Obagi Nu-derm®
48a6f977-d7b8-4da4-8bab-b3b8df5347c3
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
OBAGI NU-DERM ® Rx only For external use only
Indications and Usage
For the gradual bleaching of hyperpigmented skin conditions such as chloasma, melasma, freckles, senile lentigines and other unwanted areas of melanin hyperpigmentation.
Dosage and Administration
A thin application should be applied once or twice daily or as directed by a physician. If no improvement is seen after three (3) months of treatment, use of this product should be discontinued. Sun exposure should be limited by using a sunscreen agent or protective clothing to cover bleached skin when using and after using this product in order to prevent darkening from reoccurring.
Contraindications
People with prior history of sensitivity or allergic reaction to this product or any of its ingredients should not use it. The safety of topical hydroquinone use during pregnancy or in children (12 years and under) has not been established.
Adverse Reactions
No systemic adverse reactions have been reported. Occasional hypersensitivity (localized contact dermatitis) may occur, in which case the product should be discontinued and the physician notified immediately. To report SUSPECTED ADVERSE REACTIONS, contact Obagi Cosmeceuticals LLC, at 1-800-636-7546 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
Obagi Nu-Derm ® Clear is available as follows: Net wt. 2 oz. (57 g) bottle NDC 62032-101-36 Obagi Nu-Derm Blender ® is available as follows: Net wt. 2 oz. (57 g) bottle NDC 62032-100-36 Net wt. 1 oz. (28 g) bottle NDC 62032-100-10 Obagi Nu-Derm Sunfader ® is available as follows: Net wt. 2 oz. (57 g) bottle NDC 62032-116-36
Medication Information
Indications and Usage
For the gradual bleaching of hyperpigmented skin conditions such as chloasma, melasma, freckles, senile lentigines and other unwanted areas of melanin hyperpigmentation.
Dosage and Administration
A thin application should be applied once or twice daily or as directed by a physician. If no improvement is seen after three (3) months of treatment, use of this product should be discontinued. Sun exposure should be limited by using a sunscreen agent or protective clothing to cover bleached skin when using and after using this product in order to prevent darkening from reoccurring.
Contraindications
People with prior history of sensitivity or allergic reaction to this product or any of its ingredients should not use it. The safety of topical hydroquinone use during pregnancy or in children (12 years and under) has not been established.
Adverse Reactions
No systemic adverse reactions have been reported. Occasional hypersensitivity (localized contact dermatitis) may occur, in which case the product should be discontinued and the physician notified immediately.
To report SUSPECTED ADVERSE REACTIONS, contact Obagi Cosmeceuticals LLC, at 1-800-636-7546 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
Obagi Nu-Derm® Clear is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-101-36
Obagi Nu-Derm Blender®
is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-100-36
Net wt. 1 oz. (28 g) bottle
NDC 62032-100-10
Obagi Nu-Derm Sunfader®
is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-116-36
Description
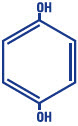
Hydroquinone, USP 4% is 1, 4-benzenediol. The drug is freely soluble in water and in alcohol. Chemically, hydroquinone is designated as p-dihydroxybenzene; the empirical formula is C6H6O2; molecular weight is 110.11 g/mol. The chemical structure is in the diagram below.
Uses
- helps prevent sunburn
- If used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun
Section 42229-5
OBAGI NU-DERM®
Rx only
For external use only
Section 44425-7
Store at controlled room temperature: 15° to 25°C (59° to 77°F). Keep out of direct sunlight.
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if rash occurs
Section 50567-7
When using this product keep out of eyes. Rinse with water to remove.
Section 50570-1
Do not use on damaged or broken skin
Section 55105-1
| Active ingredients | Purpose |
|---|---|
| Homosalate 10% | Sunscreen |
| Octisalate 5% | Sunscreen |
| Zinc Oxide 16.5% | Sunscreen |
Warnings
Hydroquinone is a skin-bleaching agent, which may produce unwanted cosmetic effects if not used as directed. The physician should be familiar with the contents of this insert before prescribing or dispensing this product.
Test for skin sensitivity before using by applying a small amount to an unbroken patch of skin and check within 24 hours. Some users of this product may experience a mild skin irritation. If skin irritation becomes severe, stop use and consult a doctor. Close patient supervision is recommended.
Warnings
For external use only
Directions
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m.–2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
- children under 6 months: Ask a doctor
Precautions
(also see WARNINGS):
Treatment should be limited to relatively small areas of the body at one time since some patients experience a transient skin reddening and a mild burning sensation, which does not preclude treatment.
Nursing Mothers
It is not known whether topical hydroquinone is absorbed or excreted in human milk. Caution is advised when topical hydroquinone is used by a nursing mother.
Pediatric Usage
Safety and effectiveness in children below the age of 12 years have not been established.
Other Information
- store at controlled room temperature: 15°C–25°C (59°F–77°F)
- protect this product from excessive heat and direct sun
Inactive Ingredients
Water (Aqua), C15-19 Alkane, Octyldodecyl Neopentanoate, Polymethylsilsesquioxane, Sorbitan Olivate, Silica, Polyglyceryl-6 Polyrininoleate, Sodium Chloride, Xanthan Gum, Glycerin, Hydroxyacetophenone, Disodium EDTA, 1,2-Hexanediol, Caprylyl Glycol, Sodium Hydroxide, Triethoxycaprylsilane, Polyhydroxystearic Acid, Disteardimonium Hectorite, Polyglyceryl-2 Isostearate, Euphorbia Cerifera (candelilla) Wax, Beeswax, Dimethicone
Pregnancy Category C
Animal reproduction studies have not been conducted with topical hydroquinone. It is also not known whether hydroquinone can cause fetal harm when used topically on a pregnant woman or affect reproductive capacity. It is not known to what degree, if any, topical hydroquinone is absorbed systemically. Topical hydroquinone should be used on pregnant women only when clearly indicated.
Clinical Pharmacology
Topical application of hydroquinone produces a reversible depigmentation of the skin by inhibition of the enzymatic oxidation of tyrosine to 3, 4-dihydroxyphenylalanine (DOPA) and suppression of other melanocyte metabolic processes. Exposure to sunlight or ultraviolet light will cause repigmentation of the bleached areas, which may be prevented by the use of sunscreen agents such as those contained in Obagi Nu-Derm Sunfader® and Obagi Nu-Derm® Sun Shield Matte Broad Spectrum SPF 50.
Questions Or Comments?
1.800.636.7546
Monday–Friday 9 a.m.–4 p.m. Pacific Time
Principal Display Panel Kit Carton
NDC# 62032-533-60
OBAGI®
MEDICAL
OBAGI NU-DERM® SYSTEM
NORMAL OILY
Skin Transformation Trial Kit
Structured Label Content
Uses
- helps prevent sunburn
- If used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun
Section 42229-5 (42229-5)
OBAGI NU-DERM®
Rx only
For external use only
Section 44425-7 (44425-7)
Store at controlled room temperature: 15° to 25°C (59° to 77°F). Keep out of direct sunlight.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if rash occurs
Section 50567-7 (50567-7)
When using this product keep out of eyes. Rinse with water to remove.
Section 50570-1 (50570-1)
Do not use on damaged or broken skin
Section 55105-1 (55105-1)
| Active ingredients | Purpose |
|---|---|
| Homosalate 10% | Sunscreen |
| Octisalate 5% | Sunscreen |
| Zinc Oxide 16.5% | Sunscreen |
Warnings (WARNINGS)
Hydroquinone is a skin-bleaching agent, which may produce unwanted cosmetic effects if not used as directed. The physician should be familiar with the contents of this insert before prescribing or dispensing this product.
Test for skin sensitivity before using by applying a small amount to an unbroken patch of skin and check within 24 hours. Some users of this product may experience a mild skin irritation. If skin irritation becomes severe, stop use and consult a doctor. Close patient supervision is recommended.
Warnings
For external use only
Directions
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m.–2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
- children under 6 months: Ask a doctor
Description (DESCRIPTION)
Hydroquinone, USP 4% is 1, 4-benzenediol. The drug is freely soluble in water and in alcohol. Chemically, hydroquinone is designated as p-dihydroxybenzene; the empirical formula is C6H6O2; molecular weight is 110.11 g/mol. The chemical structure is in the diagram below.
Precautions (PRECAUTIONS)
(also see WARNINGS):
Treatment should be limited to relatively small areas of the body at one time since some patients experience a transient skin reddening and a mild burning sensation, which does not preclude treatment.
How Supplied (HOW SUPPLIED)
Obagi Nu-Derm® Clear is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-101-36
Obagi Nu-Derm Blender®
is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-100-36
Net wt. 1 oz. (28 g) bottle
NDC 62032-100-10
Obagi Nu-Derm Sunfader®
is available as follows:
Net wt. 2 oz. (57 g) bottle
NDC 62032-116-36
Nursing Mothers (NURSING MOTHERS)
It is not known whether topical hydroquinone is absorbed or excreted in human milk. Caution is advised when topical hydroquinone is used by a nursing mother.
Pediatric Usage (PEDIATRIC USAGE)
Safety and effectiveness in children below the age of 12 years have not been established.
Adverse Reactions (ADVERSE REACTIONS)
No systemic adverse reactions have been reported. Occasional hypersensitivity (localized contact dermatitis) may occur, in which case the product should be discontinued and the physician notified immediately.
To report SUSPECTED ADVERSE REACTIONS, contact Obagi Cosmeceuticals LLC, at 1-800-636-7546 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Contraindications (CONTRAINDICATIONS)
People with prior history of sensitivity or allergic reaction to this product or any of its ingredients should not use it. The safety of topical hydroquinone use during pregnancy or in children (12 years and under) has not been established.
Other Information (Other information)
- store at controlled room temperature: 15°C–25°C (59°F–77°F)
- protect this product from excessive heat and direct sun
Inactive Ingredients (Inactive ingredients)
Water (Aqua), C15-19 Alkane, Octyldodecyl Neopentanoate, Polymethylsilsesquioxane, Sorbitan Olivate, Silica, Polyglyceryl-6 Polyrininoleate, Sodium Chloride, Xanthan Gum, Glycerin, Hydroxyacetophenone, Disodium EDTA, 1,2-Hexanediol, Caprylyl Glycol, Sodium Hydroxide, Triethoxycaprylsilane, Polyhydroxystearic Acid, Disteardimonium Hectorite, Polyglyceryl-2 Isostearate, Euphorbia Cerifera (candelilla) Wax, Beeswax, Dimethicone
Pregnancy Category C (PREGNANCY CATEGORY C)
Animal reproduction studies have not been conducted with topical hydroquinone. It is also not known whether hydroquinone can cause fetal harm when used topically on a pregnant woman or affect reproductive capacity. It is not known to what degree, if any, topical hydroquinone is absorbed systemically. Topical hydroquinone should be used on pregnant women only when clearly indicated.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Topical application of hydroquinone produces a reversible depigmentation of the skin by inhibition of the enzymatic oxidation of tyrosine to 3, 4-dihydroxyphenylalanine (DOPA) and suppression of other melanocyte metabolic processes. Exposure to sunlight or ultraviolet light will cause repigmentation of the bleached areas, which may be prevented by the use of sunscreen agents such as those contained in Obagi Nu-Derm Sunfader® and Obagi Nu-Derm® Sun Shield Matte Broad Spectrum SPF 50.
Indications and Usage (INDICATIONS AND USAGE)
For the gradual bleaching of hyperpigmented skin conditions such as chloasma, melasma, freckles, senile lentigines and other unwanted areas of melanin hyperpigmentation.
Questions Or Comments? (Questions or comments?)
1.800.636.7546
Monday–Friday 9 a.m.–4 p.m. Pacific Time
Dosage and Administration (DOSAGE AND ADMINISTRATION)
A thin application should be applied once or twice daily or as directed by a physician. If no improvement is seen after three (3) months of treatment, use of this product should be discontinued. Sun exposure should be limited by using a sunscreen agent or protective clothing to cover bleached skin when using and after using this product in order to prevent darkening from reoccurring.
Principal Display Panel Kit Carton (PRINCIPAL DISPLAY PANEL - Kit Carton)
NDC# 62032-533-60
OBAGI®
MEDICAL
OBAGI NU-DERM® SYSTEM
NORMAL OILY
Skin Transformation Trial Kit
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:08.558683 · Updated: 2026-03-14T22:24:05.784289