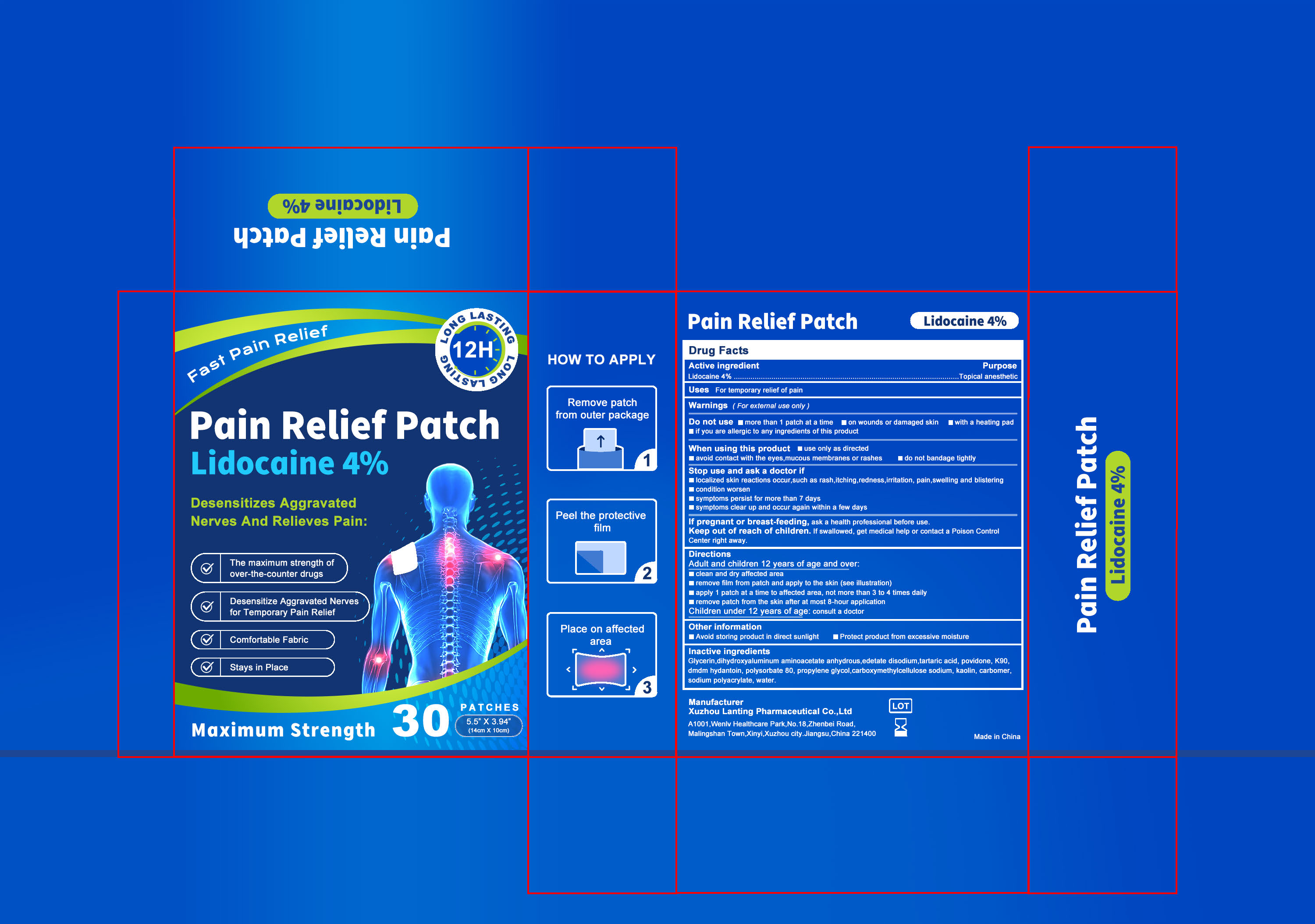
87278-003 4% Lidocaine Pain Relief Patch
4981e9c3-2a19-aed8-e063-6294a90af216
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4%
Medication Information
Warnings and Precautions
For external use only
Indications and Usage
For temporary relief of pain
Dosage and Administration
Adult and children 12 years of age and over:
clean and dry affected area
remove film from patch and apply to the skin (see illustration)
apply 1 patch at a time to affected area, not more than 3 to 4 times daily
remove patch from the skin after at most 8-hour application
Children under 12 years of age: consult a doctor
Description
Lidocaine 4%
Section 50565-1
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. lf swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
localized skin reactions occur,such as rash,itching,redness,irritation, pain,swelling and blistering
condition worsen
symptoms persist for more than 7 days
symptoms clear up and occur again within a few days
Section 50567-7
use only as directed
avoid contact with the eyes,mucous membranes or rashes
do not bandage tightly
Section 50570-1
more than 1 patch at a time
on wounds or damaged skin
with a heating pad
if you are allergic to any ingredients of this product
Section 51727-6
Glycerin
Dihydroxyaluminum aminoacetate anhydrous
Edetate disodium
Tartaric acid
Povidone K90
DMDM Hydantoin
Polysorbate 80
Propylene glycol
Carboxymethylcellulose sodium
Kaolin
Carbomer
Sodium polyacrylate
Water
Section 51945-4
Section 55105-1
Topical anesthetic
Section 55106-9
Lidocaine 4%
Structured Label Content
Indications and Usage (34067-9)
For temporary relief of pain
Dosage and Administration (34068-7)
Adult and children 12 years of age and over:
clean and dry affected area
remove film from patch and apply to the skin (see illustration)
apply 1 patch at a time to affected area, not more than 3 to 4 times daily
remove patch from the skin after at most 8-hour application
Children under 12 years of age: consult a doctor
Warnings and Precautions (34071-1)
For external use only
Section 50565-1 (50565-1)
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. lf swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
localized skin reactions occur,such as rash,itching,redness,irritation, pain,swelling and blistering
condition worsen
symptoms persist for more than 7 days
symptoms clear up and occur again within a few days
Section 50567-7 (50567-7)
use only as directed
avoid contact with the eyes,mucous membranes or rashes
do not bandage tightly
Section 50570-1 (50570-1)
more than 1 patch at a time
on wounds or damaged skin
with a heating pad
if you are allergic to any ingredients of this product
Section 51727-6 (51727-6)
Glycerin
Dihydroxyaluminum aminoacetate anhydrous
Edetate disodium
Tartaric acid
Povidone K90
DMDM Hydantoin
Polysorbate 80
Propylene glycol
Carboxymethylcellulose sodium
Kaolin
Carbomer
Sodium polyacrylate
Water
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Topical anesthetic
Section 55106-9 (55106-9)
Lidocaine 4%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:27.125438 · Updated: 2026-03-14T23:12:39.597935