Allergena Zone 9
505fe34f-07b7-42ab-a982-5ae4bca10e13
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
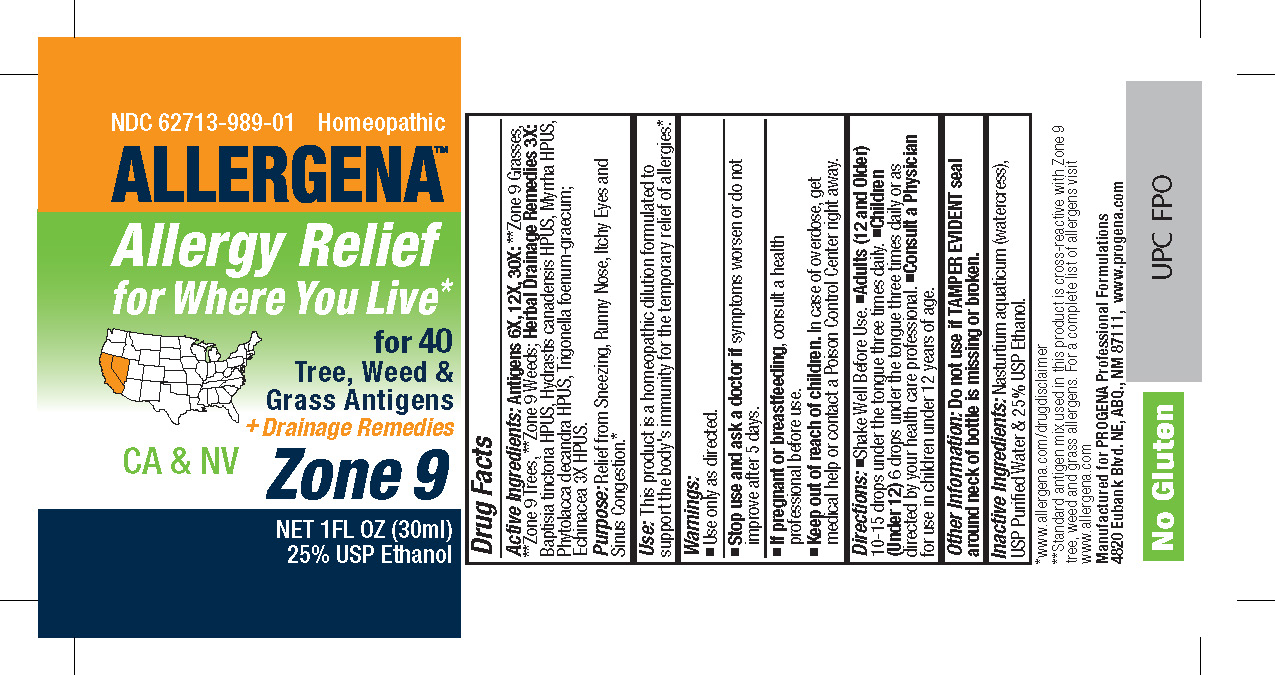
Active Ingredients: Antigens 6X, 12X, 30X: **Zone 9 Grasses, **Zone 9 Trees, **Zone 9 Weeds; Herbal Drainage Remedies 3X: Baptisia tinctoria HPUS, Hydrastis canadensis HPUS, Myrrha HPUS, Phytolacca decandra HPUS, Trigonella foenum-graecum; Echinacea 3X HPUS.
Medication Information
Warnings and Precautions
Warnings:
- Use Only as directed.
- Stop use and ask a doctorif symptoms worsen or do not improve after 5 days.
- If pregnant or breast-feeding, consult a health professional before use.
Indications and Usage
Use:This product is a Homeopathic Dilution formulated to support the body's immunity for the temporary relief of allergies.*
Dosage and Administration
Directions:•Shake Well Before Use. • Adults (12 and Older)10-15 drops under the tongue three times daily. • Children (2-12) 6-10 drops under the tongue three times daily. • Infants (0-2)3-6 drops under the tongue three times daily or as directed by your health care professional.
Description
Active Ingredients: Antigens 6X, 12X, 30X: **Zone 9 Grasses, **Zone 9 Trees, **Zone 9 Weeds; Herbal Drainage Remedies 3X: Baptisia tinctoria HPUS, Hydrastis canadensis HPUS, Myrrha HPUS, Phytolacca decandra HPUS, Trigonella foenum-graecum; Echinacea 3X HPUS.
Section 50565-1
Keep Out of Reach of Children.In case of overdose, get medical help or contact a
Poison Control Center right away.
Section 51727-6
Inactive Ingredients:Nasturtium aquaticum (watercress), USP Purified Water and 25% USP Ethanol.
Section 51945-4
*www.allergena.com/drugdisclaimer
**Standard antigen mix used in this product is cross-reactive with Zone 9 Tree, Weed, and Grass allergens. For a complete list of allergens visit
www.allergena.com
Manufactured for PROGENA Professional Formulations
4820 Eubank Blvd. NE, ABQ, NM 87111, www.progena.com
Section 55105-1
Purpose:Relief from Sneezing, Runny Nose, Itchy Eyes and Sinus Congestion.*
Section 55106-9
Active Ingredients: Antigens 6X, 12X, 30X:**Zone 9 Grasses, **Zone 9 Trees, **Zone 9 Weeds; Herbal Drainage Remedies 3X:Baptisia tinctoria HPUS, Hydrastis canadensis HPUS, Myrrha HPUS, Phytolacca decandra HPUS, Trigonella foenum-graecum; Echinacea 3X HPUS.
Section 60561-8
Other Information: Do not use if TAMPER EVIDENT seal around neck of bottle is missing or broken.
Structured Label Content
Indications and Usage (34067-9)
Use:This product is a Homeopathic Dilution formulated to support the body's immunity for the temporary relief of allergies.*
Dosage and Administration (34068-7)
Directions:•Shake Well Before Use. • Adults (12 and Older)10-15 drops under the tongue three times daily. • Children (2-12) 6-10 drops under the tongue three times daily. • Infants (0-2)3-6 drops under the tongue three times daily or as directed by your health care professional.
Warnings and Precautions (34071-1)
Warnings:
- Use Only as directed.
- Stop use and ask a doctorif symptoms worsen or do not improve after 5 days.
- If pregnant or breast-feeding, consult a health professional before use.
Section 50565-1 (50565-1)
Keep Out of Reach of Children.In case of overdose, get medical help or contact a
Poison Control Center right away.
Section 51727-6 (51727-6)
Inactive Ingredients:Nasturtium aquaticum (watercress), USP Purified Water and 25% USP Ethanol.
Section 51945-4 (51945-4)
*www.allergena.com/drugdisclaimer
**Standard antigen mix used in this product is cross-reactive with Zone 9 Tree, Weed, and Grass allergens. For a complete list of allergens visit
www.allergena.com
Manufactured for PROGENA Professional Formulations
4820 Eubank Blvd. NE, ABQ, NM 87111, www.progena.com
Section 55105-1 (55105-1)
Purpose:Relief from Sneezing, Runny Nose, Itchy Eyes and Sinus Congestion.*
Section 55106-9 (55106-9)
Active Ingredients: Antigens 6X, 12X, 30X:**Zone 9 Grasses, **Zone 9 Trees, **Zone 9 Weeds; Herbal Drainage Remedies 3X:Baptisia tinctoria HPUS, Hydrastis canadensis HPUS, Myrrha HPUS, Phytolacca decandra HPUS, Trigonella foenum-graecum; Echinacea 3X HPUS.
Section 60561-8 (60561-8)
Other Information: Do not use if TAMPER EVIDENT seal around neck of bottle is missing or broken.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:11:56.972966 · Updated: 2026-03-14T23:17:15.281880