4f4541df-76d4-351b-e054-00144ff88e88
4f4541df-76d4-351b-e054-00144ff88e88
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
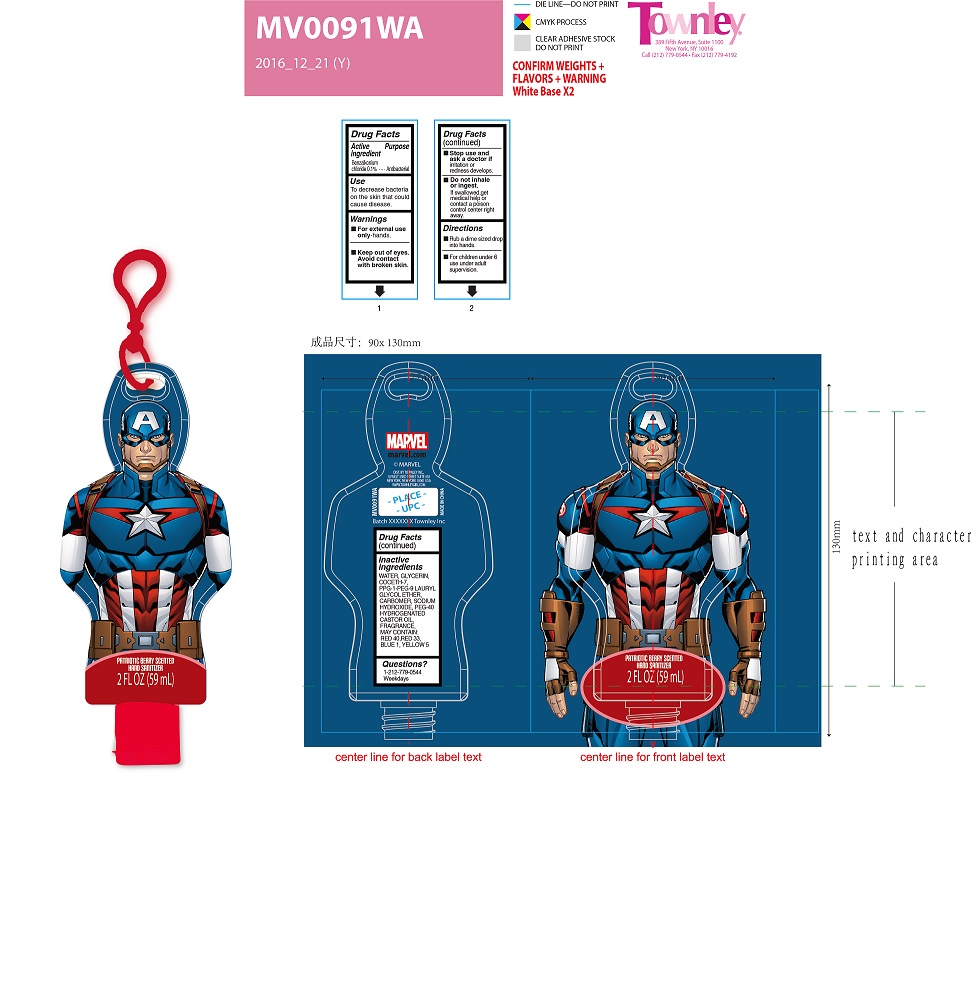
Active Ingredient Benzalkonium Chloride 0.1%
Medication Information
Warnings and Precautions
Warnings
- For external use only - hands.
- Keep out of eyes. Avoid contact with broken skin.
- Stop use and ask a doctor if irritation or redness develops.
- Do not inhale or ingest. If swallowed, get medical help or contact a poison control center right away.
Active Ingredient
Active Ingredient
Benzalkonium Chloride 0.1%
Indications and Usage
Use
To decrease bacteria on the skin that could cause disease
Dosage and Administration
Directions
- Rub a dime sized drop into hands.
- For children under 6 use under adult supervision.
Description
Active Ingredient Benzalkonium Chloride 0.1%
Section 50565-1
keep out of reach of children
Section 51727-6
Inactive ingredients
water, glycerin, coceth-7, PPG-1-PEG-9 lauryl glycol ether, carbomer, sodium hydroxide, PEG-40 hydrogenated castor oil, fragrance, may contain: red 40, red 33, blue 1, yellow 5.
Section 51945-4
Section 55105-1
Purpose
Antibacterial
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
- For external use only - hands.
- Keep out of eyes. Avoid contact with broken skin.
- Stop use and ask a doctor if irritation or redness develops.
- Do not inhale or ingest. If swallowed, get medical help or contact a poison control center right away.
Active Ingredient (55106-9)
Active Ingredient
Benzalkonium Chloride 0.1%
Indications and Usage (34067-9)
Use
To decrease bacteria on the skin that could cause disease
Dosage and Administration (34068-7)
Directions
- Rub a dime sized drop into hands.
- For children under 6 use under adult supervision.
Section 50565-1 (50565-1)
keep out of reach of children
Section 51727-6 (51727-6)
Inactive ingredients
water, glycerin, coceth-7, PPG-1-PEG-9 lauryl glycol ether, carbomer, sodium hydroxide, PEG-40 hydrogenated castor oil, fragrance, may contain: red 40, red 33, blue 1, yellow 5.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose
Antibacterial
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:19.366662 · Updated: 2026-03-14T22:54:43.254033