Topcare®
4dfe410b-9248-4549-8bc4-3d3c35e2569d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Description
Drug Facts
Use
- relieves occasional constipation
- this product generally produces bowel movement in 1 to 5 minutes
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away: 800-222-1222.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
| Active ingredients (in each 118mL delivered dose) | Purpose |
|---|---|
| Dibasic Sodium Phosphate 7 g | Saline laxative |
| Monobasic Sodium Phosphate 19 g | Saline laxative |
Warnings
For rectal use only.
Dosage Warning
Using more than one enema in 24 hours can be harmful.
Other Information
- each 118 mL contains 4.3 g sodium
- store at 15 to 30°C (59 to 86°F)
- additional liquids by mouth are recommended while using this product
- this product generally produces a bowel movement in 1 to 5 minutes
- carton sealed for safety. Do not use if top or bottom flap of carton is torn or missing
Inactive Ingredients
benzalkonium chloride, disodium EDTA, purified water
Questions Or Comments?
1-888-423-0139
When Using This Product
- do not use more than directed. Serious side effects may occur from excess dosage
- do not use for more than 3 days
Stop Use and Ask A Doctor If
- you have bleeding
- condition worsens or does not improve within 7 days
- you have no bowel movement within 30 minutes of enema use
- you have symptoms of dehydration (thirstiness, dizziness, vomiting, urinating less often than normal)
These symptoms may indicate a serious condition.
Ask A Doctor Before Use If You
- already used a laxative for more than 1 week
- have kidney disease
- have heart problems
- are dehydrated
- are 55 years old or older
- are on a sodium-restricted diet
Single Daily Dosage (per 24 Hours)
Do not use if taking another sodium phosphate product.
Do not use more unless directed by a doctor. See Warnings.
| Adults and children 12 years old and older | 1 bottle once daily |
| Children 2 to under 12 years old | Use Pediatric Enema |
| Children under 2 years | DO NOT USE |
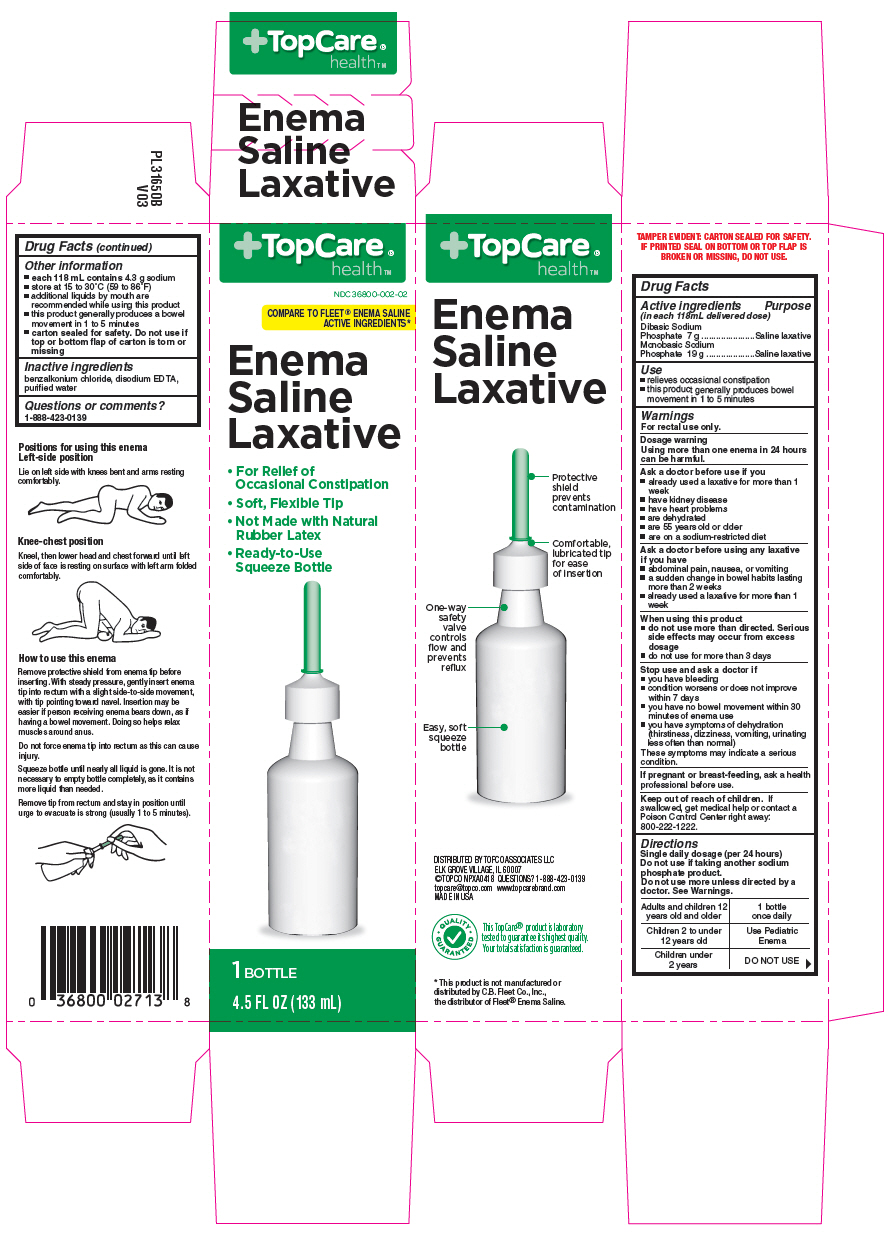
Principal Display Panel 133 Ml Bottle Carton
TopCare®
health™
NDC 36800-002-02
COMPARE TO FLEET® ENEMA SALINE
ACTIVE INGREDIENTS*
Enema
Saline
Laxative
- For Relief of
Occasional Constipation - Soft, Flexible Tip
- Not Made with Natural
Rubber Latex - Ready-to-Use
Squeeze Bottle
1 BOTTLE
4.5 FL OZ (133 mL)
Ask A Doctor Before Using Any Laxative If You Have
- abdominal pain, nausea, or vomiting
- a sudden change in bowel habits lasting more than 2 weeks
- already used a laxative for more than 1 week
Structured Label Content
Use
- relieves occasional constipation
- this product generally produces bowel movement in 1 to 5 minutes
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away: 800-222-1222.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
| Active ingredients (in each 118mL delivered dose) | Purpose |
|---|---|
| Dibasic Sodium Phosphate 7 g | Saline laxative |
| Monobasic Sodium Phosphate 19 g | Saline laxative |
Warnings
For rectal use only.
Dosage Warning (Dosage warning)
Using more than one enema in 24 hours can be harmful.
Other Information (Other information)
- each 118 mL contains 4.3 g sodium
- store at 15 to 30°C (59 to 86°F)
- additional liquids by mouth are recommended while using this product
- this product generally produces a bowel movement in 1 to 5 minutes
- carton sealed for safety. Do not use if top or bottom flap of carton is torn or missing
Inactive Ingredients (Inactive ingredients)
benzalkonium chloride, disodium EDTA, purified water
Questions Or Comments? (Questions or comments?)
1-888-423-0139
When Using This Product (When using this product)
- do not use more than directed. Serious side effects may occur from excess dosage
- do not use for more than 3 days
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- you have bleeding
- condition worsens or does not improve within 7 days
- you have no bowel movement within 30 minutes of enema use
- you have symptoms of dehydration (thirstiness, dizziness, vomiting, urinating less often than normal)
These symptoms may indicate a serious condition.
Ask A Doctor Before Use If You (Ask a doctor before use if you)
- already used a laxative for more than 1 week
- have kidney disease
- have heart problems
- are dehydrated
- are 55 years old or older
- are on a sodium-restricted diet
Single Daily Dosage (per 24 Hours) (Single daily dosage (per 24 hours))
Do not use if taking another sodium phosphate product.
Do not use more unless directed by a doctor. See Warnings.
| Adults and children 12 years old and older | 1 bottle once daily |
| Children 2 to under 12 years old | Use Pediatric Enema |
| Children under 2 years | DO NOT USE |
Principal Display Panel 133 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 133 mL Bottle Carton)
TopCare®
health™
NDC 36800-002-02
COMPARE TO FLEET® ENEMA SALINE
ACTIVE INGREDIENTS*
Enema
Saline
Laxative
- For Relief of
Occasional Constipation - Soft, Flexible Tip
- Not Made with Natural
Rubber Latex - Ready-to-Use
Squeeze Bottle
1 BOTTLE
4.5 FL OZ (133 mL)
Ask A Doctor Before Using Any Laxative If You Have (Ask a doctor before using any laxative if you have)
- abdominal pain, nausea, or vomiting
- a sudden change in bowel habits lasting more than 2 weeks
- already used a laxative for more than 1 week
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:44.190557 · Updated: 2026-03-14T22:55:24.290889