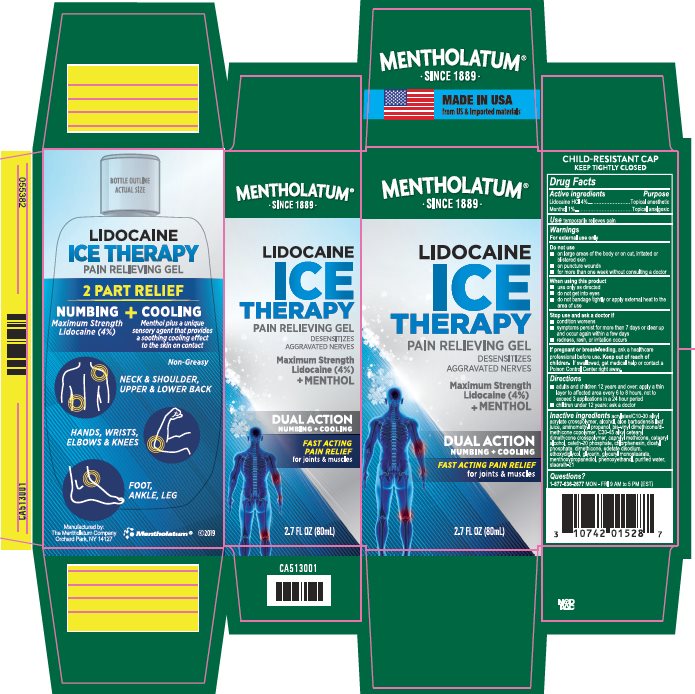
Drug Facts - Mentholatum Lidocaine Ice
4d74cbab-31cd-4579-9360-6852ee215b5e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine HCl 4% Menthol 1%
Purpose
Lidocaine HCl - Topical anesthetic Menthol – Topical analgesic
Medication Information
Purpose
Lidocaine HCl - Topical anesthetic
Menthol – Topical analgesic
Description
Lidocaine HCl 4% Menthol 1%
Uses
temporarily relieves pain
Warnings
For external use only
Directions
- adults and children 12 years and over: apply a thin layer to affected area every 6 to 8 hours, not to exceed 3 applications in a 24 hour period
- children under 12 years: ask a doctor
Do Not Use
- on large areas of the body or on cut, irritated or blistered skin
- on puncture wounds
- for more than one week without consulting a doctor
Active Ingredients
Lidocaine HCl 4%
Menthol 1%
Inactive Ingredients
acrylates/C10-30 alkyl acrylate crosspolymer, alcohol, aloe barbadensis leaf juice, aminomethyl propanol, bis-vinyl dimethicone/dimecthicone copolymer, C30-45 alkyl cetearyl dimethicone crosspolymer, caprylyl methicone, cetearyl alcohol, ceteth-20 phosphate, chlorphenesin, dicetyl phosphate, dimethicone, edetate disodium, ethoxydiglycol, glycerin, glyceryl monostearate, methoxypropanediol, phenoxyethanol, purified water, steareth-21
When Using This Product
- use only as directed
- do not get into eyes
- do not bandage tightly or apply external heat to the area of use
Stop Use and Ask A Doctor If
- condition worsens
- symptoms persist for more than 7 days or clear up and occur again within a few days
- redness, rash, or irritation occurs
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel
Structured Label Content
Uses
temporarily relieves pain
Purpose
Lidocaine HCl - Topical anesthetic
Menthol – Topical analgesic
Warnings
For external use only
Directions
- adults and children 12 years and over: apply a thin layer to affected area every 6 to 8 hours, not to exceed 3 applications in a 24 hour period
- children under 12 years: ask a doctor
Do Not Use (Do not use)
- on large areas of the body or on cut, irritated or blistered skin
- on puncture wounds
- for more than one week without consulting a doctor
Active Ingredients (Active ingredients)
Lidocaine HCl 4%
Menthol 1%
Inactive Ingredients (Inactive ingredients)
acrylates/C10-30 alkyl acrylate crosspolymer, alcohol, aloe barbadensis leaf juice, aminomethyl propanol, bis-vinyl dimethicone/dimecthicone copolymer, C30-45 alkyl cetearyl dimethicone crosspolymer, caprylyl methicone, cetearyl alcohol, ceteth-20 phosphate, chlorphenesin, dicetyl phosphate, dimethicone, edetate disodium, ethoxydiglycol, glycerin, glyceryl monostearate, methoxypropanediol, phenoxyethanol, purified water, steareth-21
When Using This Product (When using this product)
- use only as directed
- do not get into eyes
- do not bandage tightly or apply external heat to the area of use
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens
- symptoms persist for more than 7 days or clear up and occur again within a few days
- redness, rash, or irritation occurs
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel (Package/Label Principal Display Panel)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:48.370835 · Updated: 2026-03-14T23:05:48.377821