drug facts
48ffdad3-1e91-6480-e054-00144ff88e88
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Niacinamide, Adenosine
Medication Information
Warnings and Precautions
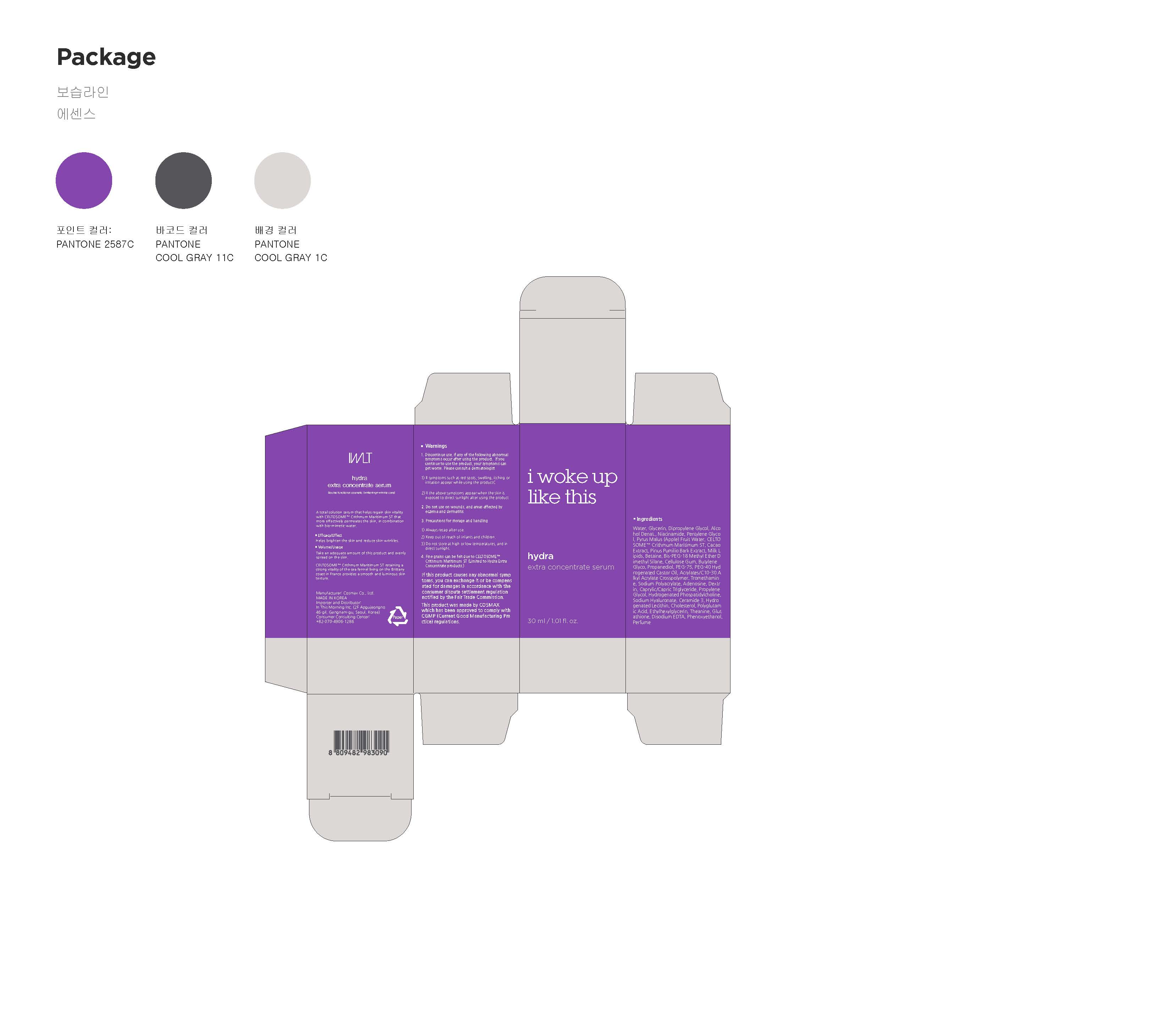
1. Discontinue use, if any of the following abnormal symptoms
occur after using the product. If you continue to use the
product, your symptoms can get worse. Please consult a
dermatologist.
1) If symptoms such as red spots, swelling, itching or irritation appear while
using the product
2) If the above symptoms appear when the skin is exposed to direct sunlight
after using the product
2. Do not use on wounds, and areas affected by eczema and
dermatitis
3. Precautions for storage and handling
1) Always recap after use.
2) Keep out of reach of infants and children.
3) Do not store at high or low temperatures, and in direct sunlight.
4. Avoid contact with the eyes.
- If you are very sensitive to a sticking plaster or a wet compress, use with
caution or discontinue use
- Limited to Whitening Concentrate Treatment Mask products.
- Fine grains can be felt due to CELTOSOME™ Crithmum Maritimum ST (Limited
to Hydra Extra Concentrate products.)
5. The SPF of this product was measured according to the
method of Cosmmedics Europe and its PA according to the
method of the Japan Cosmetic Industry Association (JCIA).
(Limited to Protective Base Sunscreen products for dry skin)
6. The SPF of this product was measured according to the
International SPF Test Method and its PA according to the
method of the Japan Cosmetic Industry Association (JCIA).
(Limited to Protective Base Sunscreen products for oily skin)
7. The SPF and PA of this product were measured according
to the methods proposed by the Regulations of Functional
Cosmetic Evaluation. (Limited to BB cream.)
8. Do not use on children under the age of 3 because this
product contains salicylic acids and salts.
(Limited to Purifying Sebum Relief Spot products.)
If this product causes any abnormal symptoms,
you can exchange it or be compensated for damages in
accordance with the consumer dispute settlement regulation
notified by the Fair Trade Commission.
This product was made by COSMAX
which has been approved to comply with
CGMP (Current Good Manufacturing Practice) regulations.
Active Ingredient
Niacinamide, Adenosine
Indications and Usage
Take an adequate amount of this product and evenly spread on the skin.
Dosage and Administration
for external use only
Description
Niacinamide, Adenosine
Section 50565-1
keep out or reach of the children
Section 51727-6
Water, Glycerin, Butylene Glycol, Crithmum Maritimum Callus Culture Filtrate, etc.
Section 51945-4
Section 55105-1
Helps brighten the skin and reduce skin wrinkles.
Structured Label Content
Warnings and Precautions (34071-1)
1. Discontinue use, if any of the following abnormal symptoms
occur after using the product. If you continue to use the
product, your symptoms can get worse. Please consult a
dermatologist.
1) If symptoms such as red spots, swelling, itching or irritation appear while
using the product
2) If the above symptoms appear when the skin is exposed to direct sunlight
after using the product
2. Do not use on wounds, and areas affected by eczema and
dermatitis
3. Precautions for storage and handling
1) Always recap after use.
2) Keep out of reach of infants and children.
3) Do not store at high or low temperatures, and in direct sunlight.
4. Avoid contact with the eyes.
- If you are very sensitive to a sticking plaster or a wet compress, use with
caution or discontinue use
- Limited to Whitening Concentrate Treatment Mask products.
- Fine grains can be felt due to CELTOSOME™ Crithmum Maritimum ST (Limited
to Hydra Extra Concentrate products.)
5. The SPF of this product was measured according to the
method of Cosmmedics Europe and its PA according to the
method of the Japan Cosmetic Industry Association (JCIA).
(Limited to Protective Base Sunscreen products for dry skin)
6. The SPF of this product was measured according to the
International SPF Test Method and its PA according to the
method of the Japan Cosmetic Industry Association (JCIA).
(Limited to Protective Base Sunscreen products for oily skin)
7. The SPF and PA of this product were measured according
to the methods proposed by the Regulations of Functional
Cosmetic Evaluation. (Limited to BB cream.)
8. Do not use on children under the age of 3 because this
product contains salicylic acids and salts.
(Limited to Purifying Sebum Relief Spot products.)
If this product causes any abnormal symptoms,
you can exchange it or be compensated for damages in
accordance with the consumer dispute settlement regulation
notified by the Fair Trade Commission.
This product was made by COSMAX
which has been approved to comply with
CGMP (Current Good Manufacturing Practice) regulations.
Active Ingredient (55106-9)
Niacinamide, Adenosine
Indications and Usage (34067-9)
Take an adequate amount of this product and evenly spread on the skin.
Dosage and Administration (34068-7)
for external use only
Section 50565-1 (50565-1)
keep out or reach of the children
Section 51727-6 (51727-6)
Water, Glycerin, Butylene Glycol, Crithmum Maritimum Callus Culture Filtrate, etc.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Helps brighten the skin and reduce skin wrinkles.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:16.071015 · Updated: 2026-03-14T22:54:38.033285