48fd73e7-1443-4ac7-bf7c-d99bf2449f51
34390-5
HUMAN OTC DRUG LABEL
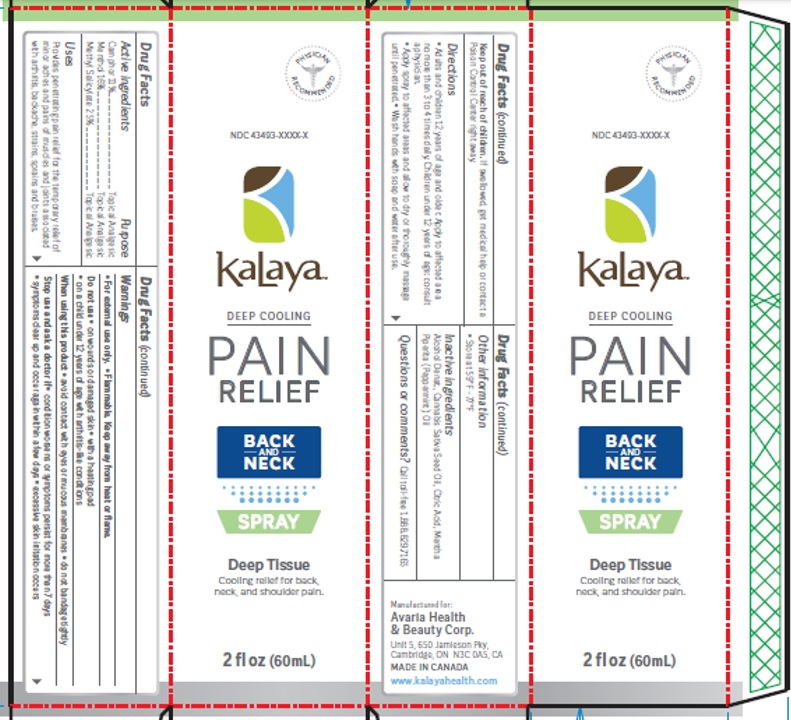
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Camphor 11% Menthol 16% Methyl salicylate 25%
Purpose
Topical Analgesic
Medication Information
Purpose
Topical Analgesic
Description
Camphor 11% Menthol 16% Methyl salicylate 25%
Uses
Provides penetrating pain relief for the temporary relief of minor aches and pains of muscles and joints
Section 50565-1
Warnings
For External Use Only. Flammable. Keep away from heat or flame.
Do not use:
• on wounds or damaged skin
• with a heating pad
• on a child under 12 years of age with arthritis-like conditions
When using this product avoid contact with eyes or mucous membranes, do not bandage tightly.
Stop use and ask a doctor if condition worsen or symptoms persist more than 7 days, symptoms clear up and occur again within a few days and/or excessive skin irritation occurs.
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away
Directions
Adults and children 12 years and over: Spray evenly onto affected areas no more than 3 to 4 times daily. Apply spray to affected areas and
allowed to dry or thoroughly massage until penetrated. Wash hands with soap and water after use.
Drug Facts
Product Label
Other Information
• Store at 59°F - 77°F
Active Ingredients
Camphor 11%
Menthol 16%
Methyl salicylate 25%
Inactive Ingredients
Alcohol Denat., Cannabis Sativa Seed Oil, Citric Acid, Mentha Piperita (Peppermint) Oil
Questions Or Comments?
Call toll-free 1.888.829.7165
Structured Label Content
Uses
Provides penetrating pain relief for the temporary relief of minor aches and pains of muscles and joints
Section 50565-1 (50565-1)
Purpose
Topical Analgesic
Warnings
For External Use Only. Flammable. Keep away from heat or flame.
Do not use:
• on wounds or damaged skin
• with a heating pad
• on a child under 12 years of age with arthritis-like conditions
When using this product avoid contact with eyes or mucous membranes, do not bandage tightly.
Stop use and ask a doctor if condition worsen or symptoms persist more than 7 days, symptoms clear up and occur again within a few days and/or excessive skin irritation occurs.
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away
Directions
Adults and children 12 years and over: Spray evenly onto affected areas no more than 3 to 4 times daily. Apply spray to affected areas and
allowed to dry or thoroughly massage until penetrated. Wash hands with soap and water after use.
Drug Facts
Product Label (Product label)
Other Information (Other information)
• Store at 59°F - 77°F
Active Ingredients (Active ingredients)
Camphor 11%
Menthol 16%
Methyl salicylate 25%
Inactive Ingredients (Inactive ingredients)
Alcohol Denat., Cannabis Sativa Seed Oil, Citric Acid, Mentha Piperita (Peppermint) Oil
Questions Or Comments? (Questions or comments?)
Call toll-free 1.888.829.7165
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:28.340910 · Updated: 2026-03-14T23:12:59.392851