These Highlights Do Not Include All The Information Needed To Use Cardiolite®
49662def-2fb9-47b2-8467-ca56f6006167
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
CARDIOLITE® is a myocardial perfusion agent indicated for: detecting coronary artery disease by localizing myocardial ischemia (reversible defects) and infarction (non-reversible defects) evaluating myocardial function and developing information for use in patient management decisions
Indications and Usage
CARDIOLITE® is a myocardial perfusion agent indicated for: detecting coronary artery disease by localizing myocardial ischemia (reversible defects) and infarction (non-reversible defects) evaluating myocardial function and developing information for use in patient management decisions
Dosage and Administration
For Myocardial Imaging: The suggested dose range for I.V. administration of CARDIOLITE® in a single dose to be employed in the average patient (70 Kg) is 370 - 1110 MBq (10 - 30 mCi). For Breast Imaging: The recommended dose range for I.V. administration of MIRALUMA® is a single dose of 740 - 1110 MBq (20 - 30 mCi).
Warnings and Precautions
Pharmacologic induction of cardiovascular stress may be associated with serious adverse events such as myocardial infarction, arrhythmia, hypotension, bronchoconstriction and cerebrovascular events. CARDIOLITE® has been rarely associated with acute severe allergic and anaphylactic events of angioedema and generalized urticaria. In some patients the allergic symptoms developed on the second injection during CARDIOLITE® imaging. Caution should be exercised and emergency equipment should be available when administering CARDIOLITE®. Before administering CARDIOLITE® patients should be asked about the possibility of allergic reactions to either drug. The contents of the vial are intended only for use in the preparation of Technetium Tc99m Sestamibi and are not to be administered directly to the patient without first undergoing the preparative procedure.
Contraindications
None known.
Adverse Reactions
Adverse events were evaluated in 3741 adults who were evaluated in clinical studies. Of these patients, 3068 (77% men, 22% women, and 0.7% of the patient's genders were not recorded) were in cardiac clinical trials and 673 (100% women) in breast imaging trials. Cases of angina, chest pain, and death have occurred (see Section 5 ). Adverse events reported at a rate of 0.5% or greater after receiving Technetium Tc99m Sestamibi administration are shown in the following table: Table 2.0 Selected Adverse Events Reported in > 0.5% of Patients Who Received Technetium Tc99m Sestamibi in Either Breast or Cardiac Clinical Studies Excludes the 22 patients whose gender was not recorded. Body System Breast Studies Cardiac Studies Women n = 673 Women n = 685 Men n = 2361 Total n = 3046 Body as a Whole 21 (3.1%) 6 (0.9%) 17 (0.7%) 23 (0.8%) Headache 11 (1.6%) 2 (0.3%) 4 (0.2%) 6 (0.2%) Cardiovascular 9 (1.3%) 24 (3.5%) 75 (3.2%) 99 (3.3%) Chest Pain/Angina 0 (0%) 18 (2.6%) 46 (1.9%) 64 (2.1%) ST segment changes 0 (0%) 11 (1.6%) 29 (1.2%) 40 (1.3%) Digestive System 8 (1.2%) 4 (0.6%) 9 (0.4%) 13 (0.4%) Nausea 4 (0.6%) 1 (0.1%) 2 (0.1%) 3 (0.1%) Special Senses 132 (19.6%) 62 (9.1%) 160 (6.8%) 222 (7.3%) Taste Perversion 129 (19.2%) 60 (8.8%) 157 (6.6%) 217 (7.1%) Parosmia 8 (1.2%) 6 (0.9%) 10 (0.4%) 16 (0.5%) In the clinical studies for breast imaging, breast pain was reported in 12 (1.7%) of the patients. In 11 of these patients the pain appears to be associated with biopsy/surgical procedures. The following adverse reactions have been reported in ≤ 0.5% of patients: signs and symptoms consistent with seizure occurring shortly after administration of the agent; transient arthritis, angioedema, arrythmia, dizziness, syncope, abdominal pain, vomiting, and severe hypersensitivity characterized by dyspnea, hypotension, bradycardia, asthenia, and vomiting within two hours after a second injection of Technetium Tc99m Sestamibi. A few cases of flushing, edema, injection site inflammation, dry mouth, fever, pruritis, rash, urticaria and fatigue have also been attributed to administration of the agent.
Drug Interactions
Specific drug-drug interactions have not been studied.
Storage and Handling
CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection is supplied as a 5 mL vial in kits of five (5) vials (NDC # 11994-001-55) and twenty (20) vials (NDC # 11994-001-20), sterile and non-pyrogenic. The patient dose should be measured by a suitable radioactivity calibration system immediately prior to patient administration. Radiochemical purity should be checked prior to patient administration. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Prior to lyophilization the pH is between 5.3-5.9. The contents of the vial are lyophilized and stored under nitrogen. Store at 15-25°C (59-77° F) before and after reconstitution. Technetium Tc99m Sestamibi contains no preservatives. Included in each five (5) vial kit is one (1) package insert, six (6) vial shield labels and six (6) radiation warning labels. Included in each twenty (20) vial kit is one (1) package insert, twenty four (24) vial shield labels and twenty four (24) radiation warning labels. This reagent kit is approved for distribution to persons licensed pursuant to the Code of Massachusetts Regulations 105 CMR 120.500 for the uses listed in 105 CMR 120.547 or 120.552, or under equivalent regulations of the U.S. Nuclear Regulatory Commission, Agreement States or Licensing States.
How Supplied
CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection is supplied as a 5 mL vial in kits of five (5) vials (NDC # 11994-001-55) and twenty (20) vials (NDC # 11994-001-20), sterile and non-pyrogenic. The patient dose should be measured by a suitable radioactivity calibration system immediately prior to patient administration. Radiochemical purity should be checked prior to patient administration. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Prior to lyophilization the pH is between 5.3-5.9. The contents of the vial are lyophilized and stored under nitrogen. Store at 15-25°C (59-77° F) before and after reconstitution. Technetium Tc99m Sestamibi contains no preservatives. Included in each five (5) vial kit is one (1) package insert, six (6) vial shield labels and six (6) radiation warning labels. Included in each twenty (20) vial kit is one (1) package insert, twenty four (24) vial shield labels and twenty four (24) radiation warning labels. This reagent kit is approved for distribution to persons licensed pursuant to the Code of Massachusetts Regulations 105 CMR 120.500 for the uses listed in 105 CMR 120.547 or 120.552, or under equivalent regulations of the U.S. Nuclear Regulatory Commission, Agreement States or Licensing States.
Medication Information
Warnings and Precautions
Pharmacologic induction of cardiovascular stress may be associated with serious adverse events such as myocardial infarction, arrhythmia, hypotension, bronchoconstriction and cerebrovascular events. CARDIOLITE® has been rarely associated with acute severe allergic and anaphylactic events of angioedema and generalized urticaria. In some patients the allergic symptoms developed on the second injection during CARDIOLITE® imaging. Caution should be exercised and emergency equipment should be available when administering CARDIOLITE®. Before administering CARDIOLITE® patients should be asked about the possibility of allergic reactions to either drug. The contents of the vial are intended only for use in the preparation of Technetium Tc99m Sestamibi and are not to be administered directly to the patient without first undergoing the preparative procedure.
Indications and Usage
CARDIOLITE® is a myocardial perfusion agent indicated for: detecting coronary artery disease by localizing myocardial ischemia (reversible defects) and infarction (non-reversible defects) evaluating myocardial function and developing information for use in patient management decisions
Dosage and Administration
For Myocardial Imaging: The suggested dose range for I.V. administration of CARDIOLITE® in a single dose to be employed in the average patient (70 Kg) is 370 - 1110 MBq (10 - 30 mCi). For Breast Imaging: The recommended dose range for I.V. administration of MIRALUMA® is a single dose of 740 - 1110 MBq (20 - 30 mCi).
Contraindications
None known.
Adverse Reactions
Adverse events were evaluated in 3741 adults who were evaluated in clinical studies. Of these patients, 3068 (77% men, 22% women, and 0.7% of the patient's genders were not recorded) were in cardiac clinical trials and 673 (100% women) in breast imaging trials. Cases of angina, chest pain, and death have occurred (see Section 5 ). Adverse events reported at a rate of 0.5% or greater after receiving Technetium Tc99m Sestamibi administration are shown in the following table: Table 2.0 Selected Adverse Events Reported in > 0.5% of Patients Who Received Technetium Tc99m Sestamibi in Either Breast or Cardiac Clinical Studies Excludes the 22 patients whose gender was not recorded. Body System Breast Studies Cardiac Studies Women n = 673 Women n = 685 Men n = 2361 Total n = 3046 Body as a Whole 21 (3.1%) 6 (0.9%) 17 (0.7%) 23 (0.8%) Headache 11 (1.6%) 2 (0.3%) 4 (0.2%) 6 (0.2%) Cardiovascular 9 (1.3%) 24 (3.5%) 75 (3.2%) 99 (3.3%) Chest Pain/Angina 0 (0%) 18 (2.6%) 46 (1.9%) 64 (2.1%) ST segment changes 0 (0%) 11 (1.6%) 29 (1.2%) 40 (1.3%) Digestive System 8 (1.2%) 4 (0.6%) 9 (0.4%) 13 (0.4%) Nausea 4 (0.6%) 1 (0.1%) 2 (0.1%) 3 (0.1%) Special Senses 132 (19.6%) 62 (9.1%) 160 (6.8%) 222 (7.3%) Taste Perversion 129 (19.2%) 60 (8.8%) 157 (6.6%) 217 (7.1%) Parosmia 8 (1.2%) 6 (0.9%) 10 (0.4%) 16 (0.5%) In the clinical studies for breast imaging, breast pain was reported in 12 (1.7%) of the patients. In 11 of these patients the pain appears to be associated with biopsy/surgical procedures. The following adverse reactions have been reported in ≤ 0.5% of patients: signs and symptoms consistent with seizure occurring shortly after administration of the agent; transient arthritis, angioedema, arrythmia, dizziness, syncope, abdominal pain, vomiting, and severe hypersensitivity characterized by dyspnea, hypotension, bradycardia, asthenia, and vomiting within two hours after a second injection of Technetium Tc99m Sestamibi. A few cases of flushing, edema, injection site inflammation, dry mouth, fever, pruritis, rash, urticaria and fatigue have also been attributed to administration of the agent.
Drug Interactions
Specific drug-drug interactions have not been studied.
Storage and Handling
CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection is supplied as a 5 mL vial in kits of five (5) vials (NDC # 11994-001-55) and twenty (20) vials (NDC # 11994-001-20), sterile and non-pyrogenic. The patient dose should be measured by a suitable radioactivity calibration system immediately prior to patient administration. Radiochemical purity should be checked prior to patient administration. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Prior to lyophilization the pH is between 5.3-5.9. The contents of the vial are lyophilized and stored under nitrogen. Store at 15-25°C (59-77° F) before and after reconstitution. Technetium Tc99m Sestamibi contains no preservatives. Included in each five (5) vial kit is one (1) package insert, six (6) vial shield labels and six (6) radiation warning labels. Included in each twenty (20) vial kit is one (1) package insert, twenty four (24) vial shield labels and twenty four (24) radiation warning labels. This reagent kit is approved for distribution to persons licensed pursuant to the Code of Massachusetts Regulations 105 CMR 120.500 for the uses listed in 105 CMR 120.547 or 120.552, or under equivalent regulations of the U.S. Nuclear Regulatory Commission, Agreement States or Licensing States.
How Supplied
CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection is supplied as a 5 mL vial in kits of five (5) vials (NDC # 11994-001-55) and twenty (20) vials (NDC # 11994-001-20), sterile and non-pyrogenic. The patient dose should be measured by a suitable radioactivity calibration system immediately prior to patient administration. Radiochemical purity should be checked prior to patient administration. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Prior to lyophilization the pH is between 5.3-5.9. The contents of the vial are lyophilized and stored under nitrogen. Store at 15-25°C (59-77° F) before and after reconstitution. Technetium Tc99m Sestamibi contains no preservatives. Included in each five (5) vial kit is one (1) package insert, six (6) vial shield labels and six (6) radiation warning labels. Included in each twenty (20) vial kit is one (1) package insert, twenty four (24) vial shield labels and twenty four (24) radiation warning labels. This reagent kit is approved for distribution to persons licensed pursuant to the Code of Massachusetts Regulations 105 CMR 120.500 for the uses listed in 105 CMR 120.547 or 120.552, or under equivalent regulations of the U.S. Nuclear Regulatory Commission, Agreement States or Licensing States.
Description
CARDIOLITE® is a myocardial perfusion agent indicated for: detecting coronary artery disease by localizing myocardial ischemia (reversible defects) and infarction (non-reversible defects) evaluating myocardial function and developing information for use in patient management decisions
Section 42229-5
Myocardial Imaging: CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection, is a myocardial perfusion agent that is indicated for detecting coronary artery disease by localizing myocardial ischemia (reversible defects) and infarction (non-reversible defects), in evaluating myocardial function and developing information for use in patient management decisions. CARDIOLITE® evaluation of myocardial ischemia can be accomplished with rest and cardiovascular stress techniques (e.g., exercise or pharmacologic stress in accordance with the pharmacologic stress agent's labeling).
It is usually not possible to determine the age of a myocardial infarction or to differentiate a recent myocardial infarction from ischemia.
5.1 Warnings
In studying patients in whom cardiac disease is known or suspected, care should be taken to assure continuous monitoring and treatment in accordance with safe, accepted clinical procedure. Infrequently, death has occurred 4 to 24 hours after Tc99m Sestamibi use and is usually associated with exercise stress testing (See Section 5.2 ).
Pharmacologic induction of cardiovascular stress may be associated with serious adverse events such as myocardial infarction, arrhythmia, hypotension, bronchoconstriction and cerebrovascular events. Caution should be used when pharmacologic stress is selected as an alternative to exercise; it should be used when indicated and in accordance with the pharmacologic stress agent's labeling.
Technetium Tc99m Sestamibi has been rarely associated with acute severe allergic and anaphylactic events of angioedema and generalized urticaria. In some patients the allergic symptoms developed on the second injection during CARDIOLITE® imaging. Patients who receive CARDIOLITE® or MIRALUMA® imaging are receiving the same drug. Caution should be exercised and emergency equipment should be available when administering Technetium Tc99m Sestamibi. Also, before administering either CARDIOLITE® or MIRALUMA®, patients should be asked about the possibility of allergic reactions to either drug.
10. Overdosage
The clinical consequences of overdosing with CARDIOLITE® are not known.
11. Description
Each 5 mL vial contains a sterile, non-pyrogenic, lyophilized mixture of:
- Tetrakis (2-methoxy isobutyl isonitrile) Copper (I) tetrafluoroborate - 1.0 mg
- Sodium Citrate Dihydrate - 2.6 mg
- L-Cysteine Hydrochloride Monohydrate - 1.0 mg
- Mannitol – 20 mg
- Stannous Chloride, Dihydrate, minimum (SnCl2∙2H2O) - 0.025 mg
- Stannous Chloride, Dihydrate, (SnCl2∙2H2O) - 0.075 mg
- Tin Chloride (stannous and stannic) Dihydrate, maximum (as SnCl2∙2H2O) - 0.086 mg
Prior to lyophilization the pH is 5.3 to 5.9. The contents of the vial are lyophilized and stored under nitrogen.
This drug is administered by intravenous injection for diagnostic use after reconstitution with sterile, non-pyrogenic, oxidant-free Sodium Pertechnetate Tc99m Injection. The pH of the reconstituted product is 5.5 (5.0 - 6.0). No bacteriostatic preservative is present.
The precise structure of the technetium complex is Tc99m[MIBI]6 + where MIBI is 2-methoxy isobutyl isonitrile.
8.4 Pediatric Use
Safety and effectiveness in the pediatric population have not been established.
No evidence of diagnostic efficacy or clinical utility of CARDIOLITE® scan was found in clinical studies of children and adolescents with Kawasaki disease.
A prospective study of 445 pediatric patients with Kawasaki disease was designed to determine the predictive value of CARDIOLITE® rest and stress myocardial perfusion imaging to define a pediatric population with Kawasaki disease that was at risk of developing cardiac events. Cardiac events were defined as cardiac death, MI, hospitalization due to cardiac etiology, heart failure, CABG or coronary angioplasty. The standard of truth was defined as cardiac events occurring 6 months following the administration of CARDIOLITE®. Only three cardiac events were observed at six months in this study. In all three cases, the scan was negative. No clinically meaningful measurements of sensitivity, specificity or other diagnostic performance parameters could be demonstrated in this study.
A ten year retrospective case history study of pediatric Kawasaki disease patients who completed CARDIOLITE® myocardial perfusion imaging and who had coronary angiography within three months of the CARDIOLITE® scan was designed to measure sensitivity and specificity of CARDIOLITE® scan. Out of 72 patients who had both evaluable CARDIOLITE® scans and evaluable angiographic images, only one patient had both an abnormal angiogram and an abnormal CARDIOLITE® scan. No clinically meaningful measurements of sensitivity, specificity or other diagnostic performance parameters could be demonstrated in this study.
In a clinical pharmacology study, 46 pediatric patients with Kawasaki disease received CARDIOLITE® administration at the following doses: 0.1 - 0.2 mCi/kg for rest, 0.3 mCi/kg for stress in one day studies; 0.2 mCi/kg for rest and 0.2 mCi/kg for stress in two day studies.
The radioactivity both in younger children and in adolescents exhibited PK profiles similar to those previously reported in adults (See Section 12).
The radiation absorbed doses in adolescents, both at rest and stress, were similar to those observed in adults (see Section 2). When comparing weight-adjusted radioactivity (up to 0.3 mCi/kg) doses administered to adolescents and younger children to the recommended dose administered to adults (up to 30 mCi), the radiation absorbed doses in both adolescents and younger children were similar to those in adults.
Adverse events were evaluated in 609 pediatric patients from the three clinical studies described above. The frequency and the type of the adverse events were similar to the ones observed in the studies of CARDIOLITE® in adults. Two of the 609 had a serious adverse event: one patient received a CARDIOLITE® overdose but remained asymptomatic, and one patient had an asthma exacerbation following administration.
8.5 Geriatric Use
Of 3068 patients in clinical studies of CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection, 693 patients were 65 or older and 121 were 75 or older.
Of 673 patients in clinical studies of MIRALUMA®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection, 138 patients were 65 or older and 30 were 75 or older.
Based on the evaluation of the frequency of adverse events and review of vital signs data, no overall differences in safety were observed between these subjects and younger subjects. Although reported clinical experience has not identified differences in response between elderly and younger patients, greater sensitivity of some older individuals cannot be ruled out.
4. Contraindications
None known.
6. Adverse Reactions
Adverse events were evaluated in 3741 adults who were evaluated in clinical studies. Of these patients, 3068 (77% men, 22% women, and 0.7% of the patient's genders were not recorded) were in cardiac clinical trials and 673 (100% women) in breast imaging trials. Cases of angina, chest pain, and death have occurred (see Section 5 ). Adverse events reported at a rate of 0.5% or greater after receiving Technetium Tc99m Sestamibi administration are shown in the following table:
| Body System | Breast Studies | Cardiac Studies | ||
|---|---|---|---|---|
| Women n = 673 |
Women n = 685 |
Men n = 2361 |
Total n = 3046 |
|
| Body as a Whole | 21 (3.1%) | 6 (0.9%) | 17 (0.7%) | 23 (0.8%) |
| Headache | 11 (1.6%) | 2 (0.3%) | 4 (0.2%) | 6 (0.2%) |
| Cardiovascular | 9 (1.3%) | 24 (3.5%) | 75 (3.2%) | 99 (3.3%) |
| Chest Pain/Angina | 0 (0%) | 18 (2.6%) | 46 (1.9%) | 64 (2.1%) |
| ST segment changes | 0 (0%) | 11 (1.6%) | 29 (1.2%) | 40 (1.3%) |
| Digestive System | 8 (1.2%) | 4 (0.6%) | 9 (0.4%) | 13 (0.4%) |
| Nausea | 4 (0.6%) | 1 (0.1%) | 2 (0.1%) | 3 (0.1%) |
| Special Senses | 132 (19.6%) | 62 (9.1%) | 160 (6.8%) | 222 (7.3%) |
| Taste Perversion | 129 (19.2%) | 60 (8.8%) | 157 (6.6%) | 217 (7.1%) |
| Parosmia | 8 (1.2%) | 6 (0.9%) | 10 (0.4%) | 16 (0.5%) |
In the clinical studies for breast imaging, breast pain was reported in 12 (1.7%) of the patients. In 11 of these patients the pain appears to be associated with biopsy/surgical procedures.
The following adverse reactions have been reported in ≤ 0.5% of patients: signs and symptoms consistent with seizure occurring shortly after administration of the agent; transient arthritis, angioedema, arrythmia, dizziness, syncope, abdominal pain, vomiting, and severe hypersensitivity characterized by dyspnea, hypotension, bradycardia, asthenia, and vomiting within two hours after a second injection of Technetium Tc99m Sestamibi. A few cases of flushing, edema, injection site inflammation, dry mouth, fever, pruritis, rash, urticaria and fatigue have also been attributed to administration of the agent.
7. Drug Interactions
Specific drug-drug interactions have not been studied.
12.3 Pharmacokinetics
Pulmonary activity is negligible even immediately after injection. Blood clearance studies indicate that the fast clearing component clears with a t1/2 of 4.3 minutes at rest, and clears with a t1/2 of 1.6 minutes under exercise conditions. At five minutes post injection about 8% of the injected dose remains in circulation. There is less than 1% protein binding of Technetium Tc99m Sestamibi in plasma. The myocardial biological half-life is approximately six hours after a rest or exercise injection. The biological half-life for the liver is approximately 30 minutes after a rest or exercise injection. The effective half-life of clearance (which includes both the biological half-life and radionuclide decay) for the heart is approximately 3 hours, and for the liver is approximately 30 minutes, after a rest or exercise injection. The ideal imaging time reflects the best compromise between heart count rate and surrounding organ uptake.
Myocardial uptake which is coronary flow dependent is 1.2% of the injected dose at rest and 1.5% of the injected dose at exercise. Table 6.0 illustrates the biological clearance as well as effective clearance (which includes biological clearance and radionuclide decay) of Tc99m Sestamibi from the heart and liver.
[Organ concentrations expressed as percentage of injected dose; data based on an average of 5 subjects at rest and 5 subjects during exercise].
| REST | STRESS | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Heart | Liver | Heart | Liver | ||||||
| Time | Biological | Effective | Biological | Effective | Biological | Effective | Biological | Effective | |
| 5 min. | 1.2 | 1.2 | 19.6 | 19.4 | 1.5 | 1.5 | 5.9 | 5.8 | |
| 30 min. | 1.1 | 1.0 | 12.2 | 11.5 | 1.4 | 1.3 | 4.5 | 4.2 | |
| 1 hour | 1.0 | 0.9 | 5.6 | 5.0 | 1.4 | 1.2 | 2.4 | 2.1 | |
| 2 hours | 1.0 | 0.8 | 2.2 | 1.7 | 1.2 | 1.0 | 0.9 | 0.7 | |
| 4 hours | 0.8 | 0.5 | 0.7 | 0.4 | 1.0 | 0.6 | 0.3 | 0.2 |
A study in a dog myocardial ischemia model reported that Technetium Tc99m Sestamibi undergoes myocardial distribution (redistribution), although more slowly and less completely than thallous chloride Tl-201. A study in a dog myocardial infarction model reported that the drug showed no redistribution of any consequence. Definitive human studies to demonstrate possible redistribution have not been reported. In patients with documented myocardial infarction, imaging revealed the infarct up to four hours post dose.
11.2 External Radiation
The specific gamma ray constant for Tc99m is 5.4 microcoulombs/Kg-MBq-hr (0.78R/mCi-hr) at 1 cm. The first half value layer is 0.017 cm of Pb. A range of values for the relative attenuation of the radiation emitted by this radionuclide that results from interposition of various thicknesses of Pb is shown in Table 4.0. To facilitate control of the radiation exposure from Megabequerel (millicurie) amounts of this radionuclide, the use of a 0.25 cm thickness of Pb will attenuate the radiation emitted by a factor of 1,000.
| Shield Thickness (Pb) cm | Coefficient of Attenuation |
|---|---|
| 0.017 | 0.5 |
| 0.08 | 10-1 |
| 0.16 | 10-2 |
| 0.25 | 10-3 |
| 0.33 | 10-4 |
To correct for physical decay of this radionuclide, the fractions that remain at selected intervals after the time of calibration are shown in Table 5.0.
| Hours | Fraction Remaining | Hours | Fraction Remaining |
|---|---|---|---|
| 0 Calibration Time
|
1.000 | 8 | .398 |
| 1 | .891 | 9 | .355 |
| 2 | .794 | 10 | .316 |
| 3 | .708 | 11 | .282 |
| 4 | .631 | 12 | .251 |
| 5 | .562 | ||
| 6 | .501 | ||
| 7 | .447 |
2.2 Radiation Dosimetry
The radiation doses to organs and tissues of an average patient (70 Kg) per 1110 MBq (30 mCi) of Technetium Tc99m Sestamibi injected intravenously are shown in Table 1.0.
| Estimated Radiation Absorbed Dose | ||||
|---|---|---|---|---|
| Radiation dosimetry calculations performed by Radiation Internal Dose Information Center, Oak Ridge Institute for Science and Education, PO Box 117, Oak Ridge, TN 37831-0117. | ||||
| REST | ||||
| 2.0 hour void | 4.8 hour void | |||
| Organ | rads/ 30 mCi |
mGy/ 1110 MBq |
rads/ 30 mCi |
mGy/ 1110 MBq |
| Breasts | 0.2 | 2.0 | 0.2 | 1.9 |
| Gallbladder Wall | 2.0 | 20.0 | 2.0 | 20.0 |
| Small Intestine | 3.0 | 30.0 | 3.0 | 30.0 |
| Upper Large Intestine Wall | 5.4 | 55.5 | 5.4 | 55.5 |
| Lower Large Intestine Wall | 3.9 | 40.0 | 4.2 | 41.1 |
| Stomach Wall | 0.6 | 6.1 | 0.6 | 5.8 |
| Heart Wall | 0.5 | 5.1 | 0.5 | 4.9 |
| Kidneys | 2.0 | 20.0 | 2.0 | 20.0 |
| Liver | 0.6 | 5.8 | 0.6 | 5.7 |
| Lungs | 0.3 | 2.8 | 0.3 | 2.7 |
| Bone Surfaces | 0.7 | 6.8 | 0.7 | 6.4 |
| Thyroid | 0.7 | 7.0 | 0.7 | 7.0 |
| Ovaries | 1.5 | 15.5 | 1.6 | 15.5 |
| Testes | 0.3 | 3.4 | 0.4 | 3.9 |
| Red Marrow | 0.5 | 5.1 | 0.5 | 5.0 |
| Urinary Bladder Wall | 2.0 | 20.0 | 4.2 | 41.1 |
| Total Body | 0.5 | 4.8 | 0.5 | 4.8 |
| STRESS | ||||
| 2.0 hour void | 4.8 hour void | |||
| Organ | rads/ 30 mCi |
mGy/ 1110 MBq |
rads/ 30 mCi |
mGy/ 1110 MBq |
| Breasts | 0.2 | 2.0 | 0.2 | 1.8 |
| Gallbladder Wall | 2.8 | 28.9 | 2.8 | 27.8 |
| Small Intestine | 2.4 | 24.4 | 2.4 | 24.4 |
| Upper Large Intestine Wall | 4.5 | 44.4 | 4.5 | 44.4 |
| Lower Large Intestine Wall | 3.3 | 32.2 | 3.3 | 32.2 |
| Stomach Wall | 0.6 | 5.3 | 0.5 | 5.2 |
| Heart Wall | 0.5 | 5.6 | 0.5 | 5.3 |
| Kidneys | 1.7 | 16.7 | 1.7 | 16.7 |
| Liver | 0.4 | 4.2 | 0.4 | 4.1 |
| Lungs | 0.3 | 2.6 | 0.2 | 2.4 |
| Bone Surfaces | 0.6 | 6.2 | 0.6 | 6.0 |
| Thyroid | 0.3 | 2.7 | 0.2 | 2.4 |
| Ovaries | 1.2 | 12.2 | 1.3 | 13.3 |
| Testes | 0.3 | 3.1 | 0.3 | 3.4 |
| Red Marrow | 0.5 | 4.6 | 0.5 | 4.4 |
| Urinary Bladder Wall | 1.5 | 15.5 | 3.0 | 30.0 |
| Total Body | 0.4 | 4.2 | 0.4 | 4.2 |
5.2 General Precautions
The contents of the vial are intended only for use in the preparation of Technetium Tc99m Sestamibi and are not to be administered directly to the patient without first undergoing the preparative procedure.
Radioactive drugs must be handled with care and appropriate safety measures should be used to minimize radiation exposure to clinical personnel. Also, care should be taken to minimize radiation exposure to the patients consistent with proper patient management.
Contents of the kit before preparation are not radioactive. However, after the Sodium Pertechnetate Tc99m Injection is added, adequate shielding of the final preparation must be maintained. The components of the kit are sterile and non-pyrogenic. It is essential to follow directions carefully and to adhere to strict aseptic procedures during preparation.
Technetium Tc99m labeling reactions depend on maintaining the stannous ion in the reduced state. Hence, Sodium Pertechnetate Tc99m Injection containing oxidants should not be used.
Technetium Tc99m Sestamibi should not be used more than six hours after preparation.
Radiopharmaceuticals should be used only by physicians who are qualified by training and experience in the safe use and handling of radionuclides and whose experience and training have been approved by the appropriate government agency authorized to license the use of radionuclides.
Stress testing should be performed only under the supervision of a qualified physician and in a laboratory equipped with appropriate resuscitation and support apparatus.
The most frequent exercise stress test endpoints sufficient to stop the test reported during controlled studies (two-thirds were cardiac patients) were:
| Fatigue | 35% |
| Dyspnea | 17% |
| Chest Pain | 16% |
| ST-depression | 7% |
| Arrhythmia | 1% |
1. Indications and Usage
CARDIOLITE® is a myocardial perfusion agent indicated for:
- detecting coronary artery disease by localizing myocardial ischemia (reversible defects) and infarction (non-reversible defects)
- evaluating myocardial function and developing information for use in patient management decisions
12.1 Mechanism of Action
Technetium Tc99m Sestamibi is a cationic Tc99m complex which has been found to accumulate in viable myocardial tissue in a manner analogous to that of thallous chloride Tl-201. Scintigraphic images obtained in humans after the intravenous administration of the drug have been comparable to those obtained with thallous chloride Tl-201 in normal and abnormal myocardial tissue.
Animal studies have shown that myocardial uptake is not blocked when the sodium pump mechanism is inhibited. Although studies of subcellular fractionation and electron micrographic analysis of heart cell aggregates suggest that Tc99m Sestamibi cellular retention occurs specifically within the mitochondria as a result of electrostatic interactions, the clinical relevance of these findings has not been determined.
The mechanism of Tc99m Sestamibi localization in various types of breast tissue (e.g., benign, inflammatory, malignant, fibrous) has not been established.
5. Warnings and Precautions
- Pharmacologic induction of cardiovascular stress may be associated with serious adverse events such as myocardial infarction, arrhythmia, hypotension, bronchoconstriction and cerebrovascular events.
- CARDIOLITE® has been rarely associated with acute severe allergic and anaphylactic events of angioedema and generalized urticaria. In some patients the allergic symptoms developed on the second injection during CARDIOLITE® imaging.
- Caution should be exercised and emergency equipment should be available when administering CARDIOLITE®.
- Before administering CARDIOLITE® patients should be asked about the possibility of allergic reactions to either drug.
- The contents of the vial are intended only for use in the preparation of Technetium Tc99m Sestamibi and are not to be administered directly to the patient without first undergoing the preparative procedure.
2. Dosage and Administration
- For Myocardial Imaging: The suggested dose range for I.V. administration of CARDIOLITE® in a single dose to be employed in the average patient (70 Kg) is 370 - 1110 MBq (10 - 30 mCi).
- For Breast Imaging: The recommended dose range for I.V. administration of MIRALUMA® is a single dose of 740 - 1110 MBq (20 - 30 mCi).
11.1 Physical Characteristics
Technetium Tc99m decays by isomeric transition with a physical half-life of 6.02 hours
| Radiation | Mean %/Disintegration | Mean Energy (KeV) |
|---|---|---|
| Gamma -2 | 89.07 | 140.5 |
3. Dosage Forms and Strengths
CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection is supplied as a lyophilized mixture in a 5 mL vial.
8. Use in Specific Populations
- In one study of 46 subjects who received CARDIOLITE® administration, the radioactivity in both children and adolescents exhibited blood PK profiles similar to those previously reported in adults.
- Lactation: Interruption of breastfeeding after exposure to Technetium Tc99m Sestamibi is not necessary, however, a lactating woman should be advised to consider restricting close contact with her breast fed infant to a maximum of 5 hours in the 24 hour period after Technetium Tc99m Sestamibi administration in order to minimize radiation exposure. (8.2)
2.3 Instructions for Preparation
Preparation of the Technetium Tc99m Sestamibi from the Kit for the Preparation of Technetium Tc99m Sestamibi is done by the following aseptic procedure:
17. Patient Counseling Information
CARDIOLITE® and MIRALUMA® are different names for the same drug. Patients should be advised to inform their health care provider if they had an allergic reaction to either drug or if they had an imaging study with either drug.
Principal Display Panel 5 Vial Box
Lantheus
Medical Imaging
®
Cardiolite ®
Kit for the Preparation of Technetium Tc99m Sestamibi for Injection
CONTENTS AND STORAGE CONDITIONS:
1 Package Insert, 12 Radiation Labels and 5 Vials, each containing:
Tetrakis (2-methoxy isobutyl isonitrile) Copper (I) tetrafluoroborate - 1.0 mg
Stannous Chloride Dihydrate - 0.075 mg; L-Cysteine Hydrochloride
Monohydrate - 1.0 mg; Sodium Citrate Dihydrate - 2.6 mg; Mannitol - 20 mg
Store at 15-25°C (59-77°F).
CONTAINS NO PRESERVATIVE. See Package Insert for dosage information.
Reconstitute with additive-free Tc99m and store at 15-25°C (59-77°F).
Use within 6 hours of reconstitution.
16. How Supplied/storage and Handling
CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection is supplied as a 5 mL vial in kits of five (5) vials (NDC # 11994-001-55) and twenty (20) vials (NDC # 11994-001-20), sterile and non-pyrogenic.
The patient dose should be measured by a suitable radioactivity calibration system immediately prior to patient administration. Radiochemical purity should be checked prior to patient administration.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Prior to lyophilization the pH is between 5.3-5.9. The contents of the vial are lyophilized and stored under nitrogen. Store at 15-25°C (59-77° F) before and after reconstitution.
Technetium Tc99m Sestamibi contains no preservatives. Included in each five (5) vial kit is one (1) package insert, six (6) vial shield labels and six (6) radiation warning labels. Included in each twenty (20) vial kit is one (1) package insert, twenty four (24) vial shield labels and twenty four (24) radiation warning labels.
This reagent kit is approved for distribution to persons licensed pursuant to the Code of Massachusetts Regulations 105 CMR 120.500 for the uses listed in 105 CMR 120.547 or 120.552, or under equivalent regulations of the U.S. Nuclear Regulatory Commission, Agreement States or Licensing States.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In comparison with most other diagnostic technetium labeled radiopharmaceuticals, the radiation dose to the ovaries (1.5 rads/30 mCi at rest, 1.2 rads/30 mCi at exercise) is high. Minimal exposure (ALARA) is necessary in women of childbearing capability. (See Section 2.)
The active intermediate, Cu(MIBI)4BF4, was evaluated for genotoxic potential in a battery of five tests. No genotoxic activity was observed in the Ames, CHO/HPRT and sister chromatid exchange tests (all in vitro). At cytotoxic concentrations (> 20 µg/mL), an increase in cells with chromosome aberrations was observed in the in vitro human lymphocyte assay. Cu(MIBI)4BF4 did not show genotoxic effects in the in vivo mouse micronucleus test at a dose which caused systemic and bone marrow toxicity (9 mg/kg, > 600 × maximal human dose).
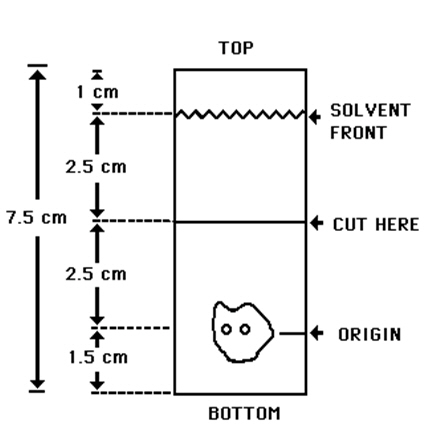
2.4 Determination of Radiochemical Purity in Technetium Tc99m Sestamibi
- Obtain a Baker-Flex Aluminum Oxide coated, plastic TLC plate, #1 B-F, pre-cut to 2.5 cm × 7.5 cm.
- Dry the plate or plates at 100°C for 1 hour and store in a desiccator. Remove pre-dried plate from the desiccator just prior to use.
- Apply 1 drop of ethanolThe ethanol used in this procedure should be 95% or greater. Absolute ethanol (99%) should remain at ≥ 95% ethanol content for one week after opening if stored tightly capped, in a cool dry place.using a 1 mL syringe with a 22-26 gauge needle, 1.5 cm from the bottom of the plate. THE SPOT SHOULD NOT BE ALLOWED TO DRY.
- Add 2 drops of Technetium Tc99m Sestamibi solution, side by side on top of the ethanol spot. Return the plate to a desiccator and allow the sample spot to dry (typically 15 minutes).
- The TLC tank is prepared by pouring ethanol to a depth of 3-4 mm. Cover the tank and let it equilibrate for ~10 minutes.
- Develop the plate in the covered TLC tank in ethanol for a distance of 5 cm from the point of application.
- Cut the TLC plate 4 cm from the bottom and measure the Tc99m activity in each piece by appropriate radiation detector.
- Calculate the % Tc99m Sestamibi as:
% Tc99m Sestamibi = µCi Top Piece × 100 µCi Both Pieces
| Figure 1.0 TLC Plate Diagram |
Structured Label Content
Section 42229-5 (42229-5)
Myocardial Imaging: CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection, is a myocardial perfusion agent that is indicated for detecting coronary artery disease by localizing myocardial ischemia (reversible defects) and infarction (non-reversible defects), in evaluating myocardial function and developing information for use in patient management decisions. CARDIOLITE® evaluation of myocardial ischemia can be accomplished with rest and cardiovascular stress techniques (e.g., exercise or pharmacologic stress in accordance with the pharmacologic stress agent's labeling).
It is usually not possible to determine the age of a myocardial infarction or to differentiate a recent myocardial infarction from ischemia.
5.1 Warnings
In studying patients in whom cardiac disease is known or suspected, care should be taken to assure continuous monitoring and treatment in accordance with safe, accepted clinical procedure. Infrequently, death has occurred 4 to 24 hours after Tc99m Sestamibi use and is usually associated with exercise stress testing (See Section 5.2 ).
Pharmacologic induction of cardiovascular stress may be associated with serious adverse events such as myocardial infarction, arrhythmia, hypotension, bronchoconstriction and cerebrovascular events. Caution should be used when pharmacologic stress is selected as an alternative to exercise; it should be used when indicated and in accordance with the pharmacologic stress agent's labeling.
Technetium Tc99m Sestamibi has been rarely associated with acute severe allergic and anaphylactic events of angioedema and generalized urticaria. In some patients the allergic symptoms developed on the second injection during CARDIOLITE® imaging. Patients who receive CARDIOLITE® or MIRALUMA® imaging are receiving the same drug. Caution should be exercised and emergency equipment should be available when administering Technetium Tc99m Sestamibi. Also, before administering either CARDIOLITE® or MIRALUMA®, patients should be asked about the possibility of allergic reactions to either drug.
10. Overdosage (10. OVERDOSAGE)
The clinical consequences of overdosing with CARDIOLITE® are not known.
11. Description (11. DESCRIPTION)
Each 5 mL vial contains a sterile, non-pyrogenic, lyophilized mixture of:
- Tetrakis (2-methoxy isobutyl isonitrile) Copper (I) tetrafluoroborate - 1.0 mg
- Sodium Citrate Dihydrate - 2.6 mg
- L-Cysteine Hydrochloride Monohydrate - 1.0 mg
- Mannitol – 20 mg
- Stannous Chloride, Dihydrate, minimum (SnCl2∙2H2O) - 0.025 mg
- Stannous Chloride, Dihydrate, (SnCl2∙2H2O) - 0.075 mg
- Tin Chloride (stannous and stannic) Dihydrate, maximum (as SnCl2∙2H2O) - 0.086 mg
Prior to lyophilization the pH is 5.3 to 5.9. The contents of the vial are lyophilized and stored under nitrogen.
This drug is administered by intravenous injection for diagnostic use after reconstitution with sterile, non-pyrogenic, oxidant-free Sodium Pertechnetate Tc99m Injection. The pH of the reconstituted product is 5.5 (5.0 - 6.0). No bacteriostatic preservative is present.
The precise structure of the technetium complex is Tc99m[MIBI]6 + where MIBI is 2-methoxy isobutyl isonitrile.
8.4 Pediatric Use
Safety and effectiveness in the pediatric population have not been established.
No evidence of diagnostic efficacy or clinical utility of CARDIOLITE® scan was found in clinical studies of children and adolescents with Kawasaki disease.
A prospective study of 445 pediatric patients with Kawasaki disease was designed to determine the predictive value of CARDIOLITE® rest and stress myocardial perfusion imaging to define a pediatric population with Kawasaki disease that was at risk of developing cardiac events. Cardiac events were defined as cardiac death, MI, hospitalization due to cardiac etiology, heart failure, CABG or coronary angioplasty. The standard of truth was defined as cardiac events occurring 6 months following the administration of CARDIOLITE®. Only three cardiac events were observed at six months in this study. In all three cases, the scan was negative. No clinically meaningful measurements of sensitivity, specificity or other diagnostic performance parameters could be demonstrated in this study.
A ten year retrospective case history study of pediatric Kawasaki disease patients who completed CARDIOLITE® myocardial perfusion imaging and who had coronary angiography within three months of the CARDIOLITE® scan was designed to measure sensitivity and specificity of CARDIOLITE® scan. Out of 72 patients who had both evaluable CARDIOLITE® scans and evaluable angiographic images, only one patient had both an abnormal angiogram and an abnormal CARDIOLITE® scan. No clinically meaningful measurements of sensitivity, specificity or other diagnostic performance parameters could be demonstrated in this study.
In a clinical pharmacology study, 46 pediatric patients with Kawasaki disease received CARDIOLITE® administration at the following doses: 0.1 - 0.2 mCi/kg for rest, 0.3 mCi/kg for stress in one day studies; 0.2 mCi/kg for rest and 0.2 mCi/kg for stress in two day studies.
The radioactivity both in younger children and in adolescents exhibited PK profiles similar to those previously reported in adults (See Section 12).
The radiation absorbed doses in adolescents, both at rest and stress, were similar to those observed in adults (see Section 2). When comparing weight-adjusted radioactivity (up to 0.3 mCi/kg) doses administered to adolescents and younger children to the recommended dose administered to adults (up to 30 mCi), the radiation absorbed doses in both adolescents and younger children were similar to those in adults.
Adverse events were evaluated in 609 pediatric patients from the three clinical studies described above. The frequency and the type of the adverse events were similar to the ones observed in the studies of CARDIOLITE® in adults. Two of the 609 had a serious adverse event: one patient received a CARDIOLITE® overdose but remained asymptomatic, and one patient had an asthma exacerbation following administration.
8.5 Geriatric Use
Of 3068 patients in clinical studies of CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection, 693 patients were 65 or older and 121 were 75 or older.
Of 673 patients in clinical studies of MIRALUMA®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection, 138 patients were 65 or older and 30 were 75 or older.
Based on the evaluation of the frequency of adverse events and review of vital signs data, no overall differences in safety were observed between these subjects and younger subjects. Although reported clinical experience has not identified differences in response between elderly and younger patients, greater sensitivity of some older individuals cannot be ruled out.
4. Contraindications (4. CONTRAINDICATIONS)
None known.
6. Adverse Reactions (6. ADVERSE REACTIONS)
Adverse events were evaluated in 3741 adults who were evaluated in clinical studies. Of these patients, 3068 (77% men, 22% women, and 0.7% of the patient's genders were not recorded) were in cardiac clinical trials and 673 (100% women) in breast imaging trials. Cases of angina, chest pain, and death have occurred (see Section 5 ). Adverse events reported at a rate of 0.5% or greater after receiving Technetium Tc99m Sestamibi administration are shown in the following table:
| Body System | Breast Studies | Cardiac Studies | ||
|---|---|---|---|---|
| Women n = 673 |
Women n = 685 |
Men n = 2361 |
Total n = 3046 |
|
| Body as a Whole | 21 (3.1%) | 6 (0.9%) | 17 (0.7%) | 23 (0.8%) |
| Headache | 11 (1.6%) | 2 (0.3%) | 4 (0.2%) | 6 (0.2%) |
| Cardiovascular | 9 (1.3%) | 24 (3.5%) | 75 (3.2%) | 99 (3.3%) |
| Chest Pain/Angina | 0 (0%) | 18 (2.6%) | 46 (1.9%) | 64 (2.1%) |
| ST segment changes | 0 (0%) | 11 (1.6%) | 29 (1.2%) | 40 (1.3%) |
| Digestive System | 8 (1.2%) | 4 (0.6%) | 9 (0.4%) | 13 (0.4%) |
| Nausea | 4 (0.6%) | 1 (0.1%) | 2 (0.1%) | 3 (0.1%) |
| Special Senses | 132 (19.6%) | 62 (9.1%) | 160 (6.8%) | 222 (7.3%) |
| Taste Perversion | 129 (19.2%) | 60 (8.8%) | 157 (6.6%) | 217 (7.1%) |
| Parosmia | 8 (1.2%) | 6 (0.9%) | 10 (0.4%) | 16 (0.5%) |
In the clinical studies for breast imaging, breast pain was reported in 12 (1.7%) of the patients. In 11 of these patients the pain appears to be associated with biopsy/surgical procedures.
The following adverse reactions have been reported in ≤ 0.5% of patients: signs and symptoms consistent with seizure occurring shortly after administration of the agent; transient arthritis, angioedema, arrythmia, dizziness, syncope, abdominal pain, vomiting, and severe hypersensitivity characterized by dyspnea, hypotension, bradycardia, asthenia, and vomiting within two hours after a second injection of Technetium Tc99m Sestamibi. A few cases of flushing, edema, injection site inflammation, dry mouth, fever, pruritis, rash, urticaria and fatigue have also been attributed to administration of the agent.
7. Drug Interactions (7. DRUG INTERACTIONS)
Specific drug-drug interactions have not been studied.
12.3 Pharmacokinetics
Pulmonary activity is negligible even immediately after injection. Blood clearance studies indicate that the fast clearing component clears with a t1/2 of 4.3 minutes at rest, and clears with a t1/2 of 1.6 minutes under exercise conditions. At five minutes post injection about 8% of the injected dose remains in circulation. There is less than 1% protein binding of Technetium Tc99m Sestamibi in plasma. The myocardial biological half-life is approximately six hours after a rest or exercise injection. The biological half-life for the liver is approximately 30 minutes after a rest or exercise injection. The effective half-life of clearance (which includes both the biological half-life and radionuclide decay) for the heart is approximately 3 hours, and for the liver is approximately 30 minutes, after a rest or exercise injection. The ideal imaging time reflects the best compromise between heart count rate and surrounding organ uptake.
Myocardial uptake which is coronary flow dependent is 1.2% of the injected dose at rest and 1.5% of the injected dose at exercise. Table 6.0 illustrates the biological clearance as well as effective clearance (which includes biological clearance and radionuclide decay) of Tc99m Sestamibi from the heart and liver.
[Organ concentrations expressed as percentage of injected dose; data based on an average of 5 subjects at rest and 5 subjects during exercise].
| REST | STRESS | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Heart | Liver | Heart | Liver | ||||||
| Time | Biological | Effective | Biological | Effective | Biological | Effective | Biological | Effective | |
| 5 min. | 1.2 | 1.2 | 19.6 | 19.4 | 1.5 | 1.5 | 5.9 | 5.8 | |
| 30 min. | 1.1 | 1.0 | 12.2 | 11.5 | 1.4 | 1.3 | 4.5 | 4.2 | |
| 1 hour | 1.0 | 0.9 | 5.6 | 5.0 | 1.4 | 1.2 | 2.4 | 2.1 | |
| 2 hours | 1.0 | 0.8 | 2.2 | 1.7 | 1.2 | 1.0 | 0.9 | 0.7 | |
| 4 hours | 0.8 | 0.5 | 0.7 | 0.4 | 1.0 | 0.6 | 0.3 | 0.2 |
A study in a dog myocardial ischemia model reported that Technetium Tc99m Sestamibi undergoes myocardial distribution (redistribution), although more slowly and less completely than thallous chloride Tl-201. A study in a dog myocardial infarction model reported that the drug showed no redistribution of any consequence. Definitive human studies to demonstrate possible redistribution have not been reported. In patients with documented myocardial infarction, imaging revealed the infarct up to four hours post dose.
11.2 External Radiation
The specific gamma ray constant for Tc99m is 5.4 microcoulombs/Kg-MBq-hr (0.78R/mCi-hr) at 1 cm. The first half value layer is 0.017 cm of Pb. A range of values for the relative attenuation of the radiation emitted by this radionuclide that results from interposition of various thicknesses of Pb is shown in Table 4.0. To facilitate control of the radiation exposure from Megabequerel (millicurie) amounts of this radionuclide, the use of a 0.25 cm thickness of Pb will attenuate the radiation emitted by a factor of 1,000.
| Shield Thickness (Pb) cm | Coefficient of Attenuation |
|---|---|
| 0.017 | 0.5 |
| 0.08 | 10-1 |
| 0.16 | 10-2 |
| 0.25 | 10-3 |
| 0.33 | 10-4 |
To correct for physical decay of this radionuclide, the fractions that remain at selected intervals after the time of calibration are shown in Table 5.0.
| Hours | Fraction Remaining | Hours | Fraction Remaining |
|---|---|---|---|
| 0 Calibration Time
|
1.000 | 8 | .398 |
| 1 | .891 | 9 | .355 |
| 2 | .794 | 10 | .316 |
| 3 | .708 | 11 | .282 |
| 4 | .631 | 12 | .251 |
| 5 | .562 | ||
| 6 | .501 | ||
| 7 | .447 |
2.2 Radiation Dosimetry
The radiation doses to organs and tissues of an average patient (70 Kg) per 1110 MBq (30 mCi) of Technetium Tc99m Sestamibi injected intravenously are shown in Table 1.0.
| Estimated Radiation Absorbed Dose | ||||
|---|---|---|---|---|
| Radiation dosimetry calculations performed by Radiation Internal Dose Information Center, Oak Ridge Institute for Science and Education, PO Box 117, Oak Ridge, TN 37831-0117. | ||||
| REST | ||||
| 2.0 hour void | 4.8 hour void | |||
| Organ | rads/ 30 mCi |
mGy/ 1110 MBq |
rads/ 30 mCi |
mGy/ 1110 MBq |
| Breasts | 0.2 | 2.0 | 0.2 | 1.9 |
| Gallbladder Wall | 2.0 | 20.0 | 2.0 | 20.0 |
| Small Intestine | 3.0 | 30.0 | 3.0 | 30.0 |
| Upper Large Intestine Wall | 5.4 | 55.5 | 5.4 | 55.5 |
| Lower Large Intestine Wall | 3.9 | 40.0 | 4.2 | 41.1 |
| Stomach Wall | 0.6 | 6.1 | 0.6 | 5.8 |
| Heart Wall | 0.5 | 5.1 | 0.5 | 4.9 |
| Kidneys | 2.0 | 20.0 | 2.0 | 20.0 |
| Liver | 0.6 | 5.8 | 0.6 | 5.7 |
| Lungs | 0.3 | 2.8 | 0.3 | 2.7 |
| Bone Surfaces | 0.7 | 6.8 | 0.7 | 6.4 |
| Thyroid | 0.7 | 7.0 | 0.7 | 7.0 |
| Ovaries | 1.5 | 15.5 | 1.6 | 15.5 |
| Testes | 0.3 | 3.4 | 0.4 | 3.9 |
| Red Marrow | 0.5 | 5.1 | 0.5 | 5.0 |
| Urinary Bladder Wall | 2.0 | 20.0 | 4.2 | 41.1 |
| Total Body | 0.5 | 4.8 | 0.5 | 4.8 |
| STRESS | ||||
| 2.0 hour void | 4.8 hour void | |||
| Organ | rads/ 30 mCi |
mGy/ 1110 MBq |
rads/ 30 mCi |
mGy/ 1110 MBq |
| Breasts | 0.2 | 2.0 | 0.2 | 1.8 |
| Gallbladder Wall | 2.8 | 28.9 | 2.8 | 27.8 |
| Small Intestine | 2.4 | 24.4 | 2.4 | 24.4 |
| Upper Large Intestine Wall | 4.5 | 44.4 | 4.5 | 44.4 |
| Lower Large Intestine Wall | 3.3 | 32.2 | 3.3 | 32.2 |
| Stomach Wall | 0.6 | 5.3 | 0.5 | 5.2 |
| Heart Wall | 0.5 | 5.6 | 0.5 | 5.3 |
| Kidneys | 1.7 | 16.7 | 1.7 | 16.7 |
| Liver | 0.4 | 4.2 | 0.4 | 4.1 |
| Lungs | 0.3 | 2.6 | 0.2 | 2.4 |
| Bone Surfaces | 0.6 | 6.2 | 0.6 | 6.0 |
| Thyroid | 0.3 | 2.7 | 0.2 | 2.4 |
| Ovaries | 1.2 | 12.2 | 1.3 | 13.3 |
| Testes | 0.3 | 3.1 | 0.3 | 3.4 |
| Red Marrow | 0.5 | 4.6 | 0.5 | 4.4 |
| Urinary Bladder Wall | 1.5 | 15.5 | 3.0 | 30.0 |
| Total Body | 0.4 | 4.2 | 0.4 | 4.2 |
5.2 General Precautions
The contents of the vial are intended only for use in the preparation of Technetium Tc99m Sestamibi and are not to be administered directly to the patient without first undergoing the preparative procedure.
Radioactive drugs must be handled with care and appropriate safety measures should be used to minimize radiation exposure to clinical personnel. Also, care should be taken to minimize radiation exposure to the patients consistent with proper patient management.
Contents of the kit before preparation are not radioactive. However, after the Sodium Pertechnetate Tc99m Injection is added, adequate shielding of the final preparation must be maintained. The components of the kit are sterile and non-pyrogenic. It is essential to follow directions carefully and to adhere to strict aseptic procedures during preparation.
Technetium Tc99m labeling reactions depend on maintaining the stannous ion in the reduced state. Hence, Sodium Pertechnetate Tc99m Injection containing oxidants should not be used.
Technetium Tc99m Sestamibi should not be used more than six hours after preparation.
Radiopharmaceuticals should be used only by physicians who are qualified by training and experience in the safe use and handling of radionuclides and whose experience and training have been approved by the appropriate government agency authorized to license the use of radionuclides.
Stress testing should be performed only under the supervision of a qualified physician and in a laboratory equipped with appropriate resuscitation and support apparatus.
The most frequent exercise stress test endpoints sufficient to stop the test reported during controlled studies (two-thirds were cardiac patients) were:
| Fatigue | 35% |
| Dyspnea | 17% |
| Chest Pain | 16% |
| ST-depression | 7% |
| Arrhythmia | 1% |
1. Indications and Usage (1. INDICATIONS AND USAGE)
CARDIOLITE® is a myocardial perfusion agent indicated for:
- detecting coronary artery disease by localizing myocardial ischemia (reversible defects) and infarction (non-reversible defects)
- evaluating myocardial function and developing information for use in patient management decisions
12.1 Mechanism of Action
Technetium Tc99m Sestamibi is a cationic Tc99m complex which has been found to accumulate in viable myocardial tissue in a manner analogous to that of thallous chloride Tl-201. Scintigraphic images obtained in humans after the intravenous administration of the drug have been comparable to those obtained with thallous chloride Tl-201 in normal and abnormal myocardial tissue.
Animal studies have shown that myocardial uptake is not blocked when the sodium pump mechanism is inhibited. Although studies of subcellular fractionation and electron micrographic analysis of heart cell aggregates suggest that Tc99m Sestamibi cellular retention occurs specifically within the mitochondria as a result of electrostatic interactions, the clinical relevance of these findings has not been determined.
The mechanism of Tc99m Sestamibi localization in various types of breast tissue (e.g., benign, inflammatory, malignant, fibrous) has not been established.
5. Warnings and Precautions (5. WARNINGS AND PRECAUTIONS)
- Pharmacologic induction of cardiovascular stress may be associated with serious adverse events such as myocardial infarction, arrhythmia, hypotension, bronchoconstriction and cerebrovascular events.
- CARDIOLITE® has been rarely associated with acute severe allergic and anaphylactic events of angioedema and generalized urticaria. In some patients the allergic symptoms developed on the second injection during CARDIOLITE® imaging.
- Caution should be exercised and emergency equipment should be available when administering CARDIOLITE®.
- Before administering CARDIOLITE® patients should be asked about the possibility of allergic reactions to either drug.
- The contents of the vial are intended only for use in the preparation of Technetium Tc99m Sestamibi and are not to be administered directly to the patient without first undergoing the preparative procedure.
2. Dosage and Administration (2. DOSAGE AND ADMINISTRATION)
- For Myocardial Imaging: The suggested dose range for I.V. administration of CARDIOLITE® in a single dose to be employed in the average patient (70 Kg) is 370 - 1110 MBq (10 - 30 mCi).
- For Breast Imaging: The recommended dose range for I.V. administration of MIRALUMA® is a single dose of 740 - 1110 MBq (20 - 30 mCi).
11.1 Physical Characteristics
Technetium Tc99m decays by isomeric transition with a physical half-life of 6.02 hours
| Radiation | Mean %/Disintegration | Mean Energy (KeV) |
|---|---|---|
| Gamma -2 | 89.07 | 140.5 |
3. Dosage Forms and Strengths (3. DOSAGE FORMS AND STRENGTHS)
CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection is supplied as a lyophilized mixture in a 5 mL vial.
8. Use in Specific Populations (8. USE IN SPECIFIC POPULATIONS)
- In one study of 46 subjects who received CARDIOLITE® administration, the radioactivity in both children and adolescents exhibited blood PK profiles similar to those previously reported in adults.
- Lactation: Interruption of breastfeeding after exposure to Technetium Tc99m Sestamibi is not necessary, however, a lactating woman should be advised to consider restricting close contact with her breast fed infant to a maximum of 5 hours in the 24 hour period after Technetium Tc99m Sestamibi administration in order to minimize radiation exposure. (8.2)
2.3 Instructions for Preparation (2.3 Instructions For Preparation)
Preparation of the Technetium Tc99m Sestamibi from the Kit for the Preparation of Technetium Tc99m Sestamibi is done by the following aseptic procedure:
17. Patient Counseling Information (17. PATIENT COUNSELING INFORMATION)
CARDIOLITE® and MIRALUMA® are different names for the same drug. Patients should be advised to inform their health care provider if they had an allergic reaction to either drug or if they had an imaging study with either drug.
Principal Display Panel 5 Vial Box (PRINCIPAL DISPLAY PANEL - 5 Vial Box)
Lantheus
Medical Imaging
®
Cardiolite ®
Kit for the Preparation of Technetium Tc99m Sestamibi for Injection
CONTENTS AND STORAGE CONDITIONS:
1 Package Insert, 12 Radiation Labels and 5 Vials, each containing:
Tetrakis (2-methoxy isobutyl isonitrile) Copper (I) tetrafluoroborate - 1.0 mg
Stannous Chloride Dihydrate - 0.075 mg; L-Cysteine Hydrochloride
Monohydrate - 1.0 mg; Sodium Citrate Dihydrate - 2.6 mg; Mannitol - 20 mg
Store at 15-25°C (59-77°F).
CONTAINS NO PRESERVATIVE. See Package Insert for dosage information.
Reconstitute with additive-free Tc99m and store at 15-25°C (59-77°F).
Use within 6 hours of reconstitution.
16. How Supplied/storage and Handling (16. HOW SUPPLIED/STORAGE AND HANDLING)
CARDIOLITE®, Kit for the Preparation of Technetium Tc99m Sestamibi for Injection is supplied as a 5 mL vial in kits of five (5) vials (NDC # 11994-001-55) and twenty (20) vials (NDC # 11994-001-20), sterile and non-pyrogenic.
The patient dose should be measured by a suitable radioactivity calibration system immediately prior to patient administration. Radiochemical purity should be checked prior to patient administration.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Prior to lyophilization the pH is between 5.3-5.9. The contents of the vial are lyophilized and stored under nitrogen. Store at 15-25°C (59-77° F) before and after reconstitution.
Technetium Tc99m Sestamibi contains no preservatives. Included in each five (5) vial kit is one (1) package insert, six (6) vial shield labels and six (6) radiation warning labels. Included in each twenty (20) vial kit is one (1) package insert, twenty four (24) vial shield labels and twenty four (24) radiation warning labels.
This reagent kit is approved for distribution to persons licensed pursuant to the Code of Massachusetts Regulations 105 CMR 120.500 for the uses listed in 105 CMR 120.547 or 120.552, or under equivalent regulations of the U.S. Nuclear Regulatory Commission, Agreement States or Licensing States.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In comparison with most other diagnostic technetium labeled radiopharmaceuticals, the radiation dose to the ovaries (1.5 rads/30 mCi at rest, 1.2 rads/30 mCi at exercise) is high. Minimal exposure (ALARA) is necessary in women of childbearing capability. (See Section 2.)
The active intermediate, Cu(MIBI)4BF4, was evaluated for genotoxic potential in a battery of five tests. No genotoxic activity was observed in the Ames, CHO/HPRT and sister chromatid exchange tests (all in vitro). At cytotoxic concentrations (> 20 µg/mL), an increase in cells with chromosome aberrations was observed in the in vitro human lymphocyte assay. Cu(MIBI)4BF4 did not show genotoxic effects in the in vivo mouse micronucleus test at a dose which caused systemic and bone marrow toxicity (9 mg/kg, > 600 × maximal human dose).
2.4 Determination of Radiochemical Purity in Technetium Tc99m Sestamibi
- Obtain a Baker-Flex Aluminum Oxide coated, plastic TLC plate, #1 B-F, pre-cut to 2.5 cm × 7.5 cm.
- Dry the plate or plates at 100°C for 1 hour and store in a desiccator. Remove pre-dried plate from the desiccator just prior to use.
- Apply 1 drop of ethanolThe ethanol used in this procedure should be 95% or greater. Absolute ethanol (99%) should remain at ≥ 95% ethanol content for one week after opening if stored tightly capped, in a cool dry place.using a 1 mL syringe with a 22-26 gauge needle, 1.5 cm from the bottom of the plate. THE SPOT SHOULD NOT BE ALLOWED TO DRY.
- Add 2 drops of Technetium Tc99m Sestamibi solution, side by side on top of the ethanol spot. Return the plate to a desiccator and allow the sample spot to dry (typically 15 minutes).
- The TLC tank is prepared by pouring ethanol to a depth of 3-4 mm. Cover the tank and let it equilibrate for ~10 minutes.
- Develop the plate in the covered TLC tank in ethanol for a distance of 5 cm from the point of application.
- Cut the TLC plate 4 cm from the bottom and measure the Tc99m activity in each piece by appropriate radiation detector.
- Calculate the % Tc99m Sestamibi as:
% Tc99m Sestamibi = µCi Top Piece × 100 µCi Both Pieces
| Figure 1.0 TLC Plate Diagram |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:31.541400 · Updated: 2026-03-14T21:50:37.524392