These Highlights Do Not Include All The Information Needed To Use Panretin Gel Safely And Effectively. See Full Prescribing Information For Panretin Gel.
49c16717-7d86-4257-80c9-baa1417e5555
34391-3
Human Prescription Drug Label
Drug Facts
Composition & Product
Identifiers & Packaging
Description
1.1 Kaposi’s Sarcoma PANRETIN GEL is indicated for topical treatment of cutaneous lesions in adults with AIDS related Kaposi’s sarcoma (KS). Limitations of Use: PANRETIN GEL is not indicated when systemic anti-KS therapy is required (including more than 10 new KS lesions in the prior month, symptomatic lymphedema, symptomatic pulmonary KS, or symptomatic visceral involvement) [see Clinical Studies ( 14.1 )] .
Indications and Usage
1.1 Kaposi’s Sarcoma PANRETIN GEL is indicated for topical treatment of cutaneous lesions in adults with AIDS related Kaposi’s sarcoma (KS). Limitations of Use: PANRETIN GEL is not indicated when systemic anti-KS therapy is required (including more than 10 new KS lesions in the prior month, symptomatic lymphedema, symptomatic pulmonary KS, or symptomatic visceral involvement) [see Clinical Studies ( 14.1 )] .
Dosage and Administration
PANRETIN GEL is for topical use only. Do not use occlusive dressings with PANRETIN GEL. Apply PANRETIN GEL twice daily to coat the entire cutaneous Kaposi sarcoma lesions. Gradually increase the application frequency up to four (4) times a day as tolerated. Continue PANRETIN GEL as long as patient is deriving benefit. Reduce application frequency for application site toxicity. Interrupt treatment for severe irritation; may resume at a reduced application frequency once symptoms improve. Avoid application of gel to normal skin and do not apply on or near mucosal surfaces. Wash hands after application unless gel is applied to Kaposi sarcoma lesions on the hands. Allow gel to dry for three to five minutes before covering with clothing.
Warnings and Precautions
• Embryo-Fetal Toxicity : Can cause fetal harm. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use an effective method of contraception. ( 5.1 . 8.1 , 8.3 ) • Photosensitivity : Minimize exposure to sunlight and sunlamps. ( 5.2 ) • DEET toxicity : Do not use DEET-containing products ( 5.3 )
Contraindications
PANRETIN GEL is contraindicated in patients with a known hypersensitivity to retinoids or to any of the ingredients of the product.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Photosensitivity [see Warnings and Precautions ( 5.2 )]
Storage and Handling
PANRETIN GEL is a clear yellow gel and is supplied in a 60-gram tubes containing 0.1% alitretinoin. Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. PANRETIN GEL is flammable; keep away from heat or flame.
How Supplied
PANRETIN GEL is a clear yellow gel and is supplied in a 60-gram tubes containing 0.1% alitretinoin. Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. PANRETIN GEL is flammable; keep away from heat or flame.
Medication Information
Warnings and Precautions
• Embryo-Fetal Toxicity : Can cause fetal harm. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use an effective method of contraception. ( 5.1 . 8.1 , 8.3 ) • Photosensitivity : Minimize exposure to sunlight and sunlamps. ( 5.2 ) • DEET toxicity : Do not use DEET-containing products ( 5.3 )
Indications and Usage
1.1 Kaposi’s Sarcoma PANRETIN GEL is indicated for topical treatment of cutaneous lesions in adults with AIDS related Kaposi’s sarcoma (KS). Limitations of Use: PANRETIN GEL is not indicated when systemic anti-KS therapy is required (including more than 10 new KS lesions in the prior month, symptomatic lymphedema, symptomatic pulmonary KS, or symptomatic visceral involvement) [see Clinical Studies ( 14.1 )] .
Dosage and Administration
PANRETIN GEL is for topical use only. Do not use occlusive dressings with PANRETIN GEL. Apply PANRETIN GEL twice daily to coat the entire cutaneous Kaposi sarcoma lesions. Gradually increase the application frequency up to four (4) times a day as tolerated. Continue PANRETIN GEL as long as patient is deriving benefit. Reduce application frequency for application site toxicity. Interrupt treatment for severe irritation; may resume at a reduced application frequency once symptoms improve. Avoid application of gel to normal skin and do not apply on or near mucosal surfaces. Wash hands after application unless gel is applied to Kaposi sarcoma lesions on the hands. Allow gel to dry for three to five minutes before covering with clothing.
Contraindications
PANRETIN GEL is contraindicated in patients with a known hypersensitivity to retinoids or to any of the ingredients of the product.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Photosensitivity [see Warnings and Precautions ( 5.2 )]
Storage and Handling
PANRETIN GEL is a clear yellow gel and is supplied in a 60-gram tubes containing 0.1% alitretinoin. Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. PANRETIN GEL is flammable; keep away from heat or flame.
How Supplied
PANRETIN GEL is a clear yellow gel and is supplied in a 60-gram tubes containing 0.1% alitretinoin. Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. PANRETIN GEL is flammable; keep away from heat or flame.
Description
1.1 Kaposi’s Sarcoma PANRETIN GEL is indicated for topical treatment of cutaneous lesions in adults with AIDS related Kaposi’s sarcoma (KS). Limitations of Use: PANRETIN GEL is not indicated when systemic anti-KS therapy is required (including more than 10 new KS lesions in the prior month, symptomatic lymphedema, symptomatic pulmonary KS, or symptomatic visceral involvement) [see Clinical Studies ( 14.1 )] .
Section 42229-5
|
PATIENT INFORMATION PANRETIN GEL (PAN-reh-tin) (alitretinoin) |
| Important information: PANRETIN GEL is for use on skin (topical use) only. Do not use PANRETIN GEL in or near your eyes, inside your nose, mouth, lips, vagina, tip of the penis, rectum, or anus. |
|
What is PANRETIN GEL?
PANRETIN GEL is a prescription medicine used to treat skin lesions in adults with AIDS-related Kaposi’s sarcoma (KS). PANRETIN GEL is not used to treat KS when a medicine taken by mouth or injection (systemic treatment) is needed. It is not known if PANRETIN GEL is safe and effective in children. |
|
Do not use PANRETIN GEL if you are allergic to retinoids or any of the ingredients in PANRETIN GEL. See the end of this Patient Information leaflet for a complete list of ingredients in PANRETIN GEL. |
|
Before using PANRETIN GEL, tell your healthcare provider about all of your medical conditions, including if you:
• are pregnant or plan to become pregnant. PANRETIN GEL can harm your unborn baby. Tell your healthcare provider right away if you become pregnant or think you may be pregnant during treatment with PANRETIN GEL. Females who are able to become pregnant: o Your healthcare provider should check to see if you are pregnant before you start treatment with PANRETIN GEL. o You should use effective birth control (contraception) during treatment and for 1 week after the last dose of PANRETIN GEL. Males with female partners who are able to become pregnant: o You should use effective birth control (contraception) during treatment and for 1 week after the last dose of PANRETIN GEL. • are breastfeeding or plan to breastfeed. It is not known if PANRETIN GEL passes into your breast milk. Do not breastfeed during treatment and for 1 week after your last dose of PANRETIN GEL. Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider about skin products that you use. You should not use products that contain DEET (N, N-diethyl-m-toluamide), during treatment with PANRETIN Gel. DEET is an ingredient commonly found in insect repellent. |
How should I use PANRETIN GEL?
|
What should I avoid while using PANRETIN GEL?
|
|
What are the possible side effects of PANRETIN GEL? The most common side effects of PANRETIN GEL at the treatment site, include:
Tell your healthcare provider about any side effect that bothers you or that does not go away. These are not all of the possible side effects of PANRETIN GEL. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
How should I store PANRETIN GEL?
|
|
General information about the safe and effective use of PANRETIN GEL.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use PANRETIN GEL for a condition for which it was not prescribed. Do not give PANRETIN GEL to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about PANRETIN GEL that is written for health professionals. |
|
What are the ingredients in PANRETIN GEL?
Active ingredient: alitretinoin Inactive ingredients: butylated hydroxytoluene NF, dehydrated alcohol USP, hydroxypropyl cellulose NF, and polyethylene glycol 400 NF. Manufactured for: Advanz Pharma (US) Corp. Bannockburn IL, 60015 PANRETIN® is a registered trademark of Mercury Pharma Group Limited. Distributed by Advanz Pharma (US) Corp. under license. |
This patient information has been approved by the U.S. Food and Drug Administration Revise 11/2025
8.1 Pregnancy
Risk Summary
Based on findings in animal studies and its mechanism of action, PANRETIN GEL can cause fetal harm when administered to a pregnant woman. Oral administration of alitretinoin to pregnant animals during the period of organogenesis was teratogenic and embryo lethal at exposures at least 5 times the estimated daily human topical dose (see Data). There are no data on the use of PANRETIN GEL in pregnant women. Advise pregnant women of the potential risk to the fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Oral administration of alitretinoin to pregnant rabbits during the period of organogenesis resulted in early resorptions, post-implantation loss, and fetal defects (limb, craniofacial, fused sternebrae) at doses ≥ 0.5 mg/kg/day (approximately 5 times the estimated daily human topical dose based on body surface area, assuming complete systemic absorption of alitretinoin, when PANRETIN GEL is administered as a 60 g tube over 1 month in a 60 kg human). Early resorptions and post-implantation loss also occurred in rats administered oral alitretinoin at doses ≥5 mg/kg/day (approximately 25 times the estimated daily human topical dose based on body surface area). Limb and craniofacial defects also occurred in mice administered oral alitretinoin on day 11 of gestation at single doses ≥50 mg/kg (approximately 127 times the estimated daily human topical dose based on body surface area).
8.2 Lactation
Risk Summary
It is not known whether alitretinoin or its metabolites are excreted in human milk. Because many drugs are excreted in human milk and because of the potential for adverse reactions from PANRETIN GEL in the nursing child, advise patients that breastfeeding is not recommended during treatment with PANRETIN GEL and for 1 week after the last dose.
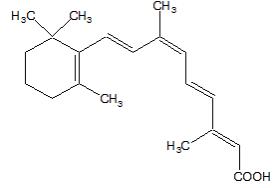
11 Description
PANRETIN GEL is a retinoid. PANRETIN GEL 0.1% contains alitretinoin and is intended for topical application only. The chemical name is (2E,4E,6Z,8E)-3,7-Dimethyl-9-(2,6,6-trimethyl-1-cyclohexen-1-yl)-2,4,6,8-nonatetraenoic acid, also known as 9-cis-retinoic acid. Chemically, alitretinoin is related to vitamin A. It is a yellow powder with a molecular weight of 300.44 and a molecular formula of C20H28O2. It is slightly soluble in ethanol (7.01 mg/g at 25℃) and insoluble in water. The structural formula of alitretinoin is as follows:
PANRETIN GEL is a clear, yellow gel containing 0.1% (w/w) alitretinoin. Each gram of PANRETIN GEL contains 1 mg alitretinoin. PANRETIN GEL contains the following inactive ingredients: butylated hydroxytoluene NF, dehydrated alcohol USP 92%, hydroxypropyl cellulose NF, and polyethylene glycol 400 NF.
8.4 Pediatric Use
The safety and effectiveness of PANRETIN GEL have not been established in pediatric patients.
8.5 Geriatric Use
Clinical studies of PANRETIN GEL did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger adult patients.
14 Clinical Studies
14.1 Kaposi’s Sarcoma
PANRETIN GEL was evaluated in two multicenter, randomized, double-blind, vehicle-controlled studies in adult patients with cutaneous lesions of AIDS-related KS. In both studies the primary efficacy objective was the patients’ cutaneous KS tumor response rate through 12 weeks of study drug treatment which was assessed by evaluating from 3 to 8 KS index lesions according to the modified AIDS Clinical Trials Group (ACTG) response criteria as applied to topical therapy (i.e., evaluation of height and area reductions of the index lesions only; progressive disease in non-index lesions and new lesions were not considered progressive disease; progressive disease was scored only in the treated index lesions). A global evaluation by physicians was also carried out. It considered all the patient’s treated lesions (index and other) compared to baseline. In this evaluation, patients with at least a 50% improvement in the KS lesions were considered responders. In addition, photographs of lesions in patients considered responders by the modified ACTG criteria were examined by the FDA for a cosmetically beneficial response, defined as at least a 50% improvement in appearance compared to baseline, considering both the KS lesions and dermal toxicity at the lesion site, in at least 50% of the index lesions and maintained for at least 3 weeks. Visceral disease was not monitored in these trials and the appearance of new KS lesions was not considered part of the response assessment.
In Trial 1, a total of 268 patients were entered from centers in the U.S. and Canada. Patients were treated topically three to four times a day with either PANRETIN GEL or a matching vehicle gel for a minimum of 12 weeks, followed by an open-label phase in patients who had not yet progressed on PANRETIN GEL. Median age of patients was 39 years, 99% were men, 75% were White, 16% Hispanic, and 7% Black. Fifty-seven percent of patients had a CD4+ lymphocyte count <200/mm3 and 36% had CD4+ lymphocyte count less than 100/mm3; 12% had visceral KS. Responses during the double-blind phase are shown in Table 2. New lesions in untreated areas were seen in about 50% of patients.
Trial 2 was an international study with a planned enrollment of 270 patients. Patients were treated topically twice a day with PANRETIN GEL or a matching vehicle for 12 weeks. The study was stopped early because of positive-interim results in the initial 82 patients. Median age of patients was 36 years for PANRETIN-GEL and 39 years for matching vehicle; all patients were men; 89% White; 5% Hispanic, and 4% Black; 67% had a CD4+ lymphocyte count ≤200/mm3 and 39% had CD4+ lymphocyte count ≤100/mm3; and 16% had visceral KS.
Results of the study are shown in Table 2. Responses to PANRETIN GEL were seen both in previously untreated patients and in patients with prior systemic and/or topical KS treatment.
Table 2: Response Rates
|
Trial 1 |
Trial 2 |
|||
|
PANRETIN GEL N=134 |
Vehicle Gel N=134 |
PANRETIN GEL N=36 |
Vehicle Gel N=45 |
|
|
Modified ACTG Response |
35%* |
16% |
36% |
7% |
|
P = 0.0012 |
|
|||
|
Physician’s Global Subjective Assessment (all treated lesions) |
19% |
4% |
47% |
11% |
|
P = 0.00014 |
|
|||
|
Photograph Response |
15% |
4% |
19% |
2% |
|
P = 0.0026 |
|
*All responses were partial responses except 1% complete response for modified ACTG response in Trial 1.
In the clinical trials, the cumulative percentage of patients who experienced a response was less than 1% at 2 weeks, 10% at 4 weeks, and 28% at 8 weeks. Photographs of some patients revealed an erythematous and edematous response, leading to a cosmetically mixed outcome.
4 Contraindications
PANRETIN GEL is contraindicated in patients with a known hypersensitivity to retinoids or to any of the ingredients of the product.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
• Photosensitivity [see Warnings and Precautions (5.2)]
5.2 Photosensitivity
Retinoids as a class have been associated with photosensitivity. Advise patients to minimize exposure of treated areas to sunlight and sunlamps during the use of PANRETIN GEL.
12.2 Pharmacodynamics
Alitretinoin exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
The range of 9-cis-retinoic acid plasma concentrations in patients with cutaneous lesions of AIDS-related KS after multiple daily applications of PANRETIN GEL for up to 60 weeks was similar to the range of circulating, naturally occurring 9-cis-retinoic acid plasma concentrations in untreated healthy participants.
Elimination
Metabolism
9-cis-retinoic acid is metabolized to 4-hydroxy-9-cis-retinoic acid and 4-oxo-9-cis-retinoic acid by CYP2C9, 3A4, 1A1, and 1A2. 4-oxo-9-cis-retinoic acid is the major circulating metabolite following oral administration of 9-cis-retinoic acid.
1 Indications and Usage
1.1 Kaposi’s Sarcoma
PANRETIN GEL is indicated for topical treatment of cutaneous lesions in adults with AIDS related Kaposi’s sarcoma (KS).
Limitations of Use: PANRETIN GEL is not indicated when systemic anti-KS therapy is required (including more than 10 new KS lesions in the prior month, symptomatic lymphedema, symptomatic pulmonary KS, or symptomatic visceral involvement) [see Clinical Studies (14.1)].
12.1 Mechanism of Action
Alitretinoin (9-cis-retinoic acid) is a naturally occurring endogenous retinoid that binds to and activates all known intracellular retinoid receptor subtypes (RARα, RARβ, RARγ, RXRα, RXRβ and RXRγ). Once activated these receptors function as transcription factors that regulate the expression of genes that control the process of cellular differentiation and proliferation in both normal and neoplastic cells. Alitretinoin inhibits the growth of Kaposi’s sarcoma (KS) cells in vitro.
5.1 Embryo Fetal Toxicity
Based on data from animal studies and its mechanism of action, PANRETIN GEL can cause fetal harm when administered to a pregnant woman. Oral administration of alitretinoin to pregnant animals during the period of organogenesis was teratogenic and embryo-lethal at exposures 5 times the estimated daily human topical dose. Advise women of the potential risk to a fetus. Advise women of reproductive potential to use effective contraception during treatment with PANRETIN GEL and for 1 week after the last dose [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions
• Embryo-Fetal Toxicity: Can cause fetal harm. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use an effective method of contraception. (5.1. 8.1,8.3)
• Photosensitivity: Minimize exposure to sunlight and sunlamps. (5.2)
• DEET toxicity: Do not use DEET-containing products (5.3)
2 Dosage and Administration
PANRETIN GEL is for topical use only.
Do not use occlusive dressings with PANRETIN GEL.
- Apply PANRETIN GEL twice daily to coat the entire cutaneous Kaposi sarcoma lesions.
- Gradually increase the application frequency up to four (4) times a day as tolerated.
- Continue PANRETIN GEL as long as patient is deriving benefit.
- Reduce application frequency for application site toxicity. Interrupt treatment for severe irritation; may resume at a reduced application frequency once symptoms improve.
- Avoid application of gel to normal skin and do not apply on or near mucosal surfaces.
- Wash hands after application unless gel is applied to Kaposi sarcoma lesions on the hands.
- Allow gel to dry for three to five minutes before covering with clothing.
3 Dosage Forms and Strengths
Topical Gel: 0.1% alitretinoin (clear yellow gel) in a 60-gram tube.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of PANRETIN GEL was assessed in two multicenter, prospective, randomized, double-blind, vehicle-controlled trials (Trial 1 and Trial 2) in patients with cutaneous lesions of AIDS-related KS [see Clinical Studies (14.1)]. In a pooled analysis of both trials, the most common adverse reactions in ≥ 5% of patients were rash, pain, paresthesia, pruritis, exfoliative dermatitis, edema, and skin disorder.
In Trial 1, severe local skin adverse reactions (erythema and edema with or without vesiculation) occurred in 10% of patients during the first 12 weeks of treatment (versus 0% in the vehicle control). Adverse reactions led to withdrawal from the study in 7% of patients.
In Trial 2, severe local skin adverse reactions (erythema and edema with or without vesiculation) occurred in 6% of patients during the first 12 weeks of treatment (versus 0% in the vehicle control) and 1 patient withdrew due to severe skin irritation.
Table 1 lists the most common application site adverse reactions that occurred in a least 5% of patients during the double-blind phase who received PANRETIN GEL in either of the two controlled studies.
TABLE 1: Adverse Reactions at Application Site in Trial 1 and 2 in ≥ 5% of Patients Treated with PANRETIN GEL
|
Adverse Event Term |
Trial 1 |
Trial 2 |
||
|
|
PANRETIN GEL N=134 % |
Vehicle Gel N=134 % |
PANRETIN GEL N=36 % |
Vehicle Gel N=46 % |
|
Rash1 |
77 |
11 |
25 |
4 |
|
Pain2 |
34 |
7 |
0 |
4 |
|
Pruritus3 |
11 |
4 |
8 |
4 |
|
Exfoliative dermatitis4 |
9 |
2 |
3 |
0 |
|
Skin disorder5 |
8 |
1 |
0 |
0 |
|
Edema6 |
8 |
3 |
3 |
0 |
|
Parethesia7 |
3 |
0 |
22 |
7 |
| Includes Investigator terms: 1 Erythema, scaling, irritation, redness, rash, dermatitis 2 Burning, pain 3 Itching, pruritus 4 Flaking, peeling, desquamation, exfoliation 5 Excoriation, cracking, scab, crusting, drainage, eschar, fissure or oozing 6 Edema, swelling, inflammation 7 Stinging, tingling |
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Embryo-Fetal Toxicity
Advise females to inform their healthcare provider if they are pregnant or become pregnant. Inform females of the risk to a fetus and potential loss of pregnancy [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
Advise females of reproductive potential to use effective contraception during treatment and for 1 week after the last dose of PANRETIN GEL [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
Advise males with female partners of reproductive potential to use effective contraception during treatment and for 1 week following the last dose of PANRETIN GEL [see Warnings and Precautions (5.1) and Use in Specific Populations (8.3)].
Lactation
Advise patients not to breastfeed during treatment with PANRETIN GEL and for 1 week after the last dose of PANRETIN GEL [see Use in Specific Populations (8.2)].
Photosensitivity
Advice patients that PANRETIN GEL can cause photosensitivity and to avoid sunlight and sunlamps and use measures such as protective clothing in PANRETIN-applied areas [see Warnings and Precautions (5.2)].
Toxicity with DEET-Containing Products
Advise patients to avoid DEET-containing products such as insect repellant while using PANRETIN GEL [see Warnings and Precautions (5.3)].
Manufactured for:
Advanz Pharma (US) Corp.
Bannockburn IL, 60015
PANRETIN® is a registered trademark of Mercury Pharma Group Limited. Distributed by Advanz Pharma (US) Corp. under license
Revised: 11/2025
16 How Supplied/storage and Handling
PANRETIN GEL is a clear yellow gel and is supplied in a 60-gram tubes containing 0.1% alitretinoin.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
PANRETIN GEL is flammable; keep away from heat or flame.
Package Label.principal Display Panel
NDC 59212-601-22
Panretin® gel
(alitretinoin) 0.1%
5.3 Toxicity With Deet Containing Products
Animal toxicology studies showed increased DEET toxicity when DEET was included as part of the formulation. Advise patients to not use PANRETIN GEL concurrently with products that contain DEET (N,N-diethyl-m-toluamide), a common component of insect repellent products.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Verify pregnancy status of females of reproductive potential prior to initiating PANRETIN GEL.
Contraception
Females
PANRETIN GEL can cause embryo-fetal harm when administered to pregnant women [see Use in Specific Populations (8.1)]. Advise females of reproductive potential to use effective contraception during treatment with PANRETIN GEL and for 1 week after the last dose.
Males
Advise males with female partners of reproductive potential to use effective contraception during treatment with PANRETIN GEL and for 1 week after the last dose.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with alitretinoin have not been conducted. Alitretinoin was not mutagenic in vitro (bacterial assay, Chinese hamster ovary cell HGPRT mutation assay) and was not clastogenic in vitro (chromosome aberration test in human lymphocytes) or in vivo (mouse micronucleus test).
Structured Label Content
Section 42229-5 (42229-5)
|
PATIENT INFORMATION PANRETIN GEL (PAN-reh-tin) (alitretinoin) |
| Important information: PANRETIN GEL is for use on skin (topical use) only. Do not use PANRETIN GEL in or near your eyes, inside your nose, mouth, lips, vagina, tip of the penis, rectum, or anus. |
|
What is PANRETIN GEL?
PANRETIN GEL is a prescription medicine used to treat skin lesions in adults with AIDS-related Kaposi’s sarcoma (KS). PANRETIN GEL is not used to treat KS when a medicine taken by mouth or injection (systemic treatment) is needed. It is not known if PANRETIN GEL is safe and effective in children. |
|
Do not use PANRETIN GEL if you are allergic to retinoids or any of the ingredients in PANRETIN GEL. See the end of this Patient Information leaflet for a complete list of ingredients in PANRETIN GEL. |
|
Before using PANRETIN GEL, tell your healthcare provider about all of your medical conditions, including if you:
• are pregnant or plan to become pregnant. PANRETIN GEL can harm your unborn baby. Tell your healthcare provider right away if you become pregnant or think you may be pregnant during treatment with PANRETIN GEL. Females who are able to become pregnant: o Your healthcare provider should check to see if you are pregnant before you start treatment with PANRETIN GEL. o You should use effective birth control (contraception) during treatment and for 1 week after the last dose of PANRETIN GEL. Males with female partners who are able to become pregnant: o You should use effective birth control (contraception) during treatment and for 1 week after the last dose of PANRETIN GEL. • are breastfeeding or plan to breastfeed. It is not known if PANRETIN GEL passes into your breast milk. Do not breastfeed during treatment and for 1 week after your last dose of PANRETIN GEL. Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider about skin products that you use. You should not use products that contain DEET (N, N-diethyl-m-toluamide), during treatment with PANRETIN Gel. DEET is an ingredient commonly found in insect repellent. |
How should I use PANRETIN GEL?
|
What should I avoid while using PANRETIN GEL?
|
|
What are the possible side effects of PANRETIN GEL? The most common side effects of PANRETIN GEL at the treatment site, include:
Tell your healthcare provider about any side effect that bothers you or that does not go away. These are not all of the possible side effects of PANRETIN GEL. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
How should I store PANRETIN GEL?
|
|
General information about the safe and effective use of PANRETIN GEL.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use PANRETIN GEL for a condition for which it was not prescribed. Do not give PANRETIN GEL to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about PANRETIN GEL that is written for health professionals. |
|
What are the ingredients in PANRETIN GEL?
Active ingredient: alitretinoin Inactive ingredients: butylated hydroxytoluene NF, dehydrated alcohol USP, hydroxypropyl cellulose NF, and polyethylene glycol 400 NF. Manufactured for: Advanz Pharma (US) Corp. Bannockburn IL, 60015 PANRETIN® is a registered trademark of Mercury Pharma Group Limited. Distributed by Advanz Pharma (US) Corp. under license. |
This patient information has been approved by the U.S. Food and Drug Administration Revise 11/2025
8.1 Pregnancy
Risk Summary
Based on findings in animal studies and its mechanism of action, PANRETIN GEL can cause fetal harm when administered to a pregnant woman. Oral administration of alitretinoin to pregnant animals during the period of organogenesis was teratogenic and embryo lethal at exposures at least 5 times the estimated daily human topical dose (see Data). There are no data on the use of PANRETIN GEL in pregnant women. Advise pregnant women of the potential risk to the fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Oral administration of alitretinoin to pregnant rabbits during the period of organogenesis resulted in early resorptions, post-implantation loss, and fetal defects (limb, craniofacial, fused sternebrae) at doses ≥ 0.5 mg/kg/day (approximately 5 times the estimated daily human topical dose based on body surface area, assuming complete systemic absorption of alitretinoin, when PANRETIN GEL is administered as a 60 g tube over 1 month in a 60 kg human). Early resorptions and post-implantation loss also occurred in rats administered oral alitretinoin at doses ≥5 mg/kg/day (approximately 25 times the estimated daily human topical dose based on body surface area). Limb and craniofacial defects also occurred in mice administered oral alitretinoin on day 11 of gestation at single doses ≥50 mg/kg (approximately 127 times the estimated daily human topical dose based on body surface area).
8.2 Lactation
Risk Summary
It is not known whether alitretinoin or its metabolites are excreted in human milk. Because many drugs are excreted in human milk and because of the potential for adverse reactions from PANRETIN GEL in the nursing child, advise patients that breastfeeding is not recommended during treatment with PANRETIN GEL and for 1 week after the last dose.
11 Description (11 DESCRIPTION)
PANRETIN GEL is a retinoid. PANRETIN GEL 0.1% contains alitretinoin and is intended for topical application only. The chemical name is (2E,4E,6Z,8E)-3,7-Dimethyl-9-(2,6,6-trimethyl-1-cyclohexen-1-yl)-2,4,6,8-nonatetraenoic acid, also known as 9-cis-retinoic acid. Chemically, alitretinoin is related to vitamin A. It is a yellow powder with a molecular weight of 300.44 and a molecular formula of C20H28O2. It is slightly soluble in ethanol (7.01 mg/g at 25℃) and insoluble in water. The structural formula of alitretinoin is as follows:
PANRETIN GEL is a clear, yellow gel containing 0.1% (w/w) alitretinoin. Each gram of PANRETIN GEL contains 1 mg alitretinoin. PANRETIN GEL contains the following inactive ingredients: butylated hydroxytoluene NF, dehydrated alcohol USP 92%, hydroxypropyl cellulose NF, and polyethylene glycol 400 NF.
8.4 Pediatric Use
The safety and effectiveness of PANRETIN GEL have not been established in pediatric patients.
8.5 Geriatric Use
Clinical studies of PANRETIN GEL did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger adult patients.
14 Clinical Studies (14 CLINICAL STUDIES)
14.1 Kaposi’s Sarcoma
PANRETIN GEL was evaluated in two multicenter, randomized, double-blind, vehicle-controlled studies in adult patients with cutaneous lesions of AIDS-related KS. In both studies the primary efficacy objective was the patients’ cutaneous KS tumor response rate through 12 weeks of study drug treatment which was assessed by evaluating from 3 to 8 KS index lesions according to the modified AIDS Clinical Trials Group (ACTG) response criteria as applied to topical therapy (i.e., evaluation of height and area reductions of the index lesions only; progressive disease in non-index lesions and new lesions were not considered progressive disease; progressive disease was scored only in the treated index lesions). A global evaluation by physicians was also carried out. It considered all the patient’s treated lesions (index and other) compared to baseline. In this evaluation, patients with at least a 50% improvement in the KS lesions were considered responders. In addition, photographs of lesions in patients considered responders by the modified ACTG criteria were examined by the FDA for a cosmetically beneficial response, defined as at least a 50% improvement in appearance compared to baseline, considering both the KS lesions and dermal toxicity at the lesion site, in at least 50% of the index lesions and maintained for at least 3 weeks. Visceral disease was not monitored in these trials and the appearance of new KS lesions was not considered part of the response assessment.
In Trial 1, a total of 268 patients were entered from centers in the U.S. and Canada. Patients were treated topically three to four times a day with either PANRETIN GEL or a matching vehicle gel for a minimum of 12 weeks, followed by an open-label phase in patients who had not yet progressed on PANRETIN GEL. Median age of patients was 39 years, 99% were men, 75% were White, 16% Hispanic, and 7% Black. Fifty-seven percent of patients had a CD4+ lymphocyte count <200/mm3 and 36% had CD4+ lymphocyte count less than 100/mm3; 12% had visceral KS. Responses during the double-blind phase are shown in Table 2. New lesions in untreated areas were seen in about 50% of patients.
Trial 2 was an international study with a planned enrollment of 270 patients. Patients were treated topically twice a day with PANRETIN GEL or a matching vehicle for 12 weeks. The study was stopped early because of positive-interim results in the initial 82 patients. Median age of patients was 36 years for PANRETIN-GEL and 39 years for matching vehicle; all patients were men; 89% White; 5% Hispanic, and 4% Black; 67% had a CD4+ lymphocyte count ≤200/mm3 and 39% had CD4+ lymphocyte count ≤100/mm3; and 16% had visceral KS.
Results of the study are shown in Table 2. Responses to PANRETIN GEL were seen both in previously untreated patients and in patients with prior systemic and/or topical KS treatment.
Table 2: Response Rates
|
Trial 1 |
Trial 2 |
|||
|
PANRETIN GEL N=134 |
Vehicle Gel N=134 |
PANRETIN GEL N=36 |
Vehicle Gel N=45 |
|
|
Modified ACTG Response |
35%* |
16% |
36% |
7% |
|
P = 0.0012 |
|
|||
|
Physician’s Global Subjective Assessment (all treated lesions) |
19% |
4% |
47% |
11% |
|
P = 0.00014 |
|
|||
|
Photograph Response |
15% |
4% |
19% |
2% |
|
P = 0.0026 |
|
*All responses were partial responses except 1% complete response for modified ACTG response in Trial 1.
In the clinical trials, the cumulative percentage of patients who experienced a response was less than 1% at 2 weeks, 10% at 4 weeks, and 28% at 8 weeks. Photographs of some patients revealed an erythematous and edematous response, leading to a cosmetically mixed outcome.
4 Contraindications (4 CONTRAINDICATIONS)
PANRETIN GEL is contraindicated in patients with a known hypersensitivity to retinoids or to any of the ingredients of the product.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
• Photosensitivity [see Warnings and Precautions (5.2)]
5.2 Photosensitivity
Retinoids as a class have been associated with photosensitivity. Advise patients to minimize exposure of treated areas to sunlight and sunlamps during the use of PANRETIN GEL.
12.2 Pharmacodynamics
Alitretinoin exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
The range of 9-cis-retinoic acid plasma concentrations in patients with cutaneous lesions of AIDS-related KS after multiple daily applications of PANRETIN GEL for up to 60 weeks was similar to the range of circulating, naturally occurring 9-cis-retinoic acid plasma concentrations in untreated healthy participants.
Elimination
Metabolism
9-cis-retinoic acid is metabolized to 4-hydroxy-9-cis-retinoic acid and 4-oxo-9-cis-retinoic acid by CYP2C9, 3A4, 1A1, and 1A2. 4-oxo-9-cis-retinoic acid is the major circulating metabolite following oral administration of 9-cis-retinoic acid.
1 Indications and Usage (1 INDICATIONS AND USAGE)
1.1 Kaposi’s Sarcoma
PANRETIN GEL is indicated for topical treatment of cutaneous lesions in adults with AIDS related Kaposi’s sarcoma (KS).
Limitations of Use: PANRETIN GEL is not indicated when systemic anti-KS therapy is required (including more than 10 new KS lesions in the prior month, symptomatic lymphedema, symptomatic pulmonary KS, or symptomatic visceral involvement) [see Clinical Studies (14.1)].
12.1 Mechanism of Action
Alitretinoin (9-cis-retinoic acid) is a naturally occurring endogenous retinoid that binds to and activates all known intracellular retinoid receptor subtypes (RARα, RARβ, RARγ, RXRα, RXRβ and RXRγ). Once activated these receptors function as transcription factors that regulate the expression of genes that control the process of cellular differentiation and proliferation in both normal and neoplastic cells. Alitretinoin inhibits the growth of Kaposi’s sarcoma (KS) cells in vitro.
5.1 Embryo Fetal Toxicity (5.1 Embryo-Fetal Toxicity)
Based on data from animal studies and its mechanism of action, PANRETIN GEL can cause fetal harm when administered to a pregnant woman. Oral administration of alitretinoin to pregnant animals during the period of organogenesis was teratogenic and embryo-lethal at exposures 5 times the estimated daily human topical dose. Advise women of the potential risk to a fetus. Advise women of reproductive potential to use effective contraception during treatment with PANRETIN GEL and for 1 week after the last dose [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
• Embryo-Fetal Toxicity: Can cause fetal harm. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use an effective method of contraception. (5.1. 8.1,8.3)
• Photosensitivity: Minimize exposure to sunlight and sunlamps. (5.2)
• DEET toxicity: Do not use DEET-containing products (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
PANRETIN GEL is for topical use only.
Do not use occlusive dressings with PANRETIN GEL.
- Apply PANRETIN GEL twice daily to coat the entire cutaneous Kaposi sarcoma lesions.
- Gradually increase the application frequency up to four (4) times a day as tolerated.
- Continue PANRETIN GEL as long as patient is deriving benefit.
- Reduce application frequency for application site toxicity. Interrupt treatment for severe irritation; may resume at a reduced application frequency once symptoms improve.
- Avoid application of gel to normal skin and do not apply on or near mucosal surfaces.
- Wash hands after application unless gel is applied to Kaposi sarcoma lesions on the hands.
- Allow gel to dry for three to five minutes before covering with clothing.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Topical Gel: 0.1% alitretinoin (clear yellow gel) in a 60-gram tube.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of PANRETIN GEL was assessed in two multicenter, prospective, randomized, double-blind, vehicle-controlled trials (Trial 1 and Trial 2) in patients with cutaneous lesions of AIDS-related KS [see Clinical Studies (14.1)]. In a pooled analysis of both trials, the most common adverse reactions in ≥ 5% of patients were rash, pain, paresthesia, pruritis, exfoliative dermatitis, edema, and skin disorder.
In Trial 1, severe local skin adverse reactions (erythema and edema with or without vesiculation) occurred in 10% of patients during the first 12 weeks of treatment (versus 0% in the vehicle control). Adverse reactions led to withdrawal from the study in 7% of patients.
In Trial 2, severe local skin adverse reactions (erythema and edema with or without vesiculation) occurred in 6% of patients during the first 12 weeks of treatment (versus 0% in the vehicle control) and 1 patient withdrew due to severe skin irritation.
Table 1 lists the most common application site adverse reactions that occurred in a least 5% of patients during the double-blind phase who received PANRETIN GEL in either of the two controlled studies.
TABLE 1: Adverse Reactions at Application Site in Trial 1 and 2 in ≥ 5% of Patients Treated with PANRETIN GEL
|
Adverse Event Term |
Trial 1 |
Trial 2 |
||
|
|
PANRETIN GEL N=134 % |
Vehicle Gel N=134 % |
PANRETIN GEL N=36 % |
Vehicle Gel N=46 % |
|
Rash1 |
77 |
11 |
25 |
4 |
|
Pain2 |
34 |
7 |
0 |
4 |
|
Pruritus3 |
11 |
4 |
8 |
4 |
|
Exfoliative dermatitis4 |
9 |
2 |
3 |
0 |
|
Skin disorder5 |
8 |
1 |
0 |
0 |
|
Edema6 |
8 |
3 |
3 |
0 |
|
Parethesia7 |
3 |
0 |
22 |
7 |
| Includes Investigator terms: 1 Erythema, scaling, irritation, redness, rash, dermatitis 2 Burning, pain 3 Itching, pruritus 4 Flaking, peeling, desquamation, exfoliation 5 Excoriation, cracking, scab, crusting, drainage, eschar, fissure or oozing 6 Edema, swelling, inflammation 7 Stinging, tingling |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Embryo-Fetal Toxicity
Advise females to inform their healthcare provider if they are pregnant or become pregnant. Inform females of the risk to a fetus and potential loss of pregnancy [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
Advise females of reproductive potential to use effective contraception during treatment and for 1 week after the last dose of PANRETIN GEL [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
Advise males with female partners of reproductive potential to use effective contraception during treatment and for 1 week following the last dose of PANRETIN GEL [see Warnings and Precautions (5.1) and Use in Specific Populations (8.3)].
Lactation
Advise patients not to breastfeed during treatment with PANRETIN GEL and for 1 week after the last dose of PANRETIN GEL [see Use in Specific Populations (8.2)].
Photosensitivity
Advice patients that PANRETIN GEL can cause photosensitivity and to avoid sunlight and sunlamps and use measures such as protective clothing in PANRETIN-applied areas [see Warnings and Precautions (5.2)].
Toxicity with DEET-Containing Products
Advise patients to avoid DEET-containing products such as insect repellant while using PANRETIN GEL [see Warnings and Precautions (5.3)].
Manufactured for:
Advanz Pharma (US) Corp.
Bannockburn IL, 60015
PANRETIN® is a registered trademark of Mercury Pharma Group Limited. Distributed by Advanz Pharma (US) Corp. under license
Revised: 11/2025
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
PANRETIN GEL is a clear yellow gel and is supplied in a 60-gram tubes containing 0.1% alitretinoin.
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
PANRETIN GEL is flammable; keep away from heat or flame.
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
NDC 59212-601-22
Panretin® gel
(alitretinoin) 0.1%
5.3 Toxicity With Deet Containing Products (5.3 Toxicity with DEET-Containing Products)
Animal toxicology studies showed increased DEET toxicity when DEET was included as part of the formulation. Advise patients to not use PANRETIN GEL concurrently with products that contain DEET (N,N-diethyl-m-toluamide), a common component of insect repellent products.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Verify pregnancy status of females of reproductive potential prior to initiating PANRETIN GEL.
Contraception
Females
PANRETIN GEL can cause embryo-fetal harm when administered to pregnant women [see Use in Specific Populations (8.1)]. Advise females of reproductive potential to use effective contraception during treatment with PANRETIN GEL and for 1 week after the last dose.
Males
Advise males with female partners of reproductive potential to use effective contraception during treatment with PANRETIN GEL and for 1 week after the last dose.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility (13.1 Carcinogenesis, Mutagenesis, Impairment Of Fertility)
Carcinogenicity studies with alitretinoin have not been conducted. Alitretinoin was not mutagenic in vitro (bacterial assay, Chinese hamster ovary cell HGPRT mutation assay) and was not clastogenic in vitro (chromosome aberration test in human lymphocytes) or in vivo (mouse micronucleus test).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:43.582424 · Updated: 2026-03-14T22:38:30.428423