Jafra Daily Antiperspirant Deodorant Roll-on
4e7a8a3b-12de-4108-a11c-4838510934a5
34390-5
HUMAN OTC DRUG LABEL
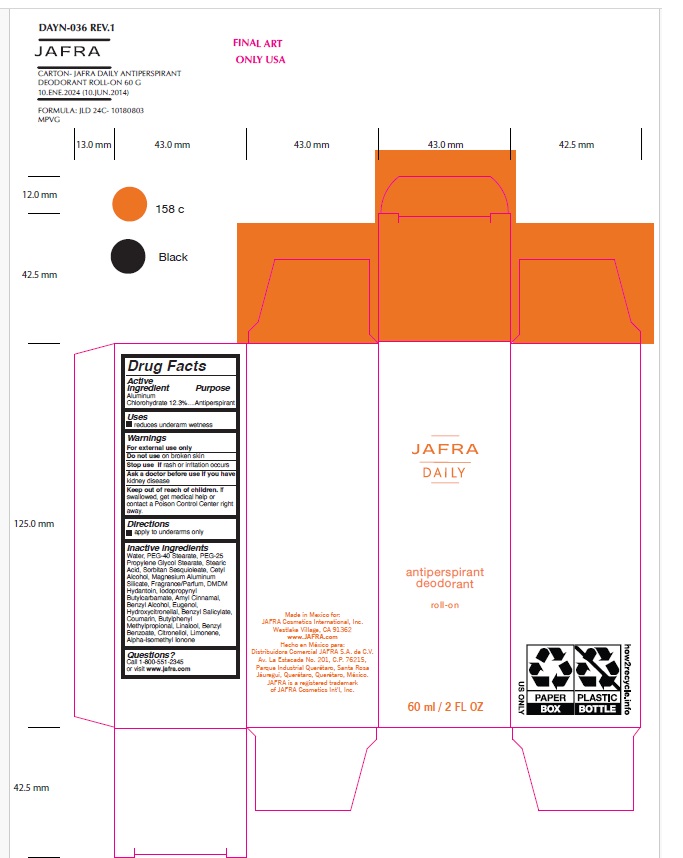
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Aluminum Chlorohydrate 12.3%
Purpose
Antiperspirant
Medication Information
Purpose
Antiperspirant
Description
Active ingredient Aluminum Chlorohydrate 12.3%
Uses
Reduces underarm wetness
Section 42229-5
Questions?
Call 1-800-551-2345 or visit
www.jafra.com
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6
Inactive Ingredients
Water, PEG-40 Stearate, PEG-25 Propylene Glycol Stearate, Stearic Acid, Sorbitan Sesquioleate, Cetyl Alcohol, Magnesium Aluminum Silicate, Fragrance/Parfum, DMDM Hydantoin, Iodopropynyl Butylcarbamate, Amyl Cinnamal, Benzyl Alcohol, Eugenol, Hydroxycitronellal, Benzyl Salicylate, Coumarin, Butylphenyl Methylpropional, Linalool, Benzyl Benzoate, Citronellol, Limonene, Alpha-Isomethyl Ionone
Section 55106-9
Active ingredient
Aluminum Chlorohydrate 12.3%
Warnings
For external use only
Do not use on broken skin
Stop use if rash or irritation occurs
Ask a doctor before use if you have kidney disease
Directions
apply to underarms only
Product Label
Structured Label Content
Uses
Reduces underarm wetness
Section 42229-5 (42229-5)
Questions?
Call 1-800-551-2345 or visit
www.jafra.com
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
Inactive Ingredients
Water, PEG-40 Stearate, PEG-25 Propylene Glycol Stearate, Stearic Acid, Sorbitan Sesquioleate, Cetyl Alcohol, Magnesium Aluminum Silicate, Fragrance/Parfum, DMDM Hydantoin, Iodopropynyl Butylcarbamate, Amyl Cinnamal, Benzyl Alcohol, Eugenol, Hydroxycitronellal, Benzyl Salicylate, Coumarin, Butylphenyl Methylpropional, Linalool, Benzyl Benzoate, Citronellol, Limonene, Alpha-Isomethyl Ionone
Section 55106-9 (55106-9)
Active ingredient
Aluminum Chlorohydrate 12.3%
Purpose
Antiperspirant
Warnings
For external use only
Do not use on broken skin
Stop use if rash or irritation occurs
Ask a doctor before use if you have kidney disease
Directions
apply to underarms only
Product Label (Product label)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:11.975486 · Updated: 2026-03-14T23:06:38.217628