloidal silicon dioxide, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, and microcrystalline cellulose. 200 mg Etravirine tablets contain the following inactive ingredients:colloidal silicon dioxide, croscarmellose sodium, hypromellose, magnesium stearate, microcrystalline cellulose, and silicified microcrystalline cellulose. Manufactured for:
For more information, call Patriot Pharmaceuticals, LLC at 1-800-667-8570. |
Section 44425-7 (44425-7)
Store Etravirine tablets at 25°C (77°F); with excursions permitted to 15° to 30°C (59° to 86°F) [see USP controlled room temperature]. Store in the original bottle. Keep the bottle tightly closed in order to protect from moisture. Do not remove the desiccant pouches.
Keep Etravirine and all medicines out of the reach of children.
10 Overdosage (10 OVERDOSAGE)
There is no specific antidote for overdose with Etravirine. Human experience of overdose with Etravirine is limited. The highest dose studied in healthy volunteers was 400 mg once daily. Treatment of overdose with Etravirine consists of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. Because etravirine is highly protein bound, dialysis is unlikely to result in significant removal of the active substance.
11 Description (11 DESCRIPTION)
Etravirine is a non-nucleoside reverse transcriptase inhibitor (NNRTI) of human immunodeficiency virus type 1 (HIV-1).
The chemical name for etravirine is 4-[[6-amino-5-bromo-2-[(4-cyanophenyl)amino]-4- pyrimidinyl]oxy]-3,5-dimethylbenzonitrile. Its molecular formula is C 20H 15BrN 6O and its molecular weight is 435.28. Etravirine has the following structural formula:
Etravirine is a white to slightly yellowish-brown powder. Etravirine is practically insoluble in water over a wide pH range. It is very slightly soluble in propylene glycol and slightly soluble in ethanol. Etravirine is soluble in polyethylene glycol (PEG)400 and freely soluble in some organic solvents (e.g., N,N-dimethylformamide and tetrahydrofuran).
Etravirine 100 mg tablets are available as white to off-white, oval tablets for oral administration. Each 100 mg tablet contains 100 mg of etravirine and the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate and microcrystalline cellulose.
Etravirine 200 mg tablets are available as white to off-white, biconvex, oblong tablets for oral administration. Each 200 mg tablet contains 200 mg of etravirine and the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, hypromellose, magnesium stearate, microcrystalline cellulose and silicified microcrystalline cellulose.
8.4 Pediatric Use
The safety and effectiveness of Etravirine have been established for the treatment of HIV-infected pediatric patients from 2 years of age to less than 18 years [see Indications and Usage (1)and Dosage and Administration (2.3)] . Use of Etravirine in pediatric patients 2 years to less than 18 years of age is supported by evidence from adequate and well-controlled studies of Etravirine in adults with additional data from two Phase 2 trials in treatment-experienced pediatric subjects, TMC125-C213, 6 years to less than 18 years of age (N=101) and TMC125-C234/IMPAACT P1090, 2 years to less than 6 years of age (N=20). Both studies were open-label, single arm trials of etravirine plus an optimized background regimen. In clinical trials, the safety, pharmacokinetics, and efficacy were comparable to that observed in adults except for rash (greater than or equal to Grade 2) which was observed more frequently in pediatric subjects [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)] . Postmarketing reports of Stevens-Johnson syndrome in pediatric patients receiving Etravirine have been reported [see Warnings and Precautions (5.1), and Adverse Reactions (6.2)] .
Treatment with Etravirine is not recommended in pediatric patients less than 2 years of age [see Clinical Pharmacology (12.3)] . Five HIV-infected subjects from 1 year to < 2 years of age were enrolled in TMC125-C234/IMPAACT P1090. Etravirine exposure was lower than reported in HIV-infected adults (AUC 12hgeometric mean ratio [90% CI] was 0.59 [0.34, 1.01] for pediatric subjects from 1 year to < 2 years of age compared to adults). Virologic failure at Week 24 (confirmed HIV-RNA greater than or equal to 400 copies/mL) occurred in 3 of 4 evaluable subjects who discontinued before or had reached Week 24. Genotypic and phenotypic resistance to etravirine developed in 1 of the 3 subjects who experienced virologic failure.
8.5 Geriatric Use
Clinical studies of Etravirine did not include sufficient numbers of subjects aged 65 years of age and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)] .
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described in greater detail in other sections:
- Severe skin and hypersensitivity reactions [see Warnings and Precautions (5.1)] .
- Immune reconstitution syndrome [see Warnings and Precautions (5.3)] .
7 Drug Interactions (7 DRUG INTERACTIONS)
8.7 Renal Impairment
Since the renal clearance of etravirine is negligible (less than 1.2%), a decrease in total body clearance is not expected in patients with renal impairment. No dose adjustments are required in patients with renal impairment. As etravirine is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis [see Clinical Pharmacology (12.3)] .
12.3 Pharmacokinetics
The pharmacokinetic properties of Etravirine were determined in healthy adult subjects and in treatment-experienced HIV-1-infected adult and pediatric subjects. The systemic exposures (AUC) to etravirine were lower in HIV-1-infected subjects (Table 5) than in healthy subjects.
| Parameter | Etravirine
N=575 |
|---|---|
| AUC 12h(ng∙h/mL) | |
| Geometric mean ± standard deviation | 4522 ± 4710 |
| Median (range) | 4380 (458–59084) |
| C 0h(ng/mL) | |
| Geometric mean ± standard deviation | 297 ± 391 |
| Median (range) | 298 (2–4852) |
Note: The median protein binding adjusted EC 50for MT4 cells infected with HIV-1/IIIB in vitroequals 4 ng/mL.
5.4 Fat Redistribution
Redistribution/accumulation of body fat, including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
8.6 Hepatic Impairment
No dose adjustment of Etravirine is required in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. The pharmacokinetics of Etravirine have not been evaluated in patients with severe hepatic impairment (Child-Pugh Class C) [see Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
Etravirine, in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-experienced adult patients and pediatric patients 2 years of age and older [see Microbiology (12.4)and Clinical Studies (14)] .
12.1 Mechanism of Action
Etravirine is an antiretroviral drug [see Microbiology (12.4)] .
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Severe, potentially life threatening and fatal skin reactions have been reported. This includes cases of Stevens-Johnson syndrome, hypersensitivity reaction, toxic epidermal necrolysis and erythema multiforme. Immediately discontinue treatment if severe hypersensitivity, severe rash or rash with systemic symptoms or liver transaminase elevations develops and monitor clinical status, including liver transaminases closely. ( 5.1)
- Monitor for immune reconstitution syndrome and fat redistribution. ( 5.3, 5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Adult patients: 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. ( 2.1, 2.2, 2.4)
- Pregnant patients: 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. ( 2.2)
- Pediatric patients (2 years to less than 18 years of age and weighing at least 10 kg): dosage of Etravirine is based on body weight and should not exceed the recommended adult dose. Etravirine tablets should be taken following a meal. ( 2.3)
2.4 Method of Administration
Instruct patients to swallow the Etravirine tablet(s) whole with liquid such as water. Patients who are unable to swallow the Etravirine tablet(s) whole may disperse the tablet(s) in water. Instruct the patient to do the following:
- place the tablet(s) in 5 mL (1 teaspoon) of water, or at least enough liquid to cover the medication,
- stir well until the water looks milky,
- add approximately 15 mL (1 tablespoon) of liquid. Water may be used but other liquids, such as orange juice or milk, may improve taste. Patients should not place the tablets in orange juice or milk without first adding water. The use of warm (temperature greater than 104°F [greater than 40°C]) or carbonated beverages should be avoided.
- drink the mixture immediately,
- rinse the glass several times with orange juice, milk or water and completely swallow the rinse each time to make sure the patient takes the entire dose.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 100 mg white to off-white oval tablets debossed with "TMC125" on one side and "100" on the other side.
- 200 mg white to off-white, biconvex, oblong tablets debossed with "T200" on one side.
6.2 Postmarketing Experience
The following events have been identified during postmarketing use of Etravirine. Because these events are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Severe hypersensitivity reactions including DRESS and cases of hepatic failure have been reported [see Warnings and Precautions (5.1)] .
Musculoskeletal and Connective Tissue Disorders: rhabdomyolysis
Skin and Subcutaneous Tissue Disorders: Fatal cases of toxic epidermal necrolysis and Stevens-Johnson syndrome have been reported [see Warnings and Precautions (5.1)] .
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Breastfeeding is not recommended due to the potential for HIV-1 transmission. ( 8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
7.3 Significant Drug Interactions
Table 4 shows significant drug interactions based on which, alterations in dose or regimen of Etravirine and/or co-administered drug may be recommended. Drugs that are not recommended for co-administration with Etravirine are also included in Table 4 [see Clinical Pharmacology (12.3)] .
| Concomitant Drug Class:
Drug Name |
Effect on Concentration of Etravirine or Concomitant Drug | Clinical Comment |
|---|---|---|
| ↑ = increase; ↓ = decrease; ↔ = no change | ||
| HIV-antiviral agents: integrase strand inhibitors | ||
| dolutegravir
The interaction between Etravirine and the drug was evaluated in a clinical study. All other drug interactions shown are predicted.
|
↓ dolutegravir
↔ etravirine |
Etravirine significantly reduced plasma concentrations of dolutegravir. Using cross -study comparisons to historical pharmacokinetic data for etravirine, dolutegravir did not appear to affect the pharmacokinetics of etravirine. |
| dolutegravir/darunavir
/ritonavir |
↓ dolutegravir
↔ etravirine |
The effect of etravirine on dolutegravir plasma concentrations was mitigated by co-administration of darunavir/ritonavir or lopinavir/ritonavir, and is expected to be mitigated by atazanavir/ritonavir. Dolutegravir should only be used with Etravirine when co-administered with atazanavir/ritonavir, darunavir/ritonavir, or lopinavir/ritonavir. |
| dolutegravir/lopinavir
/ritonavir |
↔ dolutegravir
↔ etravirine |
|
| HIV-antiviral agents: non-nucleoside reverse transcriptase inhibitors (NNRTIs) | ||
| efavirenz
nevirapine |
↓ etravirine | Combining two NNRTIs has not been shown to be beneficial. Concomitant use of Etravirine with efavirenz or nevirapine may cause a significant decrease in the plasma concentrations of etravirine and loss of therapeutic effect of Etravirine. Co-administration of Etravirine and other NNRTIs is not recommended. |
| delavirdine | ↑ etravirine | Combining two NNRTIs has not been shown to be beneficial. Etravirine and delavirdine should not be co-administered. |
| rilpivirine | ↓ rilpivirine
↔ etravirine |
Combining two NNRTIs has not been shown to be beneficial. Co-administration of Etravirine and rilpivirine is not recommended. |
| HIV-antiviral agents: protease inhibitors (PIs) | ||
| atazanavir
(without ritonavir) |
↓ atazanavir | Co-administration of Etravirine and atazanavir without low-dose ritonavir is not recommended. |
| atazanavir/ritonavir | ↓ atazanavir
↔ etravirine |
Concomitant use of Etravirine with atazanavir/ritonavir decreased atazanavir C minbut it is not considered clinically relevant. The mean systemic exposure (AUC) of etravirine after co-administration of Etravirine with atazanavir/ritonavir in HIV-infected subjects was similar to the mean systemic exposure of etravirine observed in the Phase 3 trials after co-administration of Etravirine and darunavir/ritonavir (as part of the background regimen). Etravirine and atazanavir/ritonavir can be co-administered without dose adjustments. |
| atazanavir/cobicistat | ↓ atazanavir
↓ cobicistat |
Co-administration of Etravirine with atazanavir/cobicistat is not recommended because it may result in loss of therapeutic effect and development of resistance to atazanavir. |
| darunavir/ritonavir | ↓ etravirine | The mean systemic exposure (AUC) of etravirine was reduced when Etravirine was co-administered with darunavir/ritonavir. Because all subjects in the Phase 3 trials received darunavir/ritonavir as part of the background regimen and etravirine exposures from these trials were determined to be safe and effective, Etravirine and darunavir/ritonavir can be co-administered without dose adjustments. |
| darunavir/cobicistat | ↓ cobicistat
darunavir: effect unknown |
Co-administration of Etravirine with darunavir/cobicistat is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. |
| fosamprenavir
(without ritonavir) |
↑ amprenavir | Concomitant use of Etravirine with fosamprenavir without low-dose ritonavir may cause a significant alteration in the plasma concentration of amprenavir. Co-administration of Etravirine and fosamprenavir without low-dose ritonavir is not recommended. |
| fosamprenavir/ritonavir | ↑ amprenavir | Due to a significant increase in the systemic exposure of amprenavir, the appropriate doses of the combination of Etravirine and fosamprenavir/ritonavir have not been established. Co-administration of Etravirine and fosamprenavir/ritonavir is not recommended. |
| indinavir
(without ritonavir) |
↓ indinavir | Concomitant use of Etravirine with indinavir without low-dose ritonavir may cause a significant alteration in the plasma concentration of indinavir. Co-administration of Etravirine and indinavir without low-dose ritonavir is not recommended. |
| lopinavir/ritonavir | ↓ etravirine | The mean systemic exposure (AUC) of etravirine was reduced after co-administration of Etravirine with lopinavir/ritonavir (tablet). Because the reduction in the mean systemic exposures of etravirine in the presence of lopinavir/ritonavir is similar to the reduction in mean systemic exposures of etravirine in the presence of darunavir/ritonavir, Etravirine and lopinavir/ritonavir can be co-administered without dose adjustments. |
| nelfinavir
(without ritonavir) |
↑ nelfinavir | Concomitant use of Etravirine with nelfinavir without low-dose ritonavir may cause a significant alteration in the plasma concentration of nelfinavir. Co-administration of Etravirine and nelfinavir without low-dose ritonavir is not recommended. |
| ritonavir | ↓ etravirine | Concomitant use of Etravirine with ritonavir 600 mg twice daily may cause a significant decrease in the plasma concentration of etravirine and loss of therapeutic effect of Etravirine. Co-administration of Etravirine and ritonavir 600 mg twice daily is not recommended. |
| saquinavir/ritonavir | ↓ etravirine | The mean systemic exposure (AUC) of etravirine was reduced when Etravirine was co-administered with saquinavir/ritonavir. Because the reduction in the mean systemic exposures of etravirine in the presence of saquinavir/ritonavir is similar to the reduction in mean systemic exposures of etravirine in the presence of darunavir/ritonavir, Etravirine and saquinavir/ritonavir can be co-administered without dose adjustments. |
| tipranavir/ritonavir | ↓ etravirine | Concomitant use of Etravirine with tipranavir/ritonavir may cause a significant decrease in the plasma concentrations of etravirine and loss of therapeutic effect of Etravirine. Co-administration of Etravirine and tipranavir/ritonavir is not recommended. |
| CCR5 antagonists | ||
| maraviroc | ↔ etravirine
↓ maraviroc |
When Etravirine is co-administered with maraviroc in the absence of a potent CYP3A inhibitor (e.g., ritonavir boosted protease inhibitor), the recommended dose of maraviroc is 600 mg twice daily. No dose adjustment of Etravirine is needed. |
| maraviroc/darunavir/
ritonavir The reference for etravirine exposure is the pharmacokinetic parameters of etravirine in the presence of darunavir/ritonavir.
|
↔ etravirine
↑ maraviroc |
When Etravirine is co-administered with maraviroc in the presence of a potent CYP3A inhibitor (e.g., ritonavir boosted protease inhibitor), the recommended dose of maraviroc is 150 mg twice daily. No dose adjustment of Etravirine is needed. |
| Other agents | ||
|
Antiarrhythmics:
digoxin |
↔ etravirine
↑ digoxin |
For patients who are initiating a combination of Etravirine and digoxin, the lowest dose of digoxin should initially be prescribed. For patients on a stable digoxin regimen and initiating Etravirine, no dose adjustment of either Etravirine or digoxin is needed. The serum digoxin concentrations should be monitored and used for titration of the digoxin dose to obtain the desired clinical effect. |
| amiodarone
bepridil disopyramide flecainide lidocaine (systemic) mexiletine propafenone quinidine |
↓ antiarrhythmics | Concentrations of these antiarrhythmics may be decreased when co-administered with Etravirine. Etravirine and antiarrhythmics should be co-administered with caution. Drug concentration monitoring is recommended, if available. |
|
Anticoagulant:
warfarin |
↑ anticoagulants | Warfarin concentrations may be increased when co-administered with Etravirine. The international normalized ratio (INR) should be monitored when warfarin is combined with Etravirine. |
|
Anticonvulsants:
carbamazepine phenobarbital phenytoin |
↓ etravirine | Carbamazepine, phenobarbital and phenytoin are inducers of CYP450 enzymes. Etravirine should not be used in combination with carbamazepine, phenobarbital, or phenytoin as co-administration may cause significant decreases in etravirine plasma concentrations and loss of therapeutic effect of Etravirine. |
|
Antifungals:
fluconazole |
↑ etravirine
↔ fluconazole |
Co-administration of etravirine and fluconazole significantly increased etravirine exposures. The amount of safety data at these increased etravirine exposures is limited, therefore, etravirine and fluconazole should be co-administered with caution. No dose adjustment of Etravirine or fluconazole is needed. |
| voriconazole | ↑ voriconazole | Co-administration of etravirine and voriconazole significantly increased etravirine exposures. The amount of safety data at these increased etravirine exposures is limited, therefore, etravirine and voriconazole should be co-administered with caution. No dose adjustment of Etravirine or voriconazole is needed. |
|
Antifungals:
itraconazole ketoconazole posaconazole |
↑ etravirine
↓ itraconazole ↓ ketoconazole ↔ posaconazole |
Posaconazole, a potent inhibitor of CYP3A4, may increase plasma concentrations of etravirine. Itraconazole and ketoconazole are potent inhibitors as well as substrates of CYP3A4. Concomitant systemic use of itraconazole or ketoconazole and Etravirine may increase plasma concentrations of etravirine. Simultaneously, plasma concentrations of itraconazole or ketoconazole may be decreased by Etravirine. Dose adjustments for itraconazole, ketoconazole or posaconazole may be necessary depending on the other co-administered drugs. |
|
Antiinfective:
clarithromycin |
↑ etravirine
↓ clarithromycin ↑ 14-OH-clarithromycin |
Clarithromycin exposure was decreased by Etravirine; however, concentrations of the active metabolite, 14-hydroxy-clarithromycin, were increased. Because 14-hydroxy-clarithromycin has reduced activity against Mycobacterium aviumcomplex (MAC), overall activity against this pathogen may be altered. Alternatives to clarithromycin, such as azithromycin, should be considered for the treatment of MAC. |
|
Antimalarial:
artemether/lumefantrine |
↔ etravirine
↓ artemether ↓ dihydroartemisinin ↓ lumefantrine |
Caution is warranted when co-administering Etravirine and artemether/lumefantrine as it is unknown whether the decrease in exposure of artemether or its active metabolite, dihydroartemisinin, could result in decreased antimalarial efficacy. No dose adjustment is needed for Etravirine. |
|
Antimycobacterials:
rifampin rifapentine |
↓ etravirine | Rifampin and rifapentine are potent inducers of CYP450 enzymes. Etravirine should not be used with rifampin or rifapentine as co-administration may cause significant decreases in etravirine plasma concentrations and loss of therapeutic effect of Etravirine. |
|
Antimycobacterial:
rifabutin |
↓ etravirine
↓ rifabutin ↓ 25- O-desacetylrifabutin |
If Etravirine is NOT co-administered with a protease inhibitor/ritonavir, then rifabutin at a dose of 300 mg once daily is recommended. |
| If Etravirine is co-administered with darunavir/ritonavir, lopinavir/ritonavir or saquinavir/ritonavir, then rifabutin should not be co-administered due to the potential for significant reductions in etravirine exposure. | ||
|
Benzodiazepine:
diazepam |
↑ diazepam | Concomitant use of Etravirine with diazepam may increase plasma concentrations of diazepam. A decrease in diazepam dose may be needed. |
|
Corticosteroid:
dexamethasone (systemic) |
↓ etravirine | Systemic dexamethasone induces CYP3A and can decrease etravirine plasma concentrations. This may result in loss of therapeutic effect of Etravirine. Systemic dexamethasone should be used with caution or alternatives should be considered, particularly for long-term use. |
|
Herbal products:
St. John's wort ( Hypericum perforatum) |
↓ etravirine | Concomitant use of Etravirine with products containing St. John's wort may cause significant decreases in etravirine plasma concentrations and loss of therapeutic effect of Etravirine. Etravirine and products containing St. John's wort should not be co-administered. |
|
Hepatitis C virus (HCV) direct-acting antivirals:
daclatasvir |
↓ daclatasvir | Co-administration of Etravirine with daclatasvir may decrease daclatasvir concentrations. Increase the daclatasvir dose to 90 mg once daily. |
| elbasvir/grazoprevir | ↓ elbasvir
↓ grazoprevir |
Co-administration of Etravirine with elbasvir/grazoprevir may decrease elbasvir and grazoprevir concentrations, leading to reduced therapeutic effect of elbasvir/grazoprevir. Co-administration is not recommended. |
|
HMG-CoA reductase inhibitors:
atorvastatin |
↔ etravirine
↓ atorvastatin ↑ 2-OH-atorvastatin |
The combination of Etravirine and atorvastatin can be given without dose adjustments, however, the dose of atorvastatin may need to be altered based on clinical response. |
| pravastatin
rosuvastatin |
↔ etravirine
↔ pravastatin ↔ rosuvastatin |
No interaction between pravastatin, rosuvastatin and Etravirine is expected. |
| lovastatin
simvastatin |
↓ lovastatin
↓ simvastatin |
Lovastatin and simvastatin are CYP3A substrates and co-administration with Etravirine may result in lower plasma concentrations of the HMG-CoA reductase inhibitor. |
| fluvastatin
pitavastatin |
↑ fluvastatin
↑ pitavastatin |
Fluvastatin and pitavastatin are metabolized by CYP2C9 and co-administration with Etravirine may result in higher plasma concentrations of the HMG-CoA reductase inhibitor. Dose adjustments for these HMG-CoA reductase inhibitors may be necessary. |
|
Immunosuppressants:
cyclosporine sirolimus tacrolimus |
↓ immunosuppressant | Etravirine and systemic immunosuppressants should be co-administered with caution because plasma concentrations of cyclosporine, sirolimus, or tacrolimus may be affected. |
|
Narcotic analgesics/treatment of opioid dependence:
buprenorphine buprenorphine/naloxone methadone |
↔ etravirine
↓ buprenorphine ↔ norbuprenorphine ↔ methadone |
Etravirine and buprenorphine (or buprenorphine/naloxone) can be co-administered without dose adjustments, however, clinical monitoring for withdrawal symptoms is recommended as buprenorphine (or buprenorphine/naloxone) maintenance therapy may need to be adjusted in some patients. |
| Etravirine and methadone can be co-administered without dose adjustments, however, clinical monitoring for withdrawal symptoms is recommended as methadone maintenance therapy may need to be adjusted in some patients. | ||
|
Phosphodiesterase type 5 (PDE-5) inhibitors:
sildenafil tadalafil vardenafil |
↓ sildenafil
↓ N-desmethyl-sildenafil |
Etravirine and sildenafil can be co-administered without dose adjustments, however, the dose of sildenafil may need to be altered based on clinical effect. |
|
Platelet aggregation inhibitors:
clopidogrel |
↓ clopidogrel (active) metabolite | Activation of clopidogrel to its active metabolite may be decreased when clopidogrel is co-administered with Etravirine. Alternatives to clopidogrel should be considered. |
5.3 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including Etravirine. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium aviuminfection, cytomegalovirus, Pneumocystis jirovecipneumonia (PCP) or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Etravirine 100 mg tablets are supplied as white to off-white, oval tablets containing 100 mg of etravirine. Each tablet is debossed with "TMC125" on one side and "100" on the other side.
Etravirine 200 mg tablets are supplied as white to off-white, biconvex, oblong tablets containing 200 mg of etravirine. Each tablet is debossed with "T200" on one side.
Etravirine tablets are packaged in bottles in the following configuration:
- 100 mg tablets—bottles of 120 (NDC 10147-0570-1). Each bottle contains 3 desiccant pouches.
- 200 mg tablets—bottles of 60 (NDC 10147-0571-1). Each bottle contains 3 desiccant pouches.
2.2 Recommended Dosage During Pregnancy
The recommended oral dosage of Etravirine for pregnant individuals is 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal [see Use in Specific Populations (8.1)] .
2.1 Recommended Dosage in Adult Patients
The recommended oral dosage of Etravirine for adult patients is 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. The type of food does not affect the exposure to Etravirine [see Clinical Pharmacology (12.3)] .
14.1 Treatment Experienced Adult Subjects (14.1 Treatment-Experienced Adult Subjects)
The clinical efficacy of Etravirine is derived from the analyses of 48-week data from 2 ongoing, randomized, double-blinded, placebo-controlled, Phase 3 trials, TMC125-C206 and TMC125-C216 (DUET-1 and DUET-2) in subjects with 1 or more NNRTI resistance-associated substitutions. These trials are identical in design and the results below are pooled data from the two trials.
TMC125-C206 and TMC125-C216 are Phase 3 studies designed to evaluate the safety and antiretroviral activity of Etravirine in combination with a background regimen (BR) as compared to placebo in combination with a BR. Eligible subjects were treatment-experienced HIV-1-infected subjects with plasma HIV-1 RNA greater than 5000 copies/mL while on an antiretroviral regimen for at least 8 weeks. In addition, subjects had 1 or more NNRTI resistance-associated substitutions at screening or from prior genotypic analysis, and 3 or more of the following primary PI substitutions at screening: D30N, V32I, L33F, M46I/L, I47A/V, G48V, I50L/V, V82A/F/L/S/T, I84V, N88S, or L90M. Randomization was stratified by the intended use of ENF in the BR, previous use of darunavir/ritonavir, and screening viral load. Virologic response was defined as HIV-1 RNA less than 50 copies/mL at Week 48.
All study subjects received darunavir/ritonavir as part of their BR, and at least 2 other investigator-selected antiretroviral drugs (N[t]RTIs with or without ENF). Of Etravirine-treated subjects, 25.5% used ENF for the first time ( de novo) and 20.0% re-used ENF. Of placebo-treated subjects, 26.5% used de novoENF and 20.4% re-used ENF.
In the pooled analysis for TMC125-C206 and TMC125-C216, demographics and baseline characteristics were balanced between the Etravirine arm and the placebo arm (Table 13). Table 13 displays selected demographic and baseline disease characteristics of the subjects in the Etravirine and placebo arms.
| Etravirine + BR
N=599 |
Placebo + BR
N=604 |
|
|---|---|---|
| RASs = Resistance-Associated Substitutions, BR=background regimen, FC = fold change in EC 50 | ||
| Demographic characteristics | ||
| Median age, years (range) | 46
(18–77) |
45
(18–72) |
| Sex | ||
| Male | 90.0% | 88.6% |
| Female | 10.0% | 11.4% |
| Race | ||
| White | 70.1% | 69.8% |
| Black | 13.2% | 13.0% |
| Hispanic | 11.3% | 12.2% |
| Asian | 1.3% | 0.6% |
| Other | 4.1% | 4.5% |
| Baseline disease characteristics | ||
| Median baseline plasma HIV-1 RNA (range), log 10copies/mL | 4.8
(2.7–6.8) |
4.8
(2.2–6.5) |
| Percentage of subjects with baseline viral load: | ||
| < 30,000 copies/mL | 27.5% | 28.8% |
| ≥ 30,000 copies/mL and < 100,000 copies/mL | 34.4% | 35.3% |
| ≥ 100,000 copies/mL | 38.1% | 35.9% |
| Median baseline CD4+ cell count (range), cells/mm 3 | 99
(1–789) |
109
(0–912) |
| Percentage of subjects with baseline CD4+ cell count: | ||
| < 50 cells/mm 3 | 35.6% | 34.7% |
| ≥ 50 cells/mm 3and < 200 cells/mm 3 | 34.8% | 34.5% |
| ≥ 200 cells/mm 3 | 29.6% | 30.8% |
| Median (range) number of primary PI substitutions
IAS-USA primary PI substitutions [August/September 2007]: D30N, V32I, L33F, M46I/L, I47A/V, G48V, I50L/V, I54L/M, L76V, V82A/F/L/S/T, I84V, N88S, L90M
|
4
(0–7) |
4
(0–8) |
| Percentage of subjects with previous use of NNRTIs: | ||
| 0 | 8.2% | 7.9% |
| 1 | 46.9% | 46.7% |
| > 1 | 44.9% | 45.4% |
| Percentage of subjects with previous use of the following NNRTIs: | 70.3% | 72.5% |
| Efavirenz | 57.1% | 58.6% |
| Nevirapine | 13.7% | 12.6% |
| Delavirdine | ||
| Median (range) number of NNRTI RASs
Tibotec NNRTI RASs [June 2008]: A98G, V90I, L100I, K101E/H/P/Q, K103H/N/S/T, V106A/M/I, V108I, E138A/G/K/Q, V179D/E/F/G/I/T, Y181C/I/V, Y188C/H/L, V189I, G190A/C/E/Q/S, H221Y, P255H, F227C/L, M230I/L, P236L, K238N/T, Y318F
|
2
(0–8) |
2
(0–7) |
| Median fold change of the virus for the following NNRTIs: | ||
| Delavirdine | 27.3 | 26.1 |
| Efavirenz | 63.9 | 45.4 |
| Etravirine | 1.6 | 1.5 |
| Nevirapine | 74.3 | 74.0 |
| Percentage of subjects with previous use of a fusion inhibitor | 39.6% | 42.2% |
| Percentage of subjects with a Phenotypic Sensitivity Score (PSS) for the background therapy
The PSS was calculated for the background therapy (as determined on Day 7). Percentages are based on the number of subjects with available phenotype data. For fusion inhibitors (enfuvirtide), subjects were considered resistant if the drug was used in previous therapy up to baseline. Etravirine is not included in this calculation. of:
|
||
| 0 | 17.0% | 16.2% |
| 1 | 36.5% | 38.7% |
| 2 | 26.9% | 27.8% |
| ≥ 3 | 19.7% | 17.3% |
Efficacy at Week 48 for subjects in the Etravirine and placebo arms for the pooled TMC125-C206 and TMC125-C216 study populations are shown in Table 14.
| Etravirine + BR
N=599 |
Placebo + BR
N=604 |
|
|---|---|---|
| BR=background regimen | ||
| Virologic responders at Week 48
Viral Load < 50 HIV-1 RNA copies/mL |
359 (60%) | 232 (38%) |
| Virologic failures at Week 48
Viral Load ≥ 50 HIV-1 RNA copies/mL |
123 (21%) | 201 (33%) |
| Death | 11 (2%) | 19 (3%) |
| Discontinuations before Week 48: | ||
| due to virologic failures | 58 (10%) | 110 (18%) |
| due to adverse events | 31 (5%) | 14 (2%) |
| due to other reasons | 17 (3%) | 28 (5%) |
At Week 48, 70.8% of Etravirine-treated subjects achieved HIV-1 RNA less than 400 copies/mL as compared to 46.4% of placebo-treated subjects. The mean decrease in plasma HIV-1 RNA from baseline to Week 48 was -2.23 log 10copies/mL for Etravirine-treated subjects and -1.46 log 10copies/mL for placebo-treated subjects. The mean CD4+ cell count increase from baseline for Etravirine-treated subjects was 96 cells/mm 3and 68 cells/mm 3for placebo-treated subjects.
Of the study population who either re-used or did not use ENF, 57.4% of Etravirine-treated subjects and 31.7% of placebo-treated subjects achieved HIV-1 RNA less than 50 copies/mL. Of the study population using ENF de novo,67.3% of Etravirine-treated subjects and 57.2% of placebo-treated subjects achieved HIV-1 RNA less than 50 copies/mL.
Treatment-emergent CDC category C events occurred in 4% of Etravirine-treated subjects and 8.4% of placebo-treated subjects.
Study TMC125-C227 was a randomized, exploratory, active-controlled, open-label, Phase 2b trial. Eligible subjects were treatment-experienced, PI-naïve HIV-1-infected subjects with genotypic evidence of NNRTI resistance at screening or from prior genotypic analysis. The virologic response was evaluated in 116 subjects who were randomized to Etravirine (59 subjects) or an investigator-selected PI (57 subjects), each given with 2 investigator-selected N(t)RTIs. Etravirine-treated subjects had lower antiviral responses associated with reduced susceptibility to the N(t)RTIs and to Etravirine as compared to the control PI-treated subjects.
5.1 Severe Skin and Hypersensitivity Reactions
Severe, potentially life-threatening and fatal skin reactions have been reported. In clinical trials, these include cases of Stevens-Johnson syndrome, toxic epidermal necrolysis and erythema multiforme. Hypersensitivity reactions including Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) have also been reported and were characterized by rash, constitutional findings, and sometimes organ dysfunction, including hepatic failure. In Phase 3 clinical trials, Grade 3 and 4 rashes were reported in 1.3% of subjects receiving Etravirine compared to 0.2% of placebo subjects. A total of 2.2% of HIV-1-infected subjects receiving Etravirine discontinued from Phase 3 trials due to rash [see Adverse Reactions (6.1)] . Rash occurred most commonly during the first 6 weeks of therapy. The incidence of rash was higher in females [see Adverse Reactions (6.1)] . Stevens-Johnson syndrome was reported in 1.1% (2/177) of pediatric patients less than 18 years of age receiving Etravirine in combination with other HIV-1 antiretroviral agents in an observational study.
Discontinue Etravirine immediately if signs or symptoms of severe skin reactions or hypersensitivity reactions develop (including, but not limited to, severe rash or rash accompanied by fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, hepatitis, eosinophilia, angioedema). Clinical status including liver transaminases should be monitored and appropriate therapy initiated. Delay in stopping Etravirine treatment after the onset of severe rash may result in a life-threatening reaction.
7.1 Potential for Other Drugs to Affect Etravirine
Etravirine is a substrate of CYP3A, CYP2C9, and CYP2C19. Therefore, co-administration of Etravirine with drugs that induce or inhibit CYP3A, CYP2C9, and CYP2C19 may alter the therapeutic effect or adverse reaction profile of Etravirine (see Table 4) [see Clinical Pharmacology (12.3)].
7.2 Potential for Etravirine to Affect Other Drugs
Etravirine is an inducer of CYP3A and inhibitor of CYP2C9, CYP2C19 and P-glycoprotein (P-gp). Therefore, co-administration of drugs that are substrates of CYP3A, CYP2C9 and CYP2C19 or are transported by P-gp with Etravirine may alter the therapeutic effect or adverse reaction profile of the co-administered drug(s) (see Table 4) [see Clinical Pharmacology (12.3)].
Principal Display Panel 100 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Label)
120 Tablets
NDC 10147-0570-1
Etravirine
tablets
100 mg
Each tablet contains
100 mg of etravirine.
Rx only
PATRIOT
PHARMACEUTICALS LLC
ALERT: Find out about medicines that
should NOT be taken with Etravirine tablets
from your healthcare provider.
Principal Display Panel 200 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 200 mg Tablet Bottle Label)
60 Tablets
NDC 10147-0571-1
Etravirine
tablets
200 mg
Each tablet contains
200 mg of etravirine.
Rx only
PATRIOT
PHARMACEUTICALS LLC
ALERT: Find out about medicines that
should NOT be taken with Etravirine tablets
from your healthcare provider.
7.4 Drugs Without Clinically Significant Interactions With Etravirine (7.4 Drugs Without Clinically Significant Interactions with Etravirine)
In addition to the drugs included in Table 4, the interaction between Etravirine and the following drugs were evaluated in clinical studies and no dose adjustment is needed for either drug [see Clinical Pharmacology (12.3)] : didanosine, enfuvirtide (ENF), ethinylestradiol/norethindrone, omeprazole, paroxetine, raltegravir, ranitidine, and tenofovir disoproxil fumarate.
2.3 Recommended Dosage in Pediatric Patients (2 Years to Less Than 18 Years of Age)
The recommended dosage of Etravirine for pediatric patients 2 years to less than 18 years of age and weighing at least 10 kg is based on body weight (see Table 1) not exceeding the recommended adult dosage. Etravirine should be taken orally, following a meal. The type of food does not affect the exposure to Etravirine [see Clinical Pharmacology (12.3)] .
| Body Weight kilograms (kg) | Dose |
|---|---|
| greater than or equal to 10 kg to less than 20 kg | 100 mg twice daily |
| greater than or equal to 20 kg to less than 25 kg | 125 mg twice daily |
| greater than or equal to 25 kg to less than 30 kg | 150 mg twice daily |
| greater than or equal to 30 kg | 200 mg twice daily |
14.2 Treatment Experienced Pediatric Subjects (2 Years to Less Than 18 Years of Age) (14.2 Treatment-Experienced Pediatric Subjects (2 Years to Less Than 18 Years of Age))
The efficacy of Etravirine for treatment-experienced pediatric subjects is based on two Phase 2 trials, TMC125-C213 and TMC125-C234/IMPAACT P1090.
5.2 Risk of Adverse Reactions Or Loss of Virologic Response Due to Drug Interactions (5.2 Risk of Adverse Reactions or Loss of Virologic Response Due to Drug Interactions)
The concomitant use of Etravirine and other drugs may result in potentially significant drug interactions, some of which may lead to [see Drug Interactions (7.3)] :
- Loss of therapeutic effect of concomitant drug or Etravirine and possible development of resistance.
- Possible clinically significant adverse reactions from greater exposures of Etravirine or other concomitant drugs.
See Table 4for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during Etravirine therapy and review concomitant medications during Etravirine therapy.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:29.277496 · Updated: 2026-03-14T22:24:56.522545