Bomyntra
49d25732-e05d-47e4-98cb-67df04796d4b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Bomyntra is a RANK ligand (RANKL) inhibitor indicated for: Prevention of skeletal-related events in patients with multiple myeloma and in patients with bone metastases from solid tumors. ( 1.1 ) Treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity. ( 1.2 , 14.3 ) Treatment of hypercalcemia of malignancy refractory to bisphosphonate therapy. ( 1.3 )
Dosage and Administration
Bomyntra should be administered by a healthcare provider. ( 2.1 ) Bomyntra is intended for subcutaneous route only and should not be administered intravenously, intramuscularly, or intradermally. ( 2.1 ) Multiple Myeloma and Bone Metastasis from Solid Tumors: Administer 120 mg every 4 weeks as a subcutaneous injection in the upper arm, upper thigh, or abdomen. ( 2.2 ) Giant Cell Tumor of Bone: Administer 120 mg every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen. ( 2.3 ) Administer calcium and vitamin D as necessary to treat or prevent hypocalcemia. ( 2.2 , 2.3 ) Hypercalcemia of Malignancy: Administer 120 mg every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen. ( 2.4 )
Contraindications
Hypocalcemia ( 4.1 ) Known clinically significant hypersensitivity to denosumab products ( 4.2 )
Warnings and Precautions
Drug Products with Same Active Ingredient: Patients receiving Bomyntra should not receive other denosumab products concomitantly ( 5.1 ) Hypersensitivity reactions including anaphylaxis may occur. Discontinue permanently if a clinically significant reaction occurs. ( 5.2 ) Hypocalcemia: Denosumab products can cause severe symptomatic hypocalcemia, and fatal cases have been reported. Correct hypocalcemia prior to initiating Bomyntra. Monitor calcium levels during therapy, especially in the first weeks of initiating therapy, and adequately supplement all patients with calcium and vitamin D. ( 5.3 ) Osteonecrosis of the jaw (ONJ) has been reported in patients receiving denosumab products. Perform an oral examination prior to starting Bomyntra. Monitor for symptoms. Avoid invasive dental procedures during treatment with Bomyntra. ( 5.4 ) Atypical femoral fracture: Evaluate patients with thigh or groin pain to rule out a femoral fracture. ( 5.5 ) Hypercalcemia Following Treatment Discontinuation in Patients with Giant Cell Tumor of Bone and in Patients with Growing Skeletons: Monitor patients for signs and symptoms of hypercalcemia, and manage as clinically appropriate. ( 5.6 , 8.4 ) Multiple Vertebral Fractures (MVF) Following Treatment Discontinuation: When Bomyntra treatment is discontinued, evaluate the individual patient's risk for vertebral fractures. ( 5.7 ) Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of potential risk to the fetus and to use effective contraception. ( 5.8 , 8.1 , 8.3 )
Adverse Reactions
The following adverse reactions are discussed below and elsewhere in the labeling: Hypersensitivity [see Warnings and Precautions ( 5.2 )] Hypocalcemia [see Warnings and Precautions ( 5.3 ) and Use in Specific Populations ( 8.6 )] Osteonecrosis of the Jaw [see Warnings and Precautions ( 5.4 )] Atypical Subtrochanteric and Diaphyseal Femoral Fracture [see Warnings and Precautions ( 5.5 )] Hypercalcemia following treatment discontinuation in patients with giant cell tumor of bone and in patients with growing skeletons [see Warnings and Precautions ( 5.6 ) and Use in Specific Populations ( 8.4 )] Multiple vertebral fractures (MVF) following treatment discontinuation [see Warnings and Precautions ( 5.7 )]
Description
Bomyntra is a RANK ligand (RANKL) inhibitor indicated for: Prevention of skeletal-related events in patients with multiple myeloma and in patients with bone metastases from solid tumors. ( 1.1 ) Treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity. ( 1.2 , 14.3 ) Treatment of hypercalcemia of malignancy refractory to bisphosphonate therapy. ( 1.3 )
Medication Information
Warnings and Precautions
Drug Products with Same Active Ingredient: Patients receiving Bomyntra should not receive other denosumab products concomitantly ( 5.1 ) Hypersensitivity reactions including anaphylaxis may occur. Discontinue permanently if a clinically significant reaction occurs. ( 5.2 ) Hypocalcemia: Denosumab products can cause severe symptomatic hypocalcemia, and fatal cases have been reported. Correct hypocalcemia prior to initiating Bomyntra. Monitor calcium levels during therapy, especially in the first weeks of initiating therapy, and adequately supplement all patients with calcium and vitamin D. ( 5.3 ) Osteonecrosis of the jaw (ONJ) has been reported in patients receiving denosumab products. Perform an oral examination prior to starting Bomyntra. Monitor for symptoms. Avoid invasive dental procedures during treatment with Bomyntra. ( 5.4 ) Atypical femoral fracture: Evaluate patients with thigh or groin pain to rule out a femoral fracture. ( 5.5 ) Hypercalcemia Following Treatment Discontinuation in Patients with Giant Cell Tumor of Bone and in Patients with Growing Skeletons: Monitor patients for signs and symptoms of hypercalcemia, and manage as clinically appropriate. ( 5.6 , 8.4 ) Multiple Vertebral Fractures (MVF) Following Treatment Discontinuation: When Bomyntra treatment is discontinued, evaluate the individual patient's risk for vertebral fractures. ( 5.7 ) Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of potential risk to the fetus and to use effective contraception. ( 5.8 , 8.1 , 8.3 )
Indications and Usage
Bomyntra is a RANK ligand (RANKL) inhibitor indicated for: Prevention of skeletal-related events in patients with multiple myeloma and in patients with bone metastases from solid tumors. ( 1.1 ) Treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity. ( 1.2 , 14.3 ) Treatment of hypercalcemia of malignancy refractory to bisphosphonate therapy. ( 1.3 )
Dosage and Administration
Bomyntra should be administered by a healthcare provider. ( 2.1 ) Bomyntra is intended for subcutaneous route only and should not be administered intravenously, intramuscularly, or intradermally. ( 2.1 ) Multiple Myeloma and Bone Metastasis from Solid Tumors: Administer 120 mg every 4 weeks as a subcutaneous injection in the upper arm, upper thigh, or abdomen. ( 2.2 ) Giant Cell Tumor of Bone: Administer 120 mg every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen. ( 2.3 ) Administer calcium and vitamin D as necessary to treat or prevent hypocalcemia. ( 2.2 , 2.3 ) Hypercalcemia of Malignancy: Administer 120 mg every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen. ( 2.4 )
Dosage Forms and Strengths
How Supplied
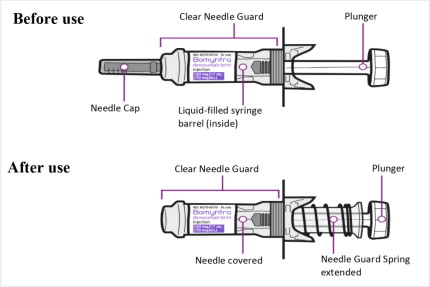
Bomyntra injection is a clear, colorless to pale yellow solution supplied in a single-dose vial and in a single-dose prefilled syringe with a safety guard.
| 120 mg/1.7 mL (70 mg/mL) in a single-dose vial | 1 vial per carton | NDC 6521967001 |
| 120 mg/1.7 mL (70 mg/mL) in a single-dose prefilled syringe | 1 syringe per carton | NDC 6521967201 |
Contraindications
Hypocalcemia ( 4.1 ) Known clinically significant hypersensitivity to denosumab products ( 4.2 )
Adverse Reactions
The following adverse reactions are discussed below and elsewhere in the labeling: Hypersensitivity [see Warnings and Precautions ( 5.2 )] Hypocalcemia [see Warnings and Precautions ( 5.3 ) and Use in Specific Populations ( 8.6 )] Osteonecrosis of the Jaw [see Warnings and Precautions ( 5.4 )] Atypical Subtrochanteric and Diaphyseal Femoral Fracture [see Warnings and Precautions ( 5.5 )] Hypercalcemia following treatment discontinuation in patients with giant cell tumor of bone and in patients with growing skeletons [see Warnings and Precautions ( 5.6 ) and Use in Specific Populations ( 8.4 )] Multiple vertebral fractures (MVF) following treatment discontinuation [see Warnings and Precautions ( 5.7 )]
Description
Bomyntra is a RANK ligand (RANKL) inhibitor indicated for: Prevention of skeletal-related events in patients with multiple myeloma and in patients with bone metastases from solid tumors. ( 1.1 ) Treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity. ( 1.2 , 14.3 ) Treatment of hypercalcemia of malignancy refractory to bisphosphonate therapy. ( 1.3 )
Section 42229-5
Instructions for Bomyntra Vial
Use a 27-gauge needle to withdraw and inject the entire contents of the vial. Do not re-enter the vial. Discard vial after single entry.
Section 44425-7
Storage and Handling
Store Bomyntra in a refrigerator at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze. Prior to administration, Bomyntra may be allowed to reach room temperature up to 25°C (77°F) in the original container. Once removed from the refrigerator, Bomyntra must not be exposed to temperatures above 77°F (25°C) or direct light and must be used within 14 days. Discard Bomyntra if not used within the 14 days. Do not use Bomyntra after the expiry date printed on the label.
Protect Bomyntra from direct light and heat.
Avoid vigorous shaking of Bomyntra.
10 Overdosage
There is no experience with overdosage of denosumab products.
11 Description
Denosumab-bnht is a human IgG2 monoclonal antibody that binds to human RANKL. Denosumab-bnht has an approximate molecular weight of 147 kDa and is produced in genetically engineered mammalian (Chinese hamster ovary) cells.
Bomyntra (denosumab-bnht) injection is a sterile, preservative-free, clear, colorless to pale yellow solution for subcutaneous use.
Each ready-to-use, single-dose prefilled syringe (PFS) and single-dose vial of Bomyntra contains 120 mg denosumab-bnht (70 mg/mL solution), 0.39 mg glacial acetic acid , 0.17 mg polysorbate 20, 1.83 mg sodium acetate, 79.9 mg sorbitol, and Water for Injection (USP). The pH is 5.2.
4.1 Hypocalcemia
Pre-existing hypocalcemia must be corrected prior to initiating therapy with Bomyntra [see Warnings and Precautions (5.3)].
5.3 Hypocalcemia
Denosumab products can cause severe symptomatic hypocalcemia, and fatal cases have been reported. Correct pre-existing hypocalcemia prior to Bomyntra treatment. Monitor calcium levels, throughout Bomyntra therapy, especially in the first weeks of initiating therapy, and administer calcium, magnesium, and vitamin D as necessary. Concomitant use of calcimimetics and other drugs that can lower calcium levels may worsen hypocalcemia risk and serum calcium should be closely monitored. Advise patients to contact a healthcare provider for symptoms of hypocalcemia [see Contraindications (4.1), Adverse Reactions (6.1, 6.2), and Patient Counseling Information (17)].
An increased risk of hypocalcemia has been observed in clinical trials of patients with increasing renal dysfunction, most commonly with severe dysfunction (creatinine clearance less than 30 mL/min and/or on dialysis), and with inadequate/no calcium supplementation. Monitor calcium levels and calcium and vitamin D intake [see Adverse Reactions (6.1), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
8.4 Pediatric Use
The safety and efficacy of Bomyntra have not been established in pediatric patients except in skeletally mature adolescents (aged 12–16 years) with giant cell tumor of bone. Bomyntra is recommended only for treatment of skeletally mature adolescents (aged 12–16 years) with giant cell tumor of bone [see Indications and Usage (1.2)]. Clinically significant hypercalcemia after treatment discontinuation has been reported in pediatric patients with growing skeletons who received denosumab products for giant cell tumor of bone or for unapproved indications [see Adverse Reactions (6.2) and Warnings and Precautions (5.6)].
Denosumab was studied in an open-label trial that enrolled a subset of 19 adolescent patients (aged 12-16 years) with giant cell tumor of bone who had reached skeletal maturity, defined by at least 1 mature long bone (e.g., closed epiphyseal growth plate of the humerus), and had a body weight ≥ 45 kg [see Indications and Usage (1.2) and Clinical Trials (14.3)]. A total of one of five (20%) evaluable adolescent patients had an objective response by retrospective independent assessment of radiographic response according to modified Response Evaluation Criteria in Solid Tumors (RECIST 1.1). The adverse reaction profile and efficacy results appeared to be similar in skeletally mature adolescents and adults [see Adverse Reactions (6.1) and Clinical Trials (14.3)].
8.5 Geriatric Use
Of the total number of patients in clinical studies that received denosumab (n = 2841) in Studies 20050136, 20050244, and 20050103, 1271 (44%) were ≥ 65 years old, while 473 patients (17%) were ≥ 75 years old. Of the 859 patients in Study 20090482 that received denosumab, 387 patients (45%) were ≥ 65 years old, while 141 patients (16%) were ≥ 75 years old. No overall differences in safety or efficacy were observed between older and younger patients.
12.6 Immunogenicity
Drug Interactions
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of denosumab or of other denosumab products. Using an electrochemiluminescent bridging immunoassay, less than 1% (55 out of 8113) of patients treated with denosumab for up to 5 years tested positive for binding antibodies (including pre-existing, transient, and developing antibodies). None of the patients tested positive for neutralizing antibodies, as was assessed using a chemiluminescent cell-based in vitro biological assay. There was no identified clinically significant effect of anti-drug antibodies on pharmacokinetics, pharmacodynamics, safety, or effectiveness of denosumab.
4 Contraindications
6 Adverse Reactions
The following adverse reactions are discussed below and elsewhere in the labeling:
- Hypersensitivity [see Warnings and Precautions (5.2)]
- Hypocalcemia [see Warnings and Precautions (5.3) and Use in Specific Populations (8.6)]
- Osteonecrosis of the Jaw [see Warnings and Precautions (5.4)]
- Atypical Subtrochanteric and Diaphyseal Femoral Fracture [see Warnings and Precautions (5.5)]
- Hypercalcemia following treatment discontinuation in patients with giant cell tumor of bone and in patients with growing skeletons [see Warnings and Precautions (5.6) and Use in Specific Populations (8.4)]
- Multiple vertebral fractures (MVF) following treatment discontinuation [see Warnings and Precautions (5.7)]
4.2 Hypersensitivity
5.2 Hypersensitivity
Clinically significant hypersensitivity including anaphylaxis has been reported with use of denosumab products. Reactions may include hypotension, dyspnea, upper airway edema, lip swelling, rash, pruritus, and urticaria. If an anaphylactic or other clinically significant allergic reaction occurs, initiate appropriate therapy and discontinue Bomyntra therapy permanently [see Contraindications (4.2) and Adverse Reactions (6.2)].
8.6 Renal Impairment
Two clinical trials were conducted in patients without cancer and with varying degrees of renal function.
In one study, patients (N = 55) with varying degrees of renal function (ranging from normal through end-stage renal disease requiring dialysis) received a single 60 mg subcutaneous dose of denosumab. In a second study, patients (N = 32) with severe renal dysfunction (creatinine clearance less than 30 mL/min and/or on dialysis) were given two 120 mg subcutaneous doses of denosumab. In both studies, greater risk of developing hypocalcemia was observed with increasing renal impairment, and with inadequate/no calcium supplementation. Hypocalcemia was mild to moderate in severity in 96% of patients. Monitor calcium levels and calcium and vitamin D intake [see Warnings and Precautions (5.3), Adverse Reactions (6.1), and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In patients with breast cancer and bone metastases, the median reduction in uNTx/Cr was 82% within 1 week following initiation of denosumab 120 mg administered subcutaneously. In Studies 20050136, 20050244, and 20050103, the median reduction in uNTx/Cr from baseline to Month 3 was approximately 80% in 2075 denosumab-treated patients.
In a phase 3 study of patients with newly diagnosed multiple myeloma who received subcutaneous doses of denosumab 120 mg every 4 weeks (Q4W), median reductions in uNTx/Cr of approximately 75% were observed by week 5. Reductions in bone turnover markers were maintained, with median reductions of 74% to 79% for uNTx/Cr from weeks 9 to 49 of continued 120 mg Q4W dosing.
In adult and skeletally mature adolescent patients with giant cell tumor of bone who received subcutaneous doses of denosumab 120 mg Q4W with a 120 mg loading dose on Days 8 and 15, median reductions in uNTx/Cr from baseline were 84% at Week 13 and 82% at Week 25.
12.3 Pharmacokinetics
Following subcutaneous administration, bioavailability was 62%. Denosumab displayed nonlinear pharmacokinetics at doses below 60 mg, but approximately dose-proportional increases in exposure at higher doses.
With multiple subcutaneous doses of 120 mg once every 4 weeks, up to 2.8-fold accumulation in serum denosumab concentrations was observed and steady-state was achieved by 6 months. A mean (± standard deviation) serum steady-state trough concentration of 20.5 (± 13.5) mcg/mL was achieved by 6 months. The mean elimination half-life was 28 days.
In patients with newly diagnosed multiple myeloma who received 120 mg every 4 weeks, denosumab concentrations appear to reach steady-state by month 6. In patients with giant cell tumor of bone, after administration of subcutaneous doses of 120 mg once every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy, mean (± standard deviation) serum trough concentrations on Day 8, 15, and one month after the first dose were 19.0 (± 24.1), 31.6 (± 27.3), 36.4 (± 20.6) mcg/mL, respectively. Steady-state was achieved in 3 months after initiation of treatment with a mean serum trough concentration of 23.4 (± 12.1) mcg/mL.
14.2 Multiple Myeloma
The efficacy of denosumab for the prevention of skeletal-related events in newly diagnosed multiple myeloma patients with treatment through disease progression, was evaluated in Study 20090482 (NCT01345019), an international, randomized (1:1), double-blind, active-controlled, noninferiority trial comparing denosumab with zoledronic acid. In this trial, patients were randomized to receive 120 mg denosumab subcutaneously every 4 weeks or 4 mg zoledronic acid intravenously (IV) every 4 weeks (dose adjusted for reduced renal function). Patients with creatinine clearance less than 30 mL/min were excluded. In this trial, the main efficacy outcome measure was noninferiority of time to first skeletal-related event (SRE). Additional efficacy outcome measures were superiority of time to first SRE, time to first and subsequent SRE, and overall survival. An SRE was defined as any of the following: pathologic fracture, radiation therapy to bone, surgery to bone, or spinal cord compression.
Study 20090482 enrolled 1718 newly diagnosed multiple myeloma patients with bone lesions. Randomization was stratified by a history of prior SRE (yes or no), the anti-myeloma agent being utilized/planned to be utilized in first-line therapy (novel therapy-based or non-novel therapy-based [novel therapies include bortezomib, lenalidomide, or thalidomide]), intent to undergo autologous PBSC transplantation (yes or no), stage at diagnosis (International Staging System I or II or III) and region Japan (yes or no). At study enrollment, 96% of the patients were receiving or planning to receive novel therapy-based first-line anti-myeloma therapy, 55% of the patients intended to undergo autologous PBSC transplantation, 61% of patients had a previous SRE, 32% were at ISS stage I, 38% were at ISS stage II and 29% were at ISS Stage III, and 2% were enrolled from Japan. Median age was 63 years, 82% of patients were White, and 46% of patients were women. The median number of doses administered was 16 for denosumab and 15 for zoledronic acid.
Denosumab was noninferior to zoledronic acid in delaying the time to first SRE following randomization (HR = 0.98, 95% CI, 0.85-1.14). The results for overall survival (OS) were comparable between denosumab and zoledronic acid treatment groups with a hazard ratio of 0.90 (95% CI: 0.70, 1.16).
|
a NE = not estimable |
||
|
Study 20090482
Multiple Myeloma |
||
|
Denosumab
N = 859 |
Zoledronic Acid
N = 859 |
|
| First On-study SRE | ||
| Number of Patients who had SREs (%) | 376 (43.8) | 383 (44.6) |
| Components of First SRE | ||
| Radiation to Bone | 47 (5.5) | 62 (7.2) |
| Pathological Fracture | 342 (39.8) | 338 (39.3) |
| Surgery to Bone | 37 (4.3) | 48 (5.6) |
| Spinal Cord Compression | 6 (0.7) | 4 (0.5) |
| Median Time to SRE (months) (95% CI) |
22.8 (14.7, NEa) |
24 (16.6, 33.3) |
| Hazard Ratio (95% CI) | 0.98 (0.85, 1.14) |
1 Indications and Usage
Bomyntra is a RANK ligand (RANKL) inhibitor indicated for:
- Prevention of skeletal-related events in patients with multiple myeloma and in patients with bone metastases from solid tumors. (1.1)
- Treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity. (1.2, 14.3)
- Treatment of hypercalcemia of malignancy refractory to bisphosphonate therapy. (1.3)
12.1 Mechanism of Action
Denosumab products bind to RANKL, a transmembrane or soluble protein essential for the formation, function, and survival of osteoclasts, the cells responsible for bone resorption, thereby modulating calcium release from bone. Increased osteoclast activity, stimulated by RANKL, is a mediator of bone pathology in solid tumors with osseous metastases.Similarly, giant cell tumors of bone consist of stromal cells expressing RANKL and osteoclast-like giant cells expressing RANK receptor, and signaling through the RANK receptor contributes to osteolysis and tumor growth. Denosumab products prevent RANKL from activating its receptor, RANK, on the surface of osteoclasts, their precursors, and osteoclast-like giant cells.
5.8 Embryo Fetal Toxicity
Based on data from animal studies and its mechanism of action, denosumab products can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of denosumab to cynomolgus monkeys throughout pregnancy at a dose 25-fold higher than the recommended human dose of denosumab based on body weight resulted in increased fetal loss, stillbirths, and postnatal mortality, along with evidence of absent peripheral lymph nodes, abnormal bone growth and decreased neonatal growth.
Verify the pregnancy status of females of reproductive potential prior to the initiation of Bomyntra. Advise pregnant women and females of reproductive potential that exposure to Bomyntra during pregnancy or within 5 months prior to conception can result in fetal harm. Advise females of reproductive potential to use effective contraception during therapy, and for at least 5 months after the last dose of Bomyntra [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions
- Drug Products with Same Active Ingredient: Patients receiving Bomyntra should not receive other denosumab products concomitantly (5.1)
- Hypersensitivity reactions including anaphylaxis may occur. Discontinue permanently if a clinically significant reaction occurs. (5.2)
- Hypocalcemia: Denosumab products can cause severe symptomatic hypocalcemia, and fatal cases have been reported. Correct hypocalcemia prior to initiating Bomyntra. Monitor calcium levels during therapy, especially in the first weeks of initiating therapy, and adequately supplement all patients with calcium and vitamin D. (5.3)
- Osteonecrosis of the jaw (ONJ) has been reported in patients receiving denosumab products. Perform an oral examination prior to starting Bomyntra. Monitor for symptoms. Avoid invasive dental procedures during treatment with Bomyntra. (5.4)
- Atypical femoral fracture: Evaluate patients with thigh or groin pain to rule out a femoral fracture. (5.5)
- Hypercalcemia Following Treatment Discontinuation in Patients with Giant Cell Tumor of Bone and in Patients with Growing Skeletons: Monitor patients for signs and symptoms of hypercalcemia, and manage as clinically appropriate. (5.6, 8.4)
- Multiple Vertebral Fractures (MVF) Following Treatment Discontinuation: When Bomyntra treatment is discontinued, evaluate the individual patient's risk for vertebral fractures. (5.7)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of potential risk to the fetus and to use effective contraception. (5.8, 8.1, 8.3)
2 Dosage and Administration
- Bomyntra should be administered by a healthcare provider. (2.1)
- Bomyntra is intended for subcutaneous route only and should not be administered intravenously, intramuscularly, or intradermally. (2.1)
- Multiple Myeloma and Bone Metastasis from Solid Tumors: Administer 120 mg every 4 weeks as a subcutaneous injection in the upper arm, upper thigh, or abdomen. (2.2)
- Giant Cell Tumor of Bone: Administer 120 mg every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen. (2.3)
- Administer calcium and vitamin D as necessary to treat or prevent hypocalcemia. (2.2, 2.3)
- Hypercalcemia of Malignancy: Administer 120 mg every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen. (2.4)
1.2 Giant Cell Tumor of Bone
Bomyntra is indicated for the treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity [see Clinical Trials (14.2)].
2.3 Giant Cell Tumor of Bone
The recommended dose of Bomyntra is 120 mg administered every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen.
Administer calcium and vitamin D as necessary to treat or prevent hypocalcemia [see Warnings and Precautions (5.3)].
3 Dosage Forms and Strengths
- Injection: 120 mg/1.7 mL (70 mg/mL) clear, colorless to pale yellow solution in a single-dose vial.
- Injection: 120 mg/1.7 mL (70 mg/mL) clear, colorless to pale yellow solution in a single-dose prefilled syringe.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of denosumab products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hypocalcemia: Severe symptomatic hypocalcemia, including fatal cases [see Contraindications (4.1) and Warnings and Precautions (5.3)].
- Hypercalcemia: Severe symptomatic hypercalcemia following treatment discontinuation can occur [see Adverse Reactions (6) and Warnings and Precautions (5.6)].
- Hypersensitivity, including anaphylactic reactions [see Contraindications (4.2) and Warnings and Precautions (5.2)].
- Musculoskeletal pain, including severe musculoskeletal pain. Positive re-challenge has been reported.
- Lichenoid drug eruptions (e.g., lichen planus-like reactions).
- Alopecia.
14.3 Giant Cell Tumor of Bone
The safety and efficacy of denosumab for the treatment of giant cell tumor of bone in adults or skeletally mature adolescents were demonstrated in two open-label trials [Study 20040215 (NCT00396279) and Study 20062004 (NCT00680992)] that enrolled patients with histologically confirmed measurable giant cell tumor of bone that was either recurrent, unresectable, or for which planned surgery was likely to result in severe morbidity. Patients received 120 mg denosumab subcutaneously every 4 weeks with a loading dose on Days 8 and 15 of the first cycle of therapy. Patients who discontinued denosumab then entered the safety follow-up phase for a minimum of 60 months. Retreatment with denosumab while in safety follow-up was allowed for patients who initially demonstrated a response to denosumab (e.g., in the case of recurrent disease).
Study 20040215 was a single-arm, pharmacodynamic, and proof of concept trial conducted in 37 adult patients with unresectable or recurrent giant cell tumor of bone. Patients were required to have histologically confirmed giant cell tumor of bone and radiologic evidence of measurable disease from a computed tomography (CT) or magnetic resonance imaging (MRI) obtained within 28 days prior to study enrollment. Patients enrolled in Study 20040215 underwent CT or MRI assessment of giant cell tumor of bone at baseline and quarterly during denosumab treatment.
Study 20062004 was a parallel-cohort, proof of concept, and safety trial conducted in 535 adult or skeletally mature adolescent patients with histologically confirmed giant cell tumor of bone and evidence of measurable active disease. Study 20062004 enrolled 19 patients who were 12-16 years of age [see Use in Specific Populations (8.4)]. Patients enrolled into one of three cohorts: Cohort 1 enrolled 268 patients with surgically unsalvageable disease (e.g., sacral or spinal sites of disease, or pulmonary metastases); Cohort 2 enrolled 252 patients with surgically salvageable disease where the investigator determined that the planned surgery was likely to result in severe morbidity (e.g., joint resection, limb amputation, or hemipelvectomy); Cohort 3 enrolled 15 patients who previously participated in Study 20040215. Patients underwent imaging assessment of disease status at intervals determined by their treating physician.
A retrospective interim analysis concluded by an independent review committee evaluated objective response in 187 patients enrolled and treated in Study 20040215 and Study 20062004 for whom baseline and at least one post-baseline radiographic assessment were available (27 of 37 patients enrolled in Study 20040215 and 160 of 270 patients enrolled in Cohorts 1 and 2 of Study 20062004). The primary efficacy outcome measure was objective response rate using Response Evaluation Criteria in Solid Tumors (RECIST) v 1.1.
The overall objective response rate (RECIST 1.1) was 25% (95% CI: 19, 32). All responses were partial responses. The estimated median time to response was 3 months. In the 47 patients with an objective response, the median duration of follow-up was 20 months (range: 2-44 months), and 51% (24/47) had a duration of response lasting at least 8 months. Three patients experienced disease progression following an objective response.
8 Use in Specific Populations
- Pediatric patients: Recommended only for treatment of skeletally mature adolescents with giant cell tumor of bone. (8.4)
- Renal impairment: Patients with creatinine clearance less than 30 mL/min or receiving dialysis are at risk for hypocalcemia. Adequately supplement with calcium and vitamin D. (8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.3 Hypercalcemia of Malignancy
Bomyntra is indicated for the treatment of hypercalcemia of malignancy refractory to bisphosphonate therapy.
2.4 Hypercalcemia of Malignancy
The recommended dose of Bomyntra is 120 mg administered every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen.
14.4 Hypercalcemia of Malignancy
The safety and efficacy of denosumab was demonstrated in an open-label, single-arm trial [Study 20070315 (NCT00896454)] that enrolled 33 patients with hypercalcemia of malignancy (with or without bone metastases) refractory to treatment with intravenous bisphosphonate therapy. Patients received denosumab subcutaneously every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy.
In this trial, refractory hypercalcemia of malignancy was defined as an albumin-corrected calcium of > 12.5 mg/dL (3.1 mmol/L) despite treatment with intravenous bisphosphonate therapy in 7-30 days prior to initiation of denosumab therapy. The primary outcome measure was the proportion of patients achieving a response, defined as corrected serum calcium (CSC) ≤ 11.5 mg/dL (2.9 mmol/L), within 10 days after denosumab administration. Efficacy data are summarized in Figure 1 and Table 4. Concurrent chemotherapy did not appear to affect response to denosumab.
Figure 1. Corrected Serum Calcium by Visit in Responders (Median and Interquartile Range)
N= Number of responders who received ≥ 1 dose of investigational product
n = Number of responders who had no missing data at baseline and the time point of interest
|
N = 33 |
Proportion (%) (95% CI) |
|
| All Responders (CSC ≤ 11.5 mg/dL) by Day 10 | 21 | 63.6 (45.1, 79.6) |
| All Responders by Day 57 | 23 | 69.7 (51.3, 84.4) |
| Complete Responders (CSC ≤ 10.8 mg/dL) by Day 10 | 12 | 36.4 (20.4, 54.9) |
| All Complete Responders by Day 57 | 21 | 63.6 (45.1, 79.6) |
Median time to response (CSC ≤ 11.5 mg/dL) was 9 days (95% CI: 8, 19), and the median duration of response was 104 days (95% CI: 7, not estimable). Median time to complete response (CSC ≤ 10.8 mg/dL) was 23 days (95% CI: 9, 36), and the median duration of complete response was 34 days (95% CI: 1, 134).
2.5 Preparation and Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Bomyntra is a clear, colorless to pale yellow solution that is free from visible particles. Do not use if the solution is discolored or cloudy or if the solution contains particles or foreign particulate matter.
Prior to administration, Bomyntra may be removed from the refrigerator and brought to room temperature up to 25°C (77°F) by standing in the original container. This generally takes 15 to 30 minutes. Do not warm Bomyntra in any other way [see How Supplied/Storage and Handling (16)].
5.4 Osteonecrosis of the Jaw (onj)
Osteonecrosis of the jaw (ONJ) has been reported in patients receiving denosumab products, manifesting as jaw pain, osteomyelitis, osteitis, bone erosion, tooth or periodontal infection, toothache, gingival ulceration, or gingival erosion. Persistent pain or slow healing of the mouth or jaw after dental surgery may also be manifestations of ONJ. In clinical trials in patients with cancer, the incidence of ONJ was higher with longer duration of exposure [see Adverse Reactions (6.1)]. Seventy-nine percent of patients with ONJ had a history of tooth extraction, poor oral hygiene, or use of a dental appliance as a predisposing factor. Other risk factors for the development of ONJ include immunosuppressive therapy, treatment with angiogenesis inhibitors, systemic corticosteroids, diabetes, and gingival infections. Similarly, for denosumab-treated patients with multiple myeloma that developed ONJ, 58% had a history of invasive dental procedures as a predisposing factor.
Perform an oral examination and appropriate preventive dentistry prior to the initiation of Bomyntra and periodically during Bomyntra therapy. Advise patients regarding oral hygiene practices. Avoid invasive dental procedures during treatment with Bomyntra. Consider temporary discontinuation of Bomyntra therapy if an invasive dental procedure must be performed. There are no data available to suggest the optimal duration of treatment interruption.
Patients who are suspected of having or who develop ONJ while on Bomyntra should receive care by a dentist or an oral surgeon. In these patients, extensive dental surgery to treat ONJ may exacerbate the condition. Clinical judgment of the treating healthcare provider should guide the management plan of each patient based on individual risk/benefit assessment.
14.1 Bone Metastasis From Solid Tumors
The safety and efficacy of denosumab for the prevention of skeletal-related events in patients with bone metastases from solid tumors was demonstrated in three international, randomized (1:1), double-blind, active-controlled, noninferiority trials comparing denosumab with zoledronic acid. In all three trials, patients were randomized to receive 120 mg denosumab subcutaneously every 4 weeks or 4 mg zoledronic acid intravenously (IV) every 4 weeks (dose adjusted for reduced renal function). Patients with creatinine clearance less than 30 mL/min were excluded. In each trial, the main outcome measure was demonstration of noninferiority of time to first skeletal-related event (SRE) as compared to zoledronic acid. Supportive outcome measures were superiority of time to first SRE and superiority of time to first and subsequent SRE; testing for these outcome measures occurred if the main outcome measure was statistically significant. An SRE was defined as any of the following: pathologic fracture, radiation therapy to bone, surgery to bone, or spinal cord compression.
Study 20050136 (NCT00321464) enrolled 2046 patients with advanced breast cancer and bone metastasis. Randomization was stratified by a history of prior SRE (yes or no), receipt of chemotherapy within 6 weeks prior to randomization (yes or no), prior oral bisphosphonate use (yes or no), and region (Japan or other countries). Forty percent of patients had a previous SRE, 40% received chemotherapy within 6 weeks prior to randomization, 5% received prior oral bisphosphonates, and 7% were enrolled from Japan. Median age was 57 years, 80% of patients were White, and 99% of patients were women. The median number of doses administered was 18 for denosumab and 17 for zoledronic acid.
Study 20050244 (NCT00330759) enrolled 1776 adults with solid tumors other than breast and castrate-resistant prostate cancer with bone metastasis and multiple myeloma. Randomization was stratified by previous SRE (yes or no), systemic anticancer therapy at time of randomization (yes or no), and tumor type (non-small cell lung cancer, myeloma, or other). Eighty-seven percent were receiving systemic anticancer therapy at the time of randomization, 52% had a previous SRE, 64% of patients were men, 87% were White, and the median age was 60 years. A total of 40% of patients had non-small cell lung cancer, 10% had multiple myeloma, 9% had renal cell carcinoma, and 6% had small cell lung cancer. Other tumor types each comprised less than 5% of the enrolled population. The median number of doses administered was 7 for both denosumab and zoledronic acid.
Study 20050103 (NCT00321620) enrolled 1901 men with castrate-resistant prostate cancer and bone metastasis. Randomization was stratified by previous SRE, PSA level (less than 10 ng/mL or 10 ng/mL or greater) and receipt of chemotherapy within 6 weeks prior to randomization (yes or no). Twenty-six percent of patients had a previous SRE, 15% of patients had PSA less than 10 ng/mL, and 14% received chemotherapy within 6 weeks prior to randomization. Median age was 71 years and 86% of patients were White. The median number of doses administered was 13 for denosumab and 11 for zoledronic acid.
Denosumab delayed the time to first SRE following randomization as compared to zoledronic acid in patients with breast or castrate-resistant prostate cancer (CRPC) with osseous metastases (Table 2). In patients with bone metastasis due to other solid tumors or lytic lesions due to multiple myeloma, denosumab was noninferior to zoledronic acid in delaying the time to first SRE following randomization.
Overall survival and progression-free survival were similar between arms in all three trials.
|
a CRPC = castrate-resistant prostate cancer. |
||||||
|
b NR = not reached. |
||||||
|
c Superiority testing performed only after denosumab demonstrated to be noninferior to zoledronic acid within trial. |
||||||
|
d All skeletal events postrandomization; new events defined by occurrence ≥ 21 days after preceding event. |
||||||
|
e Adjusted p-values are presented. |
||||||
| Study 20050136 Metastatic Breast Cancer | Study 20050244 Metastatic Solid Tumors or Multiple Myeloma | Study 20050103 Metastatic CRPC a | ||||
|
Denosumab
N = 1026 |
Zoledronic Acid
N = 1020 |
Denosumab
N = 886 |
Zoledronic Acid
N = 890 |
Denosumab
N = 950 |
Zoledronic Acid
N = 951 |
|
| First On-study SRE | ||||||
| Number of Patients who had SREs (%) | 315 (30.7) | 372 (36.5) | 278 (31.4) | 323 (36.3) | 341 (35.9) | 386 (40.6) |
| Components of First SRE | ||||||
| Radiation to Bone | 82 (8.0) | 119 (11.7) | 119 (13.4) | 144 (16.2) | 177 (18.6) | 203 (21.3) |
| Pathological Fracture | 212 (20.7) | 238 (23.3) | 122 (13.8) | 139 (15.6) | 137 (14.4) | 143 (15.0) |
| Surgery to Bone | 12 (1.2) | 8 (0.8) | 13 (1.5) | 19 (2.1) | 1 (0.1) | 4 (0.4) |
| Spinal Cord Compression | 9 (0.9) | 7 (0.7) | 24 (2.7) | 21 (2.4) | 26 (2.7) | 36 (3.8) |
| Median Time to SRE (months) | NRb | 26.4 | 20.5 | 16.3 | 20.7 | 17.1 |
| Hazard Ratio (95% CI) | 0.82 (0.71, 0.95) | 0.84 (0.71, 0.98) | 0.82 (0.71, 0.95) | |||
| Noninferiority p-value | < 0.001 | < 0.001 | < 0.001 | |||
| Superiority p-valuec | 0.010 | 0.060 | 0.008 | |||
| First and Subsequent SRE d | ||||||
| Mean Number/Patient | 0.46 | 0.60 | 0.44 | 0.49 | 0.52 | 0.61 |
| Rate Ratio (95% CI) | 0.77 (0.66, 0.89) | 0.90 (0.77, 1.04) | 0.82 (0.71, 0.94) | |||
| Superiority p-valuee | 0.001 | 0.145 | 0.009 |
2.1 Important Administration Instructions
Bomyntra should be administered by a healthcare provider.
Bomyntra is intended for subcutaneous route only and should not be administered intravenously, intramuscularly, or intradermally.
13.2 Animal Toxicology And/or Pharmacology
Denosumab products are inhibitors of osteoclastic bone resorption via inhibition of RANKL.
Because the biological activity of denosumab in animals is specific to nonhuman primates, evaluation of genetically engineered (knockout) mice or use of other biological inhibitors of the RANK/RANKL pathway, OPG-Fc and RANK-Fc, provided additional information on the pharmacodynamic properties of denosumab. RANK/RANKL knockout mice exhibited absence of lymph node formation, as well as an absence of lactation due to inhibition of mammary gland maturation (lobulo-alveolar gland development during pregnancy). Neonatal RANK/RANKL knockout mice exhibited reduced bone growth and lack of tooth eruption. A corroborative study in 2-week-old rats given the RANKL inhibitor OPG-Fc also showed reduced bone growth, altered growth plates, and impaired tooth eruption. These changes were partially reversible in this model when dosing with the RANKL inhibitors was discontinued.
5.1 Drug Products With Same Active Ingredient
Patients receiving Bomyntra should not receive other denosumab products concomitantly.
8.3 Females and Males of Reproductive Potential
Based on findings in animals and its mechanism of action, denosumab products can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Principal Display Panel – 120 Mg/1.7 Ml Carton Label
NDC 65219-670-01 Rx only
Scan for additional information
Bomyntra
denosumab-bnht
Injection
120 mg/1.7 mL (70 mg/mL)
For Subcutaneous use only
Sterile Solution - No Preservative
Bomyntra should be administered by a healthcare provider
1 Single-dose vial. Discard unused portion.
FRESENIUS
Kabi
Principal Display Panel – 120 Mg/1.7 Ml Syringe Label
NDC 65219-672-01 Rx only
Bomyntra
denosumab-bnht
Injection
120 mg/1.7 mL
(70 mg/mL)
For Subcutaneous use only
Fresenius Kabi
USA, LLC
U.S. License
No. 2146
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity
The carcinogenic potential of denosumab products has not been evaluated in long-term animal studies.
Mutagenicity
The genotoxic potential of denosumab products has not been evaluated.
Impairment of Fertility
Denosumab had no effect on female fertility or male reproductive organs in monkeys at doses that were 6.5- to 25-fold higher than the recommended human dose of 120 mg subcutaneously administered once every 4 weeks, based on body weight (mg/kg).
Carcinogenicity
1.1 Multiple Myeloma and Bone Metastasis From Solid Tumors
Bomyntra is indicated for the prevention of skeletal-related events in patients with multiple myeloma and in patients with bone metastases from solid tumors.
2.2 Multiple Myeloma and Bone Metastasis From Solid Tumors
The recommended dose of Bomyntra is 120 mg administered as a subcutaneous injection every 4 weeks in the upper arm, upper thigh, or abdomen.
Administer calcium and vitamin D as necessary to treat or prevent hypocalcemia [see Warnings and Precautions (5.3)].
Principal Display Panel – 120 Mg/1.7 Ml Pfs Carton Label
NDC 65219-672-01 Rx only
Scan for additional information
Bomyntra
denosumab-bnht
Injection
120 mg/1.7 mL (70 mg/mL)
For Subcutaneous use only
Single-dose prefilled syringe.
Discard unused portion
Sterile Solution - No Preservative
Carton contains:
1 Single- dose prefilled syringe
1 Prescribing Information
Bomyntra should be administered by a healthcare provider
FRESENIUS
Kabi
5.5 Atypical Subtrochanteric and Diaphyseal Femoral Fracture
Atypical femoral fracture has been reported with denosumab products [see Adverse Reactions (6.1)]. These fractures can occur anywhere in the femoral shaft from just below the lesser trochanter to above the supracondylar flare and are transverse or short oblique in orientation without evidence of comminution.
Atypical femoral fractures most commonly occur with minimal or no trauma to the affected area. They may be bilateral and many patients report prodromal pain in the affected area, usually presenting as dull, aching thigh pain, weeks to months before a complete fracture occurs. A number of reports note that patients were also receiving treatment with glucocorticoids (e.g. prednisone) at the time of fracture.
During Bomyntra treatment, patients should be advised to report new or unusual thigh, hip, or groin pain. Any patient who presents with thigh or groin pain should be suspected of having an atypical fracture and should be evaluated to rule out an incomplete femur fracture. Patient presenting with an atypical femur fracture should also be assessed for symptoms and signs of fracture in the contralateral limb. Interruption of Bomyntra therapy should be considered, pending a risk/benefit assessment, on an individual basis.
5.7 Multiple Vertebral Fractures (mvf) Following Treatment Discontinuation
Multiple vertebral fractures (MVF) have been reported following discontinuation of treatment with denosumab products. Patients at higher risk for MVF include those with risk factors for or a history of osteoporosis or prior fractures.
When Bomyntra treatment is discontinued, evaluate the individual patient's risk for vertebral fractures [see Patient Counseling Information (17)].
5.6 Hypercalcemia Following Treatment Discontinuation in Patients With Giant Cell Tumor of Bone and in Patients With Growing Skeletons
Clinically significant hypercalcemia requiring hospitalization and complicated by acute renal injury has been reported in denosumab product-treated patients with giant cell tumor of bone and patients with growing skeletons. Hypercalcemia has been reported within the first year after treatment discontinuation. After treatment is discontinued, monitor patients for signs and symptoms of hypercalcemia, assess serum calcium periodically, reevaluate the patient's calcium and vitamin D supplementation requirements and manage patients as clinically appropriate [see Adverse Reactions (6) and Use in Specific Populations (8.4)].
Structured Label Content
Dosage Forms and Strengths (34069-5)
How Supplied
Bomyntra injection is a clear, colorless to pale yellow solution supplied in a single-dose vial and in a single-dose prefilled syringe with a safety guard.
| 120 mg/1.7 mL (70 mg/mL) in a single-dose vial | 1 vial per carton | NDC 6521967001 |
| 120 mg/1.7 mL (70 mg/mL) in a single-dose prefilled syringe | 1 syringe per carton | NDC 6521967201 |
Section 42229-5 (42229-5)
Instructions for Bomyntra Vial
Use a 27-gauge needle to withdraw and inject the entire contents of the vial. Do not re-enter the vial. Discard vial after single entry.
Section 44425-7 (44425-7)
Storage and Handling
Store Bomyntra in a refrigerator at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze. Prior to administration, Bomyntra may be allowed to reach room temperature up to 25°C (77°F) in the original container. Once removed from the refrigerator, Bomyntra must not be exposed to temperatures above 77°F (25°C) or direct light and must be used within 14 days. Discard Bomyntra if not used within the 14 days. Do not use Bomyntra after the expiry date printed on the label.
Protect Bomyntra from direct light and heat.
Avoid vigorous shaking of Bomyntra.
10 Overdosage (10 OVERDOSAGE)
There is no experience with overdosage of denosumab products.
11 Description (11 DESCRIPTION)
Denosumab-bnht is a human IgG2 monoclonal antibody that binds to human RANKL. Denosumab-bnht has an approximate molecular weight of 147 kDa and is produced in genetically engineered mammalian (Chinese hamster ovary) cells.
Bomyntra (denosumab-bnht) injection is a sterile, preservative-free, clear, colorless to pale yellow solution for subcutaneous use.
Each ready-to-use, single-dose prefilled syringe (PFS) and single-dose vial of Bomyntra contains 120 mg denosumab-bnht (70 mg/mL solution), 0.39 mg glacial acetic acid , 0.17 mg polysorbate 20, 1.83 mg sodium acetate, 79.9 mg sorbitol, and Water for Injection (USP). The pH is 5.2.
4.1 Hypocalcemia
Pre-existing hypocalcemia must be corrected prior to initiating therapy with Bomyntra [see Warnings and Precautions (5.3)].
5.3 Hypocalcemia
Denosumab products can cause severe symptomatic hypocalcemia, and fatal cases have been reported. Correct pre-existing hypocalcemia prior to Bomyntra treatment. Monitor calcium levels, throughout Bomyntra therapy, especially in the first weeks of initiating therapy, and administer calcium, magnesium, and vitamin D as necessary. Concomitant use of calcimimetics and other drugs that can lower calcium levels may worsen hypocalcemia risk and serum calcium should be closely monitored. Advise patients to contact a healthcare provider for symptoms of hypocalcemia [see Contraindications (4.1), Adverse Reactions (6.1, 6.2), and Patient Counseling Information (17)].
An increased risk of hypocalcemia has been observed in clinical trials of patients with increasing renal dysfunction, most commonly with severe dysfunction (creatinine clearance less than 30 mL/min and/or on dialysis), and with inadequate/no calcium supplementation. Monitor calcium levels and calcium and vitamin D intake [see Adverse Reactions (6.1), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
8.4 Pediatric Use
The safety and efficacy of Bomyntra have not been established in pediatric patients except in skeletally mature adolescents (aged 12–16 years) with giant cell tumor of bone. Bomyntra is recommended only for treatment of skeletally mature adolescents (aged 12–16 years) with giant cell tumor of bone [see Indications and Usage (1.2)]. Clinically significant hypercalcemia after treatment discontinuation has been reported in pediatric patients with growing skeletons who received denosumab products for giant cell tumor of bone or for unapproved indications [see Adverse Reactions (6.2) and Warnings and Precautions (5.6)].
Denosumab was studied in an open-label trial that enrolled a subset of 19 adolescent patients (aged 12-16 years) with giant cell tumor of bone who had reached skeletal maturity, defined by at least 1 mature long bone (e.g., closed epiphyseal growth plate of the humerus), and had a body weight ≥ 45 kg [see Indications and Usage (1.2) and Clinical Trials (14.3)]. A total of one of five (20%) evaluable adolescent patients had an objective response by retrospective independent assessment of radiographic response according to modified Response Evaluation Criteria in Solid Tumors (RECIST 1.1). The adverse reaction profile and efficacy results appeared to be similar in skeletally mature adolescents and adults [see Adverse Reactions (6.1) and Clinical Trials (14.3)].
8.5 Geriatric Use
Of the total number of patients in clinical studies that received denosumab (n = 2841) in Studies 20050136, 20050244, and 20050103, 1271 (44%) were ≥ 65 years old, while 473 patients (17%) were ≥ 75 years old. Of the 859 patients in Study 20090482 that received denosumab, 387 patients (45%) were ≥ 65 years old, while 141 patients (16%) were ≥ 75 years old. No overall differences in safety or efficacy were observed between older and younger patients.
12.6 Immunogenicity
Drug Interactions
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of denosumab or of other denosumab products. Using an electrochemiluminescent bridging immunoassay, less than 1% (55 out of 8113) of patients treated with denosumab for up to 5 years tested positive for binding antibodies (including pre-existing, transient, and developing antibodies). None of the patients tested positive for neutralizing antibodies, as was assessed using a chemiluminescent cell-based in vitro biological assay. There was no identified clinically significant effect of anti-drug antibodies on pharmacokinetics, pharmacodynamics, safety, or effectiveness of denosumab.
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed below and elsewhere in the labeling:
- Hypersensitivity [see Warnings and Precautions (5.2)]
- Hypocalcemia [see Warnings and Precautions (5.3) and Use in Specific Populations (8.6)]
- Osteonecrosis of the Jaw [see Warnings and Precautions (5.4)]
- Atypical Subtrochanteric and Diaphyseal Femoral Fracture [see Warnings and Precautions (5.5)]
- Hypercalcemia following treatment discontinuation in patients with giant cell tumor of bone and in patients with growing skeletons [see Warnings and Precautions (5.6) and Use in Specific Populations (8.4)]
- Multiple vertebral fractures (MVF) following treatment discontinuation [see Warnings and Precautions (5.7)]
4.2 Hypersensitivity
5.2 Hypersensitivity
Clinically significant hypersensitivity including anaphylaxis has been reported with use of denosumab products. Reactions may include hypotension, dyspnea, upper airway edema, lip swelling, rash, pruritus, and urticaria. If an anaphylactic or other clinically significant allergic reaction occurs, initiate appropriate therapy and discontinue Bomyntra therapy permanently [see Contraindications (4.2) and Adverse Reactions (6.2)].
8.6 Renal Impairment
Two clinical trials were conducted in patients without cancer and with varying degrees of renal function.
In one study, patients (N = 55) with varying degrees of renal function (ranging from normal through end-stage renal disease requiring dialysis) received a single 60 mg subcutaneous dose of denosumab. In a second study, patients (N = 32) with severe renal dysfunction (creatinine clearance less than 30 mL/min and/or on dialysis) were given two 120 mg subcutaneous doses of denosumab. In both studies, greater risk of developing hypocalcemia was observed with increasing renal impairment, and with inadequate/no calcium supplementation. Hypocalcemia was mild to moderate in severity in 96% of patients. Monitor calcium levels and calcium and vitamin D intake [see Warnings and Precautions (5.3), Adverse Reactions (6.1), and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In patients with breast cancer and bone metastases, the median reduction in uNTx/Cr was 82% within 1 week following initiation of denosumab 120 mg administered subcutaneously. In Studies 20050136, 20050244, and 20050103, the median reduction in uNTx/Cr from baseline to Month 3 was approximately 80% in 2075 denosumab-treated patients.
In a phase 3 study of patients with newly diagnosed multiple myeloma who received subcutaneous doses of denosumab 120 mg every 4 weeks (Q4W), median reductions in uNTx/Cr of approximately 75% were observed by week 5. Reductions in bone turnover markers were maintained, with median reductions of 74% to 79% for uNTx/Cr from weeks 9 to 49 of continued 120 mg Q4W dosing.
In adult and skeletally mature adolescent patients with giant cell tumor of bone who received subcutaneous doses of denosumab 120 mg Q4W with a 120 mg loading dose on Days 8 and 15, median reductions in uNTx/Cr from baseline were 84% at Week 13 and 82% at Week 25.
12.3 Pharmacokinetics
Following subcutaneous administration, bioavailability was 62%. Denosumab displayed nonlinear pharmacokinetics at doses below 60 mg, but approximately dose-proportional increases in exposure at higher doses.
With multiple subcutaneous doses of 120 mg once every 4 weeks, up to 2.8-fold accumulation in serum denosumab concentrations was observed and steady-state was achieved by 6 months. A mean (± standard deviation) serum steady-state trough concentration of 20.5 (± 13.5) mcg/mL was achieved by 6 months. The mean elimination half-life was 28 days.
In patients with newly diagnosed multiple myeloma who received 120 mg every 4 weeks, denosumab concentrations appear to reach steady-state by month 6. In patients with giant cell tumor of bone, after administration of subcutaneous doses of 120 mg once every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy, mean (± standard deviation) serum trough concentrations on Day 8, 15, and one month after the first dose were 19.0 (± 24.1), 31.6 (± 27.3), 36.4 (± 20.6) mcg/mL, respectively. Steady-state was achieved in 3 months after initiation of treatment with a mean serum trough concentration of 23.4 (± 12.1) mcg/mL.
14.2 Multiple Myeloma
The efficacy of denosumab for the prevention of skeletal-related events in newly diagnosed multiple myeloma patients with treatment through disease progression, was evaluated in Study 20090482 (NCT01345019), an international, randomized (1:1), double-blind, active-controlled, noninferiority trial comparing denosumab with zoledronic acid. In this trial, patients were randomized to receive 120 mg denosumab subcutaneously every 4 weeks or 4 mg zoledronic acid intravenously (IV) every 4 weeks (dose adjusted for reduced renal function). Patients with creatinine clearance less than 30 mL/min were excluded. In this trial, the main efficacy outcome measure was noninferiority of time to first skeletal-related event (SRE). Additional efficacy outcome measures were superiority of time to first SRE, time to first and subsequent SRE, and overall survival. An SRE was defined as any of the following: pathologic fracture, radiation therapy to bone, surgery to bone, or spinal cord compression.
Study 20090482 enrolled 1718 newly diagnosed multiple myeloma patients with bone lesions. Randomization was stratified by a history of prior SRE (yes or no), the anti-myeloma agent being utilized/planned to be utilized in first-line therapy (novel therapy-based or non-novel therapy-based [novel therapies include bortezomib, lenalidomide, or thalidomide]), intent to undergo autologous PBSC transplantation (yes or no), stage at diagnosis (International Staging System I or II or III) and region Japan (yes or no). At study enrollment, 96% of the patients were receiving or planning to receive novel therapy-based first-line anti-myeloma therapy, 55% of the patients intended to undergo autologous PBSC transplantation, 61% of patients had a previous SRE, 32% were at ISS stage I, 38% were at ISS stage II and 29% were at ISS Stage III, and 2% were enrolled from Japan. Median age was 63 years, 82% of patients were White, and 46% of patients were women. The median number of doses administered was 16 for denosumab and 15 for zoledronic acid.
Denosumab was noninferior to zoledronic acid in delaying the time to first SRE following randomization (HR = 0.98, 95% CI, 0.85-1.14). The results for overall survival (OS) were comparable between denosumab and zoledronic acid treatment groups with a hazard ratio of 0.90 (95% CI: 0.70, 1.16).
|
a NE = not estimable |
||
|
Study 20090482
Multiple Myeloma |
||
|
Denosumab
N = 859 |
Zoledronic Acid
N = 859 |
|
| First On-study SRE | ||
| Number of Patients who had SREs (%) | 376 (43.8) | 383 (44.6) |
| Components of First SRE | ||
| Radiation to Bone | 47 (5.5) | 62 (7.2) |
| Pathological Fracture | 342 (39.8) | 338 (39.3) |
| Surgery to Bone | 37 (4.3) | 48 (5.6) |
| Spinal Cord Compression | 6 (0.7) | 4 (0.5) |
| Median Time to SRE (months) (95% CI) |
22.8 (14.7, NEa) |
24 (16.6, 33.3) |
| Hazard Ratio (95% CI) | 0.98 (0.85, 1.14) |
1 Indications and Usage (1 INDICATIONS AND USAGE)
Bomyntra is a RANK ligand (RANKL) inhibitor indicated for:
- Prevention of skeletal-related events in patients with multiple myeloma and in patients with bone metastases from solid tumors. (1.1)
- Treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity. (1.2, 14.3)
- Treatment of hypercalcemia of malignancy refractory to bisphosphonate therapy. (1.3)
12.1 Mechanism of Action
Denosumab products bind to RANKL, a transmembrane or soluble protein essential for the formation, function, and survival of osteoclasts, the cells responsible for bone resorption, thereby modulating calcium release from bone. Increased osteoclast activity, stimulated by RANKL, is a mediator of bone pathology in solid tumors with osseous metastases.Similarly, giant cell tumors of bone consist of stromal cells expressing RANKL and osteoclast-like giant cells expressing RANK receptor, and signaling through the RANK receptor contributes to osteolysis and tumor growth. Denosumab products prevent RANKL from activating its receptor, RANK, on the surface of osteoclasts, their precursors, and osteoclast-like giant cells.
5.8 Embryo Fetal Toxicity (5.8 Embryo-Fetal Toxicity)
Based on data from animal studies and its mechanism of action, denosumab products can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of denosumab to cynomolgus monkeys throughout pregnancy at a dose 25-fold higher than the recommended human dose of denosumab based on body weight resulted in increased fetal loss, stillbirths, and postnatal mortality, along with evidence of absent peripheral lymph nodes, abnormal bone growth and decreased neonatal growth.
Verify the pregnancy status of females of reproductive potential prior to the initiation of Bomyntra. Advise pregnant women and females of reproductive potential that exposure to Bomyntra during pregnancy or within 5 months prior to conception can result in fetal harm. Advise females of reproductive potential to use effective contraception during therapy, and for at least 5 months after the last dose of Bomyntra [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Drug Products with Same Active Ingredient: Patients receiving Bomyntra should not receive other denosumab products concomitantly (5.1)
- Hypersensitivity reactions including anaphylaxis may occur. Discontinue permanently if a clinically significant reaction occurs. (5.2)
- Hypocalcemia: Denosumab products can cause severe symptomatic hypocalcemia, and fatal cases have been reported. Correct hypocalcemia prior to initiating Bomyntra. Monitor calcium levels during therapy, especially in the first weeks of initiating therapy, and adequately supplement all patients with calcium and vitamin D. (5.3)
- Osteonecrosis of the jaw (ONJ) has been reported in patients receiving denosumab products. Perform an oral examination prior to starting Bomyntra. Monitor for symptoms. Avoid invasive dental procedures during treatment with Bomyntra. (5.4)
- Atypical femoral fracture: Evaluate patients with thigh or groin pain to rule out a femoral fracture. (5.5)
- Hypercalcemia Following Treatment Discontinuation in Patients with Giant Cell Tumor of Bone and in Patients with Growing Skeletons: Monitor patients for signs and symptoms of hypercalcemia, and manage as clinically appropriate. (5.6, 8.4)
- Multiple Vertebral Fractures (MVF) Following Treatment Discontinuation: When Bomyntra treatment is discontinued, evaluate the individual patient's risk for vertebral fractures. (5.7)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of potential risk to the fetus and to use effective contraception. (5.8, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Bomyntra should be administered by a healthcare provider. (2.1)
- Bomyntra is intended for subcutaneous route only and should not be administered intravenously, intramuscularly, or intradermally. (2.1)
- Multiple Myeloma and Bone Metastasis from Solid Tumors: Administer 120 mg every 4 weeks as a subcutaneous injection in the upper arm, upper thigh, or abdomen. (2.2)
- Giant Cell Tumor of Bone: Administer 120 mg every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen. (2.3)
- Administer calcium and vitamin D as necessary to treat or prevent hypocalcemia. (2.2, 2.3)
- Hypercalcemia of Malignancy: Administer 120 mg every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen. (2.4)
1.2 Giant Cell Tumor of Bone
Bomyntra is indicated for the treatment of adults and skeletally mature adolescents with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity [see Clinical Trials (14.2)].
2.3 Giant Cell Tumor of Bone
The recommended dose of Bomyntra is 120 mg administered every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen.
Administer calcium and vitamin D as necessary to treat or prevent hypocalcemia [see Warnings and Precautions (5.3)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Injection: 120 mg/1.7 mL (70 mg/mL) clear, colorless to pale yellow solution in a single-dose vial.
- Injection: 120 mg/1.7 mL (70 mg/mL) clear, colorless to pale yellow solution in a single-dose prefilled syringe.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of denosumab products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hypocalcemia: Severe symptomatic hypocalcemia, including fatal cases [see Contraindications (4.1) and Warnings and Precautions (5.3)].
- Hypercalcemia: Severe symptomatic hypercalcemia following treatment discontinuation can occur [see Adverse Reactions (6) and Warnings and Precautions (5.6)].
- Hypersensitivity, including anaphylactic reactions [see Contraindications (4.2) and Warnings and Precautions (5.2)].
- Musculoskeletal pain, including severe musculoskeletal pain. Positive re-challenge has been reported.
- Lichenoid drug eruptions (e.g., lichen planus-like reactions).
- Alopecia.
14.3 Giant Cell Tumor of Bone
The safety and efficacy of denosumab for the treatment of giant cell tumor of bone in adults or skeletally mature adolescents were demonstrated in two open-label trials [Study 20040215 (NCT00396279) and Study 20062004 (NCT00680992)] that enrolled patients with histologically confirmed measurable giant cell tumor of bone that was either recurrent, unresectable, or for which planned surgery was likely to result in severe morbidity. Patients received 120 mg denosumab subcutaneously every 4 weeks with a loading dose on Days 8 and 15 of the first cycle of therapy. Patients who discontinued denosumab then entered the safety follow-up phase for a minimum of 60 months. Retreatment with denosumab while in safety follow-up was allowed for patients who initially demonstrated a response to denosumab (e.g., in the case of recurrent disease).
Study 20040215 was a single-arm, pharmacodynamic, and proof of concept trial conducted in 37 adult patients with unresectable or recurrent giant cell tumor of bone. Patients were required to have histologically confirmed giant cell tumor of bone and radiologic evidence of measurable disease from a computed tomography (CT) or magnetic resonance imaging (MRI) obtained within 28 days prior to study enrollment. Patients enrolled in Study 20040215 underwent CT or MRI assessment of giant cell tumor of bone at baseline and quarterly during denosumab treatment.
Study 20062004 was a parallel-cohort, proof of concept, and safety trial conducted in 535 adult or skeletally mature adolescent patients with histologically confirmed giant cell tumor of bone and evidence of measurable active disease. Study 20062004 enrolled 19 patients who were 12-16 years of age [see Use in Specific Populations (8.4)]. Patients enrolled into one of three cohorts: Cohort 1 enrolled 268 patients with surgically unsalvageable disease (e.g., sacral or spinal sites of disease, or pulmonary metastases); Cohort 2 enrolled 252 patients with surgically salvageable disease where the investigator determined that the planned surgery was likely to result in severe morbidity (e.g., joint resection, limb amputation, or hemipelvectomy); Cohort 3 enrolled 15 patients who previously participated in Study 20040215. Patients underwent imaging assessment of disease status at intervals determined by their treating physician.
A retrospective interim analysis concluded by an independent review committee evaluated objective response in 187 patients enrolled and treated in Study 20040215 and Study 20062004 for whom baseline and at least one post-baseline radiographic assessment were available (27 of 37 patients enrolled in Study 20040215 and 160 of 270 patients enrolled in Cohorts 1 and 2 of Study 20062004). The primary efficacy outcome measure was objective response rate using Response Evaluation Criteria in Solid Tumors (RECIST) v 1.1.
The overall objective response rate (RECIST 1.1) was 25% (95% CI: 19, 32). All responses were partial responses. The estimated median time to response was 3 months. In the 47 patients with an objective response, the median duration of follow-up was 20 months (range: 2-44 months), and 51% (24/47) had a duration of response lasting at least 8 months. Three patients experienced disease progression following an objective response.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pediatric patients: Recommended only for treatment of skeletally mature adolescents with giant cell tumor of bone. (8.4)
- Renal impairment: Patients with creatinine clearance less than 30 mL/min or receiving dialysis are at risk for hypocalcemia. Adequately supplement with calcium and vitamin D. (8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1.3 Hypercalcemia of Malignancy
Bomyntra is indicated for the treatment of hypercalcemia of malignancy refractory to bisphosphonate therapy.
2.4 Hypercalcemia of Malignancy
The recommended dose of Bomyntra is 120 mg administered every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy. Administer subcutaneously in the upper arm, upper thigh, or abdomen.
14.4 Hypercalcemia of Malignancy
The safety and efficacy of denosumab was demonstrated in an open-label, single-arm trial [Study 20070315 (NCT00896454)] that enrolled 33 patients with hypercalcemia of malignancy (with or without bone metastases) refractory to treatment with intravenous bisphosphonate therapy. Patients received denosumab subcutaneously every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy.
In this trial, refractory hypercalcemia of malignancy was defined as an albumin-corrected calcium of > 12.5 mg/dL (3.1 mmol/L) despite treatment with intravenous bisphosphonate therapy in 7-30 days prior to initiation of denosumab therapy. The primary outcome measure was the proportion of patients achieving a response, defined as corrected serum calcium (CSC) ≤ 11.5 mg/dL (2.9 mmol/L), within 10 days after denosumab administration. Efficacy data are summarized in Figure 1 and Table 4. Concurrent chemotherapy did not appear to affect response to denosumab.
Figure 1. Corrected Serum Calcium by Visit in Responders (Median and Interquartile Range)
N= Number of responders who received ≥ 1 dose of investigational product
n = Number of responders who had no missing data at baseline and the time point of interest
|
N = 33 |
Proportion (%) (95% CI) |
|
| All Responders (CSC ≤ 11.5 mg/dL) by Day 10 | 21 | 63.6 (45.1, 79.6) |
| All Responders by Day 57 | 23 | 69.7 (51.3, 84.4) |
| Complete Responders (CSC ≤ 10.8 mg/dL) by Day 10 | 12 | 36.4 (20.4, 54.9) |
| All Complete Responders by Day 57 | 21 | 63.6 (45.1, 79.6) |
Median time to response (CSC ≤ 11.5 mg/dL) was 9 days (95% CI: 8, 19), and the median duration of response was 104 days (95% CI: 7, not estimable). Median time to complete response (CSC ≤ 10.8 mg/dL) was 23 days (95% CI: 9, 36), and the median duration of complete response was 34 days (95% CI: 1, 134).
2.5 Preparation and Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Bomyntra is a clear, colorless to pale yellow solution that is free from visible particles. Do not use if the solution is discolored or cloudy or if the solution contains particles or foreign particulate matter.
Prior to administration, Bomyntra may be removed from the refrigerator and brought to room temperature up to 25°C (77°F) by standing in the original container. This generally takes 15 to 30 minutes. Do not warm Bomyntra in any other way [see How Supplied/Storage and Handling (16)].
5.4 Osteonecrosis of the Jaw (onj) (5.4 Osteonecrosis of the Jaw (ONJ))
Osteonecrosis of the jaw (ONJ) has been reported in patients receiving denosumab products, manifesting as jaw pain, osteomyelitis, osteitis, bone erosion, tooth or periodontal infection, toothache, gingival ulceration, or gingival erosion. Persistent pain or slow healing of the mouth or jaw after dental surgery may also be manifestations of ONJ. In clinical trials in patients with cancer, the incidence of ONJ was higher with longer duration of exposure [see Adverse Reactions (6.1)]. Seventy-nine percent of patients with ONJ had a history of tooth extraction, poor oral hygiene, or use of a dental appliance as a predisposing factor. Other risk factors for the development of ONJ include immunosuppressive therapy, treatment with angiogenesis inhibitors, systemic corticosteroids, diabetes, and gingival infections. Similarly, for denosumab-treated patients with multiple myeloma that developed ONJ, 58% had a history of invasive dental procedures as a predisposing factor.
Perform an oral examination and appropriate preventive dentistry prior to the initiation of Bomyntra and periodically during Bomyntra therapy. Advise patients regarding oral hygiene practices. Avoid invasive dental procedures during treatment with Bomyntra. Consider temporary discontinuation of Bomyntra therapy if an invasive dental procedure must be performed. There are no data available to suggest the optimal duration of treatment interruption.
Patients who are suspected of having or who develop ONJ while on Bomyntra should receive care by a dentist or an oral surgeon. In these patients, extensive dental surgery to treat ONJ may exacerbate the condition. Clinical judgment of the treating healthcare provider should guide the management plan of each patient based on individual risk/benefit assessment.
14.1 Bone Metastasis From Solid Tumors (14.1 Bone Metastasis from Solid Tumors)
The safety and efficacy of denosumab for the prevention of skeletal-related events in patients with bone metastases from solid tumors was demonstrated in three international, randomized (1:1), double-blind, active-controlled, noninferiority trials comparing denosumab with zoledronic acid. In all three trials, patients were randomized to receive 120 mg denosumab subcutaneously every 4 weeks or 4 mg zoledronic acid intravenously (IV) every 4 weeks (dose adjusted for reduced renal function). Patients with creatinine clearance less than 30 mL/min were excluded. In each trial, the main outcome measure was demonstration of noninferiority of time to first skeletal-related event (SRE) as compared to zoledronic acid. Supportive outcome measures were superiority of time to first SRE and superiority of time to first and subsequent SRE; testing for these outcome measures occurred if the main outcome measure was statistically significant. An SRE was defined as any of the following: pathologic fracture, radiation therapy to bone, surgery to bone, or spinal cord compression.
Study 20050136 (NCT00321464) enrolled 2046 patients with advanced breast cancer and bone metastasis. Randomization was stratified by a history of prior SRE (yes or no), receipt of chemotherapy within 6 weeks prior to randomization (yes or no), prior oral bisphosphonate use (yes or no), and region (Japan or other countries). Forty percent of patients had a previous SRE, 40% received chemotherapy within 6 weeks prior to randomization, 5% received prior oral bisphosphonates, and 7% were enrolled from Japan. Median age was 57 years, 80% of patients were White, and 99% of patients were women. The median number of doses administered was 18 for denosumab and 17 for zoledronic acid.
Study 20050244 (NCT00330759) enrolled 1776 adults with solid tumors other than breast and castrate-resistant prostate cancer with bone metastasis and multiple myeloma. Randomization was stratified by previous SRE (yes or no), systemic anticancer therapy at time of randomization (yes or no), and tumor type (non-small cell lung cancer, myeloma, or other). Eighty-seven percent were receiving systemic anticancer therapy at the time of randomization, 52% had a previous SRE, 64% of patients were men, 87% were White, and the median age was 60 years. A total of 40% of patients had non-small cell lung cancer, 10% had multiple myeloma, 9% had renal cell carcinoma, and 6% had small cell lung cancer. Other tumor types each comprised less than 5% of the enrolled population. The median number of doses administered was 7 for both denosumab and zoledronic acid.
Study 20050103 (NCT00321620) enrolled 1901 men with castrate-resistant prostate cancer and bone metastasis. Randomization was stratified by previous SRE, PSA level (less than 10 ng/mL or 10 ng/mL or greater) and receipt of chemotherapy within 6 weeks prior to randomization (yes or no). Twenty-six percent of patients had a previous SRE, 15% of patients had PSA less than 10 ng/mL, and 14% received chemotherapy within 6 weeks prior to randomization. Median age was 71 years and 86% of patients were White. The median number of doses administered was 13 for denosumab and 11 for zoledronic acid.
Denosumab delayed the time to first SRE following randomization as compared to zoledronic acid in patients with breast or castrate-resistant prostate cancer (CRPC) with osseous metastases (Table 2). In patients with bone metastasis due to other solid tumors or lytic lesions due to multiple myeloma, denosumab was noninferior to zoledronic acid in delaying the time to first SRE following randomization.
Overall survival and progression-free survival were similar between arms in all three trials.
|
a CRPC = castrate-resistant prostate cancer. |
||||||
|
b NR = not reached. |
||||||
|
c Superiority testing performed only after denosumab demonstrated to be noninferior to zoledronic acid within trial. |
||||||
|
d All skeletal events postrandomization; new events defined by occurrence ≥ 21 days after preceding event. |
||||||
|
e Adjusted p-values are presented. |
||||||
| Study 20050136 Metastatic Breast Cancer | Study 20050244 Metastatic Solid Tumors or Multiple Myeloma | Study 20050103 Metastatic CRPC a | ||||
|
Denosumab
N = 1026 |
Zoledronic Acid
N = 1020 |
Denosumab
N = 886 |
Zoledronic Acid
N = 890 |
Denosumab
N = 950 |
Zoledronic Acid
N = 951 |
|
| First On-study SRE | ||||||
| Number of Patients who had SREs (%) | 315 (30.7) | 372 (36.5) | 278 (31.4) | 323 (36.3) | 341 (35.9) | 386 (40.6) |
| Components of First SRE | ||||||
| Radiation to Bone | 82 (8.0) | 119 (11.7) | 119 (13.4) | 144 (16.2) | 177 (18.6) | 203 (21.3) |
| Pathological Fracture | 212 (20.7) | 238 (23.3) | 122 (13.8) | 139 (15.6) | 137 (14.4) | 143 (15.0) |
| Surgery to Bone | 12 (1.2) | 8 (0.8) | 13 (1.5) | 19 (2.1) | 1 (0.1) | 4 (0.4) |
| Spinal Cord Compression | 9 (0.9) | 7 (0.7) | 24 (2.7) | 21 (2.4) | 26 (2.7) | 36 (3.8) |
| Median Time to SRE (months) | NRb | 26.4 | 20.5 | 16.3 | 20.7 | 17.1 |
| Hazard Ratio (95% CI) | 0.82 (0.71, 0.95) | 0.84 (0.71, 0.98) | 0.82 (0.71, 0.95) | |||
| Noninferiority p-value | < 0.001 | < 0.001 | < 0.001 | |||
| Superiority p-valuec | 0.010 | 0.060 | 0.008 | |||
| First and Subsequent SRE d | ||||||
| Mean Number/Patient | 0.46 | 0.60 | 0.44 | 0.49 | 0.52 | 0.61 |
| Rate Ratio (95% CI) | 0.77 (0.66, 0.89) | 0.90 (0.77, 1.04) | 0.82 (0.71, 0.94) | |||
| Superiority p-valuee | 0.001 | 0.145 | 0.009 |
2.1 Important Administration Instructions
Bomyntra should be administered by a healthcare provider.
Bomyntra is intended for subcutaneous route only and should not be administered intravenously, intramuscularly, or intradermally.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Denosumab products are inhibitors of osteoclastic bone resorption via inhibition of RANKL.
Because the biological activity of denosumab in animals is specific to nonhuman primates, evaluation of genetically engineered (knockout) mice or use of other biological inhibitors of the RANK/RANKL pathway, OPG-Fc and RANK-Fc, provided additional information on the pharmacodynamic properties of denosumab. RANK/RANKL knockout mice exhibited absence of lymph node formation, as well as an absence of lactation due to inhibition of mammary gland maturation (lobulo-alveolar gland development during pregnancy). Neonatal RANK/RANKL knockout mice exhibited reduced bone growth and lack of tooth eruption. A corroborative study in 2-week-old rats given the RANKL inhibitor OPG-Fc also showed reduced bone growth, altered growth plates, and impaired tooth eruption. These changes were partially reversible in this model when dosing with the RANKL inhibitors was discontinued.
5.1 Drug Products With Same Active Ingredient (5.1 Drug Products with Same Active Ingredient)
Patients receiving Bomyntra should not receive other denosumab products concomitantly.
8.3 Females and Males of Reproductive Potential
Based on findings in animals and its mechanism of action, denosumab products can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Principal Display Panel – 120 Mg/1.7 Ml Carton Label (Principal Display Panel – 120 mg/1.7 mL Carton Label)
NDC 65219-670-01 Rx only
Scan for additional information
Bomyntra
denosumab-bnht
Injection
120 mg/1.7 mL (70 mg/mL)
For Subcutaneous use only
Sterile Solution - No Preservative
Bomyntra should be administered by a healthcare provider
1 Single-dose vial. Discard unused portion.
FRESENIUS
Kabi
Principal Display Panel – 120 Mg/1.7 Ml Syringe Label (Principal Display Panel – 120 mg/1.7 mL Syringe Label)
NDC 65219-672-01 Rx only
Bomyntra
denosumab-bnht
Injection
120 mg/1.7 mL
(70 mg/mL)
For Subcutaneous use only
Fresenius Kabi
USA, LLC
U.S. License
No. 2146
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity
The carcinogenic potential of denosumab products has not been evaluated in long-term animal studies.
Mutagenicity
The genotoxic potential of denosumab products has not been evaluated.
Impairment of Fertility
Denosumab had no effect on female fertility or male reproductive organs in monkeys at doses that were 6.5- to 25-fold higher than the recommended human dose of 120 mg subcutaneously administered once every 4 weeks, based on body weight (mg/kg).
Carcinogenicity
1.1 Multiple Myeloma and Bone Metastasis From Solid Tumors (1.1 Multiple Myeloma and Bone Metastasis from Solid Tumors)
Bomyntra is indicated for the prevention of skeletal-related events in patients with multiple myeloma and in patients with bone metastases from solid tumors.
2.2 Multiple Myeloma and Bone Metastasis From Solid Tumors (2.2 Multiple Myeloma and Bone Metastasis from Solid Tumors)
The recommended dose of Bomyntra is 120 mg administered as a subcutaneous injection every 4 weeks in the upper arm, upper thigh, or abdomen.
Administer calcium and vitamin D as necessary to treat or prevent hypocalcemia [see Warnings and Precautions (5.3)].
Principal Display Panel – 120 Mg/1.7 Ml Pfs Carton Label (Principal Display Panel – 120 mg/1.7 mL PFS Carton Label)
NDC 65219-672-01 Rx only
Scan for additional information
Bomyntra
denosumab-bnht
Injection
120 mg/1.7 mL (70 mg/mL)
For Subcutaneous use only
Single-dose prefilled syringe.
Discard unused portion
Sterile Solution - No Preservative
Carton contains:
1 Single- dose prefilled syringe
1 Prescribing Information
Bomyntra should be administered by a healthcare provider
FRESENIUS
Kabi
5.5 Atypical Subtrochanteric and Diaphyseal Femoral Fracture
Atypical femoral fracture has been reported with denosumab products [see Adverse Reactions (6.1)]. These fractures can occur anywhere in the femoral shaft from just below the lesser trochanter to above the supracondylar flare and are transverse or short oblique in orientation without evidence of comminution.
Atypical femoral fractures most commonly occur with minimal or no trauma to the affected area. They may be bilateral and many patients report prodromal pain in the affected area, usually presenting as dull, aching thigh pain, weeks to months before a complete fracture occurs. A number of reports note that patients were also receiving treatment with glucocorticoids (e.g. prednisone) at the time of fracture.
During Bomyntra treatment, patients should be advised to report new or unusual thigh, hip, or groin pain. Any patient who presents with thigh or groin pain should be suspected of having an atypical fracture and should be evaluated to rule out an incomplete femur fracture. Patient presenting with an atypical femur fracture should also be assessed for symptoms and signs of fracture in the contralateral limb. Interruption of Bomyntra therapy should be considered, pending a risk/benefit assessment, on an individual basis.
5.7 Multiple Vertebral Fractures (mvf) Following Treatment Discontinuation (5.7 Multiple Vertebral Fractures (MVF) Following Treatment Discontinuation)
Multiple vertebral fractures (MVF) have been reported following discontinuation of treatment with denosumab products. Patients at higher risk for MVF include those with risk factors for or a history of osteoporosis or prior fractures.
When Bomyntra treatment is discontinued, evaluate the individual patient's risk for vertebral fractures [see Patient Counseling Information (17)].
5.6 Hypercalcemia Following Treatment Discontinuation in Patients With Giant Cell Tumor of Bone and in Patients With Growing Skeletons (5.6 Hypercalcemia Following Treatment Discontinuation in Patients with Giant Cell Tumor of Bone and in Patients with Growing Skeletons)
Clinically significant hypercalcemia requiring hospitalization and complicated by acute renal injury has been reported in denosumab product-treated patients with giant cell tumor of bone and patients with growing skeletons. Hypercalcemia has been reported within the first year after treatment discontinuation. After treatment is discontinued, monitor patients for signs and symptoms of hypercalcemia, assess serum calcium periodically, reevaluate the patient's calcium and vitamin D supplementation requirements and manage patients as clinically appropriate [see Adverse Reactions (6) and Use in Specific Populations (8.4)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:20.336736 · Updated: 2026-03-14T22:40:03.259578