These Highlights Do Not Include All The Information Needed To Use Vyscoxa Safely And Effectively. See Full Prescribing Information For Vyscoxa.
5094d47e-f791-4190-9397-b7500143a74d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS See full prescribing information for complete boxed warning. • Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in the treatment and may increase with duration of use. ( 5.1 ) • VYSCOXA is contraindicated in the setting of coronary artery bypass graft (CABG) surgery. ( 4 , 5.1 ) • NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events. ( 5.2 )
Indications and Usage
VYSCOXA is indicated In adults for the management of the signs and symptoms of:
Dosage and Administration
• Use the lowest effective dosage for shortest duration consistent with individual patient treatment goals. ( 2.1 ) • OA: 200 mg (20 mL) once daily or 100 mg (10 mL) twice daily. ( 2.2 , 14.1 ) • RA: 100 mg (10 mL) to 200 mg (20 mL) twice daily. ( 2.3 , 14.2 ) • AS: 200 mg (20 mL) once daily single dose or 100 mg (10 mL) twice daily. If no effect is observed after 6 weeks, a trial of 200 mg (20 mL) twice daily may be of benefit. ( 2.4 , 14.3 ) • JRA: 50 mg (5 mL) twice daily in patients 10 kg to 25 kg. 100 mg (10 mL) twice daily in patients more than 25 kg. ( 2.5 , 14.4 ) Important: VYSCOXA is not recommended at a single dose greater than 200 mg (20 mL). Single doses of the suspension greater than 200 mg (20 mL) may result in celecoxib concentrations higher than expected. For patients who require a single dose over 200 mg (20 mL), use a different celecoxib formulation ( 2.1 , 12.3 ) Hepatic Impairment: Reduce daily dose by 50% in patients with moderate hepatic impairment (Child-Pugh Class B). ( 2.6 , 8.6 , 12.3 ) Poor Metabolizers of CYP2C9 Substrates: Consider a dose reduction by 50% (or alternative management for JRA) in patients who are known or suspected to be CYP2C9 poor metabolizers. ( 2.6 , 8.8 , 12.3 ).
Warnings and Precautions
• Hepatotoxicity : Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. ( 5.3 ) • Hypertension : Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure. ( 5.4 , 7 ) • Heart Failure and Edema : Avoid use of VYSCOXA in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure. ( 5.5 ) • Renal Toxicity : Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of VYSCOXA in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function. ( 5.6 ) • Anaphylactic Reactions : Seek emergency help if an anaphylactic reaction occurs. ( 5.7 ) • Exacerbation of Asthma Related to Aspirin Sensitivity : VYSCOXA is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with preexisting asthma (without aspirin sensitivity). ( 5.8 ) • Serious Skin Reactions : Discontinue VYSCOXA at first appearance of skin rash or other signs of hypersensitivity. ( 5.9 ) • Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) : Discontinue and evaluate clinically. ( 5.10 ) • Fetal Toxicity : Limit use of NSAIDs, including VYSCOXA, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the fetal ductus arteriosus. ( 5.11 , 8.1 ) • Hematologic Toxicity : Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia. ( 5.12 , 7 )
Contraindications
VYSCOXA is contraindicated in the following patients: • Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to celecoxib, any components of the drug product [ see Warnings and Precautions (5.7 , 5.9) ]. • History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs, have been reported in such patients [ see Warnings and Precautions (5.7 , 5.8) ]. • In the setting of CABG surgery [ see Warnings and Precautions (5.1) ]. • In patients who have demonstrated allergic-type reactions to sulfonamides [ see Warnings and Precautions (5.7) ].
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: • Cardiovascular Thrombotic Events [ see Warnings and Precautions (5.1) ] • GI Bleeding, Ulceration and Perforation [ see Warnings and Precautions (5.2) ] • Hepatotoxicity [ see Warnings and Precautions (5.3) ] • Hypertension [ see Warnings and Precautions (5.4) ] • Heart Failure and Edema [ see Warnings and Precautions (5.5) ] • Renal Toxicity and Hyperkalemia [ see Warnings and Precautions (5.6) ] • Anaphylactic Reactions [ see Warnings and Precautions (5.7) ] • Serious Skin Reactions [ see Warnings and Precautions (5.9) ] • Hematologic Toxicity [ see Warnings and Precautions (5.12) ]
Drug Interactions
See Table 3 for clinically significant drug interactions with celecoxib. Table 3: Clinically Significant Drug Interactions with Celecoxib Drugs That Interfere with Hemostasis Clinical Impact: • Celecoxib and anticoagulants such as warfarin have a synergistic effect on bleeding. The concomitant use of Celecoxib and anticoagulants have an increased risk of serious bleeding compared to the use of either drug alone. • Serotonin release by platelets plays an important role in hemostasis. Case-control and cohort epidemiological studies showed that concomitant use of drugs that interfere with serotonin reuptake and an NSAID may potentiate the risk of bleeding more than an NSAID alone. Intervention: Monitor patients with concomitant use of VYSCOXA with anticoagulants (e.g., warfarin), antiplatelet drugs (e.g., aspirin), SSRIs, and SNRIs for signs of bleeding [ see Warnings and Precautions (5.12) ]. Aspirin Clinical Impact: Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [ see Warnings and Precautions (5.2) ]. In two studies in healthy volunteers, and in patients with osteoarthritis and established heart disease respectively, celecoxib (200 mg to 400 mg daily) has demonstrated a lack of interference with the cardioprotective antiplatelet effect of aspirin (100 mg to 325 mg). Intervention: Concomitant use of VYSCOXA and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [ see Warnings and Precautions (5.12) ]. VYSCOXA is not a substitute for low dose aspirin for cardiovascular protection. ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers Clinical Impact: • NSAIDs may diminish the antihypertensive effect of ACE inhibitors, ARBs, or beta-blockers (including propranolol). • In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, co-administration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Intervention: • During concomitant use of VYSCOXA and ACE inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. • During concomitant use of VYSCOXA and ACE inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function [ see Warnings and Precautions (5.6) ] . • When these drugs are administered concomitantly, patients should be adequately hydrated. Assess renal function at the beginning of the concomitant treatment and periodically thereafter. Diuretics Clinical Impact: Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of VYSCOXA with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [ see Warnings and Precautions (5.6) ]. Digoxin Clinical Impact: The concomitant use of Celecoxib with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. Intervention: During concomitant use of VYSCOXA and digoxin, monitor serum digoxin levels. Lithium Clinical Impact: NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance . The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of VYSCOXA and lithium, monitor patients for signs of lithium toxicity. Methotrexate Clinical Impact: Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). Celecoxib has no effect on methotrexate pharmacokinetics. Intervention: During concomitant use of VYSCOXA and methotrexate, monitor patients for methotrexate toxicity. Cyclosporine Clinical Impact: Concomitant use of celecoxib and cyclosporine may increase cyclosporine's nephrotoxicity. Intervention: During concomitant use of VYSCOXA and cyclosporine, monitor patients for signs of worsening renal function. NSAIDs and Salicylates Clinical Impact: Concomitant use of Celecoxib with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy [ see Warnings and Precautions (5.2) ] . Intervention: The concomitant use of Celecoxib with other NSAIDs or salicylates is not recommended. Pemetrexed Clinical Impact: Concomitant use of celecoxib and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). Intervention: During concomitant use of VYSCOXA and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. NSAIDs with short elimination half-lives (e.g., diclofenac, indomethacin) should be avoided for a period of two days before, the day of, and two days following administration of pemetrexed. In the absence of data regarding potential interaction between pemetrexed and NSAIDs with longer half-lives (e.g., meloxicam, nabumetone), patients taking these NSAIDs should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. CYP2C9 Inhibitors or inducers Clinical Impact: Celecoxib metabolism is predominantly mediated via cytochrome P450 (CYP) 2C9 in the liver. Co-administration of celecoxib with drugs that are known to inhibit CYP2C9 (e.g., fluconazole) may enhance the exposure and toxicity of celecoxib whereas co-administration with CYP2C9 inducers (e.g., rifampin) may lead to compromised efficacy of celecoxib. Intervention Evaluate each patient's medical history when consideration is given to prescribing VYSCOXA. A dosage adjustment may be warranted when VYSCOXA is administered with CYP2C9 inhibitors or inducers [ see Clinical Pharmacology (12.3) ]. CYP2D6 substrates Clinical Impact: In vitro studies indicate that celecoxib, although not a substrate, is an inhibitor of CYP2D6. Therefore, there is a potential for an in vivo drug interaction with drugs that are metabolized by CYP2D6 (e.g., atomoxetine), and celecoxib may enhance the exposure and toxicity of these drugs. Intervention Evaluate each patient's medical history when consideration is given to prescribing VYSCOXA. A dosage adjustment may be warranted when VYSCOXA is administered with CYP2D6 substrates [ see Clinical Pharmacology (12.3) ]. Corticosteroids Clinical Impact: Concomitant use of corticosteroids with celecoxib may increase the risk of GI ulceration or bleeding. Intervention Monitor patients with concomitant use of VYSCOXA with corticosteroids for signs of bleeding [ see Warnings and Precautions (5.2) ] .
Storage and Handling
VYSCOXA (celecoxib) oral suspension, 10 mg/mL celecoxib is a white, opaque, unflavored suspension, supplied as: NDC Number Size 15370-501-16 473 mL Bottle
How Supplied
VYSCOXA (celecoxib) oral suspension, 10 mg/mL celecoxib is a white, opaque, unflavored suspension, supplied as: NDC Number Size 15370-501-16 473 mL Bottle
Medication Information
Warnings and Precautions
• Hepatotoxicity : Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. ( 5.3 ) • Hypertension : Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure. ( 5.4 , 7 ) • Heart Failure and Edema : Avoid use of VYSCOXA in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure. ( 5.5 ) • Renal Toxicity : Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of VYSCOXA in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function. ( 5.6 ) • Anaphylactic Reactions : Seek emergency help if an anaphylactic reaction occurs. ( 5.7 ) • Exacerbation of Asthma Related to Aspirin Sensitivity : VYSCOXA is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with preexisting asthma (without aspirin sensitivity). ( 5.8 ) • Serious Skin Reactions : Discontinue VYSCOXA at first appearance of skin rash or other signs of hypersensitivity. ( 5.9 ) • Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) : Discontinue and evaluate clinically. ( 5.10 ) • Fetal Toxicity : Limit use of NSAIDs, including VYSCOXA, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the fetal ductus arteriosus. ( 5.11 , 8.1 ) • Hematologic Toxicity : Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia. ( 5.12 , 7 )
Indications and Usage
VYSCOXA is indicated In adults for the management of the signs and symptoms of:
Dosage and Administration
• Use the lowest effective dosage for shortest duration consistent with individual patient treatment goals. ( 2.1 ) • OA: 200 mg (20 mL) once daily or 100 mg (10 mL) twice daily. ( 2.2 , 14.1 ) • RA: 100 mg (10 mL) to 200 mg (20 mL) twice daily. ( 2.3 , 14.2 ) • AS: 200 mg (20 mL) once daily single dose or 100 mg (10 mL) twice daily. If no effect is observed after 6 weeks, a trial of 200 mg (20 mL) twice daily may be of benefit. ( 2.4 , 14.3 ) • JRA: 50 mg (5 mL) twice daily in patients 10 kg to 25 kg. 100 mg (10 mL) twice daily in patients more than 25 kg. ( 2.5 , 14.4 ) Important: VYSCOXA is not recommended at a single dose greater than 200 mg (20 mL). Single doses of the suspension greater than 200 mg (20 mL) may result in celecoxib concentrations higher than expected. For patients who require a single dose over 200 mg (20 mL), use a different celecoxib formulation ( 2.1 , 12.3 ) Hepatic Impairment: Reduce daily dose by 50% in patients with moderate hepatic impairment (Child-Pugh Class B). ( 2.6 , 8.6 , 12.3 ) Poor Metabolizers of CYP2C9 Substrates: Consider a dose reduction by 50% (or alternative management for JRA) in patients who are known or suspected to be CYP2C9 poor metabolizers. ( 2.6 , 8.8 , 12.3 ).
Contraindications
VYSCOXA is contraindicated in the following patients: • Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to celecoxib, any components of the drug product [ see Warnings and Precautions (5.7 , 5.9) ]. • History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs, have been reported in such patients [ see Warnings and Precautions (5.7 , 5.8) ]. • In the setting of CABG surgery [ see Warnings and Precautions (5.1) ]. • In patients who have demonstrated allergic-type reactions to sulfonamides [ see Warnings and Precautions (5.7) ].
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: • Cardiovascular Thrombotic Events [ see Warnings and Precautions (5.1) ] • GI Bleeding, Ulceration and Perforation [ see Warnings and Precautions (5.2) ] • Hepatotoxicity [ see Warnings and Precautions (5.3) ] • Hypertension [ see Warnings and Precautions (5.4) ] • Heart Failure and Edema [ see Warnings and Precautions (5.5) ] • Renal Toxicity and Hyperkalemia [ see Warnings and Precautions (5.6) ] • Anaphylactic Reactions [ see Warnings and Precautions (5.7) ] • Serious Skin Reactions [ see Warnings and Precautions (5.9) ] • Hematologic Toxicity [ see Warnings and Precautions (5.12) ]
Drug Interactions
See Table 3 for clinically significant drug interactions with celecoxib. Table 3: Clinically Significant Drug Interactions with Celecoxib Drugs That Interfere with Hemostasis Clinical Impact: • Celecoxib and anticoagulants such as warfarin have a synergistic effect on bleeding. The concomitant use of Celecoxib and anticoagulants have an increased risk of serious bleeding compared to the use of either drug alone. • Serotonin release by platelets plays an important role in hemostasis. Case-control and cohort epidemiological studies showed that concomitant use of drugs that interfere with serotonin reuptake and an NSAID may potentiate the risk of bleeding more than an NSAID alone. Intervention: Monitor patients with concomitant use of VYSCOXA with anticoagulants (e.g., warfarin), antiplatelet drugs (e.g., aspirin), SSRIs, and SNRIs for signs of bleeding [ see Warnings and Precautions (5.12) ]. Aspirin Clinical Impact: Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [ see Warnings and Precautions (5.2) ]. In two studies in healthy volunteers, and in patients with osteoarthritis and established heart disease respectively, celecoxib (200 mg to 400 mg daily) has demonstrated a lack of interference with the cardioprotective antiplatelet effect of aspirin (100 mg to 325 mg). Intervention: Concomitant use of VYSCOXA and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [ see Warnings and Precautions (5.12) ]. VYSCOXA is not a substitute for low dose aspirin for cardiovascular protection. ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers Clinical Impact: • NSAIDs may diminish the antihypertensive effect of ACE inhibitors, ARBs, or beta-blockers (including propranolol). • In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, co-administration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Intervention: • During concomitant use of VYSCOXA and ACE inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. • During concomitant use of VYSCOXA and ACE inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function [ see Warnings and Precautions (5.6) ] . • When these drugs are administered concomitantly, patients should be adequately hydrated. Assess renal function at the beginning of the concomitant treatment and periodically thereafter. Diuretics Clinical Impact: Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of VYSCOXA with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [ see Warnings and Precautions (5.6) ]. Digoxin Clinical Impact: The concomitant use of Celecoxib with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. Intervention: During concomitant use of VYSCOXA and digoxin, monitor serum digoxin levels. Lithium Clinical Impact: NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance . The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. Intervention: During concomitant use of VYSCOXA and lithium, monitor patients for signs of lithium toxicity. Methotrexate Clinical Impact: Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). Celecoxib has no effect on methotrexate pharmacokinetics. Intervention: During concomitant use of VYSCOXA and methotrexate, monitor patients for methotrexate toxicity. Cyclosporine Clinical Impact: Concomitant use of celecoxib and cyclosporine may increase cyclosporine's nephrotoxicity. Intervention: During concomitant use of VYSCOXA and cyclosporine, monitor patients for signs of worsening renal function. NSAIDs and Salicylates Clinical Impact: Concomitant use of Celecoxib with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy [ see Warnings and Precautions (5.2) ] . Intervention: The concomitant use of Celecoxib with other NSAIDs or salicylates is not recommended. Pemetrexed Clinical Impact: Concomitant use of celecoxib and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). Intervention: During concomitant use of VYSCOXA and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. NSAIDs with short elimination half-lives (e.g., diclofenac, indomethacin) should be avoided for a period of two days before, the day of, and two days following administration of pemetrexed. In the absence of data regarding potential interaction between pemetrexed and NSAIDs with longer half-lives (e.g., meloxicam, nabumetone), patients taking these NSAIDs should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. CYP2C9 Inhibitors or inducers Clinical Impact: Celecoxib metabolism is predominantly mediated via cytochrome P450 (CYP) 2C9 in the liver. Co-administration of celecoxib with drugs that are known to inhibit CYP2C9 (e.g., fluconazole) may enhance the exposure and toxicity of celecoxib whereas co-administration with CYP2C9 inducers (e.g., rifampin) may lead to compromised efficacy of celecoxib. Intervention Evaluate each patient's medical history when consideration is given to prescribing VYSCOXA. A dosage adjustment may be warranted when VYSCOXA is administered with CYP2C9 inhibitors or inducers [ see Clinical Pharmacology (12.3) ]. CYP2D6 substrates Clinical Impact: In vitro studies indicate that celecoxib, although not a substrate, is an inhibitor of CYP2D6. Therefore, there is a potential for an in vivo drug interaction with drugs that are metabolized by CYP2D6 (e.g., atomoxetine), and celecoxib may enhance the exposure and toxicity of these drugs. Intervention Evaluate each patient's medical history when consideration is given to prescribing VYSCOXA. A dosage adjustment may be warranted when VYSCOXA is administered with CYP2D6 substrates [ see Clinical Pharmacology (12.3) ]. Corticosteroids Clinical Impact: Concomitant use of corticosteroids with celecoxib may increase the risk of GI ulceration or bleeding. Intervention Monitor patients with concomitant use of VYSCOXA with corticosteroids for signs of bleeding [ see Warnings and Precautions (5.2) ] .
Storage and Handling
VYSCOXA (celecoxib) oral suspension, 10 mg/mL celecoxib is a white, opaque, unflavored suspension, supplied as: NDC Number Size 15370-501-16 473 mL Bottle
How Supplied
VYSCOXA (celecoxib) oral suspension, 10 mg/mL celecoxib is a white, opaque, unflavored suspension, supplied as: NDC Number Size 15370-501-16 473 mL Bottle
Description
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS See full prescribing information for complete boxed warning. • Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in the treatment and may increase with duration of use. ( 5.1 ) • VYSCOXA is contraindicated in the setting of coronary artery bypass graft (CABG) surgery. ( 4 , 5.1 ) • NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events. ( 5.2 )
Section 42229-5
Cardiovascular Thrombotic Events
-
•Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction, and stroke, which can be fatal. This risk may occur early in the treatment and may increase with duration of use [see Warnings and Precautions (5.1) ].
-
•VYSCOXA is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications (4) and Warnings and Precautions (5.1) ].
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | ||
| Issued: 07/2025 | ||
|
Medication Guide |
||
|
VYSCOXA is a prescription medicine that contains celecoxib (a nonsteroidal anti-inflammatory drug [NSAID]). Do not take VYSCOXA with food. VYSCOXA must be taken on an empty stomach at least 2 hours before or 1 hour after food. If you cannot tolerate VYSCOXA on an empty stomach, stop taking VYSCOXA and tell your healthcare provider. |
||
|
What is the most important information I should know about VYSCOXA, and medicines called nonsteroidal anti-inflammatory drugs (NSAIDs)?
Do not take VYSCOXA right before or after a heart surgery called a “coronary artery bypass graft (CABG)".
|
||
|
||
|
|
|
|
||
|
What is VYSCOXA?
VYSCOXA is not used for management of the signs and symptoms of short-term pain or treatment of menstrual cramps. |
||
|
Do not take VYSCOXA:
|
||
|
Before taking VYSCOXA tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all of the medicines you or your child take, including prescription or over-the-counter medicines, vitamins or herbal supplements.
|
||
|
How should I take VYSCOXA?
|
||
|
What are the possible side effects of VYSCOXA?
Get emergency help right away if you get any of the following symptoms: |
||
|
|
|
|
Stop taking your VYSCOXA and call your healthcare provider right away if you get any of the following symptoms: |
||
|
|
|
|
These are not all the possible side effects of VYSCOXA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
Other information about NSAIDs
|
||
|
How should I store VYSCOXA?
Keep VYSCOXA and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of VYSCOXA. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use VYSCOXA for a condition for which it was not prescribed. Do not give VYSCOXA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about VYSCOSA that is written for health professionals. |
||
|
What are the ingredients in VYSCOXA? Active ingredient: celecoxib Inactive ingredients: anhydrous citric acid, glycerin, magnesium aluminometasilicate, methylparaben, propylparaben, purified water, sodium citrate dihydrate, sucralose, and xanthan gum. Manufactured for and Distributed by: Carwin Pharmaceutical Associates, LLC, Hazlet, NJ 07730 For more information, go to www.carwinpharma.com or call: 844-700-5011 |
Section 44425-7
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from Light. Shake well before use. Discard 21 days after opening.
10. Overdosage
Symptoms following acute NSAID overdosages have been typically limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which have been generally reversible with supportive care. Gastrointestinal bleeding has occurred. Hypertension, acute renal failure, respiratory depression, and coma have occurred, but were rare [see Warnings and Precautions (5.2, 5.4, 5.6) ].
No overdoses of celecoxib were reported during clinical trials. Doses up to 2400 mg/day for up to 10 days in 12 patients did not result in serious toxicity. No information is available regarding the removal of celecoxib by hemodialysis, but based on its high degree of plasma protein binding (>97%) dialysis is unlikely to be useful in overdose.
Manage patients with symptomatic and supportive care following an NSAID overdosage. There are no specific antidotes. Consider emesis and/or activated charcoal (60 to 100 grams in adults, 1 to 2 grams per kg of body weight in pediatric patients) and/or osmotic cathartic in symptomatic patients seen within four hours of ingestion or in patients with a large overdosage (5 to 10 times the recommended dosage). Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
For additional information about overdosage treatment contact a poison control center (1-800-222-1222).
11. Description
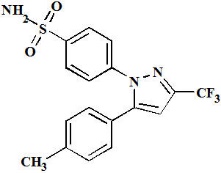
VYSCOXA contains celecoxib, a nonsteroidal anti-inflammatory agent. The chemical name of celecoxib is 4-[5-(4-methylphenyl)- 3-(trifluoromethyl)-1H-pyrazol-1-yl] benzenesulfonamide. Celecoxib has a molecular weight of is 381.38 g/mol, a molecular formula of C17H14F3N3O2S, and the following structural formula:
Celecoxib is a white to off-white powder with a pKa of 11.1 (sulfonamide moiety). Celecoxib is hydrophobic (log P is 3.5) and is practically insoluble in aqueous media at physiological pH range.
The inactive ingredients in VYSCOXA oral suspension include: anhydrous citric acid, glycerin, magnesium aluminometasilicate, methylparaben, propylparaben, purified water, sodium citrate dihydrate, sucralose, and xanthan gum. Sodium hydroxide and hydrochloric acid may be used to adjust the pH to 4.5 – 5.5.
5.4 Hypertension
NSAIDs, including VYSCOXA, can lead to new onset of hypertension or worsening of preexisting hypertension, either of which may contribute to the increased incidence of CV events. Patients taking angiotensin converting enzyme (ACE) inhibitors, thiazide diuretics or loop diuretics may have impaired response to these therapies when taking NSAIDs [see Drug Interactions (7) ].
See Clinical Studies (14.5, 14.6) for additional blood pressure data for VYSCOXA.
Monitor blood pressure (BP) during the initiation of NSAID treatment and throughout the course of therapy.
8.4 Pediatric Use
The safety and effectiveness of celecoxib for the management of the signs and symptoms of Juvenile Rheumatoid Arthritis have been established in pediatric patients 2 years of age and older. The use of celecoxib in patients 2 years of age and older is supported by evidence from a 12-week, double-blind, active-controlled, pharmacokinetic, safety and efficacy study, with a 12-week open-label extension conducted in patients 2 to 17 years of age. Safety and efficacy have not been studied beyond six months in pediatric patients.
Patients with systemic onset JRA (without active systemic features) appear to be at risk for the development of abnormal coagulation laboratory tests. In some patients with systemic onset JRA, both celecoxib and naproxen were associated with mild prolongation of activated partial thromboplastin time (APTT) but not prothrombin time (PT). When NSAIDs including celecoxib are used in patients with systemic onset JRA, monitor patients for signs and symptoms of abnormal clotting or bleeding, due to the risk of disseminated intravascular coagulation. Patients with systemic onset JRA should be monitored for the development of abnormal coagulation tests. The long-term cardiovascular toxicity in children exposed to celecoxib has not been evaluated and it is unknown if long-term risks may be similar to that seen in adults exposed to celecoxib or other COX-2 selective and non- selective NSAIDs [see Boxed Warning, Dosage and Administration (2.3), Warnings and Precautions (5.15), Warnings and Precautions (5.5), Adverse Reactions (6.1), Animal Toxicology (13.2), Clinical Studies (14.4) ].
Alternative therapies for treatment of JRA should be considered in pediatric patients identified to be CYP2C9 poor metabolizers [see Poor Metabolizers of CYP2C9 substrates (8.8) ].
The safety and effectiveness of VYSCOXA have not been established in pediatric patients with JRA less than 2 years of age, in patients with a body weight less than 10 kg, and in patients with active systemic features.
The safety and effectiveness of VYSCOXA have not been established in pediatric patients for osteoarthritis or ankylosing spondylitis.
8.5 Geriatric Use
Elderly patients, compared to younger patients, are at greater risk for NSAID-associated serious cardiovascular, gastrointestinal, and/or renal adverse reactions. If the anticipated benefit for the elderly patient outweighs these potential risks, start dosing at the low end of the dosing range, and monitor patients for adverse effects [see Warnings and Precautions (5.1, 5.2, 5.3, 5.6, 5.14) ].
Of the total number of patients who received celecoxib in pre-approval clinical trials, more than 3,300 were 65–74 years of age, while approximately 1,300 additional patients were 75 years and over. No substantial differences in effectiveness were observed between these subjects and younger subjects. In clinical studies comparing renal function as measured by the GFR, BUN and creatinine, and platelet function as measured by bleeding time and platelet aggregation, the results were not different between elderly and young volunteers. However, as with other NSAIDs, including those that selectively inhibit COX-2, there have been more spontaneous post-marketing reports of fatal GI events and acute renal failure in the elderly than in younger patients [see Warnings and Precautions (5.2, 5.6) ].
1.1 Osteoarthritis
(OA) [see Clinical Studies (14.1) ].
2.2 Osteoarthritis
For OA, the dosage is 200 mg (20 mL) per day administered as a single dose or as 100 mg (10 mL) twice daily.
5.3 Hepatotoxicity
Elevations of ALT or AST (three or more times the upper limit of normal [ULN]) have been reported in approximately 1% of NSAID-treated patients in clinical trials. In addition, rare, sometimes fatal, cases of severe hepatic injury, including fulminant hepatitis, liver necrosis, and hepatic failure have been reported.
Elevations of ALT or AST (less than three times ULN) may occur in up to 15% of patients treated with NSAIDs including celecoxib.
In controlled clinical trials of celecoxib, the incidence of borderline elevations (greater than or equal to 1.2 times and less than 3 times the upper limit of normal) of liver associated enzymes was 6% for celecoxib and 5% for placebo, and approximately 0.2% of patients taking celecoxib and 0.3% of patients taking placebo had notable elevations of ALT and AST.
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, diarrhea, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash), discontinue VYSCOXA immediately, and perform a clinical evaluation of the patient.
14.1 Osteoarthritis
Celecoxib has demonstrated significant reduction in joint pain compared to placebo. Celecoxib was evaluated for treatment of the signs and the symptoms of OA of the knee and hip in placebo- and active-controlled clinical trials of up to 12 weeks duration. In patients with OA, treatment with celecoxib 100 mg twice daily or 200 mg once daily resulted in improvement in WOMAC (Western Ontario and McMaster Universities) osteoarthritis index, a composite of pain, stiffness, and functional measures in OA. In three 12-week studies of pain accompanying OA flare, celecoxib doses of 100 mg twice daily and 200 mg twice daily provided significant reduction of pain within 24 to 48 hours of initiation of dosing. At doses of 100 mg twice daily or 200 mg twice daily the effectiveness of celecoxib was shown to be similar to that of naproxen 500 mg twice daily. Doses of 200 mg twice daily provided no additional benefit above that seen with 100 mg twice daily. A total daily dose of 200 mg has been shown to be equally effective whether administered as 100 mg twice daily or 200 mg once daily.
14. Clinical Studies
The effectiveness of VYSCOXA has been established based on adequate and well-controlled studies of another formulation of celecoxib [see Clinical Pharmacology (12.3) ]. Below is a display of the efficacy results of the adequate and well-controlled studies of celecoxib.
4. Contraindications
VYSCOXA is contraindicated in the following patients:
-
•Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to celecoxib, any components of the drug product [see Warnings and Precautions (5.7, 5.9) ].
-
•History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs, have been reported in such patients [see Warnings and Precautions (5.7, 5.8) ].
-
•In the setting of CABG surgery [see Warnings and Precautions (5.1) ].
-
•In patients who have demonstrated allergic-type reactions to sulfonamides [see Warnings and Precautions (5.7) ].
6. Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling:
-
•Cardiovascular Thrombotic Events [see Warnings and Precautions (5.1) ]
-
•GI Bleeding, Ulceration and Perforation [see Warnings and Precautions (5.2) ]
-
•Hepatotoxicity [see Warnings and Precautions (5.3) ]
-
•Hypertension [see Warnings and Precautions (5.4) ]
-
•Heart Failure and Edema [see Warnings and Precautions (5.5) ]
-
•Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.6) ]
-
•Anaphylactic Reactions [see Warnings and Precautions (5.7) ]
-
•Serious Skin Reactions [see Warnings and Precautions (5.9) ]
-
•Hematologic Toxicity [see Warnings and Precautions (5.12) ]
7. Drug Interactions
See Table 3 for clinically significant drug interactions with celecoxib.
|
Drugs That Interfere with Hemostasis |
|
|
Clinical Impact: |
|
|
Intervention: |
Monitor patients with concomitant use of VYSCOXA with anticoagulants (e.g., warfarin), antiplatelet drugs (e.g., aspirin), SSRIs, and SNRIs for signs of bleeding [see Warnings and Precautions (5.12) ]. |
|
Aspirin |
|
|
Clinical Impact: |
Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2)
]. |
|
Intervention: |
Concomitant use of VYSCOXA and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [see Warnings and Precautions (5.12)
]. |
|
ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers |
|
|
Clinical Impact: |
|
|
Intervention: |
|
|
Diuretics |
|
|
Clinical Impact: |
Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. |
|
Intervention: |
During concomitant use of VYSCOXA with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [see Warnings and Precautions (5.6) ]. |
|
Digoxin |
|
|
Clinical Impact: |
The concomitant use of Celecoxib with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. |
|
Intervention: |
During concomitant use of VYSCOXA and digoxin, monitor serum digoxin levels. |
|
Lithium |
|
|
Clinical Impact: |
NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. |
|
Intervention: |
During concomitant use of VYSCOXA and lithium, monitor patients for signs of lithium toxicity. |
|
Methotrexate |
|
|
Clinical Impact: |
Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). |
|
Intervention: |
During concomitant use of VYSCOXA and methotrexate, monitor patients for methotrexate toxicity. |
|
Cyclosporine |
|
|
Clinical Impact: |
Concomitant use of celecoxib and cyclosporine may increase cyclosporine's nephrotoxicity. |
|
Intervention: |
During concomitant use of VYSCOXA and cyclosporine, monitor patients for signs of worsening renal function. |
|
NSAIDs and Salicylates |
|
|
Clinical Impact: |
Concomitant use of Celecoxib with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy [see Warnings and Precautions (5.2) ]. |
|
Intervention: |
The concomitant use of Celecoxib with other NSAIDs or salicylates is not recommended. |
|
Pemetrexed |
|
|
Clinical Impact: |
Concomitant use of celecoxib and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). |
|
Intervention: |
During concomitant use of VYSCOXA and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. |
|
CYP2C9 Inhibitors or inducers |
|
|
Clinical Impact: |
Celecoxib metabolism is predominantly mediated via cytochrome P450 (CYP) 2C9 in the liver. Co-administration of celecoxib with drugs that are known to inhibit CYP2C9 (e.g., fluconazole) may enhance the exposure and toxicity of celecoxib whereas co-administration with CYP2C9 inducers (e.g., rifampin) may lead to compromised efficacy of celecoxib. |
|
Intervention |
Evaluate each patient's medical history when consideration is given to prescribing VYSCOXA. A dosage adjustment may be warranted when VYSCOXA is administered with CYP2C9 inhibitors or inducers [see Clinical Pharmacology (12.3) ]. |
|
CYP2D6 substrates |
|
|
Clinical Impact: |
In vitro studies indicate that celecoxib, although not a substrate, is an inhibitor of CYP2D6. Therefore, there is a potential for an in vivo drug interaction with drugs that are metabolized by CYP2D6 (e.g., atomoxetine), and celecoxib may enhance the exposure and toxicity of these drugs. |
|
Intervention |
Evaluate each patient's medical history when consideration is given to prescribing VYSCOXA. A dosage adjustment may be warranted when VYSCOXA is administered with CYP2D6 substrates [see Clinical Pharmacology (12.3) ]. |
|
Corticosteroids |
|
|
Clinical Impact: |
Concomitant use of corticosteroids with celecoxib may increase the risk of GI ulceration or bleeding. |
|
Intervention |
Monitor patients with concomitant use of VYSCOXA with corticosteroids for signs of bleeding [see Warnings and Precautions (5.2)]. |
8.7 Renal Impairment
VYSCOXA is not recommended in patients with severe renal insufficiency [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3) ].
12.3 Pharmacokinetics
Celecoxib has extensive distribution and high protein binding. It is primarily metabolized by CYP2C9 with a half-life of approximately 11 hours.
12.5 Pharmacogenomics
CYP2C9 activity is reduced in individuals with genetic polymorphisms that lead to reduced enzyme activity, such as those homozygous for the CYP2C9*2 and CYP2C9*3 polymorphisms. Limited data from 4 published reports that included a total of 8 subjects with the homozygous CYP2C9*3/*3 genotype showed celecoxib systemic levels that were 3- to 7-fold higher in these subjects compared to subjects with CYP2C9*1/*1 or *I/*3 genotypes. The pharmacokinetics of celecoxib have not been evaluated in subjects with other CYP2C9 polymorphisms, such as *2, *5, *6, *9 and *11. It is estimated that the frequency of the homozygous *3/*3 genotype is 0.3% to 1.0% in various ethnic groups [see Dosage and Administration (2.6), Use in Specific Populations (8.8) ].
13.2 Animal Toxicology
An increase in the incidence of background findings of spermatocele with or without secondary changes such as epididymal hypospermia as well as minimal to slight dilation of the seminiferous tubules was seen in the juvenile rat. These reproductive findings while apparently treatment-related did not increase in incidence or severity with dose and may indicate an exacerbation of a spontaneous condition. Similar reproductive findings were not observed in studies of juvenile or adult dogs or in adult rats treated with celecoxib. The clinical significance of this observation is unknown.
8.6 Hepatic Impairment
The daily recommended dose of VYSCOXA oral suspension in patients with moderate hepatic impairment (Child-Pugh Class B) should be reduced by 50%. The use of VYSCOXA in patients with severe hepatic impairment is not recommended [see Dosage and Administration (2.6) and Clinical Pharmacology (12.3) ].
1. Indications and Usage
VYSCOXA is indicated
In adults for the management of the signs and symptoms of:
1.2 Rheumatoid Arthritis
(RA) [see Clinical Studies (14.2) ].
12.1 Mechanism of Action
Celecoxib has analgesic, anti-inflammatory, and antipyretic properties.
The mechanism of action of celecoxib is believed to be due to inhibition of prostaglandin synthesis, primarily via inhibition of COX-2. Celecoxib is a potent inhibitor of prostaglandin synthesis in vitro. Celecoxib concentrations reached during therapy have produced in vivo effects. Prostaglandins sensitize afferent nerves and potentiate the action of bradykinin in inducing pain in animal models. Prostaglandins are mediators of inflammation. Since celecoxib is an inhibitor of prostaglandin synthesis, its mode of action may be due to a decrease of prostaglandins in peripheral tissues.
2.3 Rheumatoid Arthritis
For RA, the dosage is 100 mg (10 mL) to 200 mg (20 mL) twice daily.
14.2 Rheumatoid Arthritis
Celecoxib has demonstrated significant reduction in joint tenderness/pain and joint swelling compared to placebo. Celecoxib was evaluated for treatment of the signs and symptoms of RA in placebo- and active-controlled clinical trials of up to 24 weeks in duration. Celecoxib was shown to be superior to placebo in these studies, using the ACR20 Responder Index, a composite of clinical, laboratory, and functional measures in RA. Celecoxib doses of 100 mg twice daily and 200 mg twice daily were similar in effectiveness and both were comparable to naproxen 500 mg twice daily.
Although celecoxib 100 mg twice daily and 200 mg twice daily provided similar overall effectiveness, some patients derived additional benefit from the 200 mg twice daily dose. Doses of 400 mg twice daily provided no additional benefit above that seen with 100 mg to 200 mg twice daily.
1.3 Ankylosing Spondylitis
(AS) [see Clinical Studies (14.3) ].
In pediatric patients two years of age and older for the management of the signs and symptoms of:
2.4 Ankylosing Spondylitis
For AS, the dosage is 200 mg (20 mL) daily in single (once per day) dose or 100 mg (10 mL) twice daily. If no effect is observed after 6 weeks, a trial of 200 mg (20 mL) twice daily may be worthwhile. If no effect is observed after 6 weeks on 200 mg (20 mL) twice daily, a response is not likely and consideration should be given to alternate treatment options.
5.14 Laboratory Monitoring
Because serious GI bleeding, hepatotoxicity, and renal injury can occur without warning symptoms or signs, consider monitoring patients on long-term NSAID treatment with a CBC and a chemistry profile periodically [see Warnings and Precautions (5.2, 5.3, 5.6) ].
In controlled clinical trials, elevated BUN occurred more frequently in patients receiving celecoxib compared with patients on placebo. This laboratory abnormality was also seen in patients who received comparator NSAIDs in these studies. The clinical significance of this abnormality has not been established.
5.7 Anaphylactic Reactions
Celecoxib has been associated with anaphylactic reactions in patients with and without known hypersensitivity to celecoxib and in patients with aspirin sensitive asthma. Celecoxib is a sulfonamide and both NSAIDs and sulfonamides may cause allergic type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people [see Contraindications (4) and Warnings and Precautions (5.8) ].
Seek emergency help if any anaphylactic reaction occurs.
5.9 Serious Skin Reactions
Serious skin reactions have occurred following treatment with celecoxib, including erythema multiforme, exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalized exanthematous pustulosis (AGEP), and fixed drug eruption (FDE) which may present as a more severe variant known as generalized bullous fixed drug eruption (GBFDE). These serious events may occur without warning and can be fatal.
Inform patients about the signs and symptoms of serious skin reactions, and to discontinue the use of VYSCOXA at the first appearance of skin rash or any other sign of hypersensitivity. VYSCOXA is contraindicated in patients with previous serious skin reactions to NSAIDs [see Contraindications (4) ].
14.3 Ankylosing Spondylitis
Celecoxib was evaluated in AS patients in two placebo- and active-controlled clinical trials of 6 and 12 weeks duration. Celecoxib at doses of 100 mg twice daily, 200 mg once daily and 400 mg once daily was shown to be statistically superior to placebo in these studies for all three co-primary efficacy measures assessing global pain intensity (Visual Analogue Scale), global disease activity (Visual Analogue Scale) and functional impairment (Bath Ankylosing Spondylitis Functional Index). In the 12-week study, there was no difference in the extent of improvement between the 200 mg and 400 mg celecoxib doses in a comparison of mean change from baseline, but there was a greater percentage of patients who responded to celecoxib 400 mg, 53%, than to celecoxib 200 mg, 44%, using the Assessment in Ankylosing Spondylitis response criteria (ASAS 20). The ASAS 20 defines a responder as improvement from baseline of at least 20% and an absolute improvement of at least 10 mm, on a 0 mm to 100 mm scale, in at least three of the four following domains: patient global pain, Bath Ankylosing Spondylitis Functional Index, and inflammation. The responder analysis also demonstrated no change in the responder rates beyond 6 weeks.
5. Warnings and Precautions
-
•Hepatotoxicity: Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. (5.3)
-
•Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure. (5.4, 7)
-
•Heart Failure and Edema: Avoid use of VYSCOXA in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure. (5.5)
-
•Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of VYSCOXA in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function. (5.6)
-
•Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs. (5.7)
-
•Exacerbation of Asthma Related to Aspirin Sensitivity: VYSCOXA is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with preexisting asthma (without aspirin sensitivity). (5.8)
-
•Serious Skin Reactions: Discontinue VYSCOXA at first appearance of skin rash or other signs of hypersensitivity. (5.9)
-
•Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue and evaluate clinically. (5.10)
-
•Fetal Toxicity: Limit use of NSAIDs, including VYSCOXA, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the fetal ductus arteriosus. (5.11, 8.1)
-
•Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia. (5.12, 7)
5.12 Hematological Toxicity
Anemia has occurred in NSAID-treated patients. This may be due to occult or gross blood loss, fluid retention, or an incompletely described effect on erythropoiesis. If a patient treated with VYSCOXA has any signs or symptoms of anemia, monitor hemoglobin or hematocrit.
In controlled clinical trials the incidence of anemia was 0.6% with celecoxib and 0.4% with placebo. Patients on long-term treatment with VYSCOXA should have their hemoglobin or hematocrit checked if they exhibit any signs or symptoms of anemia or blood loss.
NSAIDs, including VYSCOXA, may increase the risk of bleeding events. Co-morbid conditions such as coagulation disorders or concomitant use of warfarin, other anticoagulants, antiplatelet drugs (e.g., aspirin), SSRIs and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients for signs of bleeding [see Drug Interactions (7) ].
5.5 Heart Failure and Edema
The Coxib and traditional NSAID Trialists' Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of celecoxib may blunt the CV effects of several therapeutic agents used to treat these medical conditions (e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers [ARBs]) [see Drug Interactions (7) ].
In the CLASS study [see Clinical Studies (14.6) ], the Kaplan-Meier cumulative rates at 9 months of peripheral edema in patients on celecoxib 400 mg twice daily (4-fold and 2-fold the recommended OA and RA doses, respectively), ibuprofen 800 mg three times daily and diclofenac 75 mg twice daily were 4.5%, 6.9% and 4.7%, respectively.
Avoid the use of VYSCOXA in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If VYSCOXA is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
2. Dosage and Administration
-
•Use the lowest effective dosage for shortest duration consistent with individual patient treatment goals. (2.1)
-
•OA: 200 mg (20 mL) once daily or 100 mg (10 mL) twice daily. (2.2, 14.1)
-
•RA: 100 mg (10 mL) to 200 mg (20 mL) twice daily. (2.3, 14.2)
-
•AS: 200 mg (20 mL) once daily single dose or 100 mg (10 mL) twice daily. If no effect is observed after 6 weeks, a trial of 200 mg (20 mL) twice daily may be of benefit. (2.4, 14.3)
-
•JRA: 50 mg (5 mL) twice daily in patients 10 kg to 25 kg. 100 mg (10 mL) twice daily in patients more than 25 kg. (2.5, 14.4)
Important:VYSCOXA is not recommended at a single dose greater than 200 mg (20 mL). Single doses of the suspension greater than 200 mg (20 mL) may result in celecoxib concentrations higher than expected. For patients who require a single dose over 200 mg (20 mL), use a different celecoxib formulation (2.1, 12.3)
Hepatic Impairment: Reduce daily dose by 50% in patients with moderate hepatic impairment (Child-Pugh Class B). (2.6, 8.6, 12.3)
Poor Metabolizers of CYP2C9 Substrates: Consider a dose reduction by 50% (or alternative management for JRA) in patients who are known or suspected to be CYP2C9 poor metabolizers. (2.6, 8.8, 12.3).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of celecoxib. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure
Cardiovascular: Vasculitis, deep venous thrombosis
General: Anaphylactoid reaction, angioedema
Liver and biliary: Liver necrosis, hepatitis, jaundice, hepatic failure
Hemic and lymphatic: Agranulocytosis, aplastic anemia, pancytopenia, leucopenia
Metabolic: Hypoglycemia, hyponatremia
Nervous: Aseptic meningitis, ageusia, anosmia, fatal intracranial hemorrhage
Renal: Interstitial nephritis
Skin and Appendages: Erythema multiforme, exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalized exanthematous pustulosis (AGEP), and fixed drug eruption (FDE).
3. Dosage Forms and Strengths
Oral suspension: 10 mg/mL of celecoxib as a white, opaque, unflavored suspension.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The adverse reaction information from clinical trials does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating rates.
Of the celecoxib-treated patients in the pre-marketing controlled clinical trials of another formulation of celecoxib, approximately 4,250 were patients with OA, approximately 2,100 were patients with RA, and approximately 1,050 were patients with post-surgical pain. More than 8,500 patients received a total daily dose of celecoxib of 200 mg (100 mg twice daily or 200 mg once daily) or more, including more than 400 treated at 800 mg (400 mg twice daily). Approximately 3,900 patients received celecoxib at these doses for 6 months or more; approximately 2,300 of these have received it for 1 year or more and 124 of these have received it for 2 years or more.
8. Use in Specific Populations
Infertility: NSAIDs are associated with reversible infertility. Consider withdrawal of VYSCOXA in women who have difficulties conceiving. (8.3)
2.1 General Dosing Instructions
Carefully consider the potential benefits and risks of VYSCOXA and other treatment options before deciding to use VYSCOXA. Use the lowest effective dosage for the shortest duration consistent with individual patient treatment goals [see Warnings and Precautions (5) ].
The maximum single dose of VYSCOXA is 200 mg (20 mL). Administering more than 200 mg (20 mL) in a single dose of the VYXCOXA suspension may result in higher than intended plasma concentrations of celecoxib. In patients requiring a single dose greater than 200 mg (20 mL), use a different product.
VYSCOXA must be taken on an empty stomach at least 2 hours before or 1 hour after food [see Clinical Pharmacology (12.3) ].
For patients who cannot tolerate dosing with VYSCOXA on an empty stomach, discontinue the use of this product. Do not advise the patient to take VYSCOXA with food.
1.4 Juvenile Rheumatoid Arthritis
(JRA) [see Clinical Studies (14.4) ].
Limitations of Use
VYSCOXA must be administered on an empty stomach at least 2 hours before or 1 hour after food. Taking VYSCOXA with food results in plasma exposures of celecoxib up to 50% higher than intended. If patients cannot tolerate VYSCOXA in the fasted state, discontinue use of VYSCOXA (reference Sections 2.1 , 5, 12).
VYSCOXA is NOT indicated for the management of acute pain or treatment of primary dysmenorrhea.
2.5 Juvenile Rheumatoid Arthritis
For JRA, the dosage for pediatric patients (age 2 years and older) is based on weight. For patients weighing 10 kg to 25 kg the recommended dose is 50 mg (5 mL) twice daily. For patients greater than 25 kg the recommended dose is 100 mg (10 mL) twice daily.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide) that accompanies each prescription dispensed. Inform patients, families, or their caregivers of the following information before initiating therapy with VYSCOXA and periodically during the course of ongoing therapy.
5.1 Cardiovascular Thrombotic Events
Clinical trials of several cyclooxygenase-2 (COX-2) selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.
In the APC (Adenoma Prevention with Celecoxib) trial, there was about a threefold increased risk of the composite endpoint of cardiovascular death, MI, or stroke for the celecoxib 400 mg twice daily and celecoxib 200 mg twice daily treatment arms compared to placebo. The increases in both celecoxib dose groups versus placebo-treated patients were mainly due to an increased incidence of myocardial infarction [see Clinical Studies (14.6) ].
A randomized controlled trial entitled the Prospective Randomized Evaluation of Celecoxib Integrated Safety vs. Ibuprofen Or Naproxen (PRECISION) was conducted to assess the relative cardiovascular thrombotic risk of a COX-2 inhibitor, celecoxib, compared to the non-selective NSAIDs naproxen and ibuprofen. Celecoxib 100 mg twice daily was non-inferior to naproxen 375 to 500 mg twice daily and ibuprofen 600 to 800 mg three times daily for the composite endpoint of the Antiplatelet Trialists' Collaboration (APTC), which consists of cardiovascular death (including hemorrhagic death), non-fatal myocardial infarction, and non-fatal stroke [see Clinical Studies (14.5) ].
To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as celecoxib, increases the risk of serious gastrointestinal (GI) events [see Warnings and Precautions (5.2) ].
16. How Supplied/storage and Handling
VYSCOXA (celecoxib) oral suspension, 10 mg/mL celecoxib is a white, opaque, unflavored suspension, supplied as:
| NDC Number | Size |
|---|---|
|
15370-501-16 |
473 mL Bottle |
5.13 Masking of Inflammation and Fever
The pharmacological activity of VYSCOXA in reducing inflammation, and possibly fever, may diminish the utility of diagnostic signs in detecting infections.
8.8 Poor Metabolizers of Cyp2c9 Substrates
In patients who are known or suspected to be poor CYP2C9 metabolizers (i.e., CYP2C9*3/*3), based on genotype or previous history/experience with other CYP2C9 substrates (such as warfarin, phenytoin) administer VYSCOXA starting with half the lowest recommended dose. Alternative management should be considered in JRA patients identified to be CYP2C9 poor metabolizers [see Dosage and Administration (2.6) and Clinical Pharmacology (12.5) ].
14.4 Juvenile Rheumatoid Arthritis (nct00652925)
In a 12-week, randomized, double-blind active-controlled, parallel-group, multicenter, noninferiority study, patients from 2 years to 17 years of age with pauciarticular, polyarticular course JRA or systemic onset JRA (with currently inactive systemic features), received one of the following treatments: celecoxib 3 mg/kg (to a maximum of 150 mg) twice daily; celecoxib 6 mg/kg (to a maximum of 300 mg) twice daily; or naproxen 7.5 mg/kg (to a maximum of 500 mg) twice daily. The response rates were based upon the JRA Definition of Improvement greater than or equal to 30% (JRA DOI 30) criterion, which is a composite of clinical, laboratory, and functional measures of JRA. The JRA DOI 30 response rates at week 12 were 69%, 80% and 67% in the celecoxib 3 mg/kg twice daily, celecoxib 6 mg/kg twice daily, and naproxen 7.5 mg/kg twice daily treatment groups, respectively.
The efficacy and safety of celecoxib for JRA have not been studied beyond six months. The long-term cardiovascular toxicity in children exposed to celecoxib has not been evaluated and it is unknown if the long-term risk may be similar to that seen in adults exposed to celecoxib or other COX-2 selective and non-selective NSAIDs [see Boxed Warning, Warnings and Precautions (5.1, 5.15) ].
5.15 Disseminated Intravascular Coagulation (dic)
Because of the risk of disseminated intravascular coagulation with use of VYSCOXA in pediatric patients with systemic onset JRA, monitor patients for signs and symptoms of abnormal clotting or bleeding, and inform patients and their caregivers to report symptoms as soon as possible.
5.8 Exacerbation of Asthma Related to Aspirin Sensitivity
A subpopulation of patients with asthma may have aspirin-sensitive asthma which may include chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other NSAIDs. Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, VYSCOXA is contraindicated in patients with this form of aspirin sensitivity [see Contraindications (4) ]. When VYSCOXA is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for changes in the signs and symptoms of asthma.
5.2 Gastrointestinal Bleeding, Ulceration, and Perforation
NSAIDs, including celecoxib cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with VYSCOXA. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occurred in approximately 1% of patients treated for 3 to 6 months, and in about 2% to 4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
5.10 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as celecoxib. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue VYSCOXA and evaluate the patient immediately.
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS
See full prescribing information for complete boxed warning.
-
•Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in the treatment and may increase with duration of use. (5.1)
-
•VYSCOXA is contraindicated in the setting of coronary artery bypass graft (CABG) surgery. (4, 5.1)
-
•NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events. (5.2)
Principal Display Panel Celecoxib Oral Suspension, 10 Mg/ml (container Label)
Structured Label Content
Section 42229-5 (42229-5)
Cardiovascular Thrombotic Events
-
•Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction, and stroke, which can be fatal. This risk may occur early in the treatment and may increase with duration of use [see Warnings and Precautions (5.1) ].
-
•VYSCOXA is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications (4) and Warnings and Precautions (5.1) ].
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | ||
| Issued: 07/2025 | ||
|
Medication Guide |
||
|
VYSCOXA is a prescription medicine that contains celecoxib (a nonsteroidal anti-inflammatory drug [NSAID]). Do not take VYSCOXA with food. VYSCOXA must be taken on an empty stomach at least 2 hours before or 1 hour after food. If you cannot tolerate VYSCOXA on an empty stomach, stop taking VYSCOXA and tell your healthcare provider. |
||
|
What is the most important information I should know about VYSCOXA, and medicines called nonsteroidal anti-inflammatory drugs (NSAIDs)?
Do not take VYSCOXA right before or after a heart surgery called a “coronary artery bypass graft (CABG)".
|
||
|
||
|
|
|
|
||
|
What is VYSCOXA?
VYSCOXA is not used for management of the signs and symptoms of short-term pain or treatment of menstrual cramps. |
||
|
Do not take VYSCOXA:
|
||
|
Before taking VYSCOXA tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all of the medicines you or your child take, including prescription or over-the-counter medicines, vitamins or herbal supplements.
|
||
|
How should I take VYSCOXA?
|
||
|
What are the possible side effects of VYSCOXA?
Get emergency help right away if you get any of the following symptoms: |
||
|
|
|
|
Stop taking your VYSCOXA and call your healthcare provider right away if you get any of the following symptoms: |
||
|
|
|
|
These are not all the possible side effects of VYSCOXA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
Other information about NSAIDs
|
||
|
How should I store VYSCOXA?
Keep VYSCOXA and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of VYSCOXA. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use VYSCOXA for a condition for which it was not prescribed. Do not give VYSCOXA to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about VYSCOSA that is written for health professionals. |
||
|
What are the ingredients in VYSCOXA? Active ingredient: celecoxib Inactive ingredients: anhydrous citric acid, glycerin, magnesium aluminometasilicate, methylparaben, propylparaben, purified water, sodium citrate dihydrate, sucralose, and xanthan gum. Manufactured for and Distributed by: Carwin Pharmaceutical Associates, LLC, Hazlet, NJ 07730 For more information, go to www.carwinpharma.com or call: 844-700-5011 |
Section 44425-7 (44425-7)
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from Light. Shake well before use. Discard 21 days after opening.
10. Overdosage (10. OVERDOSAGE)
Symptoms following acute NSAID overdosages have been typically limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which have been generally reversible with supportive care. Gastrointestinal bleeding has occurred. Hypertension, acute renal failure, respiratory depression, and coma have occurred, but were rare [see Warnings and Precautions (5.2, 5.4, 5.6) ].
No overdoses of celecoxib were reported during clinical trials. Doses up to 2400 mg/day for up to 10 days in 12 patients did not result in serious toxicity. No information is available regarding the removal of celecoxib by hemodialysis, but based on its high degree of plasma protein binding (>97%) dialysis is unlikely to be useful in overdose.
Manage patients with symptomatic and supportive care following an NSAID overdosage. There are no specific antidotes. Consider emesis and/or activated charcoal (60 to 100 grams in adults, 1 to 2 grams per kg of body weight in pediatric patients) and/or osmotic cathartic in symptomatic patients seen within four hours of ingestion or in patients with a large overdosage (5 to 10 times the recommended dosage). Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
For additional information about overdosage treatment contact a poison control center (1-800-222-1222).
11. Description (11. DESCRIPTION)
VYSCOXA contains celecoxib, a nonsteroidal anti-inflammatory agent. The chemical name of celecoxib is 4-[5-(4-methylphenyl)- 3-(trifluoromethyl)-1H-pyrazol-1-yl] benzenesulfonamide. Celecoxib has a molecular weight of is 381.38 g/mol, a molecular formula of C17H14F3N3O2S, and the following structural formula:
Celecoxib is a white to off-white powder with a pKa of 11.1 (sulfonamide moiety). Celecoxib is hydrophobic (log P is 3.5) and is practically insoluble in aqueous media at physiological pH range.
The inactive ingredients in VYSCOXA oral suspension include: anhydrous citric acid, glycerin, magnesium aluminometasilicate, methylparaben, propylparaben, purified water, sodium citrate dihydrate, sucralose, and xanthan gum. Sodium hydroxide and hydrochloric acid may be used to adjust the pH to 4.5 – 5.5.
5.4 Hypertension
NSAIDs, including VYSCOXA, can lead to new onset of hypertension or worsening of preexisting hypertension, either of which may contribute to the increased incidence of CV events. Patients taking angiotensin converting enzyme (ACE) inhibitors, thiazide diuretics or loop diuretics may have impaired response to these therapies when taking NSAIDs [see Drug Interactions (7) ].
See Clinical Studies (14.5, 14.6) for additional blood pressure data for VYSCOXA.
Monitor blood pressure (BP) during the initiation of NSAID treatment and throughout the course of therapy.
8.4 Pediatric Use
The safety and effectiveness of celecoxib for the management of the signs and symptoms of Juvenile Rheumatoid Arthritis have been established in pediatric patients 2 years of age and older. The use of celecoxib in patients 2 years of age and older is supported by evidence from a 12-week, double-blind, active-controlled, pharmacokinetic, safety and efficacy study, with a 12-week open-label extension conducted in patients 2 to 17 years of age. Safety and efficacy have not been studied beyond six months in pediatric patients.
Patients with systemic onset JRA (without active systemic features) appear to be at risk for the development of abnormal coagulation laboratory tests. In some patients with systemic onset JRA, both celecoxib and naproxen were associated with mild prolongation of activated partial thromboplastin time (APTT) but not prothrombin time (PT). When NSAIDs including celecoxib are used in patients with systemic onset JRA, monitor patients for signs and symptoms of abnormal clotting or bleeding, due to the risk of disseminated intravascular coagulation. Patients with systemic onset JRA should be monitored for the development of abnormal coagulation tests. The long-term cardiovascular toxicity in children exposed to celecoxib has not been evaluated and it is unknown if long-term risks may be similar to that seen in adults exposed to celecoxib or other COX-2 selective and non- selective NSAIDs [see Boxed Warning, Dosage and Administration (2.3), Warnings and Precautions (5.15), Warnings and Precautions (5.5), Adverse Reactions (6.1), Animal Toxicology (13.2), Clinical Studies (14.4) ].
Alternative therapies for treatment of JRA should be considered in pediatric patients identified to be CYP2C9 poor metabolizers [see Poor Metabolizers of CYP2C9 substrates (8.8) ].
The safety and effectiveness of VYSCOXA have not been established in pediatric patients with JRA less than 2 years of age, in patients with a body weight less than 10 kg, and in patients with active systemic features.
The safety and effectiveness of VYSCOXA have not been established in pediatric patients for osteoarthritis or ankylosing spondylitis.
8.5 Geriatric Use
Elderly patients, compared to younger patients, are at greater risk for NSAID-associated serious cardiovascular, gastrointestinal, and/or renal adverse reactions. If the anticipated benefit for the elderly patient outweighs these potential risks, start dosing at the low end of the dosing range, and monitor patients for adverse effects [see Warnings and Precautions (5.1, 5.2, 5.3, 5.6, 5.14) ].
Of the total number of patients who received celecoxib in pre-approval clinical trials, more than 3,300 were 65–74 years of age, while approximately 1,300 additional patients were 75 years and over. No substantial differences in effectiveness were observed between these subjects and younger subjects. In clinical studies comparing renal function as measured by the GFR, BUN and creatinine, and platelet function as measured by bleeding time and platelet aggregation, the results were not different between elderly and young volunteers. However, as with other NSAIDs, including those that selectively inhibit COX-2, there have been more spontaneous post-marketing reports of fatal GI events and acute renal failure in the elderly than in younger patients [see Warnings and Precautions (5.2, 5.6) ].
1.1 Osteoarthritis
(OA) [see Clinical Studies (14.1) ].
2.2 Osteoarthritis
For OA, the dosage is 200 mg (20 mL) per day administered as a single dose or as 100 mg (10 mL) twice daily.
5.3 Hepatotoxicity
Elevations of ALT or AST (three or more times the upper limit of normal [ULN]) have been reported in approximately 1% of NSAID-treated patients in clinical trials. In addition, rare, sometimes fatal, cases of severe hepatic injury, including fulminant hepatitis, liver necrosis, and hepatic failure have been reported.
Elevations of ALT or AST (less than three times ULN) may occur in up to 15% of patients treated with NSAIDs including celecoxib.
In controlled clinical trials of celecoxib, the incidence of borderline elevations (greater than or equal to 1.2 times and less than 3 times the upper limit of normal) of liver associated enzymes was 6% for celecoxib and 5% for placebo, and approximately 0.2% of patients taking celecoxib and 0.3% of patients taking placebo had notable elevations of ALT and AST.
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, diarrhea, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash), discontinue VYSCOXA immediately, and perform a clinical evaluation of the patient.
14.1 Osteoarthritis
Celecoxib has demonstrated significant reduction in joint pain compared to placebo. Celecoxib was evaluated for treatment of the signs and the symptoms of OA of the knee and hip in placebo- and active-controlled clinical trials of up to 12 weeks duration. In patients with OA, treatment with celecoxib 100 mg twice daily or 200 mg once daily resulted in improvement in WOMAC (Western Ontario and McMaster Universities) osteoarthritis index, a composite of pain, stiffness, and functional measures in OA. In three 12-week studies of pain accompanying OA flare, celecoxib doses of 100 mg twice daily and 200 mg twice daily provided significant reduction of pain within 24 to 48 hours of initiation of dosing. At doses of 100 mg twice daily or 200 mg twice daily the effectiveness of celecoxib was shown to be similar to that of naproxen 500 mg twice daily. Doses of 200 mg twice daily provided no additional benefit above that seen with 100 mg twice daily. A total daily dose of 200 mg has been shown to be equally effective whether administered as 100 mg twice daily or 200 mg once daily.
14. Clinical Studies (14. CLINICAL STUDIES)
The effectiveness of VYSCOXA has been established based on adequate and well-controlled studies of another formulation of celecoxib [see Clinical Pharmacology (12.3) ]. Below is a display of the efficacy results of the adequate and well-controlled studies of celecoxib.
4. Contraindications (4. CONTRAINDICATIONS)
VYSCOXA is contraindicated in the following patients:
-
•Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to celecoxib, any components of the drug product [see Warnings and Precautions (5.7, 5.9) ].
-
•History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs, have been reported in such patients [see Warnings and Precautions (5.7, 5.8) ].
-
•In the setting of CABG surgery [see Warnings and Precautions (5.1) ].
-
•In patients who have demonstrated allergic-type reactions to sulfonamides [see Warnings and Precautions (5.7) ].
6. Adverse Reactions (6. ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the labeling:
-
•Cardiovascular Thrombotic Events [see Warnings and Precautions (5.1) ]
-
•GI Bleeding, Ulceration and Perforation [see Warnings and Precautions (5.2) ]
-
•Hepatotoxicity [see Warnings and Precautions (5.3) ]
-
•Hypertension [see Warnings and Precautions (5.4) ]
-
•Heart Failure and Edema [see Warnings and Precautions (5.5) ]
-
•Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.6) ]
-
•Anaphylactic Reactions [see Warnings and Precautions (5.7) ]
-
•Serious Skin Reactions [see Warnings and Precautions (5.9) ]
-
•Hematologic Toxicity [see Warnings and Precautions (5.12) ]
7. Drug Interactions (7. DRUG INTERACTIONS)
See Table 3 for clinically significant drug interactions with celecoxib.
|
Drugs That Interfere with Hemostasis |
|
|
Clinical Impact: |
|
|
Intervention: |
Monitor patients with concomitant use of VYSCOXA with anticoagulants (e.g., warfarin), antiplatelet drugs (e.g., aspirin), SSRIs, and SNRIs for signs of bleeding [see Warnings and Precautions (5.12) ]. |
|
Aspirin |
|
|
Clinical Impact: |
Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2)
]. |
|
Intervention: |
Concomitant use of VYSCOXA and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [see Warnings and Precautions (5.12)
]. |
|
ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers |
|
|
Clinical Impact: |
|
|
Intervention: |
|
|
Diuretics |
|
|
Clinical Impact: |
Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. |
|
Intervention: |
During concomitant use of VYSCOXA with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [see Warnings and Precautions (5.6) ]. |
|
Digoxin |
|
|
Clinical Impact: |
The concomitant use of Celecoxib with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. |
|
Intervention: |
During concomitant use of VYSCOXA and digoxin, monitor serum digoxin levels. |
|
Lithium |
|
|
Clinical Impact: |
NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. |
|
Intervention: |
During concomitant use of VYSCOXA and lithium, monitor patients for signs of lithium toxicity. |
|
Methotrexate |
|
|
Clinical Impact: |
Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). |
|
Intervention: |
During concomitant use of VYSCOXA and methotrexate, monitor patients for methotrexate toxicity. |
|
Cyclosporine |
|
|
Clinical Impact: |
Concomitant use of celecoxib and cyclosporine may increase cyclosporine's nephrotoxicity. |
|
Intervention: |
During concomitant use of VYSCOXA and cyclosporine, monitor patients for signs of worsening renal function. |
|
NSAIDs and Salicylates |
|
|
Clinical Impact: |
Concomitant use of Celecoxib with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy [see Warnings and Precautions (5.2) ]. |
|
Intervention: |
The concomitant use of Celecoxib with other NSAIDs or salicylates is not recommended. |
|
Pemetrexed |
|
|
Clinical Impact: |
Concomitant use of celecoxib and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). |
|
Intervention: |
During concomitant use of VYSCOXA and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. |
|
CYP2C9 Inhibitors or inducers |
|
|
Clinical Impact: |
Celecoxib metabolism is predominantly mediated via cytochrome P450 (CYP) 2C9 in the liver. Co-administration of celecoxib with drugs that are known to inhibit CYP2C9 (e.g., fluconazole) may enhance the exposure and toxicity of celecoxib whereas co-administration with CYP2C9 inducers (e.g., rifampin) may lead to compromised efficacy of celecoxib. |
|
Intervention |
Evaluate each patient's medical history when consideration is given to prescribing VYSCOXA. A dosage adjustment may be warranted when VYSCOXA is administered with CYP2C9 inhibitors or inducers [see Clinical Pharmacology (12.3) ]. |
|
CYP2D6 substrates |
|
|
Clinical Impact: |
In vitro studies indicate that celecoxib, although not a substrate, is an inhibitor of CYP2D6. Therefore, there is a potential for an in vivo drug interaction with drugs that are metabolized by CYP2D6 (e.g., atomoxetine), and celecoxib may enhance the exposure and toxicity of these drugs. |
|
Intervention |
Evaluate each patient's medical history when consideration is given to prescribing VYSCOXA. A dosage adjustment may be warranted when VYSCOXA is administered with CYP2D6 substrates [see Clinical Pharmacology (12.3) ]. |
|
Corticosteroids |
|
|
Clinical Impact: |
Concomitant use of corticosteroids with celecoxib may increase the risk of GI ulceration or bleeding. |
|
Intervention |
Monitor patients with concomitant use of VYSCOXA with corticosteroids for signs of bleeding [see Warnings and Precautions (5.2)]. |
8.7 Renal Impairment
VYSCOXA is not recommended in patients with severe renal insufficiency [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3) ].
12.3 Pharmacokinetics
Celecoxib has extensive distribution and high protein binding. It is primarily metabolized by CYP2C9 with a half-life of approximately 11 hours.
12.5 Pharmacogenomics
CYP2C9 activity is reduced in individuals with genetic polymorphisms that lead to reduced enzyme activity, such as those homozygous for the CYP2C9*2 and CYP2C9*3 polymorphisms. Limited data from 4 published reports that included a total of 8 subjects with the homozygous CYP2C9*3/*3 genotype showed celecoxib systemic levels that were 3- to 7-fold higher in these subjects compared to subjects with CYP2C9*1/*1 or *I/*3 genotypes. The pharmacokinetics of celecoxib have not been evaluated in subjects with other CYP2C9 polymorphisms, such as *2, *5, *6, *9 and *11. It is estimated that the frequency of the homozygous *3/*3 genotype is 0.3% to 1.0% in various ethnic groups [see Dosage and Administration (2.6), Use in Specific Populations (8.8) ].
13.2 Animal Toxicology
An increase in the incidence of background findings of spermatocele with or without secondary changes such as epididymal hypospermia as well as minimal to slight dilation of the seminiferous tubules was seen in the juvenile rat. These reproductive findings while apparently treatment-related did not increase in incidence or severity with dose and may indicate an exacerbation of a spontaneous condition. Similar reproductive findings were not observed in studies of juvenile or adult dogs or in adult rats treated with celecoxib. The clinical significance of this observation is unknown.
8.6 Hepatic Impairment
The daily recommended dose of VYSCOXA oral suspension in patients with moderate hepatic impairment (Child-Pugh Class B) should be reduced by 50%. The use of VYSCOXA in patients with severe hepatic impairment is not recommended [see Dosage and Administration (2.6) and Clinical Pharmacology (12.3) ].
1. Indications and Usage (1. INDICATIONS AND USAGE)
VYSCOXA is indicated
In adults for the management of the signs and symptoms of:
1.2 Rheumatoid Arthritis
(RA) [see Clinical Studies (14.2) ].
12.1 Mechanism of Action
Celecoxib has analgesic, anti-inflammatory, and antipyretic properties.
The mechanism of action of celecoxib is believed to be due to inhibition of prostaglandin synthesis, primarily via inhibition of COX-2. Celecoxib is a potent inhibitor of prostaglandin synthesis in vitro. Celecoxib concentrations reached during therapy have produced in vivo effects. Prostaglandins sensitize afferent nerves and potentiate the action of bradykinin in inducing pain in animal models. Prostaglandins are mediators of inflammation. Since celecoxib is an inhibitor of prostaglandin synthesis, its mode of action may be due to a decrease of prostaglandins in peripheral tissues.
2.3 Rheumatoid Arthritis
For RA, the dosage is 100 mg (10 mL) to 200 mg (20 mL) twice daily.
14.2 Rheumatoid Arthritis
Celecoxib has demonstrated significant reduction in joint tenderness/pain and joint swelling compared to placebo. Celecoxib was evaluated for treatment of the signs and symptoms of RA in placebo- and active-controlled clinical trials of up to 24 weeks in duration. Celecoxib was shown to be superior to placebo in these studies, using the ACR20 Responder Index, a composite of clinical, laboratory, and functional measures in RA. Celecoxib doses of 100 mg twice daily and 200 mg twice daily were similar in effectiveness and both were comparable to naproxen 500 mg twice daily.
Although celecoxib 100 mg twice daily and 200 mg twice daily provided similar overall effectiveness, some patients derived additional benefit from the 200 mg twice daily dose. Doses of 400 mg twice daily provided no additional benefit above that seen with 100 mg to 200 mg twice daily.
1.3 Ankylosing Spondylitis
(AS) [see Clinical Studies (14.3) ].
In pediatric patients two years of age and older for the management of the signs and symptoms of:
2.4 Ankylosing Spondylitis
For AS, the dosage is 200 mg (20 mL) daily in single (once per day) dose or 100 mg (10 mL) twice daily. If no effect is observed after 6 weeks, a trial of 200 mg (20 mL) twice daily may be worthwhile. If no effect is observed after 6 weeks on 200 mg (20 mL) twice daily, a response is not likely and consideration should be given to alternate treatment options.
5.14 Laboratory Monitoring
Because serious GI bleeding, hepatotoxicity, and renal injury can occur without warning symptoms or signs, consider monitoring patients on long-term NSAID treatment with a CBC and a chemistry profile periodically [see Warnings and Precautions (5.2, 5.3, 5.6) ].
In controlled clinical trials, elevated BUN occurred more frequently in patients receiving celecoxib compared with patients on placebo. This laboratory abnormality was also seen in patients who received comparator NSAIDs in these studies. The clinical significance of this abnormality has not been established.
5.7 Anaphylactic Reactions
Celecoxib has been associated with anaphylactic reactions in patients with and without known hypersensitivity to celecoxib and in patients with aspirin sensitive asthma. Celecoxib is a sulfonamide and both NSAIDs and sulfonamides may cause allergic type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people [see Contraindications (4) and Warnings and Precautions (5.8) ].
Seek emergency help if any anaphylactic reaction occurs.
5.9 Serious Skin Reactions
Serious skin reactions have occurred following treatment with celecoxib, including erythema multiforme, exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalized exanthematous pustulosis (AGEP), and fixed drug eruption (FDE) which may present as a more severe variant known as generalized bullous fixed drug eruption (GBFDE). These serious events may occur without warning and can be fatal.
Inform patients about the signs and symptoms of serious skin reactions, and to discontinue the use of VYSCOXA at the first appearance of skin rash or any other sign of hypersensitivity. VYSCOXA is contraindicated in patients with previous serious skin reactions to NSAIDs [see Contraindications (4) ].
14.3 Ankylosing Spondylitis
Celecoxib was evaluated in AS patients in two placebo- and active-controlled clinical trials of 6 and 12 weeks duration. Celecoxib at doses of 100 mg twice daily, 200 mg once daily and 400 mg once daily was shown to be statistically superior to placebo in these studies for all three co-primary efficacy measures assessing global pain intensity (Visual Analogue Scale), global disease activity (Visual Analogue Scale) and functional impairment (Bath Ankylosing Spondylitis Functional Index). In the 12-week study, there was no difference in the extent of improvement between the 200 mg and 400 mg celecoxib doses in a comparison of mean change from baseline, but there was a greater percentage of patients who responded to celecoxib 400 mg, 53%, than to celecoxib 200 mg, 44%, using the Assessment in Ankylosing Spondylitis response criteria (ASAS 20). The ASAS 20 defines a responder as improvement from baseline of at least 20% and an absolute improvement of at least 10 mm, on a 0 mm to 100 mm scale, in at least three of the four following domains: patient global pain, Bath Ankylosing Spondylitis Functional Index, and inflammation. The responder analysis also demonstrated no change in the responder rates beyond 6 weeks.
5. Warnings and Precautions (5. WARNINGS AND PRECAUTIONS)
-
•Hepatotoxicity: Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. (5.3)
-
•Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure. (5.4, 7)
-
•Heart Failure and Edema: Avoid use of VYSCOXA in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure. (5.5)
-
•Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of VYSCOXA in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function. (5.6)
-
•Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs. (5.7)
-
•Exacerbation of Asthma Related to Aspirin Sensitivity: VYSCOXA is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with preexisting asthma (without aspirin sensitivity). (5.8)
-
•Serious Skin Reactions: Discontinue VYSCOXA at first appearance of skin rash or other signs of hypersensitivity. (5.9)
-
•Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue and evaluate clinically. (5.10)
-
•Fetal Toxicity: Limit use of NSAIDs, including VYSCOXA, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the fetal ductus arteriosus. (5.11, 8.1)
-
•Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia. (5.12, 7)
5.12 Hematological Toxicity
Anemia has occurred in NSAID-treated patients. This may be due to occult or gross blood loss, fluid retention, or an incompletely described effect on erythropoiesis. If a patient treated with VYSCOXA has any signs or symptoms of anemia, monitor hemoglobin or hematocrit.
In controlled clinical trials the incidence of anemia was 0.6% with celecoxib and 0.4% with placebo. Patients on long-term treatment with VYSCOXA should have their hemoglobin or hematocrit checked if they exhibit any signs or symptoms of anemia or blood loss.
NSAIDs, including VYSCOXA, may increase the risk of bleeding events. Co-morbid conditions such as coagulation disorders or concomitant use of warfarin, other anticoagulants, antiplatelet drugs (e.g., aspirin), SSRIs and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients for signs of bleeding [see Drug Interactions (7) ].
5.5 Heart Failure and Edema
The Coxib and traditional NSAID Trialists' Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of celecoxib may blunt the CV effects of several therapeutic agents used to treat these medical conditions (e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers [ARBs]) [see Drug Interactions (7) ].
In the CLASS study [see Clinical Studies (14.6) ], the Kaplan-Meier cumulative rates at 9 months of peripheral edema in patients on celecoxib 400 mg twice daily (4-fold and 2-fold the recommended OA and RA doses, respectively), ibuprofen 800 mg three times daily and diclofenac 75 mg twice daily were 4.5%, 6.9% and 4.7%, respectively.
Avoid the use of VYSCOXA in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If VYSCOXA is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
2. Dosage and Administration (2. DOSAGE AND ADMINISTRATION)
-
•Use the lowest effective dosage for shortest duration consistent with individual patient treatment goals. (2.1)
-
•OA: 200 mg (20 mL) once daily or 100 mg (10 mL) twice daily. (2.2, 14.1)
-
•RA: 100 mg (10 mL) to 200 mg (20 mL) twice daily. (2.3, 14.2)
-
•AS: 200 mg (20 mL) once daily single dose or 100 mg (10 mL) twice daily. If no effect is observed after 6 weeks, a trial of 200 mg (20 mL) twice daily may be of benefit. (2.4, 14.3)
-
•JRA: 50 mg (5 mL) twice daily in patients 10 kg to 25 kg. 100 mg (10 mL) twice daily in patients more than 25 kg. (2.5, 14.4)
Important:VYSCOXA is not recommended at a single dose greater than 200 mg (20 mL). Single doses of the suspension greater than 200 mg (20 mL) may result in celecoxib concentrations higher than expected. For patients who require a single dose over 200 mg (20 mL), use a different celecoxib formulation (2.1, 12.3)
Hepatic Impairment: Reduce daily dose by 50% in patients with moderate hepatic impairment (Child-Pugh Class B). (2.6, 8.6, 12.3)
Poor Metabolizers of CYP2C9 Substrates: Consider a dose reduction by 50% (or alternative management for JRA) in patients who are known or suspected to be CYP2C9 poor metabolizers. (2.6, 8.8, 12.3).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of celecoxib. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure
Cardiovascular: Vasculitis, deep venous thrombosis
General: Anaphylactoid reaction, angioedema
Liver and biliary: Liver necrosis, hepatitis, jaundice, hepatic failure
Hemic and lymphatic: Agranulocytosis, aplastic anemia, pancytopenia, leucopenia
Metabolic: Hypoglycemia, hyponatremia
Nervous: Aseptic meningitis, ageusia, anosmia, fatal intracranial hemorrhage
Renal: Interstitial nephritis
Skin and Appendages: Erythema multiforme, exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalized exanthematous pustulosis (AGEP), and fixed drug eruption (FDE).
3. Dosage Forms and Strengths (3. DOSAGE FORMS AND STRENGTHS)
Oral suspension: 10 mg/mL of celecoxib as a white, opaque, unflavored suspension.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The adverse reaction information from clinical trials does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating rates.
Of the celecoxib-treated patients in the pre-marketing controlled clinical trials of another formulation of celecoxib, approximately 4,250 were patients with OA, approximately 2,100 were patients with RA, and approximately 1,050 were patients with post-surgical pain. More than 8,500 patients received a total daily dose of celecoxib of 200 mg (100 mg twice daily or 200 mg once daily) or more, including more than 400 treated at 800 mg (400 mg twice daily). Approximately 3,900 patients received celecoxib at these doses for 6 months or more; approximately 2,300 of these have received it for 1 year or more and 124 of these have received it for 2 years or more.
8. Use in Specific Populations (8. USE IN SPECIFIC POPULATIONS)
Infertility: NSAIDs are associated with reversible infertility. Consider withdrawal of VYSCOXA in women who have difficulties conceiving. (8.3)
2.1 General Dosing Instructions
Carefully consider the potential benefits and risks of VYSCOXA and other treatment options before deciding to use VYSCOXA. Use the lowest effective dosage for the shortest duration consistent with individual patient treatment goals [see Warnings and Precautions (5) ].
The maximum single dose of VYSCOXA is 200 mg (20 mL). Administering more than 200 mg (20 mL) in a single dose of the VYXCOXA suspension may result in higher than intended plasma concentrations of celecoxib. In patients requiring a single dose greater than 200 mg (20 mL), use a different product.
VYSCOXA must be taken on an empty stomach at least 2 hours before or 1 hour after food [see Clinical Pharmacology (12.3) ].
For patients who cannot tolerate dosing with VYSCOXA on an empty stomach, discontinue the use of this product. Do not advise the patient to take VYSCOXA with food.
1.4 Juvenile Rheumatoid Arthritis
(JRA) [see Clinical Studies (14.4) ].
Limitations of Use
VYSCOXA must be administered on an empty stomach at least 2 hours before or 1 hour after food. Taking VYSCOXA with food results in plasma exposures of celecoxib up to 50% higher than intended. If patients cannot tolerate VYSCOXA in the fasted state, discontinue use of VYSCOXA (reference Sections 2.1 , 5, 12).
VYSCOXA is NOT indicated for the management of acute pain or treatment of primary dysmenorrhea.
2.5 Juvenile Rheumatoid Arthritis
For JRA, the dosage for pediatric patients (age 2 years and older) is based on weight. For patients weighing 10 kg to 25 kg the recommended dose is 50 mg (5 mL) twice daily. For patients greater than 25 kg the recommended dose is 100 mg (10 mL) twice daily.
17. Patient Counseling Information (17. PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide) that accompanies each prescription dispensed. Inform patients, families, or their caregivers of the following information before initiating therapy with VYSCOXA and periodically during the course of ongoing therapy.
5.1 Cardiovascular Thrombotic Events
Clinical trials of several cyclooxygenase-2 (COX-2) selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.
In the APC (Adenoma Prevention with Celecoxib) trial, there was about a threefold increased risk of the composite endpoint of cardiovascular death, MI, or stroke for the celecoxib 400 mg twice daily and celecoxib 200 mg twice daily treatment arms compared to placebo. The increases in both celecoxib dose groups versus placebo-treated patients were mainly due to an increased incidence of myocardial infarction [see Clinical Studies (14.6) ].
A randomized controlled trial entitled the Prospective Randomized Evaluation of Celecoxib Integrated Safety vs. Ibuprofen Or Naproxen (PRECISION) was conducted to assess the relative cardiovascular thrombotic risk of a COX-2 inhibitor, celecoxib, compared to the non-selective NSAIDs naproxen and ibuprofen. Celecoxib 100 mg twice daily was non-inferior to naproxen 375 to 500 mg twice daily and ibuprofen 600 to 800 mg three times daily for the composite endpoint of the Antiplatelet Trialists' Collaboration (APTC), which consists of cardiovascular death (including hemorrhagic death), non-fatal myocardial infarction, and non-fatal stroke [see Clinical Studies (14.5) ].
To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as celecoxib, increases the risk of serious gastrointestinal (GI) events [see Warnings and Precautions (5.2) ].
16. How Supplied/storage and Handling (16. HOW SUPPLIED/STORAGE AND HANDLING)
VYSCOXA (celecoxib) oral suspension, 10 mg/mL celecoxib is a white, opaque, unflavored suspension, supplied as:
| NDC Number | Size |
|---|---|
|
15370-501-16 |
473 mL Bottle |
5.13 Masking of Inflammation and Fever
The pharmacological activity of VYSCOXA in reducing inflammation, and possibly fever, may diminish the utility of diagnostic signs in detecting infections.
8.8 Poor Metabolizers of Cyp2c9 Substrates (8.8 Poor Metabolizers of CYP2C9 Substrates)
In patients who are known or suspected to be poor CYP2C9 metabolizers (i.e., CYP2C9*3/*3), based on genotype or previous history/experience with other CYP2C9 substrates (such as warfarin, phenytoin) administer VYSCOXA starting with half the lowest recommended dose. Alternative management should be considered in JRA patients identified to be CYP2C9 poor metabolizers [see Dosage and Administration (2.6) and Clinical Pharmacology (12.5) ].
14.4 Juvenile Rheumatoid Arthritis (nct00652925) (14.4 Juvenile Rheumatoid Arthritis (NCT00652925))
In a 12-week, randomized, double-blind active-controlled, parallel-group, multicenter, noninferiority study, patients from 2 years to 17 years of age with pauciarticular, polyarticular course JRA or systemic onset JRA (with currently inactive systemic features), received one of the following treatments: celecoxib 3 mg/kg (to a maximum of 150 mg) twice daily; celecoxib 6 mg/kg (to a maximum of 300 mg) twice daily; or naproxen 7.5 mg/kg (to a maximum of 500 mg) twice daily. The response rates were based upon the JRA Definition of Improvement greater than or equal to 30% (JRA DOI 30) criterion, which is a composite of clinical, laboratory, and functional measures of JRA. The JRA DOI 30 response rates at week 12 were 69%, 80% and 67% in the celecoxib 3 mg/kg twice daily, celecoxib 6 mg/kg twice daily, and naproxen 7.5 mg/kg twice daily treatment groups, respectively.
The efficacy and safety of celecoxib for JRA have not been studied beyond six months. The long-term cardiovascular toxicity in children exposed to celecoxib has not been evaluated and it is unknown if the long-term risk may be similar to that seen in adults exposed to celecoxib or other COX-2 selective and non-selective NSAIDs [see Boxed Warning, Warnings and Precautions (5.1, 5.15) ].
5.15 Disseminated Intravascular Coagulation (dic) (5.15 Disseminated Intravascular Coagulation (DIC))
Because of the risk of disseminated intravascular coagulation with use of VYSCOXA in pediatric patients with systemic onset JRA, monitor patients for signs and symptoms of abnormal clotting or bleeding, and inform patients and their caregivers to report symptoms as soon as possible.
5.8 Exacerbation of Asthma Related to Aspirin Sensitivity
A subpopulation of patients with asthma may have aspirin-sensitive asthma which may include chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other NSAIDs. Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, VYSCOXA is contraindicated in patients with this form of aspirin sensitivity [see Contraindications (4) ]. When VYSCOXA is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for changes in the signs and symptoms of asthma.
5.2 Gastrointestinal Bleeding, Ulceration, and Perforation
NSAIDs, including celecoxib cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with VYSCOXA. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occurred in approximately 1% of patients treated for 3 to 6 months, and in about 2% to 4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
5.10 Drug Reaction With Eosinophilia and Systemic Symptoms (dress) (5.10 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS))
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as celecoxib. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue VYSCOXA and evaluate the patient immediately.
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events (WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS)
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS
See full prescribing information for complete boxed warning.
-
•Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in the treatment and may increase with duration of use. (5.1)
-
•VYSCOXA is contraindicated in the setting of coronary artery bypass graft (CABG) surgery. (4, 5.1)
-
•NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events. (5.2)
Principal Display Panel Celecoxib Oral Suspension, 10 Mg/ml (container Label) (PRINCIPAL DISPLAY PANEL - Celecoxib Oral Suspension, 10 mg/mL (Container Label))
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:47.508894 · Updated: 2026-03-14T22:30:51.485924