Qlosi
4ee2a840-1203-473a-aa42-316a3ddf2fbe
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
QLOSI is indicated for the treatment of presbyopia in adults.
Indications and Usage
QLOSI is indicated for the treatment of presbyopia in adults.
Dosage and Administration
Instill one drop of QLOSI in each eye. This can be repeated a second time after 2 to 3 hours for an effect up to 8 hours. QLOSI can be administered on a daily basis, or as needed, up to twice each day. If more than one topical ophthalmic medication is being used, the medicines must be administered at least 5 minutes apart. Discard single-patient-use vial after use.
Warnings and Precautions
Blurred Vision : Advise patients to not drive or operate machinery if vision is not clear (e.g., blurred vision). Exercise caution in night driving and other hazardous occupations in poor illumination. ( 5.1 ) Risk of Retinal Detachment : Rare cases of retinal detachment have been reported with miotics. Examination of the retina is advised in all patients prior to initiation of therapy. Advise patients to seek immediate medical care with sudden onset of flashes of lights, floaters, or vision loss. ( 5.2 ) Iritis : Caution is advised in patients with iritis. ( 5.3 )
Contraindications
QLOSI is contraindicated in patients with known hypersensitivity to the active ingredient or to any of the excipients.
Adverse Reactions
Most common adverse reactions (5% to 8%) are instillation site pain and headaches. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Orasis Pharmaceuticals Inc. at 1-866-ORASIS1 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
QLOSI is supplied as a sterile, clear, colorless to slightly yellowish and slightly viscous ophthalmic solution in configurations of 5 single-patient-use vials of 0.4 mL fill. Each single-patient-use vial is comprised of transparent low-density polyethylene (LDPE). 5 single-patient-use vials are packaged in a foil pouch. QLOSI is supplied in: NDC 83661-018-10: carton box of 10 single-patient-use vials (2 pouches containing 5 vials) NDC 83661-018-20: carton box of 20 single-patient-use vials (4 pouches containing 5 vials) NDC 83661-018-30: carton box of 30 single-patient-use vials (6 pouches containing 5 vials) NDC 83661-018-60: carton box of 60 single-patient-use vials (12 pouches containing 5 vials)
Medication Information
Warnings and Precautions
Blurred Vision : Advise patients to not drive or operate machinery if vision is not clear (e.g., blurred vision). Exercise caution in night driving and other hazardous occupations in poor illumination. ( 5.1 ) Risk of Retinal Detachment : Rare cases of retinal detachment have been reported with miotics. Examination of the retina is advised in all patients prior to initiation of therapy. Advise patients to seek immediate medical care with sudden onset of flashes of lights, floaters, or vision loss. ( 5.2 ) Iritis : Caution is advised in patients with iritis. ( 5.3 )
Indications and Usage
QLOSI is indicated for the treatment of presbyopia in adults.
Dosage and Administration
Instill one drop of QLOSI in each eye. This can be repeated a second time after 2 to 3 hours for an effect up to 8 hours. QLOSI can be administered on a daily basis, or as needed, up to twice each day. If more than one topical ophthalmic medication is being used, the medicines must be administered at least 5 minutes apart. Discard single-patient-use vial after use.
Contraindications
QLOSI is contraindicated in patients with known hypersensitivity to the active ingredient or to any of the excipients.
Adverse Reactions
Most common adverse reactions (5% to 8%) are instillation site pain and headaches. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Orasis Pharmaceuticals Inc. at 1-866-ORASIS1 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
QLOSI is supplied as a sterile, clear, colorless to slightly yellowish and slightly viscous ophthalmic solution in configurations of 5 single-patient-use vials of 0.4 mL fill. Each single-patient-use vial is comprised of transparent low-density polyethylene (LDPE). 5 single-patient-use vials are packaged in a foil pouch. QLOSI is supplied in: NDC 83661-018-10: carton box of 10 single-patient-use vials (2 pouches containing 5 vials) NDC 83661-018-20: carton box of 20 single-patient-use vials (4 pouches containing 5 vials) NDC 83661-018-30: carton box of 30 single-patient-use vials (6 pouches containing 5 vials) NDC 83661-018-60: carton box of 60 single-patient-use vials (12 pouches containing 5 vials)
Description
QLOSI is indicated for the treatment of presbyopia in adults.
Section 42229-5
Risk Summary
There are no adequate and well-controlled studies of QLOSI administration in pregnant women to inform a drug associated risk. Oral administration of pilocarpine to pregnant rats throughout organogenesis and lactation did not produce adverse effects at clinically relevant doses.
5.3 Iritis
QLOSI is not recommended to be used when iritis is present because adhesions (synechiae) may form between the iris and the lens.
16.2 Storage
Store refrigerated at 36°F to 46°F (2°C to 8°C). Once a pouch is open, the single-patient-use vials may be stored at room temperature [up to 77°F (25°C)] and used within 30 days.
Store unused product away from light. Discard opened vials after use.
10 Overdosage
Systemic toxicity following topical ocular administration of pilocarpine is rare, but occasionally patients who are sensitive may develop sweating and gastrointestinal overactivity.
Accidental ingestion can produce sweating, salivation, nausea, tremors and slowing of the pulse and a decrease in blood pressure.
In moderate overdosage, spontaneous recovery is to be expected and is aided by intravenous fluids to compensate for dehydration. For patients demonstrating severe poisoning, atropine, the pharmacologic antagonist to pilocarpine, should be used.
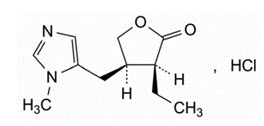
11 Description
QLOSI (pilocarpine hydrochloride ophthalmic solution) 0.4% is a sterile, clear, colorless to slightly yellowish and slightly viscous ophthalmic solution for topical ophthalmic use which has a pH between 5.1-6.1.
The structural formula of pilocarpine hydrochloride is:
The chemical name for pilocarpine hydrochloride is: 2(3 H)-Furanone, 3-ethyldihydro-4-[(1-methyl-1 H-imidazol-5-yl)- methyl]-monohydrochloride, (3 S-cis). Its molecular weight is 244.72 g/mol and its molecular formula is C 11H 16N 2O 2∙ HCl.
Each mL contains 0.4% (4 mg) of pilocarpine hydrochloride as the active ingredient, equivalent to 0.3% (3.4 mg) of pilocarpine free-base. The inactive ingredients in QLOSI are: hypromellose, sodium chloride, sodium hyaluronate, edetate disodium dihydrate, water for injection, and may also include sodium hydroxide and/or hydrochloric acid for pH adjustment. QLOSI does not contain an anti-microbial preservative.
16.1 How Supplied
QLOSI is supplied as a sterile, clear, colorless to slightly yellowish and slightly viscous ophthalmic solution in configurations of 5 single-patient-use vials of 0.4 mL fill. Each single-patient-use vial is comprised of transparent low-density polyethylene (LDPE).
5 single-patient-use vials are packaged in a foil pouch.
QLOSI is supplied in:
NDC 83661-018-10: carton box of 10 single-patient-use vials (2 pouches containing 5 vials)
NDC 83661-018-20: carton box of 20 single-patient-use vials (4 pouches containing 5 vials)
NDC 83661-018-30: carton box of 30 single-patient-use vials (6 pouches containing 5 vials)
NDC 83661-018-60: carton box of 60 single-patient-use vials (12 pouches containing 5 vials)
8.4 Pediatric Use
Presbyopia is an age-related condition which does not appear in the pediatric population.
8.5 Geriatric Use
Clinical studies of QLOSI did not include subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience with ophthalmic pilocarpine solutions have not identified overall differences in safety between elderly and younger patients. Safety and effectiveness have not been established in patients over the age of 65.
5.1 Blurred Vision
Miotics, including QLOSI, may cause accommodative spasm. Advise patients to not drive or operate machinery if vision is not clear (e.g., blurred vision).
In addition, patients may experience temporary dim or dark vision with miotics, including QLOSI. Advise patients to exercise caution in night driving and other hazardous activities in poor illumination.
14 Clinical Studies
The efficacy of QLOSI for the treatment of presbyopia was demonstrated in two Phase 3, randomized, double-masked, vehicle-controlled studies, namely NEAR-1 (NCT04599933) and NEAR-2 (NCT04599972). A total of 613 participants aged 45 to 64 years old with presbyopia were randomized (309 to QLOSI group) in these two studies. Participants were instructed to instill one drop of QLOSI or vehicle, in each eye, once in the morning and to repeat the instillation 2 to 3 hours later. Participants were treated for two weeks. Ophthalmic assessments were conducted on Day 1, 8 and 15 of the study at various timepoints.
Responders demonstrated improvement by achieving a gain from baseline of 3 lines or more in near BDCVA at 40 centimeters without a loss of 1 line or more (≥ 5 letters) in BDCVA at 4 meters. Overall, the percentages of responders were consistently higher in the QLOSI group than in the Vehicle group at each assessment day.
| NEAR-1 | NEAR-2 | |||||
|---|---|---|---|---|---|---|
| Assessment timing | QLOSI N=155 | Vehicle N=154 | p-value | QLOSI N=154 | Vehicle N=150 | p-value |
| Abbreviations: N: number of participants in this study arm | ||||||
| 1 hour post Dose 1 | 39% | 17% | <0.01 | 42% | 21% | <0.01 |
| 2 hours post Dose 1 | 39% | 17% | <0.01 | 40% | 21% | <0.01 |
| 1 hour post Dose 2 | 48% | 16% | <0.01 | 52% | 17% | <0.01 |
| 2 hours post Dose 2 | 39% | 15% | <0.01 | 46% | 19% | <0.01 |
Figure 1 presents the percentage of responders for each time point at Day 15. (Standard analysis of pooled data NEAR-1 and NEAR-2)
Abbreviations: HRS: hours; min: minutes
This figure demonstrates the onset of the QLOSI effect on presbyopia, from 20 minutes post dose 1, and lasting up to 8 hours (i.e., 5 hours from the second dose).
4 Contraindications
QLOSI is contraindicated in patients with known hypersensitivity to the active ingredient or to any of the excipients.
6 Adverse Reactions
Most common adverse reactions (5% to 8%) are instillation site pain and headaches. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Orasis Pharmaceuticals Inc. at 1-866-ORASIS1 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
12.3 Pharmacokinetics
Systemic absorption of pilocarpine was evaluated in 12 healthy subjects following QLOSI administration (2 doses daily in each eye, 2 hours apart, for 8 days). The overall median T maxin plasma after Day 8 dosing was 2.20 hr and the mean (SD) overall plasma C maxand AUC 0-tafter Day 8 dosing were 897.2 (287.2) pg/mL and 2699 (741.4) hr*pg/mL, respectively. The mean T 1/2after Day 8 dosing was 3.96 hr.
5.4 Contact Lens Wear
Contact lens wearers should be advised to remove their lenses prior to the instillation of QLOSI and to wait 10 minutes after dosing before reinserting their contact lenses.
1 Indications and Usage
QLOSI is indicated for the treatment of presbyopia in adults.
12.1 Mechanism of Action
Pilocarpine hydrochloride is a cholinergic muscarinic agonist which activates muscarinic receptors located at smooth muscles such as the iris sphincter muscle and ciliary muscle. QLOSI contracts the iris sphincter muscle, constricting the pupil to enhance the depth of focus and improve near visual acuity while maintaining some pupillary response to light.
5 Warnings and Precautions
Blurred Vision: Advise patients to not drive or operate machinery if vision is not clear (e.g., blurred vision). Exercise caution in night driving and other hazardous occupations in poor illumination. ( 5.1)
Risk of Retinal Detachment: Rare cases of retinal detachment have been reported with miotics. Examination of the retina is advised in all patients prior to initiation of therapy. Advise patients to seek immediate medical care with sudden onset of flashes of lights, floaters, or vision loss. ( 5.2)
Iritis: Caution is advised in patients with iritis. ( 5.3)
2 Dosage and Administration
Instill one drop of QLOSI in each eye. This can be repeated a second time after 2 to 3 hours for an effect up to 8 hours. QLOSI can be administered on a daily basis, or as needed, up to twice each day.
If more than one topical ophthalmic medication is being used, the medicines must be administered at least 5 minutes apart.
Discard single-patient-use vial after use.
3 Dosage Forms and Strengths
QLOSI is a clear, colorless to slightly yellowish ophthalmic solution containing pilocarpine hydrochloride 0.4% (4 mg /mL) in a single-patient-use vial.
5.2 Risk of Retinal Detachment
Rare cases of retinal detachment have been reported with miotics when used in susceptible individuals and those with pre-existing retinal disease. Examination of the retina is advised in all patients prior to the initiation of therapy. Advise patients to seek immediate medical care with sudden onset of flashes of lights, floaters, or vision loss.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
QLOSI was evaluated in 309 patients with presbyopia in two randomized, double-masked, vehicle-controlled studies (NEAR-1 and NEAR-2) of 15 days duration. The most commonly reported treatment-related adverse events in 5-8% of patients were instillation site pain, and headache. Ocular adverse reaction reported in 2-5% of patients was blurred vision. The majority of the adverse events were mild, transient and self-resolving.
Principal Display Panel 0.4 Ml Vial Box
Qlosi
™
(pilocarpine HCl
ophthalmic
solution) 0.4%
Sterile / Rx only
For topical application in the eye
6 pouches containing 5 single-
patient-use vials (0.4 mL each)
NDC 83661-018-30
5.5 Potential for Eye Injury Or Contamination
To prevent eye injury or contamination, avoid touching the tip of the single-patient-use vial to the eye or to any other surface.
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
There are no adequate and well-controlled studies of QLOSI administration in pregnant women to inform a drug associated risk. Oral administration of pilocarpine to pregnant rats throughout organogenesis and lactation did not produce adverse effects at clinically relevant doses.
5.3 Iritis
QLOSI is not recommended to be used when iritis is present because adhesions (synechiae) may form between the iris and the lens.
16.2 Storage
Store refrigerated at 36°F to 46°F (2°C to 8°C). Once a pouch is open, the single-patient-use vials may be stored at room temperature [up to 77°F (25°C)] and used within 30 days.
Store unused product away from light. Discard opened vials after use.
10 Overdosage (10 OVERDOSAGE)
Systemic toxicity following topical ocular administration of pilocarpine is rare, but occasionally patients who are sensitive may develop sweating and gastrointestinal overactivity.
Accidental ingestion can produce sweating, salivation, nausea, tremors and slowing of the pulse and a decrease in blood pressure.
In moderate overdosage, spontaneous recovery is to be expected and is aided by intravenous fluids to compensate for dehydration. For patients demonstrating severe poisoning, atropine, the pharmacologic antagonist to pilocarpine, should be used.
11 Description (11 DESCRIPTION)
QLOSI (pilocarpine hydrochloride ophthalmic solution) 0.4% is a sterile, clear, colorless to slightly yellowish and slightly viscous ophthalmic solution for topical ophthalmic use which has a pH between 5.1-6.1.
The structural formula of pilocarpine hydrochloride is:
The chemical name for pilocarpine hydrochloride is: 2(3 H)-Furanone, 3-ethyldihydro-4-[(1-methyl-1 H-imidazol-5-yl)- methyl]-monohydrochloride, (3 S-cis). Its molecular weight is 244.72 g/mol and its molecular formula is C 11H 16N 2O 2∙ HCl.
Each mL contains 0.4% (4 mg) of pilocarpine hydrochloride as the active ingredient, equivalent to 0.3% (3.4 mg) of pilocarpine free-base. The inactive ingredients in QLOSI are: hypromellose, sodium chloride, sodium hyaluronate, edetate disodium dihydrate, water for injection, and may also include sodium hydroxide and/or hydrochloric acid for pH adjustment. QLOSI does not contain an anti-microbial preservative.
16.1 How Supplied
QLOSI is supplied as a sterile, clear, colorless to slightly yellowish and slightly viscous ophthalmic solution in configurations of 5 single-patient-use vials of 0.4 mL fill. Each single-patient-use vial is comprised of transparent low-density polyethylene (LDPE).
5 single-patient-use vials are packaged in a foil pouch.
QLOSI is supplied in:
NDC 83661-018-10: carton box of 10 single-patient-use vials (2 pouches containing 5 vials)
NDC 83661-018-20: carton box of 20 single-patient-use vials (4 pouches containing 5 vials)
NDC 83661-018-30: carton box of 30 single-patient-use vials (6 pouches containing 5 vials)
NDC 83661-018-60: carton box of 60 single-patient-use vials (12 pouches containing 5 vials)
8.4 Pediatric Use
Presbyopia is an age-related condition which does not appear in the pediatric population.
8.5 Geriatric Use
Clinical studies of QLOSI did not include subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience with ophthalmic pilocarpine solutions have not identified overall differences in safety between elderly and younger patients. Safety and effectiveness have not been established in patients over the age of 65.
5.1 Blurred Vision
Miotics, including QLOSI, may cause accommodative spasm. Advise patients to not drive or operate machinery if vision is not clear (e.g., blurred vision).
In addition, patients may experience temporary dim or dark vision with miotics, including QLOSI. Advise patients to exercise caution in night driving and other hazardous activities in poor illumination.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of QLOSI for the treatment of presbyopia was demonstrated in two Phase 3, randomized, double-masked, vehicle-controlled studies, namely NEAR-1 (NCT04599933) and NEAR-2 (NCT04599972). A total of 613 participants aged 45 to 64 years old with presbyopia were randomized (309 to QLOSI group) in these two studies. Participants were instructed to instill one drop of QLOSI or vehicle, in each eye, once in the morning and to repeat the instillation 2 to 3 hours later. Participants were treated for two weeks. Ophthalmic assessments were conducted on Day 1, 8 and 15 of the study at various timepoints.
Responders demonstrated improvement by achieving a gain from baseline of 3 lines or more in near BDCVA at 40 centimeters without a loss of 1 line or more (≥ 5 letters) in BDCVA at 4 meters. Overall, the percentages of responders were consistently higher in the QLOSI group than in the Vehicle group at each assessment day.
| NEAR-1 | NEAR-2 | |||||
|---|---|---|---|---|---|---|
| Assessment timing | QLOSI N=155 | Vehicle N=154 | p-value | QLOSI N=154 | Vehicle N=150 | p-value |
| Abbreviations: N: number of participants in this study arm | ||||||
| 1 hour post Dose 1 | 39% | 17% | <0.01 | 42% | 21% | <0.01 |
| 2 hours post Dose 1 | 39% | 17% | <0.01 | 40% | 21% | <0.01 |
| 1 hour post Dose 2 | 48% | 16% | <0.01 | 52% | 17% | <0.01 |
| 2 hours post Dose 2 | 39% | 15% | <0.01 | 46% | 19% | <0.01 |
Figure 1 presents the percentage of responders for each time point at Day 15. (Standard analysis of pooled data NEAR-1 and NEAR-2)
Abbreviations: HRS: hours; min: minutes
This figure demonstrates the onset of the QLOSI effect on presbyopia, from 20 minutes post dose 1, and lasting up to 8 hours (i.e., 5 hours from the second dose).
4 Contraindications (4 CONTRAINDICATIONS)
QLOSI is contraindicated in patients with known hypersensitivity to the active ingredient or to any of the excipients.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (5% to 8%) are instillation site pain and headaches. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Orasis Pharmaceuticals Inc. at 1-866-ORASIS1 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
12.3 Pharmacokinetics
Systemic absorption of pilocarpine was evaluated in 12 healthy subjects following QLOSI administration (2 doses daily in each eye, 2 hours apart, for 8 days). The overall median T maxin plasma after Day 8 dosing was 2.20 hr and the mean (SD) overall plasma C maxand AUC 0-tafter Day 8 dosing were 897.2 (287.2) pg/mL and 2699 (741.4) hr*pg/mL, respectively. The mean T 1/2after Day 8 dosing was 3.96 hr.
5.4 Contact Lens Wear
Contact lens wearers should be advised to remove their lenses prior to the instillation of QLOSI and to wait 10 minutes after dosing before reinserting their contact lenses.
1 Indications and Usage (1 INDICATIONS AND USAGE)
QLOSI is indicated for the treatment of presbyopia in adults.
12.1 Mechanism of Action
Pilocarpine hydrochloride is a cholinergic muscarinic agonist which activates muscarinic receptors located at smooth muscles such as the iris sphincter muscle and ciliary muscle. QLOSI contracts the iris sphincter muscle, constricting the pupil to enhance the depth of focus and improve near visual acuity while maintaining some pupillary response to light.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Blurred Vision: Advise patients to not drive or operate machinery if vision is not clear (e.g., blurred vision). Exercise caution in night driving and other hazardous occupations in poor illumination. ( 5.1)
Risk of Retinal Detachment: Rare cases of retinal detachment have been reported with miotics. Examination of the retina is advised in all patients prior to initiation of therapy. Advise patients to seek immediate medical care with sudden onset of flashes of lights, floaters, or vision loss. ( 5.2)
Iritis: Caution is advised in patients with iritis. ( 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Instill one drop of QLOSI in each eye. This can be repeated a second time after 2 to 3 hours for an effect up to 8 hours. QLOSI can be administered on a daily basis, or as needed, up to twice each day.
If more than one topical ophthalmic medication is being used, the medicines must be administered at least 5 minutes apart.
Discard single-patient-use vial after use.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
QLOSI is a clear, colorless to slightly yellowish ophthalmic solution containing pilocarpine hydrochloride 0.4% (4 mg /mL) in a single-patient-use vial.
5.2 Risk of Retinal Detachment
Rare cases of retinal detachment have been reported with miotics when used in susceptible individuals and those with pre-existing retinal disease. Examination of the retina is advised in all patients prior to the initiation of therapy. Advise patients to seek immediate medical care with sudden onset of flashes of lights, floaters, or vision loss.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
QLOSI was evaluated in 309 patients with presbyopia in two randomized, double-masked, vehicle-controlled studies (NEAR-1 and NEAR-2) of 15 days duration. The most commonly reported treatment-related adverse events in 5-8% of patients were instillation site pain, and headache. Ocular adverse reaction reported in 2-5% of patients was blurred vision. The majority of the adverse events were mild, transient and self-resolving.
Principal Display Panel 0.4 Ml Vial Box (PRINCIPAL DISPLAY PANEL - 0.4 mL Vial Box)
Qlosi
™
(pilocarpine HCl
ophthalmic
solution) 0.4%
Sterile / Rx only
For topical application in the eye
6 pouches containing 5 single-
patient-use vials (0.4 mL each)
NDC 83661-018-30
5.5 Potential for Eye Injury Or Contamination (5.5 Potential for Eye Injury or Contamination)
To prevent eye injury or contamination, avoid touching the tip of the single-patient-use vial to the eye or to any other surface.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:21.698965 · Updated: 2026-03-14T22:27:03.737378