Cetirizine Hydrochloride Tablets Usp, 10 Mg, Allergy
4b192084-dfdc-47bc-a968-6f49e137b248
34390-5
HUMAN OTC DRUG LABEL
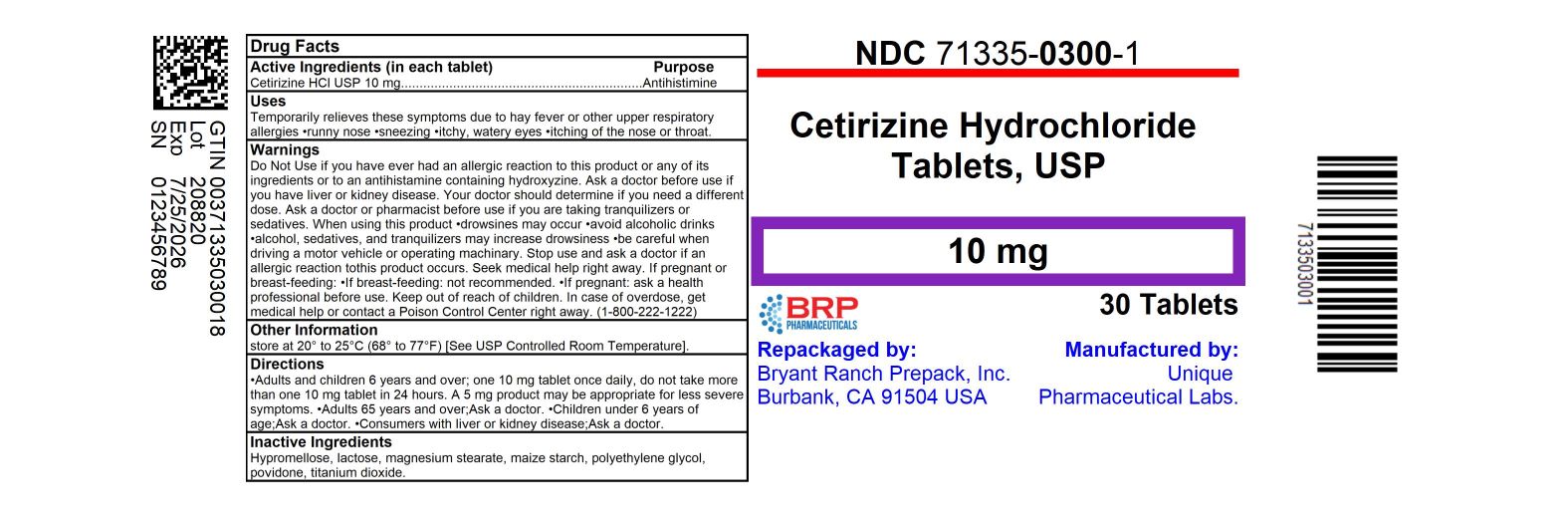
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredient (in each tablet) Cetirizine HCl USP 10 mg
Purpose
Antihistamine
How Supplied
NDC: 71335-0300-1: 30 Tablets in a BOTTLE NDC: 71335-0300-2: 14 Tablets in a BOTTLE NDC: 71335-0300-3: 7 Tablets in a BOTTLE NDC: 71335-0300-4: 10 Tablets in a BOTTLE NDC: 71335-0300-5: 15 Tablets in a BOTTLE NDC: 71335-0300-6: 90 Tablets in a BOTTLE NDC: 71335-0300-7: 20 Tablets in a BOTTLE NDC: 71335-0300-8: 60 Tablets in a BOTTLE NDC: 71335-0300-9: 100 Tablets in a BOTTLE Repackaged/Relabeled by: Bryant Ranch Prepack, Inc. Burbank, CA 91504
Medication Information
Purpose
Antihistamine
How Supplied
NDC: 71335-0300-1: 30 Tablets in a BOTTLE
NDC: 71335-0300-2: 14 Tablets in a BOTTLE
NDC: 71335-0300-3: 7 Tablets in a BOTTLE
NDC: 71335-0300-4: 10 Tablets in a BOTTLE
NDC: 71335-0300-5: 15 Tablets in a BOTTLE
NDC: 71335-0300-6: 90 Tablets in a BOTTLE
NDC: 71335-0300-7: 20 Tablets in a BOTTLE
NDC: 71335-0300-8: 60 Tablets in a BOTTLE
NDC: 71335-0300-9: 100 Tablets in a BOTTLE
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
Description
Active Ingredient (in each tablet) Cetirizine HCl USP 10 mg
Uses:
Temporarily relieves these symptoms due to hay fever or other upper respiratory allergies
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
Section 42229-5
Manufactured by:
Unique Pharmaceutical Labs.
(A Div. of J. B. Chemicals & Pharmaceuticals Ltd.),
Mumbai 400 030, India
Distributed by:
Rising Pharma Holdings, Inc.
East Brunswick, NJ 08816
M. L. G/1430 Jul. 2020
126406
Section 50566-9
Stop use and ask a doctor if an allergic reaction tothis product occurs. Seek medical help right away.
Section 50568-5
Ask a doctor or pharmacist before use if you are taking tranquilizers or sedatives.
Section 50569-3
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Section 51945-4
Cetirizine Hydrochloride 10 mg Tablet
Section 55106-9
Active Ingredient (in each tablet)
Cetirizine HCl USP 10 mg
Warnings:
Do Not Use if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Directions
| Adults and children 6 years and over |
one 10 mg tablet once daily, do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. |
| Adults 65 years and over | Ask a doctor |
| Children under 6 years of age | Ask a doctor |
| Consumers with liver or kidney disease | Ask a doctor |
Questions?
Call 1-844-874-7464
Other Information:
store at 20° to 25°C (68° to 77°F)
[See USP Controlled Room Temperature].
Inactive Ingredients
Hypromellose, lactose, magnesium stearate, maize starch, polyethylene glycol, povidone, titanium dioxide.
When Using This Product
- drowsines may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinary.
If Pregnant Or Breast Feeding:
- if breast-feeding: not recommended
- if pregnant: ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Structured Label Content
Uses:
Temporarily relieves these symptoms due to hay fever or other upper respiratory allergies
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
Section 42229-5 (42229-5)
Manufactured by:
Unique Pharmaceutical Labs.
(A Div. of J. B. Chemicals & Pharmaceuticals Ltd.),
Mumbai 400 030, India
Distributed by:
Rising Pharma Holdings, Inc.
East Brunswick, NJ 08816
M. L. G/1430 Jul. 2020
126406
Section 50566-9 (50566-9)
Stop use and ask a doctor if an allergic reaction tothis product occurs. Seek medical help right away.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you are taking tranquilizers or sedatives.
Section 50569-3 (50569-3)
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Section 51945-4 (51945-4)
Cetirizine Hydrochloride 10 mg Tablet
Section 55106-9 (55106-9)
Active Ingredient (in each tablet)
Cetirizine HCl USP 10 mg
Purpose
Antihistamine
Warnings:
Do Not Use if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Directions
| Adults and children 6 years and over |
one 10 mg tablet once daily, do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. |
| Adults 65 years and over | Ask a doctor |
| Children under 6 years of age | Ask a doctor |
| Consumers with liver or kidney disease | Ask a doctor |
Questions?
Call 1-844-874-7464
How Supplied (HOW SUPPLIED)
NDC: 71335-0300-1: 30 Tablets in a BOTTLE
NDC: 71335-0300-2: 14 Tablets in a BOTTLE
NDC: 71335-0300-3: 7 Tablets in a BOTTLE
NDC: 71335-0300-4: 10 Tablets in a BOTTLE
NDC: 71335-0300-5: 15 Tablets in a BOTTLE
NDC: 71335-0300-6: 90 Tablets in a BOTTLE
NDC: 71335-0300-7: 20 Tablets in a BOTTLE
NDC: 71335-0300-8: 60 Tablets in a BOTTLE
NDC: 71335-0300-9: 100 Tablets in a BOTTLE
Repackaged/Relabeled by:
Bryant Ranch Prepack, Inc.
Burbank, CA 91504
Other Information:
store at 20° to 25°C (68° to 77°F)
[See USP Controlled Room Temperature].
Inactive Ingredients (Inactive ingredients)
Hypromellose, lactose, magnesium stearate, maize starch, polyethylene glycol, povidone, titanium dioxide.
When Using This Product (When using this product)
- drowsines may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinary.
If Pregnant Or Breast Feeding: (If pregnant or breast-feeding:)
- if breast-feeding: not recommended
- if pregnant: ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:20.505377 · Updated: 2026-03-14T23:03:21.577672