Aerowound Blood Clotting Spray
4a38237f-9353-4c12-8bee-cc53f57d750b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
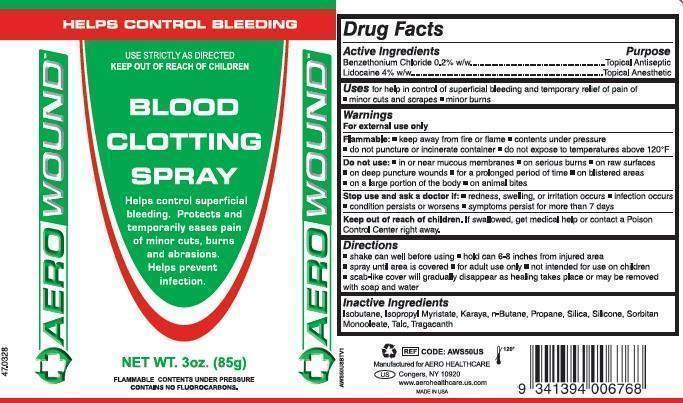
Benzethonium Chloride 0.2% w/w Lidocaine 4% w/w
Purpose
Topical Antiseptic Topical Anesthetic
Medication Information
Purpose
Topical Antiseptic
Topical Anesthetic
Description
Benzethonium Chloride 0.2% w/w Lidocaine 4% w/w
Uses
for help in control of superficial bleeding and temporary relief of pain of:
- minor cuts and scrapes
- minor burns
Warnings
For external use only
Flammable
- keep away from flame
- contents under pressure
- do not puncture or incinerate
- do not expose to temperatures above 120°F
directions
- shake can well before using
- hold 6-8 inches from moist injured area
- spray until area is covered
- for adult use only
- not intended for use on children
- scab-like cover will gradually disappear as healing takes place or may be removed with soap and water
do Not Use
- in or near mucous membranes
- on serious burns
- on raw surfaces
- on deep puncture wounds
- for a prolonged period of time
- on blistered areas
- on a large portion of the body
- on animal bites
Principal Display
Active Ingredients
Benzethonium Chloride 0.2% w/w
Lidocaine 4% w/w
Inactive Ingredients
Isobutane, Isopropyl Myristate, Karaya, n-Butane, Propane, Silica, Silicone, Sorbitan Monooleate, Talc, Tragacanth
Stop Use and Ask Doctor If
- redness, swelling, or irritation occurs
- infection occurs
- condition persists or worsens
- symptoms persist for more than 7 days
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
for help in control of superficial bleeding and temporary relief of pain of:
- minor cuts and scrapes
- minor burns
Purpose
Topical Antiseptic
Topical Anesthetic
Warnings
For external use only
Flammable
- keep away from flame
- contents under pressure
- do not puncture or incinerate
- do not expose to temperatures above 120°F
directions (Directions)
- shake can well before using
- hold 6-8 inches from moist injured area
- spray until area is covered
- for adult use only
- not intended for use on children
- scab-like cover will gradually disappear as healing takes place or may be removed with soap and water
do Not Use (Do not use)
- in or near mucous membranes
- on serious burns
- on raw surfaces
- on deep puncture wounds
- for a prolonged period of time
- on blistered areas
- on a large portion of the body
- on animal bites
Principal Display
Active Ingredients
Benzethonium Chloride 0.2% w/w
Lidocaine 4% w/w
Inactive Ingredients
Isobutane, Isopropyl Myristate, Karaya, n-Butane, Propane, Silica, Silicone, Sorbitan Monooleate, Talc, Tragacanth
Stop Use and Ask Doctor If (Stop use and ask doctor if)
- redness, swelling, or irritation occurs
- infection occurs
- condition persists or worsens
- symptoms persist for more than 7 days
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:49.456688 · Updated: 2026-03-14T22:53:47.887144