These Highlights Do Not Include All The Information Needed To Use Gleolan Safely And Effectively. See Full Prescribing Information For Gleolan.
4873911e-a5d7-47e3-9679-fa5c95999323
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Gleolan is indicated in patients with glioma [suspected World Health Organization (WHO) Grades III or IV on preoperative imaging] as an adjunct for the visualization of malignant tissue during surgery.
Indications and Usage
Gleolan is indicated in patients with glioma [suspected World Health Organization (WHO) Grades III or IV on preoperative imaging] as an adjunct for the visualization of malignant tissue during surgery.
Dosage and Administration
For oral use only ( 2.1 ) Recommended reconstituted oral dose of Gleolan is 20 mg/kg. ( 2.1 ) Administer Gleolan to patient orally 3 hours (range 2 to 4 hours) before anesthesia. ( 2.1 ) See Full Prescribing Information for reconstitution information. ( 2.2 ) Use appropriate visualization techniques with appropriate surgical microscopes and light source filters. ( 2.4 )
Warnings and Precautions
Phototoxic reactions: Do not administer phototoxic drugs (St. John's wort, griseofulvin, thiazide diuretics, sulfonylureas, phenothiazines, sulphonamides, quinolones and tetracyclines), and topical preparations containing ALA for 24 hours during the perioperative period. Reduce exposure to sunlight or room lights for 48 hours after oral administration of Gleolan. ( 5.1 , 7 ) Risk of misinterpretation: Non-fluorescing tissue in the surgical field does not rule out the presence of tumor. ( 5.2 , 14 )
Contraindications
Hypersensitivity to the aminolevulinic acid (ALA) or porphyrins. [see Warnings and Precautions (5.3) ] Acute or chronic types of porphyria, due to potential ineffectiveness of the drug in these patients.
Adverse Reactions
Adverse reactions occurring in >1% of patients in the week following surgery were pyrexia, hypotension, nausea, and vomiting. ( 6.1 ) Adverse reactions occurring in < 1% of patients in the first 6 weeks after surgery were: chills, photosensitivity reaction, solar dermatitis, hypotension, abnormal liver function test, and diarrhea. ( 6.1 ) Neurologic events related to the surgical procedure occurred in 29% of patients and included: aphasia, hemiparesis, hemianopia, headache, seizure, hemiplegia, monoparesis, hypoesthesia, and brain edema. ( 6.1 ) Elevated liver enzymes occurred in clinical studies. There were no cases of liver failure. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact NXDC toll-free at (844) 517-5252 and [email protected] or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
Store at 25 °C (77 °F); excursions permitted to 15-30 °C (59-86 °F).
How Supplied
Gleolan (NDC 59137-231-01) is supplied as 1,500 mg of lyophilized aminolevulinic acid hydrochloride powder (equivalent to 1,170 mg aminolevulinic acid), for oral solution in a 50-mL clear, colorless, glass vial with a rubber stopper and an aluminum crimp seal.
Medication Information
Warnings and Precautions
Phototoxic reactions: Do not administer phototoxic drugs (St. John's wort, griseofulvin, thiazide diuretics, sulfonylureas, phenothiazines, sulphonamides, quinolones and tetracyclines), and topical preparations containing ALA for 24 hours during the perioperative period. Reduce exposure to sunlight or room lights for 48 hours after oral administration of Gleolan. ( 5.1 , 7 ) Risk of misinterpretation: Non-fluorescing tissue in the surgical field does not rule out the presence of tumor. ( 5.2 , 14 )
Indications and Usage
Gleolan is indicated in patients with glioma [suspected World Health Organization (WHO) Grades III or IV on preoperative imaging] as an adjunct for the visualization of malignant tissue during surgery.
Dosage and Administration
For oral use only ( 2.1 ) Recommended reconstituted oral dose of Gleolan is 20 mg/kg. ( 2.1 ) Administer Gleolan to patient orally 3 hours (range 2 to 4 hours) before anesthesia. ( 2.1 ) See Full Prescribing Information for reconstitution information. ( 2.2 ) Use appropriate visualization techniques with appropriate surgical microscopes and light source filters. ( 2.4 )
Contraindications
Hypersensitivity to the aminolevulinic acid (ALA) or porphyrins. [see Warnings and Precautions (5.3) ] Acute or chronic types of porphyria, due to potential ineffectiveness of the drug in these patients.
Adverse Reactions
Adverse reactions occurring in >1% of patients in the week following surgery were pyrexia, hypotension, nausea, and vomiting. ( 6.1 ) Adverse reactions occurring in < 1% of patients in the first 6 weeks after surgery were: chills, photosensitivity reaction, solar dermatitis, hypotension, abnormal liver function test, and diarrhea. ( 6.1 ) Neurologic events related to the surgical procedure occurred in 29% of patients and included: aphasia, hemiparesis, hemianopia, headache, seizure, hemiplegia, monoparesis, hypoesthesia, and brain edema. ( 6.1 ) Elevated liver enzymes occurred in clinical studies. There were no cases of liver failure. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact NXDC toll-free at (844) 517-5252 and [email protected] or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
Store at 25 °C (77 °F); excursions permitted to 15-30 °C (59-86 °F).
How Supplied
Gleolan (NDC 59137-231-01) is supplied as 1,500 mg of lyophilized aminolevulinic acid hydrochloride powder (equivalent to 1,170 mg aminolevulinic acid), for oral solution in a 50-mL clear, colorless, glass vial with a rubber stopper and an aluminum crimp seal.
Description
Gleolan is indicated in patients with glioma [suspected World Health Organization (WHO) Grades III or IV on preoperative imaging] as an adjunct for the visualization of malignant tissue during surgery.
Section 42229-5
Neurologic Events
Nervous system disorders occurred in 29% of patients within the first week after surgery. Events occurring in > 1% of patients included aphasia (8%), hemiparesis (7.8%), hemianopsia (3.2%), headache (2.7%), seizure (1.9%), hemiplegia (1.9%), monoparesis (1.3%) and hypoesthesia (1.1%). Brain edema occurred in < 1 % of patients in the first 6 weeks after surgery. In a randomized clinical trial (Study 3), the numbers of serious neurologic adverse events in the post operative period were higher in patients randomized to ALA fluorescence arm compared to the control arm. An imbalance was notable for the adverse events aphasia, ataxia, convulsion and hemianopsia, and is likely related to the higher amount of brain resection performed in the ALA arm. At longer follow up periods, the numbers between the two arms appeared similar[seeClinical Trials (14)].
10 Overdosage
Overdosage has been associated with respiratory insufficiency and erythema. In the event of overdose, supportive measures should be provided as necessary, including protection from strong light sources.
16.1 How Supplied
Gleolan (NDC 59137-231-01) is supplied as 1,500 mg of lyophilized aminolevulinic acid hydrochloride powder (equivalent to 1,170 mg aminolevulinic acid), for oral solution in a 50-mL clear, colorless, glass vial with a rubber stopper and an aluminum crimp seal.
8.4 Pediatric Use
The safety and effectiveness of Gleolan in pediatric patients have not been established.
8.5 Geriatric Use
Of 527 subjects in clinical studies of Gleolan, 182 were 65 to < 75 years of age and 7 were ≥ 75 years of age. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. No dose adjustment is required in elderly patients.
14 Clinical Studies
The efficacy of 20 mg / kg ALA HCl was evaluated in 3 clinical studies (Study 1-3) involving patients, ages 18 to 75 years old, who had a preoperative MRI compatible with high-grade glioma (WHO Grade III or IV) and were undergoing surgical resection.
Study 1 was an open-label study of 33 patients with newly diagnosed high-grade glioma and Study 2 was an open-label study of 36 patients with recurrent high-grade glioma. In Studies 1 and 2, after initial debulking was carried out under white light, biopsies were obtained under fluorescent light from fluorescent and nonfluorescent sites. Presence of fluorescence (positive/negative) was compared to tumor status (true/false) using histopathology as the reference standard. True positives and false positives among fluorescent biopsies and true negatives and false negatives among nonfluorescent biopsies are provided in Table 1.
Study 3 was a randomized, multicenter study in 415 patients with a preoperative diagnosis of high-grade glioma by MRI. Patients were randomized in 1:1 ratio to ALA fluorescence arm or to white light control arm. Biopsies were obtained from tumor-core, tumor-margin and regions just distant to the tumor margins. In 349 patients high grade glioma was confirmed by a blinded central read and histopathology. The remaining patients were diagnosed with metastatic disease, abscess, low-grade glioma or other conditions.
In patients with confirmed high-grade glioma randomized to the ALA fluorescence arm, presence of fluorescence at a biopsy level was compared to tumor status using histopathology as the reference standard (Table 1). In 4 patients with low-grade glioma (WHO Grade I or II) who received ALA HCl, 9 out of 10 biopsies were false negative.
The extent of resection among patients with confirmed high-grade glioma in the ALA fluorescence arm was compared to that among patient in the control arm, with the "completeness" of resection being determined by a central blinded read of early post-surgical MRI. Percentage of patients who had "completeness" of resection was 64% in the ALA arm and 38% in the control arm, with the difference of 26% [95% CI: (16%, 36%)].
| Study 1 (N=297) N is Number of total (fluorescent and non-fluorescent) biopsies
|
Study 2 (N=370) |
Study 3 (N=479) |
|
|---|---|---|---|
| Number of Fluorescent Biopsies | 185 | 354 | 319 |
| True Positive | 178 | 342 | 312 |
| False Positive | 7 | 12 | 7 |
| Number of Nonfluorescent Biopsies | 112 | 16 | 160 |
| True Negative | 27 | 3 | 30 |
| False Negative | 85 | 13 | 130 |
4 Contraindications
- Hypersensitivity to the aminolevulinic acid (ALA) or porphyrins.[seeWarnings and Precautions (5.3)]
- Acute or chronic types of porphyria, due to potential ineffectiveness of the drug in these patients.
6 Adverse Reactions
- Adverse reactions occurring in >1% of patients in the week following surgery were pyrexia, hypotension, nausea, and vomiting. (6.1)
- Adverse reactions occurring in < 1% of patients in the first 6 weeks after surgery were: chills, photosensitivity reaction, solar dermatitis, hypotension, abnormal liver function test, and diarrhea. (6.1)
- Neurologic events related to the surgical procedure occurred in 29% of patients and included: aphasia, hemiparesis, hemianopia, headache, seizure, hemiplegia, monoparesis, hypoesthesia, and brain edema. (6.1)
- Elevated liver enzymes occurred in clinical studies. There were no cases of liver failure. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact NXDC toll-free at (844) 517-5252 and [email protected] or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
2.1 Recommended Dose
- For oral use only
- The recommended oral dose of reconstituted Gleolan is 20 mg / kg body weight. More than 1 vial may be required.
12.2 Pharmacodynamics
The effect of the timing of the Gleolan dosing on fluorescence intensity in brain tissue is unknown. The relationship between systemic ALA plasma concentrations at the time of visualization and fluorescence intensity in brain is also unknown. The dose of 20 mg / kg provided stronger ALA-induced fluorescence in glioma tissue by both visual and spectrophotometric assessment compared to lower doses tested.
12.3 Pharmacokinetics
In 12 healthy subjects, the mean half-life of ALA following the recommended dose of Gleolan solution was 0.9 ± 1.2 hours (mean ± std dev) with a range of 0.8 to 1.3 hours. Maximum concentrations of the PpIX metabolite (Tmaxfor PpIX) occurred with a median of 4 hours and a range of 1.2 to 7.8 hours. The elimination half-life of PpIX was 3.6 ± 1.8 hours (mean ± std dev) with a range of 1.2 to 7.8 hours.
1 Indications and Usage
Gleolan is indicated in patients with glioma [suspected World Health Organization (WHO) Grades III or IV on preoperative imaging] as an adjunct for the visualization of malignant tissue during surgery.
11.1 Chemical Properties
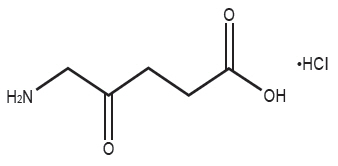
Gleolan (aminolevulinic acid hydrochloride) is an optical imaging agent for oral solution. The 50-mL, clear vial contains 1,500 mg of lyophilized aminolevulinic acid hydrochloride powder (equivalent to 1,170 mg aminolevulinic acid). After reconstitution, the product has a concentration of 30 mg aminolevulinic acid hydrochloride per mL (equivalent to 23.4 mg aminolevulinic acid per mL). The chemical name is 5-amino-4-oxo-pentanoic acid hydrochloride. The chemical formula for aminolevulinic acid hydrochloride is C5H10ClNO3. Its molecular weight is 167.59 g/mol with the following structural formula:
12.1 Mechanism of Action
ALA occurs endogenously as a metabolite that is formed in the mitochondria from succinyl-CoA and glycine. Exogenous administration of ALA leads to accumulation of the ALA metabolite PpIX in tumor cells. The reason for the accumulation of PpIX in neoplastic brain tissue is not known.
During glioma surgery, Gleolan is used with an operating microscope adapted with a blue emitting light source (power density 40-80 mW/cm2) and filters for excitation light of wavelength 375 to 440 nm, and observation at wavelengths of 620 to 710 nm. This allows tumor tissue to be visualized as red fluorescence. Tissue lacking sufficient PpIX concentrations appears blue.
2.4 Imaging Instructions
- Gleolan should be used with a standard surgical operating microscope adapted with a blue light emitting light source (power density 40-80 mW/cm2) and ancillary excitation and emission filters to visualize fluorescence excitation in the wavelength of 375 to 440 nm and for observation from 620 to 710 nm. Filters transmit porphyrin fluorescence as red-violet, as well as a fraction of backscattered blue excitation light necessary for distinguishing nonfluorescing tissue.
- Gleolan should only be used by neurosurgeons who have completed a training program on use of fluorescence in surgery. Training is provided by the distributor.
16.2 Storage and Handling
Store at 25 °C (77 °F); excursions permitted to 15-30 °C (59-86 °F).
2.3 Gleolan Administration
Gleolan is for ORAL USE ONLY. The reconstituted Gleolan solution is administered according to the following steps:
- Calculate the administration volume, in mL, to achieve the intended dose according to the following equation:
Administration Volume (mL) = Patient Body Weight (kg)* 20mg/kg 30mg/mL - Transfer the entire contents of the prepared vial(s) into an appropriate dosing container (e.g., oral medicine bottle); ensure the entire contents of the vials are transferred.
- After transfer, discard the empty vial(s).
- Using a disposable volumetric syringe, remove the administration volume of reconstituted Gleolan solution from the dosing container and transfer to a separate oral dosing container.
- Discard unneeded volume of Gleolan solution.
- Administer orally 3 hours (range 2 to 4 hours) prior to induction of anesthesia.
5 Warnings and Precautions
- Phototoxic reactions: Do not administer phototoxic drugs (St. John's wort, griseofulvin, thiazide diuretics, sulfonylureas, phenothiazines, sulphonamides, quinolones and tetracyclines), and topical preparations containing ALA for 24 hours during the perioperative period. Reduce exposure to sunlight or room lights for 48 hours after oral administration of Gleolan. (5.1,7)
- Risk of misinterpretation: Non-fluorescing tissue in the surgical field does not rule out the presence of tumor. (5.2,14)
2 Dosage and Administration
- For oral use only (2.1)
- Recommended reconstituted oral dose of Gleolan is 20 mg/kg. (2.1)
- Administer Gleolan to patient orally 3 hours (range 2 to 4 hours) before anesthesia. (2.1)
- See Full Prescribing Information for reconstitution information. (2.2)
- Use appropriate visualization techniques with appropriate surgical microscopes and light source filters. (2.4)
3 Dosage Forms and Strengths
For oral solution: 1,500 mg aminolevulinic acid hydrochloride (ALA HCl) lyophilized powder, equivalent to 1,170 mg aminolevulinic acid (ALA) , in a 50 mL single-dose clear, colorless, glass vial with rubber stopper. After reconstitution with 50 mL drinking water, the solution contains 30 mg per mL of aminolevulinic acid hydrochloride (equivalent to 23.4 mg per mL of aminolevulinic acid) and is clear and colorless to slightly yellowish in color.
2.2 Reconstitution of Gleolan
Gleolan powder must be reconstituted prior to administration by a healthcare provider according to the following instructions:
- Determine the total number of vials needed to achieve the intended dose for the patient according to the equation below (rounded up to the nearest whole vial):
# of vials= Patient Body Weight (kg) 75kg / vial - Completely remove the white cap and aluminum crimp seal from each vial.
- Remove and retain the rubber stopper from the vial.
- Using an appropriate volumetric measuring device (e.g., flask, graduated cylinder, dosing syringe), measure 50 mL of drinking water and add to each vial containing 1,500 mg of Gleolan.
- Gently swirl the vial to completely dissolve the powder.
- The resulting reconstituted solution (30 mg of Gleolan per mL) is clear and colorless to slightly yellowish.
- If required, replace the stopper and store reconstituted solution for up to 24 hours at room temperature prior to administration.
5.2 Risk of Misinterpretation
Errors may occur with the use of Gleolan for intraoperative visualization of malignant glioma, including false negatives and false positives. Non-fluorescing tissue in the surgical field does not rule out the presence of tumor in patients with glioma[seeClinical Studies (14)]. Fluorescence may be seen in areas of inflammation or metastases from other tumor types.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of Gleolan is supported by data from 5 open label clinical studies, which included 527 patients with glioma who received ALA HCl. Adverse reactions that occurred in > 1% of patients in the week following surgery were pyrexia, hypotension, nausea, and vomiting. Adverse reactions occurring in the first 6 weeks after surgery in < 1% of patients were: chills, photosensitivity reaction, solar dermatitis, hypotension, abnormal liver function test, and diarrhea. One patient experienced respiratory failure due to drug overdose[seeOverdosage (10)].
6.2 Post Marketing Experience
The following adverse reactions are among those that have been identified during post-approval use of Gleolan outside of the United States. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune Disorders: anaphylactic shock, angioedema, drug eruption, urticaria, erythema.
Metabolism and Nutrition Disorders: metabolic acidosis.
5.3 Hypersensitivity Reactions
Hypersensitivity reactions, including serious hypersensitivity reactions have occurred; these reactions include anaphylactic shock, swelling, and urticaria[seeContraindications (4),Adverse Reactions (6.2)]. Always have cardiopulmonary resuscitation personnel and equipment readily available and monitor all patients for hypersensitivity reactions.
5.1 Risk of Phototoxic Reaction
Due to the risk of phototoxic reactions, do not administer phototoxic drugs (St. John's wort, griseofulvin, thiazide diuretics, sulfonylureas, phenothiazines, sulphonamides, quinolones and tetracyclines), and topical preparations containing ALA for 24 hours during the perioperative period[seeDrug Interactions (7)]. Reduce exposure to sunlight or room lights for 48 hours after administration of Gleolan.
17 Patient Counseling Information
Advise patients that they may experience elevated liver enzymes (ALT and GGT) within the first week after surgery. This elevation may persist after 6 weeks.
Advise patients to reduce exposure to sunlight or room lights for 48 hours after administration of Gleolan due to risk of phototoxic reactions.
8.6 Patients With Renal Impairment
Because approximately one third of the ALA dose is excreted in urine as parent drug, ALA clearance may be reduced in patients with renal impairment; it is not known if dose adjustment is needed[seeClinical Pharmacology (12.3)].
8.7 Patients With Hepatic Impairment
The contribution of the liver to the elimination of ALA following Gleolan dosing is unknown. ALA clearance may be reduced in patients with hepatic impairment; it is not known if dose adjustment is needed[seeClinical Pharmacology (12.3)].
Principal Display Panel 1,500 Mg Vial Carton
NDC 59137-231-01
Rx only
Gleolan®
(aminolevulinic
acid hydrochloride)
for oral solution
1,500 mg
Reconstitute Prior To Use.
For Oral Use Only
SINGLE-DOSE VIAL.
Discard Unused Portion
One Vial
MEDEXUS
PHARMA
Structured Label Content
Section 42229-5 (42229-5)
Neurologic Events
Nervous system disorders occurred in 29% of patients within the first week after surgery. Events occurring in > 1% of patients included aphasia (8%), hemiparesis (7.8%), hemianopsia (3.2%), headache (2.7%), seizure (1.9%), hemiplegia (1.9%), monoparesis (1.3%) and hypoesthesia (1.1%). Brain edema occurred in < 1 % of patients in the first 6 weeks after surgery. In a randomized clinical trial (Study 3), the numbers of serious neurologic adverse events in the post operative period were higher in patients randomized to ALA fluorescence arm compared to the control arm. An imbalance was notable for the adverse events aphasia, ataxia, convulsion and hemianopsia, and is likely related to the higher amount of brain resection performed in the ALA arm. At longer follow up periods, the numbers between the two arms appeared similar[seeClinical Trials (14)].
10 Overdosage (10 OVERDOSAGE)
Overdosage has been associated with respiratory insufficiency and erythema. In the event of overdose, supportive measures should be provided as necessary, including protection from strong light sources.
16.1 How Supplied
Gleolan (NDC 59137-231-01) is supplied as 1,500 mg of lyophilized aminolevulinic acid hydrochloride powder (equivalent to 1,170 mg aminolevulinic acid), for oral solution in a 50-mL clear, colorless, glass vial with a rubber stopper and an aluminum crimp seal.
8.4 Pediatric Use
The safety and effectiveness of Gleolan in pediatric patients have not been established.
8.5 Geriatric Use
Of 527 subjects in clinical studies of Gleolan, 182 were 65 to < 75 years of age and 7 were ≥ 75 years of age. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. No dose adjustment is required in elderly patients.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of 20 mg / kg ALA HCl was evaluated in 3 clinical studies (Study 1-3) involving patients, ages 18 to 75 years old, who had a preoperative MRI compatible with high-grade glioma (WHO Grade III or IV) and were undergoing surgical resection.
Study 1 was an open-label study of 33 patients with newly diagnosed high-grade glioma and Study 2 was an open-label study of 36 patients with recurrent high-grade glioma. In Studies 1 and 2, after initial debulking was carried out under white light, biopsies were obtained under fluorescent light from fluorescent and nonfluorescent sites. Presence of fluorescence (positive/negative) was compared to tumor status (true/false) using histopathology as the reference standard. True positives and false positives among fluorescent biopsies and true negatives and false negatives among nonfluorescent biopsies are provided in Table 1.
Study 3 was a randomized, multicenter study in 415 patients with a preoperative diagnosis of high-grade glioma by MRI. Patients were randomized in 1:1 ratio to ALA fluorescence arm or to white light control arm. Biopsies were obtained from tumor-core, tumor-margin and regions just distant to the tumor margins. In 349 patients high grade glioma was confirmed by a blinded central read and histopathology. The remaining patients were diagnosed with metastatic disease, abscess, low-grade glioma or other conditions.
In patients with confirmed high-grade glioma randomized to the ALA fluorescence arm, presence of fluorescence at a biopsy level was compared to tumor status using histopathology as the reference standard (Table 1). In 4 patients with low-grade glioma (WHO Grade I or II) who received ALA HCl, 9 out of 10 biopsies were false negative.
The extent of resection among patients with confirmed high-grade glioma in the ALA fluorescence arm was compared to that among patient in the control arm, with the "completeness" of resection being determined by a central blinded read of early post-surgical MRI. Percentage of patients who had "completeness" of resection was 64% in the ALA arm and 38% in the control arm, with the difference of 26% [95% CI: (16%, 36%)].
| Study 1 (N=297) N is Number of total (fluorescent and non-fluorescent) biopsies
|
Study 2 (N=370) |
Study 3 (N=479) |
|
|---|---|---|---|
| Number of Fluorescent Biopsies | 185 | 354 | 319 |
| True Positive | 178 | 342 | 312 |
| False Positive | 7 | 12 | 7 |
| Number of Nonfluorescent Biopsies | 112 | 16 | 160 |
| True Negative | 27 | 3 | 30 |
| False Negative | 85 | 13 | 130 |
4 Contraindications (4 CONTRAINDICATIONS)
- Hypersensitivity to the aminolevulinic acid (ALA) or porphyrins.[seeWarnings and Precautions (5.3)]
- Acute or chronic types of porphyria, due to potential ineffectiveness of the drug in these patients.
6 Adverse Reactions (6 ADVERSE REACTIONS)
- Adverse reactions occurring in >1% of patients in the week following surgery were pyrexia, hypotension, nausea, and vomiting. (6.1)
- Adverse reactions occurring in < 1% of patients in the first 6 weeks after surgery were: chills, photosensitivity reaction, solar dermatitis, hypotension, abnormal liver function test, and diarrhea. (6.1)
- Neurologic events related to the surgical procedure occurred in 29% of patients and included: aphasia, hemiparesis, hemianopia, headache, seizure, hemiplegia, monoparesis, hypoesthesia, and brain edema. (6.1)
- Elevated liver enzymes occurred in clinical studies. There were no cases of liver failure. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact NXDC toll-free at (844) 517-5252 and [email protected] or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
2.1 Recommended Dose
- For oral use only
- The recommended oral dose of reconstituted Gleolan is 20 mg / kg body weight. More than 1 vial may be required.
12.2 Pharmacodynamics
The effect of the timing of the Gleolan dosing on fluorescence intensity in brain tissue is unknown. The relationship between systemic ALA plasma concentrations at the time of visualization and fluorescence intensity in brain is also unknown. The dose of 20 mg / kg provided stronger ALA-induced fluorescence in glioma tissue by both visual and spectrophotometric assessment compared to lower doses tested.
12.3 Pharmacokinetics
In 12 healthy subjects, the mean half-life of ALA following the recommended dose of Gleolan solution was 0.9 ± 1.2 hours (mean ± std dev) with a range of 0.8 to 1.3 hours. Maximum concentrations of the PpIX metabolite (Tmaxfor PpIX) occurred with a median of 4 hours and a range of 1.2 to 7.8 hours. The elimination half-life of PpIX was 3.6 ± 1.8 hours (mean ± std dev) with a range of 1.2 to 7.8 hours.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Gleolan is indicated in patients with glioma [suspected World Health Organization (WHO) Grades III or IV on preoperative imaging] as an adjunct for the visualization of malignant tissue during surgery.
11.1 Chemical Properties
Gleolan (aminolevulinic acid hydrochloride) is an optical imaging agent for oral solution. The 50-mL, clear vial contains 1,500 mg of lyophilized aminolevulinic acid hydrochloride powder (equivalent to 1,170 mg aminolevulinic acid). After reconstitution, the product has a concentration of 30 mg aminolevulinic acid hydrochloride per mL (equivalent to 23.4 mg aminolevulinic acid per mL). The chemical name is 5-amino-4-oxo-pentanoic acid hydrochloride. The chemical formula for aminolevulinic acid hydrochloride is C5H10ClNO3. Its molecular weight is 167.59 g/mol with the following structural formula:
12.1 Mechanism of Action
ALA occurs endogenously as a metabolite that is formed in the mitochondria from succinyl-CoA and glycine. Exogenous administration of ALA leads to accumulation of the ALA metabolite PpIX in tumor cells. The reason for the accumulation of PpIX in neoplastic brain tissue is not known.
During glioma surgery, Gleolan is used with an operating microscope adapted with a blue emitting light source (power density 40-80 mW/cm2) and filters for excitation light of wavelength 375 to 440 nm, and observation at wavelengths of 620 to 710 nm. This allows tumor tissue to be visualized as red fluorescence. Tissue lacking sufficient PpIX concentrations appears blue.
2.4 Imaging Instructions
- Gleolan should be used with a standard surgical operating microscope adapted with a blue light emitting light source (power density 40-80 mW/cm2) and ancillary excitation and emission filters to visualize fluorescence excitation in the wavelength of 375 to 440 nm and for observation from 620 to 710 nm. Filters transmit porphyrin fluorescence as red-violet, as well as a fraction of backscattered blue excitation light necessary for distinguishing nonfluorescing tissue.
- Gleolan should only be used by neurosurgeons who have completed a training program on use of fluorescence in surgery. Training is provided by the distributor.
16.2 Storage and Handling
Store at 25 °C (77 °F); excursions permitted to 15-30 °C (59-86 °F).
2.3 Gleolan Administration
Gleolan is for ORAL USE ONLY. The reconstituted Gleolan solution is administered according to the following steps:
- Calculate the administration volume, in mL, to achieve the intended dose according to the following equation:
Administration Volume (mL) = Patient Body Weight (kg)* 20mg/kg 30mg/mL - Transfer the entire contents of the prepared vial(s) into an appropriate dosing container (e.g., oral medicine bottle); ensure the entire contents of the vials are transferred.
- After transfer, discard the empty vial(s).
- Using a disposable volumetric syringe, remove the administration volume of reconstituted Gleolan solution from the dosing container and transfer to a separate oral dosing container.
- Discard unneeded volume of Gleolan solution.
- Administer orally 3 hours (range 2 to 4 hours) prior to induction of anesthesia.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Phototoxic reactions: Do not administer phototoxic drugs (St. John's wort, griseofulvin, thiazide diuretics, sulfonylureas, phenothiazines, sulphonamides, quinolones and tetracyclines), and topical preparations containing ALA for 24 hours during the perioperative period. Reduce exposure to sunlight or room lights for 48 hours after oral administration of Gleolan. (5.1,7)
- Risk of misinterpretation: Non-fluorescing tissue in the surgical field does not rule out the presence of tumor. (5.2,14)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For oral use only (2.1)
- Recommended reconstituted oral dose of Gleolan is 20 mg/kg. (2.1)
- Administer Gleolan to patient orally 3 hours (range 2 to 4 hours) before anesthesia. (2.1)
- See Full Prescribing Information for reconstitution information. (2.2)
- Use appropriate visualization techniques with appropriate surgical microscopes and light source filters. (2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For oral solution: 1,500 mg aminolevulinic acid hydrochloride (ALA HCl) lyophilized powder, equivalent to 1,170 mg aminolevulinic acid (ALA) , in a 50 mL single-dose clear, colorless, glass vial with rubber stopper. After reconstitution with 50 mL drinking water, the solution contains 30 mg per mL of aminolevulinic acid hydrochloride (equivalent to 23.4 mg per mL of aminolevulinic acid) and is clear and colorless to slightly yellowish in color.
2.2 Reconstitution of Gleolan
Gleolan powder must be reconstituted prior to administration by a healthcare provider according to the following instructions:
- Determine the total number of vials needed to achieve the intended dose for the patient according to the equation below (rounded up to the nearest whole vial):
# of vials= Patient Body Weight (kg) 75kg / vial - Completely remove the white cap and aluminum crimp seal from each vial.
- Remove and retain the rubber stopper from the vial.
- Using an appropriate volumetric measuring device (e.g., flask, graduated cylinder, dosing syringe), measure 50 mL of drinking water and add to each vial containing 1,500 mg of Gleolan.
- Gently swirl the vial to completely dissolve the powder.
- The resulting reconstituted solution (30 mg of Gleolan per mL) is clear and colorless to slightly yellowish.
- If required, replace the stopper and store reconstituted solution for up to 24 hours at room temperature prior to administration.
5.2 Risk of Misinterpretation
Errors may occur with the use of Gleolan for intraoperative visualization of malignant glioma, including false negatives and false positives. Non-fluorescing tissue in the surgical field does not rule out the presence of tumor in patients with glioma[seeClinical Studies (14)]. Fluorescence may be seen in areas of inflammation or metastases from other tumor types.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of Gleolan is supported by data from 5 open label clinical studies, which included 527 patients with glioma who received ALA HCl. Adverse reactions that occurred in > 1% of patients in the week following surgery were pyrexia, hypotension, nausea, and vomiting. Adverse reactions occurring in the first 6 weeks after surgery in < 1% of patients were: chills, photosensitivity reaction, solar dermatitis, hypotension, abnormal liver function test, and diarrhea. One patient experienced respiratory failure due to drug overdose[seeOverdosage (10)].
6.2 Post Marketing Experience
The following adverse reactions are among those that have been identified during post-approval use of Gleolan outside of the United States. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune Disorders: anaphylactic shock, angioedema, drug eruption, urticaria, erythema.
Metabolism and Nutrition Disorders: metabolic acidosis.
5.3 Hypersensitivity Reactions
Hypersensitivity reactions, including serious hypersensitivity reactions have occurred; these reactions include anaphylactic shock, swelling, and urticaria[seeContraindications (4),Adverse Reactions (6.2)]. Always have cardiopulmonary resuscitation personnel and equipment readily available and monitor all patients for hypersensitivity reactions.
5.1 Risk of Phototoxic Reaction
Due to the risk of phototoxic reactions, do not administer phototoxic drugs (St. John's wort, griseofulvin, thiazide diuretics, sulfonylureas, phenothiazines, sulphonamides, quinolones and tetracyclines), and topical preparations containing ALA for 24 hours during the perioperative period[seeDrug Interactions (7)]. Reduce exposure to sunlight or room lights for 48 hours after administration of Gleolan.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients that they may experience elevated liver enzymes (ALT and GGT) within the first week after surgery. This elevation may persist after 6 weeks.
Advise patients to reduce exposure to sunlight or room lights for 48 hours after administration of Gleolan due to risk of phototoxic reactions.
8.6 Patients With Renal Impairment (8.6 Patients with Renal Impairment)
Because approximately one third of the ALA dose is excreted in urine as parent drug, ALA clearance may be reduced in patients with renal impairment; it is not known if dose adjustment is needed[seeClinical Pharmacology (12.3)].
8.7 Patients With Hepatic Impairment (8.7 Patients with Hepatic Impairment)
The contribution of the liver to the elimination of ALA following Gleolan dosing is unknown. ALA clearance may be reduced in patients with hepatic impairment; it is not known if dose adjustment is needed[seeClinical Pharmacology (12.3)].
Principal Display Panel 1,500 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 1,500 mg Vial Carton)
NDC 59137-231-01
Rx only
Gleolan®
(aminolevulinic
acid hydrochloride)
for oral solution
1,500 mg
Reconstitute Prior To Use.
For Oral Use Only
SINGLE-DOSE VIAL.
Discard Unused Portion
One Vial
MEDEXUS
PHARMA
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:40:57.253240 · Updated: 2026-03-14T22:04:41.081691