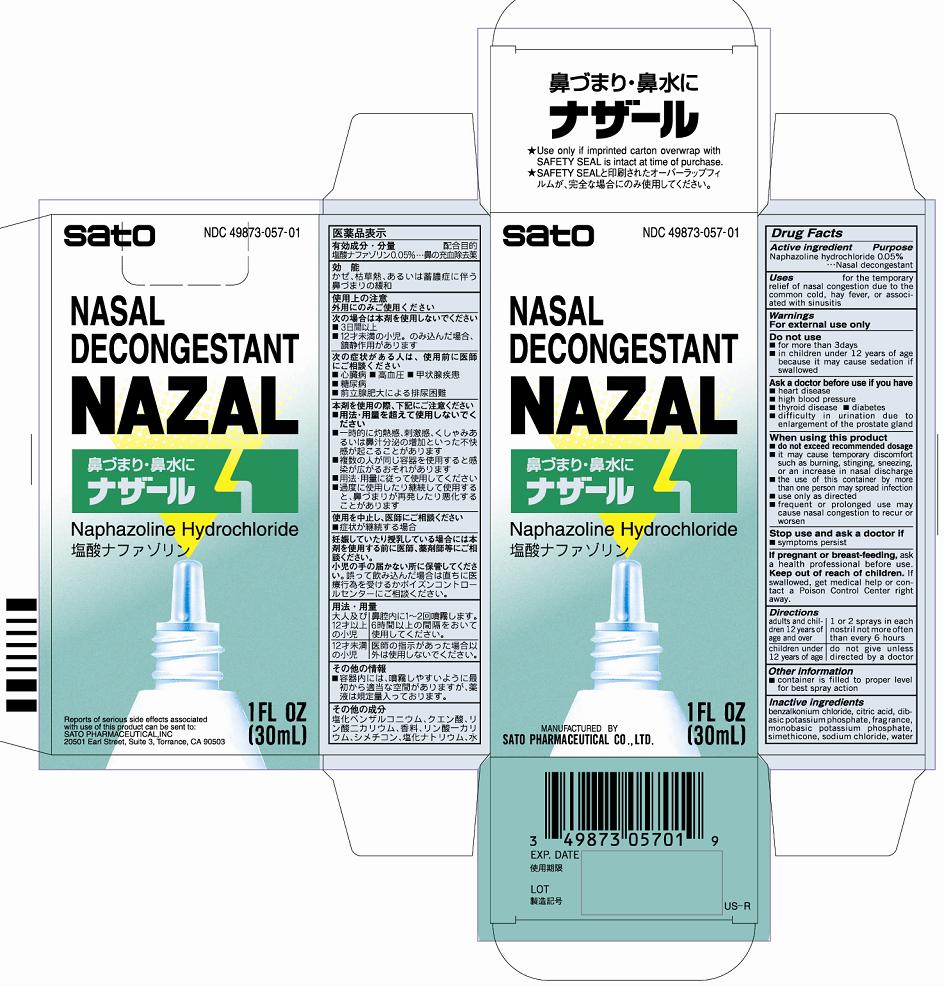
Nazal
4d4043f8-5ed3-4027-beb6-687db1308202
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients Naphazoline hydrochloride 0.05%
Medication Information
Warnings and Precautions
Warnings
For external use only
Indications and Usage
Uses for the temporary relief of nasal congestion due to the common cold, hay fever, or associated with sinusitis
Dosage and Administration
Directions
Adults and children 12 years of age and older - 1 or 2 sprays in each nostril not more often than every 6 hours
Children under 12 years of age - do not give unless directed by a doctor
Description
Active ingredients Naphazoline hydrochloride 0.05%
Section 50565-1
Keep our of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
■symptoms persist
Section 50567-7
When using this product
■do not exceed recommended dosage
■if may cause temporary discomfort such as burning, stinging, sneezing, or an increase in nasal discharge
■the use of this container by more than one person may spread infection
■use only as directed
■frequent or prolonged use may cause nasal congestion to recur or worsen
Section 50569-3
Ask a doctor before use if you have
■heart disease
■high blood pressure
■thyroid disease ■diabetes
■difficulty in urination due to enlargement of the prostate gland
Section 50570-1
Do not use
■for more than 3 days
■in children under 12 years of age because it may cause sedation if swallowed
Section 51727-6
Inactive ingredients
benzalkonium chloride, citric acid, dibasic potassium phosphate, fragrance, monobasic potassium phosphate, simethicone, sodium chloride, water
Section 51945-4
nazalcart.jpg
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purpose Nasal decongestant
Section 55106-9
Active ingredients
Naphazoline hydrochloride 0.05%
Section 60561-8
Other information
■container is filled to proper level for best spray action
Structured Label Content
Indications and Usage (34067-9)
Uses for the temporary relief of nasal congestion due to the common cold, hay fever, or associated with sinusitis
Dosage and Administration (34068-7)
Directions
Adults and children 12 years of age and older - 1 or 2 sprays in each nostril not more often than every 6 hours
Children under 12 years of age - do not give unless directed by a doctor
Warnings and Precautions (34071-1)
Warnings
For external use only
Section 50565-1 (50565-1)
Keep our of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
■symptoms persist
Section 50567-7 (50567-7)
When using this product
■do not exceed recommended dosage
■if may cause temporary discomfort such as burning, stinging, sneezing, or an increase in nasal discharge
■the use of this container by more than one person may spread infection
■use only as directed
■frequent or prolonged use may cause nasal congestion to recur or worsen
Section 50569-3 (50569-3)
Ask a doctor before use if you have
■heart disease
■high blood pressure
■thyroid disease ■diabetes
■difficulty in urination due to enlargement of the prostate gland
Section 50570-1 (50570-1)
Do not use
■for more than 3 days
■in children under 12 years of age because it may cause sedation if swallowed
Section 51727-6 (51727-6)
Inactive ingredients
benzalkonium chloride, citric acid, dibasic potassium phosphate, fragrance, monobasic potassium phosphate, simethicone, sodium chloride, water
Section 51945-4 (51945-4)
nazalcart.jpg
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purpose Nasal decongestant
Section 55106-9 (55106-9)
Active ingredients
Naphazoline hydrochloride 0.05%
Section 60561-8 (60561-8)
Other information
■container is filled to proper level for best spray action
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:26.945025 · Updated: 2026-03-14T23:02:00.533418