These Highlights Do Not Include All The Information Needed To Use Nexplanon Safely And Effectively. See Full Prescribing Information For Nexplanon.
487f8a62-e142-457c-97cc-2e398fde7594
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Improper insertion of NEXPLANON increases the risk of complications [see Warnings and Precautions (5.1) ] . Proper training prior to first use of NEXPLANON can minimize the risk of improper NEXPLANON insertion [see Warnings and Precautions (5.1) ] . Because of the risk of complications due to improper insertion and removal NEXPLANON is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the NEXPLANON REMS [see Warnings and Precautions (5.2) ] .
Indications and Usage
NEXPLANON ® is indicated for prevention of pregnancy in women of reproductive potential for up to 5 years.
Dosage and Administration
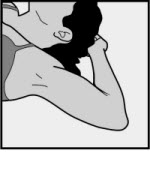
The efficacy of NEXPLANON does not depend on daily, weekly, or monthly administration. All healthcare professionals should receive instruction and training prior to performing insertion and/or removal of NEXPLANON. A single NEXPLANON implant is inserted subdermally just under the skin at the inner side of the non-dominant upper arm. The insertion site is overlying the triceps muscle about 8-10 cm (3-4 inches) from the medial epicondyle of the humerus and 3-5 cm (1.25-2 inches) posterior to (below) the sulcus (groove) between the biceps and triceps muscles. This location is intended to avoid the large blood vessels and nerves lying within and surrounding the sulcus (see Figures 2a , 2b and 2c ). Inserting an implant more deeply than subdermally (a deep insertion) may preclude palpation and localization, making removal difficult or impossible [see Dosage and Administration (2.3) and Warnings and Precautions (5.1) ] . NEXPLANON must be inserted by the expiration date stated on the packaging. NEXPLANON is a long-acting (up to 5 years), reversible, hormonal contraceptive method. The implant must be removed by the end of the fifth year and may be replaced by a new implant at the time of removal, if continued contraceptive protection is desired.
Warnings and Precautions
The following information is based on experience with the etonogestrel implants (IMPLANON and/or NEXPLANON), other progestin-only contraceptives, or experience with combination (estrogen plus progestin) oral contraceptives.
Contraindications
NEXPLANON should not be used in women who have Known or suspected pregnancy Current or past history of thrombosis or thromboembolic disorders Liver tumors, benign or malignant, or active liver disease Undiagnosed abnormal uterine bleeding Known or suspected breast cancer, personal history of breast cancer, or other progestin-sensitive cancer, now or in the past Allergic reaction to any of the components of NEXPLANON [see Adverse Reactions (6) ]
Adverse Reactions
The following adverse reactions reported with the use of hormonal contraception are discussed elsewhere in the labeling: Changes in Menstrual Bleeding Patterns [see Warnings and Precautions (5.3) ] Ectopic Pregnancies [see Warnings and Precautions (5.4) ] Thrombotic and Other Vascular Events [see Warnings and Precautions (5.5) ] Liver Disease [see Warnings and Precautions (5.8) ]
Drug Interactions
Consult the labeling of concurrently-used drugs to obtain further information about interactions with hormonal contraceptives or the potential for enzyme alterations.
Storage and Handling
Store NEXPLANON at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted between 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature]. Avoid storing NEXPLANON at temperatures above 30ºC (86ºF).
How Supplied
NEXPLANON is supplied as follows: NDC 78206-145-01 One NEXPLANON package consists of a single implant containing 68 mg etonogestrel, 15 mg of barium sulfate and 0.1 mg of magnesium stearate that is 4 cm in length and 2 mm in diameter, which is pre-loaded in the needle of a disposable applicator. The sterile applicator containing the implant is packed in a blister pack.
Medication Information
Warnings and Precautions
The following information is based on experience with the etonogestrel implants (IMPLANON and/or NEXPLANON), other progestin-only contraceptives, or experience with combination (estrogen plus progestin) oral contraceptives.
Indications and Usage
NEXPLANON ® is indicated for prevention of pregnancy in women of reproductive potential for up to 5 years.
Dosage and Administration
The efficacy of NEXPLANON does not depend on daily, weekly, or monthly administration. All healthcare professionals should receive instruction and training prior to performing insertion and/or removal of NEXPLANON. A single NEXPLANON implant is inserted subdermally just under the skin at the inner side of the non-dominant upper arm. The insertion site is overlying the triceps muscle about 8-10 cm (3-4 inches) from the medial epicondyle of the humerus and 3-5 cm (1.25-2 inches) posterior to (below) the sulcus (groove) between the biceps and triceps muscles. This location is intended to avoid the large blood vessels and nerves lying within and surrounding the sulcus (see Figures 2a , 2b and 2c ). Inserting an implant more deeply than subdermally (a deep insertion) may preclude palpation and localization, making removal difficult or impossible [see Dosage and Administration (2.3) and Warnings and Precautions (5.1) ] . NEXPLANON must be inserted by the expiration date stated on the packaging. NEXPLANON is a long-acting (up to 5 years), reversible, hormonal contraceptive method. The implant must be removed by the end of the fifth year and may be replaced by a new implant at the time of removal, if continued contraceptive protection is desired.
Contraindications
NEXPLANON should not be used in women who have Known or suspected pregnancy Current or past history of thrombosis or thromboembolic disorders Liver tumors, benign or malignant, or active liver disease Undiagnosed abnormal uterine bleeding Known or suspected breast cancer, personal history of breast cancer, or other progestin-sensitive cancer, now or in the past Allergic reaction to any of the components of NEXPLANON [see Adverse Reactions (6) ]
Adverse Reactions
The following adverse reactions reported with the use of hormonal contraception are discussed elsewhere in the labeling: Changes in Menstrual Bleeding Patterns [see Warnings and Precautions (5.3) ] Ectopic Pregnancies [see Warnings and Precautions (5.4) ] Thrombotic and Other Vascular Events [see Warnings and Precautions (5.5) ] Liver Disease [see Warnings and Precautions (5.8) ]
Drug Interactions
Consult the labeling of concurrently-used drugs to obtain further information about interactions with hormonal contraceptives or the potential for enzyme alterations.
Storage and Handling
Store NEXPLANON at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted between 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature]. Avoid storing NEXPLANON at temperatures above 30ºC (86ºF).
How Supplied
NEXPLANON is supplied as follows: NDC 78206-145-01 One NEXPLANON package consists of a single implant containing 68 mg etonogestrel, 15 mg of barium sulfate and 0.1 mg of magnesium stearate that is 4 cm in length and 2 mm in diameter, which is pre-loaded in the needle of a disposable applicator. The sterile applicator containing the implant is packed in a blister pack.
Description
Improper insertion of NEXPLANON increases the risk of complications [see Warnings and Precautions (5.1) ] . Proper training prior to first use of NEXPLANON can minimize the risk of improper NEXPLANON insertion [see Warnings and Precautions (5.1) ] . Because of the risk of complications due to improper insertion and removal NEXPLANON is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the NEXPLANON REMS [see Warnings and Precautions (5.2) ] .
Section 42229-5
• No preceding hormonal contraceptive use in the past month
NEXPLANON should be inserted between Day 1 (first day of menstrual bleeding) and Day 5 of the menstrual cycle, even if the woman is still bleeding.
If inserted as recommended, back-up contraception is not necessary. If deviating from the recommended timing of insertion, the woman should be advised to use a barrier method until 7 days after insertion. If intercourse has already occurred, pregnancy should be excluded.
Section 42230-3
FDA-Approved Patient Labeling
NEXPLANON® (etonogestrel implant)
Radiopaque
Subdermal Use Only
NEXPLANON® does not protect against HIV infection (the virus that causes AIDS) or other sexually transmitted diseases.
Read this Patient Information leaflet carefully before you decide if NEXPLANON is right for you. This information does not take the place of talking with your healthcare professional. If you have any questions about NEXPLANON, ask your healthcare professional.
What is NEXPLANON?
NEXPLANON is a hormone-releasing birth control implant for prevention of pregnancy for up to 5 years. The implant is a flexible plastic rod about the size of a matchstick that contains a progestin hormone called etonogestrel. It contains a small amount of barium sulfate (15 mg), so that the implant can be seen by X-ray, an ethylene vinyl acetate (EVA) copolymer (28% vinyl acetate, 43 mg) core, and magnesium stearate (0.1 mg). Your healthcare professional will insert the implant just under the skin of the inner side of your upper arm. You can use a single NEXPLANON implant for up to 5 years. NEXPLANON does not contain estrogen.
What if I need birth control for more than 5 years?
The NEXPLANON implant must be removed by the end of 5 years. Your healthcare professional can insert a new implant under your skin after taking out the old one if you choose to continue using NEXPLANON for birth control.
What if I change my mind about birth control and want to stop using NEXPLANON before 5 years?
Your healthcare professional can remove the implant at any time. You may become pregnant as early as the first week after removal of the implant. If you do not want to get pregnant after your healthcare professional removes the NEXPLANON implant, you should start another birth control method right away.
How does NEXPLANON work?
NEXPLANON prevents pregnancy in several ways. The most important way is by stopping the release of an egg from your ovary. NEXPLANON also thickens the mucus in your cervix and this change may keep sperm from reaching the egg. NEXPLANON also changes the lining of your uterus.
How well does NEXPLANON work?
When the NEXPLANON implant is placed correctly, your chance of getting pregnant is very low (less than 1 pregnancy per 100 women who use NEXPLANON for 1 year).
The following chart shows the chance of getting pregnant for women who use different methods of birth control. Each box on the chart contains a list of birth control methods that are similar in effectiveness. The most effective methods are at the top of the chart. The box on the bottom of the chart shows the chance of getting pregnant for women who do not use birth control and are trying to get pregnant.
Who should not use NEXPLANON?
Do not use NEXPLANON if you have any of the following conditions:
-
Are pregnant or think you may be pregnant
- Have, or have had, blood clots, such as blood clots in your legs (deep vein thrombosis), lungs (pulmonary embolism), eyes (total or partial blindness), heart (heart attack), or brain (stroke)
- Liver disease or a liver tumor
- Unexplained vaginal bleeding
- Breast cancer or any other cancer that is sensitive to progestin (a female hormone), now or in the past
- An allergy to anything in NEXPLANON
Tell your healthcare professional if you have or have had any of the conditions listed above. Your healthcare professional can suggest a different method of birth control.
In addition, talk to your healthcare professional about using NEXPLANON if you have any of the following conditions:
- Diabetes
- High cholesterol or triglycerides
- Headaches
- Gallbladder or kidney problems
- A history of depressed mood
- High blood pressure
- An allergy to numbing medicines (anesthetics) or medicines used to clean your skin (antiseptics). These medicines will be used when the implant is placed into or removed from your arm.
Interaction with Other Medicines
Tell your healthcare professional about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Using certain medicines with NEXPLANON, including those listed below, may make NEXPLANON less effective:
- aprepitant
- barbiturates
- bosentan
- carbamazepine
- felbamate
- griseofulvin
- oxcarbazepine
- phenytoin
- rifampin
- St. John's wort
- topiramate
- HIV medicines
- Hepatitis C Virus medicines
Ask your healthcare professional if you are not sure if your medicine is one listed above.
If you are taking medicines or herbal products that might make NEXPLANON less effective, you and your healthcare professional may decide to leave NEXPLANON in place; in that case, an additional non-hormonal contraceptive should be used. It is necessary to use the additional non-hormonal contraceptive for 28 days after stopping the medicine or herbal product because its effect on NEXPLANON may last that long.
When you are using NEXPLANON, tell all your healthcare professionals that you have NEXPLANON in place in your arm.
Magnetic Resonance Imaging (MRI) Safety Information
NEXPLANON is MRI safe.
How is the NEXPLANON implant placed and removed?
Your healthcare professional will place and remove the NEXPLANON implant using a minor surgical procedure in his or her office. The implant is placed just under the skin on the inner side of your non-dominant upper arm.
The timing of insertion is important. Your healthcare professional may take the following steps to guide timing of insertion:
- Perform a pregnancy test before inserting NEXPLANON
- Schedule the insertion at a specific time of your menstrual cycle (for example, within the first 5 days of your regular menstrual bleeding). If the implant is placed after the fifth day of menses, then you should use an additional contraceptive method (such as a condom) for the first 7 days after insertion.
Your healthcare professional will cover the site where NEXPLANON was placed with 2 bandages. A small bandage will be placed over the insertion site. A larger pressure bandage will be placed over the smaller bandage. Leave the larger pressure bandage on for 24 hours. Keep the smaller bandage clean, dry, and in place for 3 to 5 days.
Immediately after the NEXPLANON implant has been placed, you and your healthcare professional should check that the implant is in your arm by feeling for it.
If you cannot feel the implant immediately after insertion, the implant may not have been inserted, or it may have been inserted deeply. A deep insertion may cause problems with locating and removing the implant. When the healthcare professional has located the implant, it should be removed.
If at any time you cannot feel the NEXPLANON implant, contact your healthcare professional immediately and use a non-hormonal birth control method (such as condoms) until your healthcare professional confirms that the implant is in place. You may need special tests to check that the implant is in place or to help find the implant when it is time to take it out. If the implant cannot be found in the arm after a thorough search, your healthcare professional may use X-rays or other imaging methods on your chest.
Depending on the exact position of the implant, removal may be difficult and may require surgery.
Keep track of the date the implant is to be removed. Schedule an appointment with your healthcare professional to remove the implant on or before the removal date.
Be sure to have checkups as advised by your healthcare professional.
What are the most common side effects I can expect while using NEXPLANON?
• Changes in Menstrual Bleeding Patterns (Menstrual Periods)
The most common side effect of NEXPLANON is a change in your normal menstrual bleeding pattern. In studies, one out of ten women stopped using the implant because of an unfavorable change in their bleeding pattern. You may experience longer or shorter bleeding during your periods or have no bleeding at all. The time between periods may vary, and you may also have spotting in between periods.
Tell your healthcare professional right away if you have the following concerns:
- You think you may be pregnant
- Your menstrual bleeding is heavy and prolonged
The following additional frequent side effects also caused women to stop using the implant:
- Mood swings
- Depressed mood
- Weight gain
- Headache
- Acne
Other common side effects include the following conditions:
- Headache
- Vaginitis (inflammation of the vagina)
- Weight gain
- Acne
- Breast pain
- Viral infections such as sore throats or flu-like symptoms
- Stomach pain
- Painful periods
- Mood swings, nervousness, or depressed mood
- Back pain
- Nausea
- Dizziness
- Pain
- Pain at the site of insertion
Implants have been reported to be found in a blood vessel, including a blood vessel in the lung which can be associated with shortness of breath, cough and/or the coughing up of blood or blood-stained mucus.
These are not all the possible side effects of NEXPLANON. Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
What are the possible risks of using NEXPLANON?
• Problems with Insertion and Removal
The implant may not be actually in your arm due to a failed insertion. If this happens, you may become pregnant. Immediately after insertion, and with help from your healthcare professional, you should be able to feel the implant under your skin. If you can’t feel the implant, tell your healthcare professional.
Location and removal of the implant may be difficult or impossible because the implant is not where it should be. Special procedures, including surgery in the hospital, may be needed to remove the implant. If the implant is not removed, then the effects of NEXPLANON will continue for a longer time.
Implants have been found in the pulmonary artery (a blood vessel in the lung). If the implant cannot be found in the arm, your healthcare professional may use X-rays or other imaging methods on the chest. If the implant is located in the chest, surgery may be needed.
The following problems related to insertion and removal may also occur:
- Vasovagal reactions (such as a drop in blood pressure, dizziness, or fainting)
- Pain, irritation, swelling, or bruising at the insertion site
- Numbness and tingling at the insertion site
- Scarring, including a thick scar called a keloid around the insertion site
- Infection
- Scar tissue may form around the implant making it difficult to remove
- The implant may come out by itself. You may become pregnant if the implant comes out by itself. Use a back-up birth control method and call your healthcare professional right away if the implant comes out.
- The need for surgery in the hospital to remove the implant
- Injury to nerves or blood vessels in your arm
- The implant breaks, making removal difficult
• Ectopic Pregnancy
If you become pregnant while using NEXPLANON, you have a slightly higher chance that the pregnancy will be ectopic (occurring outside the womb) than do women who do not use birth control. Unusual vaginal bleeding or lower stomach (abdominal) pain may be a sign of ectopic pregnancy. Ectopic pregnancy is a medical emergency that often requires surgery. Ectopic pregnancies can cause serious internal bleeding, infertility, and even death. Call your healthcare professional right away if you think you are pregnant or have unexplained lower stomach (abdominal) pain.
• Ovarian Cysts
Cysts may develop on the ovaries and usually go away without treatment, but sometimes surgery is needed to remove them.
• Breast Cancer
It is not known whether NEXPLANON use changes a woman's risk for breast cancer. If you have breast cancer now, or have had it in the past, do not use NEXPLANON because some breast cancers are sensitive to hormones.
• Serious Blood Clots
NEXPLANON may increase your chance of serious blood clots, especially if you have other risk factors such as smoking. It is possible to die from a problem caused by a blood clot, such as a heart attack or a stroke.
Serious blood clots can occur within blood vessels of different parts of the body, including the following examples:
- Legs (deep vein thrombosis)
- Lungs (pulmonary embolism)
- Brain (stroke)
- Heart (heart attack)
- Eyes (total or partial blindness)
The risk of serious blood clots is increased in women who smoke. If you smoke and want to use NEXPLANON, you should quit. Your healthcare professional may be able to help.
Tell your healthcare professional at least 4 weeks before if you are going to have surgery or will need to be on bed rest. You have an increased chance of getting blood clots during surgery or bed rest.
• Other Risks
A few women who use birth control that contains hormones may get the following conditions:
- High blood pressure
- Gallbladder problems
- Rare cancerous or noncancerous liver tumors
• Broken or Bent Implant
Breakage or bending of the implant while it is in your arm may occur due to external forces (such as manipulation of the implant or contact sports). A broken implant may move from the insertion site. If you feel that the implant may have broken or bent while in your arm, contact your healthcare professional.
When should I call my healthcare professional?
Call your healthcare professional right away if you have any of the following conditions:
- Pain in your lower leg that does not go away
- Severe chest pain or heaviness in the chest
- Sudden shortness of breath, sharp chest pain, or coughing blood
- Symptoms of a severe allergic reaction, such as swollen face, tongue or throat; trouble breathing or swallowing
- Sudden severe headache unlike your usual headaches
- Weakness or numbness in your arm, leg, or trouble speaking
- Sudden partial or complete blindness
- Yellowing of your skin or whites of your eyes, especially with fever, tiredness, loss of appetite, dark colored urine, or light-colored bowel movements
- Severe pain, swelling, or tenderness in the lower stomach (abdomen)
- Lump in your breast
- Problems sleeping, lack of energy, tiredness, or you feel very sad
- Heavy menstrual bleeding
What if I become pregnant while using NEXPLANON?
You should see your healthcare professional right away if you think that you may be pregnant. It is important to remove the implant and make sure that the pregnancy is not ectopic (occurring outside the womb). Based on experience with other hormonal contraceptives, NEXPLANON is not likely to cause birth defects.
Can I use NEXPLANON when I am breastfeeding?
If you are breastfeeding your child, you may use NEXPLANON if 4 weeks have passed since you had your baby. A small amount of the hormone contained in NEXPLANON passes into your breast milk. The health of breast-fed children whose mothers were using the implant has been studied up to 3 years of age in a small number of children. No effects on the growth and development of the children were seen. If you are breastfeeding and want to use NEXPLANON, talk with your healthcare professional for more information.
General information about the safe and effective use of NEXPLANON
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. If you would like more information, talk with your healthcare professional. You can ask your healthcare professional for information about NEXPLANON that is written for healthcare professionals.
Section 43683-2
10 Overdosage
Overdosage may result if more than one implant is inserted. In case of suspected overdose, the implant should be removed.
11 Description
NEXPLANON is a radiopaque, progestin-only, soft, flexible implant preloaded in a sterile, disposable applicator for subdermal use. The implant is white/off-white, non-biodegradable and 4 cm in length with a diameter of 2 mm (see Figure 19). Each implant consists of an ethylene vinyl acetate (EVA) copolymer (28% vinyl acetate, 43 mg) core, containing 68 mg of the synthetic progestin etonogestrel, barium sulfate ((15 mg), radiopaque ingredient), and magnesium stearate (0.1 mg), surrounded by an EVA copolymer skin. Once inserted subdermally, the release rate is 60-75 mcg/day in week 5-6 and decreases to approximately 40-50 mcg/day at the end of the first year, and then to approximately 15-20 mcg/day at the end of the fifth year. NEXPLANON is a progestin-only contraceptive and does not contain estrogen. NEXPLANON is not made with natural rubber latex.
| Figure 19 (Not to scale) |
Etonogestrel [13-Ethyl-17-hydroxy-11-methylene-18,19-dinor-17α-pregn-4-en-20-yn-3-one], structurally derived from 19-nortestosterone, is the synthetic biologically active metabolite of the synthetic progestin desogestrel. It has a molecular weight of 324.46 and the following structural formula (Figure 20).
| Figure 20 |
14.1 Pregnancy
In clinical trials of up to 3 years duration that involved 923 subjects, 18-40 years of age at entry, and 1756 women-years of use with the non-radiopaque etonogestrel implant (IMPLANON), the total exposures expressed as 28-day cycle equivalents by study year were:
Year 1: 10,866 cycles
Year 2: 8,581 cycles
Year 3: 3,442 cycles
The clinical trials excluded women who:
- Weighed more than 130% of their ideal body weight
- Were chronically taking medications that induce liver enzymes
In the subgroup of women, 18-35 years of age at entry, 6 pregnancies during 20,648 cycles of use were reported. Two pregnancies occurred in each of Years 1, 2, and 3. Each conception was likely to have occurred shortly before or within 2 weeks after removal of the non-radiopaque etonogestrel implant. With these 6 pregnancies, the cumulative Pearl Index (PI) was 0.38 pregnancies per 100 women-years of use.
The contraceptive efficacy of NEXPLANON during use from 3 to 5 years was evaluated in a multicenter, single-arm, open-label study (NCT04626596) conducted in the United States. The trial enrolled women 18 to 35 years of age who had been using NEXPLANON for 36 months (±2 weeks) from the date of insertion at the time of enrollment. A total of 399 women were evaluated, having a mean age of 27 years.
Participants were 74.2% White, 16.8% Black/African American, 3.8% Asian, 1.3% American Indian or Alaska Native, 0.5% Native Hawaiian or Other Pacific Islander, and 3.5% multiple or missing races. The mean BMI was 29.4 kg/m2 (range: 17.2-64.3 kg/m2) and the mean weight was 78.7 kg (range: 40.8-180.8 kg). One hundred fifty-two participants (38.1%) had a BMI ≥30 kg/m2, including 40 participants (10.0%) with BMI ≥40 kg/m2.
The total exposures expressed as 28-day cycle equivalents by study year were:
Year 4: 4478 cycles
Year 5: 3274 cycles
No pregnancies were reported in years 4 and 5, with a PI of 0.0 (95% CI- 0.00, 0.69) pregnancies per 100 women-years of use.
5.17 Monitoring
A woman who is using NEXPLANON should have a yearly visit with her healthcare professional for a blood pressure check and for other indicated health care.
5.9 Weight Gain
In clinical studies, mean weight gain in U.S. non-radiopaque etonogestrel implant (IMPLANON) users was 2.8 pounds after one year and 3.7 pounds after two years. How much of the weight gain was related to the non-radiopaque etonogestrel implant is unknown. In studies, 2.3% of the users reported weight gain as the reason for having the non-radiopaque etonogestrel implant removed.
16.1 How Supplied
NEXPLANON is supplied as follows:
NDC 78206-145-01
One NEXPLANON package consists of a single implant containing 68 mg etonogestrel, 15 mg of barium sulfate and 0.1 mg of magnesium stearate that is 4 cm in length and 2 mm in diameter, which is pre-loaded in the needle of a disposable applicator. The sterile applicator containing the implant is packed in a blister pack.
5.6 Ovarian Cysts
If follicular development occurs, atresia of the follicle is sometimes delayed, and the follicle may continue to grow beyond the size it would attain in a normal cycle. Generally, these enlarged follicles disappear spontaneously. On rare occasion, surgery may be required.
5.8 Liver Disease
Disturbances of liver function may necessitate the discontinuation of hormonal contraceptive use until markers of liver function return to normal. Remove NEXPLANON if jaundice develops.
Hepatic adenomas are associated with combination hormonal contraceptives use. An estimate of the attributable risk is 3.3 cases per 100,000 for combination hormonal contraceptives users. It is not known whether a similar risk exists with progestin-only methods like NEXPLANON.
The progestin in NEXPLANON may be poorly metabolized in women with liver impairment. Use of NEXPLANON in women with active liver disease or liver cancer is contraindicated [see Contraindications (4)].
8.4 Pediatric Use
The safety and effectiveness of NEXPLANON have been established in women of reproductive potential. Safety and effectiveness of NEXPLANON are expected to be the same in postpubertal adolescents as in adult women.
NEXPLANON is not indicated before menarche.
8.5 Geriatric Use
This product has not been studied in women over 65 years of age and is not indicated in this population.
5.2 Nexplanon Rems
NEXPLANON is only available through a restricted program under a REMS called NEXPLANON REMS because of the risk of complications due to improper insertion and removal [see Warnings and Precautions (5.1)].
Notable requirements of the NEXPLANON REMS include the following:
- Healthcare providers must be certified with the program by enrolling and completing training on the proper insertion and removal of NEXPLANON prior to first use.
- Pharmacies must be certified with the program and must only dispense NEXPLANON to certified healthcare providers who dispense NEXPLANON for insertion.
- Wholesalers and distributors must be registered with the program and must only distribute to certified pharmacies and certified healthcare providers.
Further information is available at www.NEXPLANONREMS.com and 1-833-697-7367.
4 Contraindications
NEXPLANON should not be used in women who have
- Known or suspected pregnancy
- Current or past history of thrombosis or thromboembolic disorders
- Liver tumors, benign or malignant, or active liver disease
- Undiagnosed abnormal uterine bleeding
- Known or suspected breast cancer, personal history of breast cancer, or other progestin-sensitive cancer, now or in the past
- Allergic reaction to any of the components of NEXPLANON [see Adverse Reactions (6)]
5.13 Depressed Mood
Women with a history of depressed mood should be carefully observed. Consideration should be given to removing NEXPLANON in patients who become significantly depressed.
5.16 Contact Lenses
Contact lens wearers who develop visual changes or changes in lens tolerance should be assessed by an ophthalmologist.
6 Adverse Reactions
The following adverse reactions reported with the use of hormonal contraception are discussed elsewhere in the labeling:
- Changes in Menstrual Bleeding Patterns [see Warnings and Precautions (5.3)]
- Ectopic Pregnancies [see Warnings and Precautions (5.4)]
- Thrombotic and Other Vascular Events [see Warnings and Precautions (5.5)]
- Liver Disease [see Warnings and Precautions (5.8)]
7 Drug Interactions
Consult the labeling of concurrently-used drugs to obtain further information about interactions with hormonal contraceptives or the potential for enzyme alterations.
5.15 Fluid Retention
Hormonal contraceptives may cause some degree of fluid retention. They should be prescribed with caution, and only with careful monitoring, in patients with conditions which might be aggravated by fluid retention. It is unknown if NEXPLANON causes fluid retention.
12.2 Pharmacodynamics
Exposure-response relationships of NEXPLANON are unknown.
8.6 Hepatic Impairment
No studies were conducted to evaluate the effect of hepatic disease on the disposition of NEXPLANON. The use of NEXPLANON in women with active liver disease is contraindicated [see Contraindications (4)].
1 Indications and Usage
NEXPLANON® is indicated for prevention of pregnancy in women of reproductive potential for up to 5 years.
2.4 Replacing Nexplanon
Immediate replacement can be done after removal of the previous implant and is similar to the insertion procedure described in section 2.2 Insertion of NEXPLANON.
The new implant may be inserted in the same arm, and through the same incision from which the previous implant was removed, if the site is in the correct location, i.e., 8-10 cm from the medial epicondyle of the humerus and 3-5 cm posterior to (below) the sulcus [see Dosage and Administration (2.2)]. If the same incision is being used to insert a new implant, anesthetize the insertion site [for example, 2 mL lidocaine (1%)] applying it just under the skin along the 'insertion canal.'
Follow the subsequent steps in the insertion instructions [see Dosage and Administration (2.2)].
5.4 Ectopic Pregnancies
As with all progestin-only contraceptive products, be alert to the possibility of an ectopic pregnancy among women using NEXPLANON who become pregnant or complain of lower abdominal pain. Although ectopic pregnancies are uncommon among women using NEXPLANON, a pregnancy that occurs in a woman using NEXPLANON may be more likely to be ectopic than a pregnancy occurring in a woman using no contraception.
12.1 Mechanism of Action
The contraceptive effect of NEXPLANON is achieved by suppression of ovulation, increased viscosity of the cervical mucus, and alterations in the endometrium.
14.2 Return to Ovulation
In clinical trials with the non-radiopaque etonogestrel implant (IMPLANON), the etonogestrel levels in blood decreased below sensitivity of the assay by one week after removal of the implant. In addition, pregnancies were observed to occur as early as 7 to 14 days after removal. Therefore, a woman should re-start contraception immediately after removal of the implant if continued contraceptive protection is desired.
5.11 Gallbladder Disease
Studies suggest a small increased relative risk of developing gallbladder disease among combination hormonal contraceptive users. It is not known whether a similar risk exists with progestin-only methods like NEXPLANON.
5.14 Return to Ovulation
In clinical trials with the non-radiopaque etonogestrel implant (IMPLANON), the etonogestrel levels in blood decreased below sensitivity of the assay by one week after removal of the implant. In addition, pregnancies were observed to occur as early as 7 to 14 days after removal. Therefore, a woman should re-start contraception immediately after removal of the implant if continued contraceptive protection is desired.
16.2 Storage and Handling
Store NEXPLANON at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted between 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature]. Avoid storing NEXPLANON at temperatures above 30ºC (86ºF).
2.2 Insertion of Nexplanon
The basis for successful use and subsequent removal of NEXPLANON is a correct and carefully performed subdermal insertion of the single, rod-shaped implant in accordance with the instructions. Both the healthcare professional and the woman should be able to feel the implant under the skin after placement.
All healthcare professionals performing insertions and/or removals of NEXPLANON must receive instructions and training prior to inserting or removing the implant.
5 Warnings and Precautions
The following information is based on experience with the etonogestrel implants (IMPLANON and/or NEXPLANON), other progestin-only contraceptives, or experience with combination (estrogen plus progestin) oral contraceptives.
2 Dosage and Administration
The efficacy of NEXPLANON does not depend on daily, weekly, or monthly administration.
All healthcare professionals should receive instruction and training prior to performing insertion and/or removal of NEXPLANON.
A single NEXPLANON implant is inserted subdermally just under the skin at the inner side of the non-dominant upper arm. The insertion site is overlying the triceps muscle about 8-10 cm (3-4 inches) from the medial epicondyle of the humerus and 3-5 cm (1.25-2 inches) posterior to (below) the sulcus (groove) between the biceps and triceps muscles. This location is intended to avoid the large blood vessels and nerves lying within and surrounding the sulcus (see Figures 2a, 2b and 2c). Inserting an implant more deeply than subdermally (a deep insertion) may preclude palpation and localization, making removal difficult or impossible [see Dosage and Administration (2.3) and Warnings and Precautions (5.1)].
NEXPLANON must be inserted by the expiration date stated on the packaging. NEXPLANON is a long-acting (up to 5 years), reversible, hormonal contraceptive method. The implant must be removed by the end of the fifth year and may be replaced by a new implant at the time of removal, if continued contraceptive protection is desired.
3 Dosage Forms and Strengths
Single, white/off-white, soft, radiopaque, flexible, ethylene vinyl acetate (EVA) copolymer implant, 4 cm in length and 2 mm in diameter containing 68 mg etonogestrel, 15 mg of barium sulfate, and 0.1 mg of magnesium stearate.
5.10 Elevated Blood Pressure
Women with a history of hypertension-related diseases or renal disease should be discouraged from using hormonal contraception. For women with well-controlled hypertension, use of NEXPLANON can be considered. Women with hypertension using NEXPLANON should be closely monitored. If sustained hypertension develops during the use of NEXPLANON, or if a significant increase in blood pressure does not respond adequately to antihypertensive therapy, NEXPLANON should be removed.
6.2 Postmarketing Experience
Adverse Reactions and Events from Postmarketing Study
Nexplanon Observational Risk Assessment Study (NORA)
A postmarketing prospective active surveillance study was conducted among 7,364 patients in the United States to characterize the frequency of insertion-, localization-, and removal-related events.
Implant Insertion
Insertion difficulty or an insertion-related event occurred in 2.6% of the study participants. The overall incidence of incorrect insertion (unrecognized non-insertion, partial insertion, and deep insertion), reported by healthcare professionals was 12.6 per 1,000 insertions (95% CI, 10.2, 15.5). Table 5 summarizes the types and frequencies of these incorrect insertions.
| Type of Incorrect Insertion Event | Number of Events Total Insertion Procedures = 7,364
|
Incidence per 1,000 Insertions (95% CI) |
|---|---|---|
| (Initially) Unrecognized Non-insertions | 1 | 0.1 (0.0-0.8) |
| Partial Insertions | 27 | 3.7 (2.4-5.3) |
| Deep Insertions | 65 | 8.8 (6.8-11.2) |
| Injury to nerve or blood vessel | 1 | 0.1 (0.0-0.8) |
| Implant located within muscle | 2 | 0.3 (0.0-1.0) |
| Implant located adjacent to fascial tissue | 56 | 7.6 (5.8-9.9) |
| Implant not palpable | 6 | 0.8 (0.3-1.8) |
Implant Removal
Implant removal information from both healthcare professionals and patients was collected for 5,159 patients (70% of the study population). Of these patients, data were available from healthcare professionals regarding 4,373 removal procedures. Healthcare professionals reported removal-related difficulties or complications in 1.5% of removal procedures. Table 6 provides a summary.
| Removal Related Events | Number of Events Total Removal Procedures: N= 4,373
|
Incidence per 1,000 Removals (95% CI) |
|---|---|---|
| Any Event Limited to one event per removal procedure
|
60 | 13.7 (10.5-17.6) |
| Encased in Fibrotic Tissue | 29 | 6.6 (4.4-9.5) |
| Implant Too Deep | 11 | 2.5 (1.3-4.5) |
| Implant Migrated Only local migrations within the arm reported
|
6 | 1.4 (0.5-3.0) |
| Multiple Attempts Required | 13 | 3.0 (1.6-5.1) |
| Other Other included fragmented or bent implants, patient-related issues, wound care required, two incisions required, and difficulty identifying end of device.
|
14 | 3.2 (1.8-5.4) |
At the time of implant removal, eighteen implants (0.4% of all localizations or removals) were not palpable by the healthcare professionals. Of these eighteen, eleven were localized and removed, and one was localized but left in situ. Removal was not attempted for six non-palpable implants due to underlying health conditions, administrative problems, or unspecified reasons.
There were no reports of implants having migrated more than a few centimeters from the insertion site and no reports of implants localized at a site other than the arm. No neurovascular injuries were reported by healthcare professionals.
Adverse Reactions Reported by Patients
Table 7 provides a summary of adverse reactions reported by patients at the time of implant insertion and after removal.
| Patient Reported Adverse Reactions | At Insertion | After Removal | ||
|---|---|---|---|---|
| N Total Insertion Procedures: N = 7,364
|
Incidence per 1000 insertions (95% CI) | N | Incidence per 1000 insertions (95% CI) | |
| Any Event Limited to one event per woman
|
49 | 6.7 (4.9-8.8) | 42 Based on 3,447 questionnaires
|
5.7 (4.1-7.7) |
| Pins and Needles/Numbness (arm/hand/fingers) | 17 | 2.3 (1.4-3.7) | 24 | 3.3 (2.1-4.9) |
| Severe Pain | 10 | 1.4 (0.7-2.5) | 11 | 1.5 (0.8-2.7) |
| Altered Strength/Movement | 3 | 0.4 (0.1-1.2) | 8 | 1.1 (0.5-2.1) |
| Injury to Blood Vessels or Blood Clots in Arm No blood clots were observed during the study
|
2 | 0.3 (0-1.0) | -- | -- |
| Other “Other” included localized or insertion site pain, soreness, tenderness, dermatological changes, itching, bruising, and infection, local migrations within the arm, and physical damage to the implant (e.g., fractured or bent implant).
|
22 | 3.0 (1.9-4.5) | 18 | 2.4 (1.5-3.9) |
In summary, this prospective active surveillance study showed that the frequency of insertion-, localization-, and removal-related events is consistent with results previously reported from clinical trials.
Adverse Reactions from Postmarketing Spontaneous Reports
The following additional adverse reactions have been identified during post-approval use of IMPLANON and NEXPLANON. It is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure because these reactions are reported voluntarily from a population of uncertain size.
Gastrointestinal disorders: constipation, diarrhea, flatulence, vomiting.
General disorders and administration site conditions: edema, fatigue, implant site reaction, pyrexia.
Immune system disorders: anaphylactic reactions.
Infections and infestations: rhinitis, urinary tract infection.
Investigations: clinically relevant rise in blood pressure, weight decreased.
Metabolism and nutrition disorders: increased appetite.
Musculoskeletal and connective tissue disorders: arthralgia, musculoskeletal pain, myalgia.
Nervous system disorders: convulsions, migraine, somnolence.
Pregnancy, puerperium and perinatal conditions: ectopic pregnancy.
Psychiatric disorders: anxiety, insomnia, libido decreased.
Renal and urinary disorders: dysuria.
Reproductive system and breast disorders: breast discharge, breast enlargement, ovarian cyst, pruritus genital, vulvovaginal discomfort.
Skin and subcutaneous tissue disorders: angioedema, aggravation of angioedema and/or aggravation of hereditary angioedema, alopecia, chloasma, hypertrichosis, pruritus, rash, seborrhea, urticaria.
Vascular disorders: hot flush.
Reported complications related to insertion or removal of the etonogestrel implants include vasovagal reactions (e.g., hypotension, dizziness, or syncope), bruising, slight local irritation, pain, itching, fibrosis at the implant site, paresthesia or paresthesia-like events, scarring, and abscesses. Implant expulsions and migrations also have been reported. In some cases, implants have migrated to the chest wall or into the vasculature, including the pulmonary artery. Some cases of implants migrating to the pulmonary artery presented with symptoms of chest pain and/or respiratory disorders (e.g., dyspnea, cough, or hemoptysis); other cases have been reported as asymptomatic. In-patient surgical interventions might be necessary when removing implants associated with complications [see Warnings and Precautions (5.1)].
8 Use in Specific Populations
- Pregnancy: Discontinue if maintaining a pregnancy. (8.1)
6.1 Clinical Trials Experience
Adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice, because clinical trials are conducted under widely varying conditions.
In clinical trials of three years duration involving 942 women who were evaluated for safety, change in menstrual bleeding patterns (irregular menses) was the most common adverse reaction causing discontinuation of use of the non-radiopaque etonogestrel implant (IMPLANON) (11.1% of women).
Adverse reactions that resulted in a rate of discontinuation of ≥1% are shown in Table 3.
| Adverse Reactions | All Studies N = 942 |
|---|---|
| Bleeding Irregularities Includes "frequent", "heavy", "prolonged", "spotting", and other patterns of bleeding irregularity.
|
11.1% |
| Emotional Lability Among US subjects (N=330), 6.1% experienced emotional lability that led to discontinuation.
|
2.3% |
| Weight Increase | 2.3% |
| Headache | 1.6% |
| Acne | 1.3% |
| Depression Among US subjects (N=330), 2.4% experienced depression that led to discontinuation.
|
1.0% |
Other adverse reactions that were reported by at least 5% of subjects in the non-radiopaque etonogestrel implant clinical trials are listed in Table 4.
| Adverse Reactions | All Studies N = 942 |
|---|---|
| Headache | 24.9% |
| Vaginitis | 14.5% |
| Weight increase | 13.7% |
| Acne | 13.5% |
| Breast pain | 12.8% |
| Abdominal pain | 10.9% |
| Pharyngitis | 10.5% |
| Leukorrhea | 9.6% |
| Influenza-like symptoms | 7.6% |
| Dizziness | 7.2% |
| Dysmenorrhea | 7.2% |
| Back pain | 6.8% |
| Emotional lability | 6.5% |
| Nausea | 6.4% |
| Pain | 5.6% |
| Nervousness | 5.6% |
| Depression | 5.5% |
| Hypersensitivity | 5.4% |
| Insertion site pain | 5.2% |
In a clinical trial of NEXPLANON, in which investigators were asked to examine the implant site after insertion, implant site reactions were reported in 8.6% of women. Erythema was the most frequent implant site complication, reported during or shortly after insertion, occurring in 3.3% of subjects. Additionally, hematoma (3.0%), bruising (2.0%), pain (1.0%), and swelling (0.7%) were reported.
In a separate clinical trial to assess contraceptive efficacy and safety of NEXPLANON beyond 3 years, up to 5 years, where a total of 498 women were evaluated for safety, a similar adverse reaction profile was observed as in Years 1 through 3.The most frequently reported adverse reaction >5% related to NEXPLANON was intermenstrual bleeding (5.4%), Changes in menstrual bleeding patterns were the most frequently reported adverse reaction leading to discontinuation occurring in 4.0% of participants.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
- Advise women about the insertion and removal procedure of the NEXPLANON implant. Provide the woman with a copy of the Patient Labeling and ensure that she understands the information in the Patient Labeling before insertion and removal.
- Advise women to contact their healthcare professional immediately if, at any time, they are unable to palpate the implant.
- Advise women that NEXPLANON does not protect against HIV infection (AIDS) or other sexually transmitted diseases.
- Advise women that the use of NEXPLANON may be associated with changes in their normal menstrual bleeding patterns so that they know what to expect.
- MRI Safety Information: NEXPLANON is MR safe.
5.18 Drug Laboratory Test Interactions
Sex hormone-binding globulin concentrations may be decreased for the first six months after NEXPLANON insertion followed by gradual recovery. Thyroxine concentrations may initially be slightly decreased followed by gradual recovery to baseline.
5.5 Thrombotic and Other Vascular Events
The use of combination hormonal contraceptives (progestin plus estrogen) increases the risk of vascular events, including arterial events (strokes and myocardial infarctions) or deep venous thrombotic events (venous thromboembolism, deep venous thrombosis, retinal vein thrombosis, and pulmonary embolism). NEXPLANON is a progestin-only contraceptive. It is unknown whether this increased risk is applicable to etonogestrel alone. It is recommended, however, that women with risk factors known to increase the risk of venous and arterial thromboembolism be carefully assessed.
There have been postmarketing reports of serious arterial thrombotic and venous thromboembolic events, including cases of pulmonary emboli (some fatal), deep vein thrombosis, myocardial infarction, and strokes, in women using etonogestrel implants. NEXPLANON should be removed in the event of a thrombosis.
Due to the risk of thromboembolism associated with pregnancy and immediately following delivery, NEXPLANON should not be used prior to 21 days postpartum. Women with a history of thromboembolic disorders should be made aware of the possibility of a recurrence.
Evaluate for retinal vein thrombosis immediately if there is unexplained loss of vision, proptosis, diplopia, papilledema, or retinal vascular lesions.
Consider removal of the NEXPLANON implant in case of long-term immobilization due to surgery or illness.
5.3 Changes in Menstrual Bleeding Patterns
After starting NEXPLANON, women are likely to have a change from their normal menstrual bleeding pattern. These may include changes in bleeding frequency (absent, less, more frequent or continuous), intensity (reduced or increased) or duration. In clinical trials of the non-radiopaque etonogestrel implant (IMPLANON), bleeding patterns ranged from amenorrhea (1 in 5 women) to frequent and/or prolonged bleeding (1 in 5 women). The bleeding pattern experienced during the first three months of NEXPLANON use is broadly predictive of the future bleeding pattern for many women. Women should be counseled regarding the bleeding pattern changes they may experience so that they know what to expect. Abnormal bleeding should be evaluated as needed to exclude pathologic conditions or pregnancy.
In clinical studies of the non-radiopaque etonogestrel implant, reports of changes in bleeding pattern were the most common reason for stopping treatment (11.1%). Irregular bleeding (10.8%) was the single most common reason women stopped treatment, while amenorrhea (0.3%) was cited less frequently. In these studies, women had an average of 17.7 days of bleeding or spotting every 90 days (based on 3,315 intervals of 90 days recorded by 780 patients). The percentages of patients having 0, 1-7, 8-21, or >21 days of spotting or bleeding over a 90-day interval while using the non-radiopaque etonogestrel implant are shown in Table 1.
| Total Days of Bleeding or Spotting | Percentage of Patients | ||
|---|---|---|---|
| Treatment Days 91-180 (N = 745) |
Treatment Days 271-360 (N = 657) |
Treatment Days 631-720 (N = 547) |
|
| 0 Days | 19% | 24% | 17% |
| 1-7 Days | 15% | 13% | 12% |
| 8-21 Days | 30% | 30% | 37% |
| >21 Days | 35% | 33% | 35% |
Bleeding patterns observed with use of the non-radiopaque etonogestrel implant for up to 2 years, and the proportion of 90-day intervals with these bleeding patterns, are summarized in Table 2.
| BLEEDING PATTERNS | DEFINITIONS | % % = Percentage of 90-day intervals with this pattern
|
|---|---|---|
| Infrequent | Less than three bleeding and/or spotting episodes in 90 days (excluding amenorrhea) | 33.6 |
| Amenorrhea | No bleeding and/or spotting in 90 days | 22.2 |
| Prolonged | Any bleeding and/or spotting episode lasting more than 14 days in 90 days | 17.7 |
| Frequent | More than 5 bleeding and/or spotting episodes in 90 days | 6.7 |
In case of undiagnosed, persistent, or recurrent abnormal vaginal bleeding, appropriate measures should be conducted to rule out malignancy.
2.1 Initiating Contraception With Nexplanon
IMPORTANT: Rule out pregnancy before inserting the implant.
Timing of insertion depends on the woman's recent contraceptive history, as follows:
5.12 Carbohydrate and Lipid Metabolic Effects
Use of NEXPLANON may induce mild insulin resistance and small changes in glucose concentrations of unknown clinical significance. Carefully monitor prediabetic and diabetic women using NEXPLANON.
Women who are being treated for hyperlipidemia should be followed closely if they elect to use NEXPLANON. Some progestins may elevate LDL levels and may render the control of hyperlipidemia more difficult.
14.3 Implant Insertion and Removal Characteristics
Out of 301 insertions of the NEXPLANON implant in a clinical trial, the mean insertion time (from the removal of the protection cap of the applicator until retraction of the needle from the arm) was 27.9 ± 29.3 seconds. After insertion, 300 out of 301 (99.7%) NEXPLANON implants were palpable that were placed by trained providers. The single, non-palpable implant was not inserted according to the instructions.
5.7 Carcinoma of the Breast and Reproductive Organs
Women who currently have or have had breast cancer should not use hormonal contraception because breast cancer may be hormonally sensitive [see Contraindications (4)]. Some studies suggest that the use of combination hormonal contraceptives might increase the incidence of breast cancer; however, other studies have not confirmed such findings.
Some studies suggest that the use of combination hormonal contraceptives is associated with an increase in the risk of cervical cancer or intraepithelial neoplasia. However, there is controversy about the extent to which these findings are due to differences in sexual behavior and other factors.
Women with a family history of breast cancer or who develop breast nodules should be carefully monitored.
7.2 Effects of Hormonal Contraceptives On Other Drugs
Hormonal contraceptives may affect the metabolism of other drugs. Consequently, plasma concentrations may either increase (for example, cyclosporine) or decrease (for example, lamotrigine). Consult the labeling of all concurrently-used drugs to obtain further information about interactions with hormonal contraceptives or the potential for enzyme alterations.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 24-month carcinogenicity study in male and female rats with subdermal implants releasing 10 and 20 mcg etonogestrel per day (equal to approximately 1.8-3.6 times the systemic steady state exposure in women using NEXPLANON), no drug-related carcinogenic potential was observed. Etonogestrel was not genotoxic in the in vitro Ames/Salmonella reverse mutation assay, the chromosomal aberration assay in Chinese hamster ovary cells or the in vivo mouse micronucleus test. Fertility in female rats returned after withdrawal from treatment.
Principal Display Panel 68 Mg Implant Blister Pack Carton
NDC 78206-145-01
1 applicator containing 1 subdermal implant
Nexplanon®
(etonogestrel implant) 68 mg
Radiopaque
Subdermal Use Only
This product is intended to prevent pregnancy. It does not protect
against HIV infection (AIDS) and other sexually transmitted diseases.
Rx only
5.1 Risk of Complications Due to Improper Insertion and Removal
Complications of Insertion and Removal
NEXPLANON should be inserted subdermally so that it will be palpable after insertion, and this should be confirmed by palpation immediately after insertion. Failure to insert NEXPLANON properly may go unnoticed unless it is palpated immediately after insertion. Undetected failure to insert the implant may lead to an unintended pregnancy. Complications related to insertion and removal procedures may occur, e.g., pain, paresthesia, bleeding, hematoma, scarring, or infection.
If NEXPLANON is inserted deeply (intramuscular or intrafascial), neural or vascular injury may occur. To help reduce the risk of neural or vascular injury, NEXPLANON should be inserted subdermally just under the skin at the inner side of the non-dominant upper arm overlying the triceps muscle, about 8-10 cm (3-4 inches) from the medial epicondyle of the humerus, and 3-5 cm (1.25-2 inches) posterior to (below) the sulcus (groove) between the biceps and triceps muscles. This location is intended to avoid the large nerves and blood vessels lying within and surrounding the sulcus. Deep insertions of NEXPLANON have been associated with paresthesia (due to neural injury), migration of the implant (due to intramuscular or fascial insertion), and intravascular insertion. If infection develops at the insertion site, start suitable treatment. If the infection persists, the implant should be removed. Incomplete insertions or infections may lead to expulsion.
Reports of implant migration within the arm may have been related to deep insertion. Postmarketing reports of implants located within the vessels of the arm and the pulmonary artery also may have been related to deep insertions or intravascular insertions. Some cases of implants found within the pulmonary artery were associated with chest pain and/or respiratory disorders (such as dyspnea, cough, or hemoptysis); others were asymptomatic. In cases where the implant has migrated to the pulmonary artery, endovascular or surgical procedures may be needed for removal.
Implant removal may be difficult or impossible if the implant is not inserted correctly, is inserted too deeply, not palpable, encased in fibrous tissue, or has migrated. If at any time the implant cannot be palpated, it should be localized, and removal is recommended. When an implant is removed, it is important to remove it in its entirety [see Dosage and Administration (2.3)].
Exploratory surgery without knowledge of the exact location of the implant is strongly discouraged. Removal of deeply inserted implants should be conducted with caution in order to prevent injury to deeper neural or vascular structures in the arm and be performed by healthcare professionals familiar with the anatomy of the arm. If the implant is located in the chest, healthcare professionals familiar with the anatomy of the chest should be consulted. Failure to remove the implant may result in continued effects of etonogestrel, such as compromised fertility, ectopic pregnancy, or persistence or occurrence of a drug-related adverse event.
Broken or Bent Implants
Cases of breakage or bending of implants while inserted within a patient’s arm have been reported. Cases of migration of a broken implant fragment within the arm have also occurred. These cases may be related to external forces, e.g., manipulation of the implant or contact sports. The release rate of etonogestrel may be slightly increased in a broken or bent implant, based on in vitro data. As noted previously, when an implant is removed, it is important to remove it in its entirety [see Dosage and Administration (2.3)].
NEXPLANON is available only through a restricted program under a REMS [see Warnings and Precautions (5.2) ].
Warning: Risk of Complications Due to Improper Insertion and Removal
- Improper insertion of NEXPLANON increases the risk of complications [see Warnings and Precautions (5.1)].
- Proper training prior to first use of NEXPLANON can minimize the risk of improper NEXPLANON insertion [see Warnings and Precautions (5.1)].
- Because of the risk of complications due to improper insertion and removal NEXPLANON is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the NEXPLANON REMS [see Warnings and Precautions (5.2)].
Structured Label Content
Section 42229-5 (42229-5)
• No preceding hormonal contraceptive use in the past month
NEXPLANON should be inserted between Day 1 (first day of menstrual bleeding) and Day 5 of the menstrual cycle, even if the woman is still bleeding.
If inserted as recommended, back-up contraception is not necessary. If deviating from the recommended timing of insertion, the woman should be advised to use a barrier method until 7 days after insertion. If intercourse has already occurred, pregnancy should be excluded.
Section 42230-3 (42230-3)
FDA-Approved Patient Labeling
NEXPLANON® (etonogestrel implant)
Radiopaque
Subdermal Use Only
NEXPLANON® does not protect against HIV infection (the virus that causes AIDS) or other sexually transmitted diseases.
Read this Patient Information leaflet carefully before you decide if NEXPLANON is right for you. This information does not take the place of talking with your healthcare professional. If you have any questions about NEXPLANON, ask your healthcare professional.
What is NEXPLANON?
NEXPLANON is a hormone-releasing birth control implant for prevention of pregnancy for up to 5 years. The implant is a flexible plastic rod about the size of a matchstick that contains a progestin hormone called etonogestrel. It contains a small amount of barium sulfate (15 mg), so that the implant can be seen by X-ray, an ethylene vinyl acetate (EVA) copolymer (28% vinyl acetate, 43 mg) core, and magnesium stearate (0.1 mg). Your healthcare professional will insert the implant just under the skin of the inner side of your upper arm. You can use a single NEXPLANON implant for up to 5 years. NEXPLANON does not contain estrogen.
What if I need birth control for more than 5 years?
The NEXPLANON implant must be removed by the end of 5 years. Your healthcare professional can insert a new implant under your skin after taking out the old one if you choose to continue using NEXPLANON for birth control.
What if I change my mind about birth control and want to stop using NEXPLANON before 5 years?
Your healthcare professional can remove the implant at any time. You may become pregnant as early as the first week after removal of the implant. If you do not want to get pregnant after your healthcare professional removes the NEXPLANON implant, you should start another birth control method right away.
How does NEXPLANON work?
NEXPLANON prevents pregnancy in several ways. The most important way is by stopping the release of an egg from your ovary. NEXPLANON also thickens the mucus in your cervix and this change may keep sperm from reaching the egg. NEXPLANON also changes the lining of your uterus.
How well does NEXPLANON work?
When the NEXPLANON implant is placed correctly, your chance of getting pregnant is very low (less than 1 pregnancy per 100 women who use NEXPLANON for 1 year).
The following chart shows the chance of getting pregnant for women who use different methods of birth control. Each box on the chart contains a list of birth control methods that are similar in effectiveness. The most effective methods are at the top of the chart. The box on the bottom of the chart shows the chance of getting pregnant for women who do not use birth control and are trying to get pregnant.
Who should not use NEXPLANON?
Do not use NEXPLANON if you have any of the following conditions:
-
Are pregnant or think you may be pregnant
- Have, or have had, blood clots, such as blood clots in your legs (deep vein thrombosis), lungs (pulmonary embolism), eyes (total or partial blindness), heart (heart attack), or brain (stroke)
- Liver disease or a liver tumor
- Unexplained vaginal bleeding
- Breast cancer or any other cancer that is sensitive to progestin (a female hormone), now or in the past
- An allergy to anything in NEXPLANON
Tell your healthcare professional if you have or have had any of the conditions listed above. Your healthcare professional can suggest a different method of birth control.
In addition, talk to your healthcare professional about using NEXPLANON if you have any of the following conditions:
- Diabetes
- High cholesterol or triglycerides
- Headaches
- Gallbladder or kidney problems
- A history of depressed mood
- High blood pressure
- An allergy to numbing medicines (anesthetics) or medicines used to clean your skin (antiseptics). These medicines will be used when the implant is placed into or removed from your arm.
Interaction with Other Medicines
Tell your healthcare professional about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Using certain medicines with NEXPLANON, including those listed below, may make NEXPLANON less effective:
- aprepitant
- barbiturates
- bosentan
- carbamazepine
- felbamate
- griseofulvin
- oxcarbazepine
- phenytoin
- rifampin
- St. John's wort
- topiramate
- HIV medicines
- Hepatitis C Virus medicines
Ask your healthcare professional if you are not sure if your medicine is one listed above.
If you are taking medicines or herbal products that might make NEXPLANON less effective, you and your healthcare professional may decide to leave NEXPLANON in place; in that case, an additional non-hormonal contraceptive should be used. It is necessary to use the additional non-hormonal contraceptive for 28 days after stopping the medicine or herbal product because its effect on NEXPLANON may last that long.
When you are using NEXPLANON, tell all your healthcare professionals that you have NEXPLANON in place in your arm.
Magnetic Resonance Imaging (MRI) Safety Information
NEXPLANON is MRI safe.
How is the NEXPLANON implant placed and removed?
Your healthcare professional will place and remove the NEXPLANON implant using a minor surgical procedure in his or her office. The implant is placed just under the skin on the inner side of your non-dominant upper arm.
The timing of insertion is important. Your healthcare professional may take the following steps to guide timing of insertion:
- Perform a pregnancy test before inserting NEXPLANON
- Schedule the insertion at a specific time of your menstrual cycle (for example, within the first 5 days of your regular menstrual bleeding). If the implant is placed after the fifth day of menses, then you should use an additional contraceptive method (such as a condom) for the first 7 days after insertion.
Your healthcare professional will cover the site where NEXPLANON was placed with 2 bandages. A small bandage will be placed over the insertion site. A larger pressure bandage will be placed over the smaller bandage. Leave the larger pressure bandage on for 24 hours. Keep the smaller bandage clean, dry, and in place for 3 to 5 days.
Immediately after the NEXPLANON implant has been placed, you and your healthcare professional should check that the implant is in your arm by feeling for it.
If you cannot feel the implant immediately after insertion, the implant may not have been inserted, or it may have been inserted deeply. A deep insertion may cause problems with locating and removing the implant. When the healthcare professional has located the implant, it should be removed.
If at any time you cannot feel the NEXPLANON implant, contact your healthcare professional immediately and use a non-hormonal birth control method (such as condoms) until your healthcare professional confirms that the implant is in place. You may need special tests to check that the implant is in place or to help find the implant when it is time to take it out. If the implant cannot be found in the arm after a thorough search, your healthcare professional may use X-rays or other imaging methods on your chest.
Depending on the exact position of the implant, removal may be difficult and may require surgery.
Keep track of the date the implant is to be removed. Schedule an appointment with your healthcare professional to remove the implant on or before the removal date.
Be sure to have checkups as advised by your healthcare professional.
What are the most common side effects I can expect while using NEXPLANON?
• Changes in Menstrual Bleeding Patterns (Menstrual Periods)
The most common side effect of NEXPLANON is a change in your normal menstrual bleeding pattern. In studies, one out of ten women stopped using the implant because of an unfavorable change in their bleeding pattern. You may experience longer or shorter bleeding during your periods or have no bleeding at all. The time between periods may vary, and you may also have spotting in between periods.
Tell your healthcare professional right away if you have the following concerns:
- You think you may be pregnant
- Your menstrual bleeding is heavy and prolonged
The following additional frequent side effects also caused women to stop using the implant:
- Mood swings
- Depressed mood
- Weight gain
- Headache
- Acne
Other common side effects include the following conditions:
- Headache
- Vaginitis (inflammation of the vagina)
- Weight gain
- Acne
- Breast pain
- Viral infections such as sore throats or flu-like symptoms
- Stomach pain
- Painful periods
- Mood swings, nervousness, or depressed mood
- Back pain
- Nausea
- Dizziness
- Pain
- Pain at the site of insertion
Implants have been reported to be found in a blood vessel, including a blood vessel in the lung which can be associated with shortness of breath, cough and/or the coughing up of blood or blood-stained mucus.
These are not all the possible side effects of NEXPLANON. Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
What are the possible risks of using NEXPLANON?
• Problems with Insertion and Removal
The implant may not be actually in your arm due to a failed insertion. If this happens, you may become pregnant. Immediately after insertion, and with help from your healthcare professional, you should be able to feel the implant under your skin. If you can’t feel the implant, tell your healthcare professional.
Location and removal of the implant may be difficult or impossible because the implant is not where it should be. Special procedures, including surgery in the hospital, may be needed to remove the implant. If the implant is not removed, then the effects of NEXPLANON will continue for a longer time.
Implants have been found in the pulmonary artery (a blood vessel in the lung). If the implant cannot be found in the arm, your healthcare professional may use X-rays or other imaging methods on the chest. If the implant is located in the chest, surgery may be needed.
The following problems related to insertion and removal may also occur:
- Vasovagal reactions (such as a drop in blood pressure, dizziness, or fainting)
- Pain, irritation, swelling, or bruising at the insertion site
- Numbness and tingling at the insertion site
- Scarring, including a thick scar called a keloid around the insertion site
- Infection
- Scar tissue may form around the implant making it difficult to remove
- The implant may come out by itself. You may become pregnant if the implant comes out by itself. Use a back-up birth control method and call your healthcare professional right away if the implant comes out.
- The need for surgery in the hospital to remove the implant
- Injury to nerves or blood vessels in your arm
- The implant breaks, making removal difficult
• Ectopic Pregnancy
If you become pregnant while using NEXPLANON, you have a slightly higher chance that the pregnancy will be ectopic (occurring outside the womb) than do women who do not use birth control. Unusual vaginal bleeding or lower stomach (abdominal) pain may be a sign of ectopic pregnancy. Ectopic pregnancy is a medical emergency that often requires surgery. Ectopic pregnancies can cause serious internal bleeding, infertility, and even death. Call your healthcare professional right away if you think you are pregnant or have unexplained lower stomach (abdominal) pain.
• Ovarian Cysts
Cysts may develop on the ovaries and usually go away without treatment, but sometimes surgery is needed to remove them.
• Breast Cancer
It is not known whether NEXPLANON use changes a woman's risk for breast cancer. If you have breast cancer now, or have had it in the past, do not use NEXPLANON because some breast cancers are sensitive to hormones.
• Serious Blood Clots
NEXPLANON may increase your chance of serious blood clots, especially if you have other risk factors such as smoking. It is possible to die from a problem caused by a blood clot, such as a heart attack or a stroke.
Serious blood clots can occur within blood vessels of different parts of the body, including the following examples:
- Legs (deep vein thrombosis)
- Lungs (pulmonary embolism)
- Brain (stroke)
- Heart (heart attack)
- Eyes (total or partial blindness)
The risk of serious blood clots is increased in women who smoke. If you smoke and want to use NEXPLANON, you should quit. Your healthcare professional may be able to help.
Tell your healthcare professional at least 4 weeks before if you are going to have surgery or will need to be on bed rest. You have an increased chance of getting blood clots during surgery or bed rest.
• Other Risks
A few women who use birth control that contains hormones may get the following conditions:
- High blood pressure
- Gallbladder problems
- Rare cancerous or noncancerous liver tumors
• Broken or Bent Implant
Breakage or bending of the implant while it is in your arm may occur due to external forces (such as manipulation of the implant or contact sports). A broken implant may move from the insertion site. If you feel that the implant may have broken or bent while in your arm, contact your healthcare professional.
When should I call my healthcare professional?
Call your healthcare professional right away if you have any of the following conditions:
- Pain in your lower leg that does not go away
- Severe chest pain or heaviness in the chest
- Sudden shortness of breath, sharp chest pain, or coughing blood
- Symptoms of a severe allergic reaction, such as swollen face, tongue or throat; trouble breathing or swallowing
- Sudden severe headache unlike your usual headaches
- Weakness or numbness in your arm, leg, or trouble speaking
- Sudden partial or complete blindness
- Yellowing of your skin or whites of your eyes, especially with fever, tiredness, loss of appetite, dark colored urine, or light-colored bowel movements
- Severe pain, swelling, or tenderness in the lower stomach (abdomen)
- Lump in your breast
- Problems sleeping, lack of energy, tiredness, or you feel very sad
- Heavy menstrual bleeding
What if I become pregnant while using NEXPLANON?
You should see your healthcare professional right away if you think that you may be pregnant. It is important to remove the implant and make sure that the pregnancy is not ectopic (occurring outside the womb). Based on experience with other hormonal contraceptives, NEXPLANON is not likely to cause birth defects.
Can I use NEXPLANON when I am breastfeeding?
If you are breastfeeding your child, you may use NEXPLANON if 4 weeks have passed since you had your baby. A small amount of the hormone contained in NEXPLANON passes into your breast milk. The health of breast-fed children whose mothers were using the implant has been studied up to 3 years of age in a small number of children. No effects on the growth and development of the children were seen. If you are breastfeeding and want to use NEXPLANON, talk with your healthcare professional for more information.
General information about the safe and effective use of NEXPLANON
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. If you would like more information, talk with your healthcare professional. You can ask your healthcare professional for information about NEXPLANON that is written for healthcare professionals.
Section 43683-2 (43683-2)
10 Overdosage (10 OVERDOSAGE)
Overdosage may result if more than one implant is inserted. In case of suspected overdose, the implant should be removed.
11 Description (11 DESCRIPTION)
NEXPLANON is a radiopaque, progestin-only, soft, flexible implant preloaded in a sterile, disposable applicator for subdermal use. The implant is white/off-white, non-biodegradable and 4 cm in length with a diameter of 2 mm (see Figure 19). Each implant consists of an ethylene vinyl acetate (EVA) copolymer (28% vinyl acetate, 43 mg) core, containing 68 mg of the synthetic progestin etonogestrel, barium sulfate ((15 mg), radiopaque ingredient), and magnesium stearate (0.1 mg), surrounded by an EVA copolymer skin. Once inserted subdermally, the release rate is 60-75 mcg/day in week 5-6 and decreases to approximately 40-50 mcg/day at the end of the first year, and then to approximately 15-20 mcg/day at the end of the fifth year. NEXPLANON is a progestin-only contraceptive and does not contain estrogen. NEXPLANON is not made with natural rubber latex.
| Figure 19 (Not to scale) |
Etonogestrel [13-Ethyl-17-hydroxy-11-methylene-18,19-dinor-17α-pregn-4-en-20-yn-3-one], structurally derived from 19-nortestosterone, is the synthetic biologically active metabolite of the synthetic progestin desogestrel. It has a molecular weight of 324.46 and the following structural formula (Figure 20).
| Figure 20 |
14.1 Pregnancy
In clinical trials of up to 3 years duration that involved 923 subjects, 18-40 years of age at entry, and 1756 women-years of use with the non-radiopaque etonogestrel implant (IMPLANON), the total exposures expressed as 28-day cycle equivalents by study year were:
Year 1: 10,866 cycles
Year 2: 8,581 cycles
Year 3: 3,442 cycles
The clinical trials excluded women who:
- Weighed more than 130% of their ideal body weight
- Were chronically taking medications that induce liver enzymes
In the subgroup of women, 18-35 years of age at entry, 6 pregnancies during 20,648 cycles of use were reported. Two pregnancies occurred in each of Years 1, 2, and 3. Each conception was likely to have occurred shortly before or within 2 weeks after removal of the non-radiopaque etonogestrel implant. With these 6 pregnancies, the cumulative Pearl Index (PI) was 0.38 pregnancies per 100 women-years of use.
The contraceptive efficacy of NEXPLANON during use from 3 to 5 years was evaluated in a multicenter, single-arm, open-label study (NCT04626596) conducted in the United States. The trial enrolled women 18 to 35 years of age who had been using NEXPLANON for 36 months (±2 weeks) from the date of insertion at the time of enrollment. A total of 399 women were evaluated, having a mean age of 27 years.
Participants were 74.2% White, 16.8% Black/African American, 3.8% Asian, 1.3% American Indian or Alaska Native, 0.5% Native Hawaiian or Other Pacific Islander, and 3.5% multiple or missing races. The mean BMI was 29.4 kg/m2 (range: 17.2-64.3 kg/m2) and the mean weight was 78.7 kg (range: 40.8-180.8 kg). One hundred fifty-two participants (38.1%) had a BMI ≥30 kg/m2, including 40 participants (10.0%) with BMI ≥40 kg/m2.
The total exposures expressed as 28-day cycle equivalents by study year were:
Year 4: 4478 cycles
Year 5: 3274 cycles
No pregnancies were reported in years 4 and 5, with a PI of 0.0 (95% CI- 0.00, 0.69) pregnancies per 100 women-years of use.
5.17 Monitoring
A woman who is using NEXPLANON should have a yearly visit with her healthcare professional for a blood pressure check and for other indicated health care.
5.9 Weight Gain
In clinical studies, mean weight gain in U.S. non-radiopaque etonogestrel implant (IMPLANON) users was 2.8 pounds after one year and 3.7 pounds after two years. How much of the weight gain was related to the non-radiopaque etonogestrel implant is unknown. In studies, 2.3% of the users reported weight gain as the reason for having the non-radiopaque etonogestrel implant removed.
16.1 How Supplied
NEXPLANON is supplied as follows:
NDC 78206-145-01
One NEXPLANON package consists of a single implant containing 68 mg etonogestrel, 15 mg of barium sulfate and 0.1 mg of magnesium stearate that is 4 cm in length and 2 mm in diameter, which is pre-loaded in the needle of a disposable applicator. The sterile applicator containing the implant is packed in a blister pack.
5.6 Ovarian Cysts
If follicular development occurs, atresia of the follicle is sometimes delayed, and the follicle may continue to grow beyond the size it would attain in a normal cycle. Generally, these enlarged follicles disappear spontaneously. On rare occasion, surgery may be required.
5.8 Liver Disease
Disturbances of liver function may necessitate the discontinuation of hormonal contraceptive use until markers of liver function return to normal. Remove NEXPLANON if jaundice develops.
Hepatic adenomas are associated with combination hormonal contraceptives use. An estimate of the attributable risk is 3.3 cases per 100,000 for combination hormonal contraceptives users. It is not known whether a similar risk exists with progestin-only methods like NEXPLANON.
The progestin in NEXPLANON may be poorly metabolized in women with liver impairment. Use of NEXPLANON in women with active liver disease or liver cancer is contraindicated [see Contraindications (4)].
8.4 Pediatric Use
The safety and effectiveness of NEXPLANON have been established in women of reproductive potential. Safety and effectiveness of NEXPLANON are expected to be the same in postpubertal adolescents as in adult women.
NEXPLANON is not indicated before menarche.
8.5 Geriatric Use
This product has not been studied in women over 65 years of age and is not indicated in this population.
5.2 Nexplanon Rems (5.2 NEXPLANON REMS)
NEXPLANON is only available through a restricted program under a REMS called NEXPLANON REMS because of the risk of complications due to improper insertion and removal [see Warnings and Precautions (5.1)].
Notable requirements of the NEXPLANON REMS include the following:
- Healthcare providers must be certified with the program by enrolling and completing training on the proper insertion and removal of NEXPLANON prior to first use.
- Pharmacies must be certified with the program and must only dispense NEXPLANON to certified healthcare providers who dispense NEXPLANON for insertion.
- Wholesalers and distributors must be registered with the program and must only distribute to certified pharmacies and certified healthcare providers.
Further information is available at www.NEXPLANONREMS.com and 1-833-697-7367.
4 Contraindications (4 CONTRAINDICATIONS)
NEXPLANON should not be used in women who have
- Known or suspected pregnancy
- Current or past history of thrombosis or thromboembolic disorders
- Liver tumors, benign or malignant, or active liver disease
- Undiagnosed abnormal uterine bleeding
- Known or suspected breast cancer, personal history of breast cancer, or other progestin-sensitive cancer, now or in the past
- Allergic reaction to any of the components of NEXPLANON [see Adverse Reactions (6)]
5.13 Depressed Mood
Women with a history of depressed mood should be carefully observed. Consideration should be given to removing NEXPLANON in patients who become significantly depressed.
5.16 Contact Lenses
Contact lens wearers who develop visual changes or changes in lens tolerance should be assessed by an ophthalmologist.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions reported with the use of hormonal contraception are discussed elsewhere in the labeling:
- Changes in Menstrual Bleeding Patterns [see Warnings and Precautions (5.3)]
- Ectopic Pregnancies [see Warnings and Precautions (5.4)]
- Thrombotic and Other Vascular Events [see Warnings and Precautions (5.5)]
- Liver Disease [see Warnings and Precautions (5.8)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Consult the labeling of concurrently-used drugs to obtain further information about interactions with hormonal contraceptives or the potential for enzyme alterations.
5.15 Fluid Retention
Hormonal contraceptives may cause some degree of fluid retention. They should be prescribed with caution, and only with careful monitoring, in patients with conditions which might be aggravated by fluid retention. It is unknown if NEXPLANON causes fluid retention.
12.2 Pharmacodynamics
Exposure-response relationships of NEXPLANON are unknown.
8.6 Hepatic Impairment
No studies were conducted to evaluate the effect of hepatic disease on the disposition of NEXPLANON. The use of NEXPLANON in women with active liver disease is contraindicated [see Contraindications (4)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
NEXPLANON® is indicated for prevention of pregnancy in women of reproductive potential for up to 5 years.
2.4 Replacing Nexplanon (2.4 Replacing NEXPLANON)
Immediate replacement can be done after removal of the previous implant and is similar to the insertion procedure described in section 2.2 Insertion of NEXPLANON.
The new implant may be inserted in the same arm, and through the same incision from which the previous implant was removed, if the site is in the correct location, i.e., 8-10 cm from the medial epicondyle of the humerus and 3-5 cm posterior to (below) the sulcus [see Dosage and Administration (2.2)]. If the same incision is being used to insert a new implant, anesthetize the insertion site [for example, 2 mL lidocaine (1%)] applying it just under the skin along the 'insertion canal.'
Follow the subsequent steps in the insertion instructions [see Dosage and Administration (2.2)].
5.4 Ectopic Pregnancies
As with all progestin-only contraceptive products, be alert to the possibility of an ectopic pregnancy among women using NEXPLANON who become pregnant or complain of lower abdominal pain. Although ectopic pregnancies are uncommon among women using NEXPLANON, a pregnancy that occurs in a woman using NEXPLANON may be more likely to be ectopic than a pregnancy occurring in a woman using no contraception.
12.1 Mechanism of Action
The contraceptive effect of NEXPLANON is achieved by suppression of ovulation, increased viscosity of the cervical mucus, and alterations in the endometrium.
14.2 Return to Ovulation
In clinical trials with the non-radiopaque etonogestrel implant (IMPLANON), the etonogestrel levels in blood decreased below sensitivity of the assay by one week after removal of the implant. In addition, pregnancies were observed to occur as early as 7 to 14 days after removal. Therefore, a woman should re-start contraception immediately after removal of the implant if continued contraceptive protection is desired.
5.11 Gallbladder Disease
Studies suggest a small increased relative risk of developing gallbladder disease among combination hormonal contraceptive users. It is not known whether a similar risk exists with progestin-only methods like NEXPLANON.
5.14 Return to Ovulation
In clinical trials with the non-radiopaque etonogestrel implant (IMPLANON), the etonogestrel levels in blood decreased below sensitivity of the assay by one week after removal of the implant. In addition, pregnancies were observed to occur as early as 7 to 14 days after removal. Therefore, a woman should re-start contraception immediately after removal of the implant if continued contraceptive protection is desired.
16.2 Storage and Handling
Store NEXPLANON at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted between 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature]. Avoid storing NEXPLANON at temperatures above 30ºC (86ºF).
2.2 Insertion of Nexplanon (2.2 Insertion of NEXPLANON)
The basis for successful use and subsequent removal of NEXPLANON is a correct and carefully performed subdermal insertion of the single, rod-shaped implant in accordance with the instructions. Both the healthcare professional and the woman should be able to feel the implant under the skin after placement.
All healthcare professionals performing insertions and/or removals of NEXPLANON must receive instructions and training prior to inserting or removing the implant.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
The following information is based on experience with the etonogestrel implants (IMPLANON and/or NEXPLANON), other progestin-only contraceptives, or experience with combination (estrogen plus progestin) oral contraceptives.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The efficacy of NEXPLANON does not depend on daily, weekly, or monthly administration.
All healthcare professionals should receive instruction and training prior to performing insertion and/or removal of NEXPLANON.
A single NEXPLANON implant is inserted subdermally just under the skin at the inner side of the non-dominant upper arm. The insertion site is overlying the triceps muscle about 8-10 cm (3-4 inches) from the medial epicondyle of the humerus and 3-5 cm (1.25-2 inches) posterior to (below) the sulcus (groove) between the biceps and triceps muscles. This location is intended to avoid the large blood vessels and nerves lying within and surrounding the sulcus (see Figures 2a, 2b and 2c). Inserting an implant more deeply than subdermally (a deep insertion) may preclude palpation and localization, making removal difficult or impossible [see Dosage and Administration (2.3) and Warnings and Precautions (5.1)].
NEXPLANON must be inserted by the expiration date stated on the packaging. NEXPLANON is a long-acting (up to 5 years), reversible, hormonal contraceptive method. The implant must be removed by the end of the fifth year and may be replaced by a new implant at the time of removal, if continued contraceptive protection is desired.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Single, white/off-white, soft, radiopaque, flexible, ethylene vinyl acetate (EVA) copolymer implant, 4 cm in length and 2 mm in diameter containing 68 mg etonogestrel, 15 mg of barium sulfate, and 0.1 mg of magnesium stearate.
5.10 Elevated Blood Pressure
Women with a history of hypertension-related diseases or renal disease should be discouraged from using hormonal contraception. For women with well-controlled hypertension, use of NEXPLANON can be considered. Women with hypertension using NEXPLANON should be closely monitored. If sustained hypertension develops during the use of NEXPLANON, or if a significant increase in blood pressure does not respond adequately to antihypertensive therapy, NEXPLANON should be removed.
6.2 Postmarketing Experience
Adverse Reactions and Events from Postmarketing Study
Nexplanon Observational Risk Assessment Study (NORA)
A postmarketing prospective active surveillance study was conducted among 7,364 patients in the United States to characterize the frequency of insertion-, localization-, and removal-related events.
Implant Insertion
Insertion difficulty or an insertion-related event occurred in 2.6% of the study participants. The overall incidence of incorrect insertion (unrecognized non-insertion, partial insertion, and deep insertion), reported by healthcare professionals was 12.6 per 1,000 insertions (95% CI, 10.2, 15.5). Table 5 summarizes the types and frequencies of these incorrect insertions.
| Type of Incorrect Insertion Event | Number of Events Total Insertion Procedures = 7,364
|
Incidence per 1,000 Insertions (95% CI) |
|---|---|---|
| (Initially) Unrecognized Non-insertions | 1 | 0.1 (0.0-0.8) |
| Partial Insertions | 27 | 3.7 (2.4-5.3) |
| Deep Insertions | 65 | 8.8 (6.8-11.2) |
| Injury to nerve or blood vessel | 1 | 0.1 (0.0-0.8) |
| Implant located within muscle | 2 | 0.3 (0.0-1.0) |
| Implant located adjacent to fascial tissue | 56 | 7.6 (5.8-9.9) |
| Implant not palpable | 6 | 0.8 (0.3-1.8) |
Implant Removal
Implant removal information from both healthcare professionals and patients was collected for 5,159 patients (70% of the study population). Of these patients, data were available from healthcare professionals regarding 4,373 removal procedures. Healthcare professionals reported removal-related difficulties or complications in 1.5% of removal procedures. Table 6 provides a summary.
| Removal Related Events | Number of Events Total Removal Procedures: N= 4,373
|
Incidence per 1,000 Removals (95% CI) |
|---|---|---|
| Any Event Limited to one event per removal procedure
|
60 | 13.7 (10.5-17.6) |
| Encased in Fibrotic Tissue | 29 | 6.6 (4.4-9.5) |
| Implant Too Deep | 11 | 2.5 (1.3-4.5) |
| Implant Migrated Only local migrations within the arm reported
|
6 | 1.4 (0.5-3.0) |
| Multiple Attempts Required | 13 | 3.0 (1.6-5.1) |
| Other Other included fragmented or bent implants, patient-related issues, wound care required, two incisions required, and difficulty identifying end of device.
|
14 | 3.2 (1.8-5.4) |
At the time of implant removal, eighteen implants (0.4% of all localizations or removals) were not palpable by the healthcare professionals. Of these eighteen, eleven were localized and removed, and one was localized but left in situ. Removal was not attempted for six non-palpable implants due to underlying health conditions, administrative problems, or unspecified reasons.
There were no reports of implants having migrated more than a few centimeters from the insertion site and no reports of implants localized at a site other than the arm. No neurovascular injuries were reported by healthcare professionals.
Adverse Reactions Reported by Patients
Table 7 provides a summary of adverse reactions reported by patients at the time of implant insertion and after removal.
| Patient Reported Adverse Reactions | At Insertion | After Removal | ||
|---|---|---|---|---|
| N Total Insertion Procedures: N = 7,364
|
Incidence per 1000 insertions (95% CI) | N | Incidence per 1000 insertions (95% CI) | |
| Any Event Limited to one event per woman
|
49 | 6.7 (4.9-8.8) | 42 Based on 3,447 questionnaires
|
5.7 (4.1-7.7) |
| Pins and Needles/Numbness (arm/hand/fingers) | 17 | 2.3 (1.4-3.7) | 24 | 3.3 (2.1-4.9) |
| Severe Pain | 10 | 1.4 (0.7-2.5) | 11 | 1.5 (0.8-2.7) |
| Altered Strength/Movement | 3 | 0.4 (0.1-1.2) | 8 | 1.1 (0.5-2.1) |
| Injury to Blood Vessels or Blood Clots in Arm No blood clots were observed during the study
|
2 | 0.3 (0-1.0) | -- | -- |
| Other “Other” included localized or insertion site pain, soreness, tenderness, dermatological changes, itching, bruising, and infection, local migrations within the arm, and physical damage to the implant (e.g., fractured or bent implant).
|
22 | 3.0 (1.9-4.5) | 18 | 2.4 (1.5-3.9) |
In summary, this prospective active surveillance study showed that the frequency of insertion-, localization-, and removal-related events is consistent with results previously reported from clinical trials.
Adverse Reactions from Postmarketing Spontaneous Reports
The following additional adverse reactions have been identified during post-approval use of IMPLANON and NEXPLANON. It is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure because these reactions are reported voluntarily from a population of uncertain size.
Gastrointestinal disorders: constipation, diarrhea, flatulence, vomiting.
General disorders and administration site conditions: edema, fatigue, implant site reaction, pyrexia.
Immune system disorders: anaphylactic reactions.
Infections and infestations: rhinitis, urinary tract infection.
Investigations: clinically relevant rise in blood pressure, weight decreased.
Metabolism and nutrition disorders: increased appetite.
Musculoskeletal and connective tissue disorders: arthralgia, musculoskeletal pain, myalgia.
Nervous system disorders: convulsions, migraine, somnolence.
Pregnancy, puerperium and perinatal conditions: ectopic pregnancy.
Psychiatric disorders: anxiety, insomnia, libido decreased.
Renal and urinary disorders: dysuria.
Reproductive system and breast disorders: breast discharge, breast enlargement, ovarian cyst, pruritus genital, vulvovaginal discomfort.
Skin and subcutaneous tissue disorders: angioedema, aggravation of angioedema and/or aggravation of hereditary angioedema, alopecia, chloasma, hypertrichosis, pruritus, rash, seborrhea, urticaria.
Vascular disorders: hot flush.
Reported complications related to insertion or removal of the etonogestrel implants include vasovagal reactions (e.g., hypotension, dizziness, or syncope), bruising, slight local irritation, pain, itching, fibrosis at the implant site, paresthesia or paresthesia-like events, scarring, and abscesses. Implant expulsions and migrations also have been reported. In some cases, implants have migrated to the chest wall or into the vasculature, including the pulmonary artery. Some cases of implants migrating to the pulmonary artery presented with symptoms of chest pain and/or respiratory disorders (e.g., dyspnea, cough, or hemoptysis); other cases have been reported as asymptomatic. In-patient surgical interventions might be necessary when removing implants associated with complications [see Warnings and Precautions (5.1)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Discontinue if maintaining a pregnancy. (8.1)
6.1 Clinical Trials Experience
Adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice, because clinical trials are conducted under widely varying conditions.
In clinical trials of three years duration involving 942 women who were evaluated for safety, change in menstrual bleeding patterns (irregular menses) was the most common adverse reaction causing discontinuation of use of the non-radiopaque etonogestrel implant (IMPLANON) (11.1% of women).
Adverse reactions that resulted in a rate of discontinuation of ≥1% are shown in Table 3.
| Adverse Reactions | All Studies N = 942 |
|---|---|
| Bleeding Irregularities Includes "frequent", "heavy", "prolonged", "spotting", and other patterns of bleeding irregularity.
|
11.1% |
| Emotional Lability Among US subjects (N=330), 6.1% experienced emotional lability that led to discontinuation.
|
2.3% |
| Weight Increase | 2.3% |
| Headache | 1.6% |
| Acne | 1.3% |
| Depression Among US subjects (N=330), 2.4% experienced depression that led to discontinuation.
|
1.0% |
Other adverse reactions that were reported by at least 5% of subjects in the non-radiopaque etonogestrel implant clinical trials are listed in Table 4.
| Adverse Reactions | All Studies N = 942 |
|---|---|
| Headache | 24.9% |
| Vaginitis | 14.5% |
| Weight increase | 13.7% |
| Acne | 13.5% |
| Breast pain | 12.8% |
| Abdominal pain | 10.9% |
| Pharyngitis | 10.5% |
| Leukorrhea | 9.6% |
| Influenza-like symptoms | 7.6% |
| Dizziness | 7.2% |
| Dysmenorrhea | 7.2% |
| Back pain | 6.8% |
| Emotional lability | 6.5% |
| Nausea | 6.4% |
| Pain | 5.6% |
| Nervousness | 5.6% |
| Depression | 5.5% |
| Hypersensitivity | 5.4% |
| Insertion site pain | 5.2% |
In a clinical trial of NEXPLANON, in which investigators were asked to examine the implant site after insertion, implant site reactions were reported in 8.6% of women. Erythema was the most frequent implant site complication, reported during or shortly after insertion, occurring in 3.3% of subjects. Additionally, hematoma (3.0%), bruising (2.0%), pain (1.0%), and swelling (0.7%) were reported.
In a separate clinical trial to assess contraceptive efficacy and safety of NEXPLANON beyond 3 years, up to 5 years, where a total of 498 women were evaluated for safety, a similar adverse reaction profile was observed as in Years 1 through 3.The most frequently reported adverse reaction >5% related to NEXPLANON was intermenstrual bleeding (5.4%), Changes in menstrual bleeding patterns were the most frequently reported adverse reaction leading to discontinuation occurring in 4.0% of participants.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
- Advise women about the insertion and removal procedure of the NEXPLANON implant. Provide the woman with a copy of the Patient Labeling and ensure that she understands the information in the Patient Labeling before insertion and removal.
- Advise women to contact their healthcare professional immediately if, at any time, they are unable to palpate the implant.
- Advise women that NEXPLANON does not protect against HIV infection (AIDS) or other sexually transmitted diseases.
- Advise women that the use of NEXPLANON may be associated with changes in their normal menstrual bleeding patterns so that they know what to expect.
- MRI Safety Information: NEXPLANON is MR safe.
5.18 Drug Laboratory Test Interactions (5.18 Drug-Laboratory Test Interactions)
Sex hormone-binding globulin concentrations may be decreased for the first six months after NEXPLANON insertion followed by gradual recovery. Thyroxine concentrations may initially be slightly decreased followed by gradual recovery to baseline.
5.5 Thrombotic and Other Vascular Events
The use of combination hormonal contraceptives (progestin plus estrogen) increases the risk of vascular events, including arterial events (strokes and myocardial infarctions) or deep venous thrombotic events (venous thromboembolism, deep venous thrombosis, retinal vein thrombosis, and pulmonary embolism). NEXPLANON is a progestin-only contraceptive. It is unknown whether this increased risk is applicable to etonogestrel alone. It is recommended, however, that women with risk factors known to increase the risk of venous and arterial thromboembolism be carefully assessed.
There have been postmarketing reports of serious arterial thrombotic and venous thromboembolic events, including cases of pulmonary emboli (some fatal), deep vein thrombosis, myocardial infarction, and strokes, in women using etonogestrel implants. NEXPLANON should be removed in the event of a thrombosis.
Due to the risk of thromboembolism associated with pregnancy and immediately following delivery, NEXPLANON should not be used prior to 21 days postpartum. Women with a history of thromboembolic disorders should be made aware of the possibility of a recurrence.
Evaluate for retinal vein thrombosis immediately if there is unexplained loss of vision, proptosis, diplopia, papilledema, or retinal vascular lesions.
Consider removal of the NEXPLANON implant in case of long-term immobilization due to surgery or illness.
5.3 Changes in Menstrual Bleeding Patterns
After starting NEXPLANON, women are likely to have a change from their normal menstrual bleeding pattern. These may include changes in bleeding frequency (absent, less, more frequent or continuous), intensity (reduced or increased) or duration. In clinical trials of the non-radiopaque etonogestrel implant (IMPLANON), bleeding patterns ranged from amenorrhea (1 in 5 women) to frequent and/or prolonged bleeding (1 in 5 women). The bleeding pattern experienced during the first three months of NEXPLANON use is broadly predictive of the future bleeding pattern for many women. Women should be counseled regarding the bleeding pattern changes they may experience so that they know what to expect. Abnormal bleeding should be evaluated as needed to exclude pathologic conditions or pregnancy.
In clinical studies of the non-radiopaque etonogestrel implant, reports of changes in bleeding pattern were the most common reason for stopping treatment (11.1%). Irregular bleeding (10.8%) was the single most common reason women stopped treatment, while amenorrhea (0.3%) was cited less frequently. In these studies, women had an average of 17.7 days of bleeding or spotting every 90 days (based on 3,315 intervals of 90 days recorded by 780 patients). The percentages of patients having 0, 1-7, 8-21, or >21 days of spotting or bleeding over a 90-day interval while using the non-radiopaque etonogestrel implant are shown in Table 1.
| Total Days of Bleeding or Spotting | Percentage of Patients | ||
|---|---|---|---|
| Treatment Days 91-180 (N = 745) |
Treatment Days 271-360 (N = 657) |
Treatment Days 631-720 (N = 547) |
|
| 0 Days | 19% | 24% | 17% |
| 1-7 Days | 15% | 13% | 12% |
| 8-21 Days | 30% | 30% | 37% |
| >21 Days | 35% | 33% | 35% |
Bleeding patterns observed with use of the non-radiopaque etonogestrel implant for up to 2 years, and the proportion of 90-day intervals with these bleeding patterns, are summarized in Table 2.
| BLEEDING PATTERNS | DEFINITIONS | % % = Percentage of 90-day intervals with this pattern
|
|---|---|---|
| Infrequent | Less than three bleeding and/or spotting episodes in 90 days (excluding amenorrhea) | 33.6 |
| Amenorrhea | No bleeding and/or spotting in 90 days | 22.2 |
| Prolonged | Any bleeding and/or spotting episode lasting more than 14 days in 90 days | 17.7 |
| Frequent | More than 5 bleeding and/or spotting episodes in 90 days | 6.7 |
In case of undiagnosed, persistent, or recurrent abnormal vaginal bleeding, appropriate measures should be conducted to rule out malignancy.
2.1 Initiating Contraception With Nexplanon (2.1 Initiating Contraception with NEXPLANON)
IMPORTANT: Rule out pregnancy before inserting the implant.
Timing of insertion depends on the woman's recent contraceptive history, as follows:
5.12 Carbohydrate and Lipid Metabolic Effects
Use of NEXPLANON may induce mild insulin resistance and small changes in glucose concentrations of unknown clinical significance. Carefully monitor prediabetic and diabetic women using NEXPLANON.
Women who are being treated for hyperlipidemia should be followed closely if they elect to use NEXPLANON. Some progestins may elevate LDL levels and may render the control of hyperlipidemia more difficult.
14.3 Implant Insertion and Removal Characteristics
Out of 301 insertions of the NEXPLANON implant in a clinical trial, the mean insertion time (from the removal of the protection cap of the applicator until retraction of the needle from the arm) was 27.9 ± 29.3 seconds. After insertion, 300 out of 301 (99.7%) NEXPLANON implants were palpable that were placed by trained providers. The single, non-palpable implant was not inserted according to the instructions.
5.7 Carcinoma of the Breast and Reproductive Organs
Women who currently have or have had breast cancer should not use hormonal contraception because breast cancer may be hormonally sensitive [see Contraindications (4)]. Some studies suggest that the use of combination hormonal contraceptives might increase the incidence of breast cancer; however, other studies have not confirmed such findings.
Some studies suggest that the use of combination hormonal contraceptives is associated with an increase in the risk of cervical cancer or intraepithelial neoplasia. However, there is controversy about the extent to which these findings are due to differences in sexual behavior and other factors.
Women with a family history of breast cancer or who develop breast nodules should be carefully monitored.
7.2 Effects of Hormonal Contraceptives On Other Drugs (7.2 Effects of Hormonal Contraceptives on Other Drugs)
Hormonal contraceptives may affect the metabolism of other drugs. Consequently, plasma concentrations may either increase (for example, cyclosporine) or decrease (for example, lamotrigine). Consult the labeling of all concurrently-used drugs to obtain further information about interactions with hormonal contraceptives or the potential for enzyme alterations.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 24-month carcinogenicity study in male and female rats with subdermal implants releasing 10 and 20 mcg etonogestrel per day (equal to approximately 1.8-3.6 times the systemic steady state exposure in women using NEXPLANON), no drug-related carcinogenic potential was observed. Etonogestrel was not genotoxic in the in vitro Ames/Salmonella reverse mutation assay, the chromosomal aberration assay in Chinese hamster ovary cells or the in vivo mouse micronucleus test. Fertility in female rats returned after withdrawal from treatment.
Principal Display Panel 68 Mg Implant Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 68 mg Implant Blister Pack Carton)
NDC 78206-145-01
1 applicator containing 1 subdermal implant
Nexplanon®
(etonogestrel implant) 68 mg
Radiopaque
Subdermal Use Only
This product is intended to prevent pregnancy. It does not protect
against HIV infection (AIDS) and other sexually transmitted diseases.
Rx only
5.1 Risk of Complications Due to Improper Insertion and Removal
Complications of Insertion and Removal
NEXPLANON should be inserted subdermally so that it will be palpable after insertion, and this should be confirmed by palpation immediately after insertion. Failure to insert NEXPLANON properly may go unnoticed unless it is palpated immediately after insertion. Undetected failure to insert the implant may lead to an unintended pregnancy. Complications related to insertion and removal procedures may occur, e.g., pain, paresthesia, bleeding, hematoma, scarring, or infection.
If NEXPLANON is inserted deeply (intramuscular or intrafascial), neural or vascular injury may occur. To help reduce the risk of neural or vascular injury, NEXPLANON should be inserted subdermally just under the skin at the inner side of the non-dominant upper arm overlying the triceps muscle, about 8-10 cm (3-4 inches) from the medial epicondyle of the humerus, and 3-5 cm (1.25-2 inches) posterior to (below) the sulcus (groove) between the biceps and triceps muscles. This location is intended to avoid the large nerves and blood vessels lying within and surrounding the sulcus. Deep insertions of NEXPLANON have been associated with paresthesia (due to neural injury), migration of the implant (due to intramuscular or fascial insertion), and intravascular insertion. If infection develops at the insertion site, start suitable treatment. If the infection persists, the implant should be removed. Incomplete insertions or infections may lead to expulsion.
Reports of implant migration within the arm may have been related to deep insertion. Postmarketing reports of implants located within the vessels of the arm and the pulmonary artery also may have been related to deep insertions or intravascular insertions. Some cases of implants found within the pulmonary artery were associated with chest pain and/or respiratory disorders (such as dyspnea, cough, or hemoptysis); others were asymptomatic. In cases where the implant has migrated to the pulmonary artery, endovascular or surgical procedures may be needed for removal.
Implant removal may be difficult or impossible if the implant is not inserted correctly, is inserted too deeply, not palpable, encased in fibrous tissue, or has migrated. If at any time the implant cannot be palpated, it should be localized, and removal is recommended. When an implant is removed, it is important to remove it in its entirety [see Dosage and Administration (2.3)].
Exploratory surgery without knowledge of the exact location of the implant is strongly discouraged. Removal of deeply inserted implants should be conducted with caution in order to prevent injury to deeper neural or vascular structures in the arm and be performed by healthcare professionals familiar with the anatomy of the arm. If the implant is located in the chest, healthcare professionals familiar with the anatomy of the chest should be consulted. Failure to remove the implant may result in continued effects of etonogestrel, such as compromised fertility, ectopic pregnancy, or persistence or occurrence of a drug-related adverse event.
Broken or Bent Implants
Cases of breakage or bending of implants while inserted within a patient’s arm have been reported. Cases of migration of a broken implant fragment within the arm have also occurred. These cases may be related to external forces, e.g., manipulation of the implant or contact sports. The release rate of etonogestrel may be slightly increased in a broken or bent implant, based on in vitro data. As noted previously, when an implant is removed, it is important to remove it in its entirety [see Dosage and Administration (2.3)].
NEXPLANON is available only through a restricted program under a REMS [see Warnings and Precautions (5.2) ].
Warning: Risk of Complications Due to Improper Insertion and Removal (WARNING: RISK OF COMPLICATIONS DUE TO IMPROPER INSERTION and REMOVAL)
- Improper insertion of NEXPLANON increases the risk of complications [see Warnings and Precautions (5.1)].
- Proper training prior to first use of NEXPLANON can minimize the risk of improper NEXPLANON insertion [see Warnings and Precautions (5.1)].
- Because of the risk of complications due to improper insertion and removal NEXPLANON is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the NEXPLANON REMS [see Warnings and Precautions (5.2)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:34.607199 · Updated: 2026-03-14T22:44:06.575433