These Highlights Do Not Include All The Information Needed To Use Brineura Safely And Effectively. See Full Prescribing Information For Brineura.
485ab3c5-0359-4878-872f-2fe1e7df4047
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Patients treated with enzyme replacement therapies have experienced life-threatening hypersensitivity reactions, including anaphylaxis. Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy. Initiate BRINEURA in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue BRINEURA and immediately initiate appropriate medical treatment, including use of epinephrine. Inform patients of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and to seek immediate medical care should symptoms occur [see Warnings and Precautions (5.1) ].
Indications and Usage
BRINEURA is indicated to slow the loss of ambulation in pediatric patients with neuronal ceroid lipofuscinosis type 2 (CLN2 disease), also known as tripeptidyl peptidase 1 (TPP1) deficiency.
Dosage and Administration
Administration of BRINEURA should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis. ( 2.1 ) BRINEURA should be administered by or under the supervision of a physician experienced in intraventricular administration. ( 2.1 ) Premedication of patients with antihistamines with or without antipyretics or corticosteroids is recommended. ( 2.1 ) Prior to each infusion, inspect the scalp for signs of intraventricular access device reservoir leakage, failure, or potential infection. ( 2.1 ) Recommended BRINEURA dosage is 300 mg administered once every other week as an intraventricular infusion. In patients less than 2 years of age, lower doses are recommended. ( 2.2 ) Dosing is not recommended in patients less than 37 weeks post-menstrual age or those weighing less than 2.5 kg. ( 2.2 ) See the full prescribing information for dosage modifications due to hypersensitivity reactions. ( 2.3 ) See the full prescribing information for preparation and administration instructions. ( 2.4 , 2.6 )
Warnings and Precautions
Meningitis and Other Intraventricular Access Device-Related Infections : Monitor the device insertion site for signs of infection. ( 4 , 5.2 ) Intraventricular Access Device-Related Complications : Consult a neurosurgeon for any complications with the implanted device. In case of device-related complication, discontinue the infusion and refer to the device labeling for further instructions. ( 4 , 5.3 ) Cardiovascular Adverse Reactions : Monitor vital signs before, during, and post-infusion. Monitor Electrocardiogram (ECG) in patients with a history of bradycardia, conduction disorder, or with structural heart disease, during the infusion. In patients without cardiac abnormalities, perform regular 12-lead ECG evaluations every 6 months. ( 2.5 , 5.4 ) Infusion-Associated Reactions (IARs) : If an IAR occurs, decreasing the infusion rate, temporarily stopping the infusion, and/or administering antihistamines and/or antipyretics may ameliorate the symptoms. ( 5.5 )
Contraindications
BRINEURA is contraindicated in patients with: any sign or symptom of acute, unresolved localized infection on or around the device insertion site (e.g., cellulitis or abscess); or suspected or confirmed CNS infection (e.g., cloudy CSF or positive CSF gram stain, or meningitis) [see Warnings and Precautions (5.2) ] . any acute intraventricular access device-related complication (e.g., leakage, extravasation of fluid, or device failure) [see Warnings and Precautions (5.3) ]. ventriculoperitoneal shunts.
Adverse Reactions
The following adverse reactions are described below and elsewhere in the labeling: Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.1) ] Meningitis and Other Intraventricular Access Device-Related Infections [see Warnings and Precautions (5.2) ] Intraventricular Access Device-Related Complications [see Warnings and Precautions (5.3) ] Cardiovascular Adverse Reactions [see Warnings and Precautions (5.4) ] Infusion-Associated Reactions [see Warnings and Precautions (5.5) ]
Storage and Handling
BRINEURA is supplied as a sterile, clear to slightly opalescent and colorless to pale yellow solution for intraventricular infusion and Intraventricular Electrolytes Injection is supplied as a clear to colorless solution for intraventricular infusion; both are included in package 1 of 2. The Administration Kit for use with BRINEURA is supplied separately as package 2 of 2 [see Dosage and Administration (2.4) ] .
How Supplied
BRINEURA is supplied as a sterile, clear to slightly opalescent and colorless to pale yellow solution for intraventricular infusion and Intraventricular Electrolytes Injection is supplied as a clear to colorless solution for intraventricular infusion; both are included in package 1 of 2. The Administration Kit for use with BRINEURA is supplied separately as package 2 of 2 [see Dosage and Administration (2.4) ] .
Medication Information
Warnings and Precautions
Meningitis and Other Intraventricular Access Device-Related Infections : Monitor the device insertion site for signs of infection. ( 4 , 5.2 ) Intraventricular Access Device-Related Complications : Consult a neurosurgeon for any complications with the implanted device. In case of device-related complication, discontinue the infusion and refer to the device labeling for further instructions. ( 4 , 5.3 ) Cardiovascular Adverse Reactions : Monitor vital signs before, during, and post-infusion. Monitor Electrocardiogram (ECG) in patients with a history of bradycardia, conduction disorder, or with structural heart disease, during the infusion. In patients without cardiac abnormalities, perform regular 12-lead ECG evaluations every 6 months. ( 2.5 , 5.4 ) Infusion-Associated Reactions (IARs) : If an IAR occurs, decreasing the infusion rate, temporarily stopping the infusion, and/or administering antihistamines and/or antipyretics may ameliorate the symptoms. ( 5.5 )
Indications and Usage
BRINEURA is indicated to slow the loss of ambulation in pediatric patients with neuronal ceroid lipofuscinosis type 2 (CLN2 disease), also known as tripeptidyl peptidase 1 (TPP1) deficiency.
Dosage and Administration
Administration of BRINEURA should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis. ( 2.1 ) BRINEURA should be administered by or under the supervision of a physician experienced in intraventricular administration. ( 2.1 ) Premedication of patients with antihistamines with or without antipyretics or corticosteroids is recommended. ( 2.1 ) Prior to each infusion, inspect the scalp for signs of intraventricular access device reservoir leakage, failure, or potential infection. ( 2.1 ) Recommended BRINEURA dosage is 300 mg administered once every other week as an intraventricular infusion. In patients less than 2 years of age, lower doses are recommended. ( 2.2 ) Dosing is not recommended in patients less than 37 weeks post-menstrual age or those weighing less than 2.5 kg. ( 2.2 ) See the full prescribing information for dosage modifications due to hypersensitivity reactions. ( 2.3 ) See the full prescribing information for preparation and administration instructions. ( 2.4 , 2.6 )
Contraindications
BRINEURA is contraindicated in patients with: any sign or symptom of acute, unresolved localized infection on or around the device insertion site (e.g., cellulitis or abscess); or suspected or confirmed CNS infection (e.g., cloudy CSF or positive CSF gram stain, or meningitis) [see Warnings and Precautions (5.2) ] . any acute intraventricular access device-related complication (e.g., leakage, extravasation of fluid, or device failure) [see Warnings and Precautions (5.3) ]. ventriculoperitoneal shunts.
Adverse Reactions
The following adverse reactions are described below and elsewhere in the labeling: Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.1) ] Meningitis and Other Intraventricular Access Device-Related Infections [see Warnings and Precautions (5.2) ] Intraventricular Access Device-Related Complications [see Warnings and Precautions (5.3) ] Cardiovascular Adverse Reactions [see Warnings and Precautions (5.4) ] Infusion-Associated Reactions [see Warnings and Precautions (5.5) ]
Storage and Handling
BRINEURA is supplied as a sterile, clear to slightly opalescent and colorless to pale yellow solution for intraventricular infusion and Intraventricular Electrolytes Injection is supplied as a clear to colorless solution for intraventricular infusion; both are included in package 1 of 2. The Administration Kit for use with BRINEURA is supplied separately as package 2 of 2 [see Dosage and Administration (2.4) ] .
How Supplied
BRINEURA is supplied as a sterile, clear to slightly opalescent and colorless to pale yellow solution for intraventricular infusion and Intraventricular Electrolytes Injection is supplied as a clear to colorless solution for intraventricular infusion; both are included in package 1 of 2. The Administration Kit for use with BRINEURA is supplied separately as package 2 of 2 [see Dosage and Administration (2.4) ] .
Description
Patients treated with enzyme replacement therapies have experienced life-threatening hypersensitivity reactions, including anaphylaxis. Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy. Initiate BRINEURA in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue BRINEURA and immediately initiate appropriate medical treatment, including use of epinephrine. Inform patients of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and to seek immediate medical care should symptoms occur [see Warnings and Precautions (5.1) ].
Section 42229-5
Missed Dose
If one or more doses are missed, restart BRINEURA treatment as soon as possible, maintaining the 2-week interval between infusions thereafter.
Section 43683-2
Section 44425-7
Storage
BRINEURA (cerliponase alfa) Injection and Intraventricular Electrolytes Injection:
Store upright in a freezer (-25°C to -15°C) in original carton to protect from light.
Administration Kit for use with BRINEURA:
Store in original carton separately from BRINEURA. Do not freeze.
11 Description
Cerliponase alfa is a purified human enzyme produced by recombinant DNA technology in a Chinese hamster ovary cell line. The active substance is a recombinant human tripeptidyl peptidase-1 (rhTPP1), a lysosomal exopeptidase. The primary activity of the mature enzyme is the cleavage of N-terminal tripeptides from a broad range of protein substrates.
Cerliponase alfa contains 544 amino acids with an average molecular mass of 59 kDa. The mature enzyme is 368 amino acids in length. There are 5 consensus N-glycosylation sites on rhTPP1 that contain high mannose, phosphorylated high mannose and complex glycosylation structures.
BRINEURA (cerliponase alfa) Injection and Intraventricular Electrolytes Injection are administered by intraventricular infusion. The solutions are sterile, nonpyrogenic, and free of foreign particulates. BRINEURA is a clear to slightly opalescent and colorless to pale yellow solution. Intraventricular Electrolytes is a clear to colorless solution.
BRINEURA and Intraventricular Electrolytes Injection are packaged in 10 mL clear Type 1 single-dose glass vials [see How Supplied/Storage and Handling (16)]. Each vial of BRINEURA provides 5 mL of solution containing 150 mg cerliponase alfa. Each vial of Intraventricular Electrolytes Injection provides 5 mL of solution. Both BRINEURA and Intraventricular Electrolytes Injection are formulated with the following excipients: calcium chloride dihydrate (1.05 mg); magnesium chloride hexahydrate (0.8 mg); potassium chloride (1.1 mg); sodium chloride (43.85 mg); sodium phosphate, dibasic, heptahydrate (0.55 mg); sodium phosphate, monobasic, monohydrate (0.4 mg); and Water for Injection, USP. The pH of the solution is between 6.2 to 6.8 for BRINEURA, and between 6.0 to 7.0 for Intraventricular Electrolytes Injection.
Each vial contains: sodium: 0.76 mEq, and potassium: 0.015 mEq.
8.4 Pediatric Use
The safety and effectiveness of BRINEURA have been established to slow the loss of ambulation in pediatric patients with CLN2 disease and the information on this use is discussed throughout the labeling.
BRINEURA is not recommended for use in patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age) or those weighing less than 2.5 kg due to physiologic immaturity which may increase risk of serious and clinically significant adverse reactions observed with BRINEURA [see Warnings and Precautions (5)].
Patients less than 3 years of age may be at increased risk for developing hypersensitivity reactions with BRINEURA use compared to patients 3 years of age and older [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies including cerliponase alfa.
In Trials 1 and 2 [see Clinical Studies (14)], 19 of 24 (79%) and 10 of 24 (42%) patients treated with BRINEURA developed anti-drug antibodies (ADAs) in serum and CSF, respectively. Drug-specific neutralizing antibodies (NAb) capable of inhibiting receptor-mediated cellular uptake of cerliponase alfa were detected in the CSF of 3 of 24 (13%) patients at a single visit and were undetectable in all other CSF samples tested in ADA positive patients. In Trial 3 [see Clinical Studies (14)], 14 of 14 (100%) and 3 of 14 (21%, all of the 3 patients were < 3 years of age) patients treated with BRINEURA developed ADAs in serum and CSF, respectively. NAb responses were not detected in the CSF of any ADA positive patients. Overall, patients younger than 3 years of age had higher ADA titers compared to patients 3 years of age and older.
In Trials 1 and 2, patients who experienced moderate to severe hypersensitivity adverse reactions were tested for drug-specific IgE and found to be negative. No association was found between serum ADA titers and incidence or severity of hypersensitivity. In Trial 3, hypersensitivity occurred in higher percentage in BRINEURA-treated patients < 3 years of age at baseline (amongst these patients, one patient experienced anaphylaxis), and higher ADA titers were also observed in this age group compared to patients ≥ 3 years of age at baseline. There was no identified clinically significant effect of ADA on pharmacokinetics or efficacy of BRINEURA. There is insufficient information to characterize the effects of ADA on safety.
14 Clinical Studies
The efficacy of BRINEURA was assessed over 96 weeks in a non-randomized single-arm dose escalation clinical study with extension in symptomatic pediatric patients with late infantile neuronal ceroid lipofuscinosis type 2 (CLN2) disease, confirmed by TPP1 deficiency. BRINEURA-treated patients were compared to untreated patients from a natural history cohort. The Motor domain of a CLN2 Clinical Rating Scale was used to assess disease progression. Scores ranged from 3 (grossly normal) to 0 (profoundly impaired) with unit decrements representing milestone events in the loss of motor function (ability to walk or crawl). Due to the inability to establish comparability for the CLN2 Language domain ratings between the clinical study with extension and the natural history cohort, efficacy of BRINEURA for the Language domain cannot be established.
Twenty-four patients, aged 3 to 8 years were enrolled in the BRINEURA single-arm clinical study (Trial 1, NCT01907087). Sixty-three percent of patients were female and 37% were male. Ninety-six percent of patients were White and 4% were Asian; for ethnicity, 4% identified as Hispanic/Latino, 96% as non-Hispanic/Latino. One patient withdrew after week 1 due to inability to continue with study procedures; 23 patients were treated with BRINEURA 300 mg every other week by intraventricular infusion for 48 weeks, and continued treatment during the 240-week extension period, Trial 2 (NCT02485899), for a total duration of 288 weeks, plus a 24-week safety follow-up.
In the clinical study with extension, patients were assessed for decline in the Motor domain of the CLN2 Clinical Rating Scale at 48, 72 and 96 weeks. Decline was defined as having an unreversed (sustained) 2-category decline or an unreversed score of 0 in the Motor domain of the CLN2 Clinical Rating Scale. Patients' responses to BRINEURA treatment were evaluated if at screening a combined Motor plus Language CLN2 score of less than 6 was recorded. Two patients with a combined Motor plus Language CLN2 score of 6 were excluded from the analyses; they maintained that score throughout the study period. The patient who terminated early was analyzed as having a decline at the time of termination. Data used in the analyses from the natural history cohort began at 36 months of age or greater and at the first time a Motor plus Language CLN2 score less than 6 was recorded.
Motor scores of the 22 BRINEURA-treated patients in the clinical study with extension were compared to scores of the independent natural history cohort that included 42 untreated patients who satisfied inclusion criteria for the clinical study. The results of logistic modeling with covariates (screening age, screening motor score, genotype: 0 key mutations (yes/no)), demonstrated the odds of BRINEURA-treated patients not having a decline by 96 weeks were 13 times the odds of natural history cohort patients not having a decline (Odds Ratio (95% CI): 13.1 (1.2, 146.9)).
4 Contraindications
BRINEURA is contraindicated in patients with:
- any sign or symptom of acute, unresolved localized infection on or around the device insertion site (e.g., cellulitis or abscess); or suspected or confirmed CNS infection (e.g., cloudy CSF or positive CSF gram stain, or meningitis) [see Warnings and Precautions (5.2)].
- any acute intraventricular access device-related complication (e.g., leakage, extravasation of fluid, or device failure) [see Warnings and Precautions (5.3)].
- ventriculoperitoneal shunts.
6 Adverse Reactions
The following adverse reactions are described below and elsewhere in the labeling:
- Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.1)]
- Meningitis and Other Intraventricular Access Device-Related Infections [see Warnings and Precautions (5.2)]
- Intraventricular Access Device-Related Complications [see Warnings and Precautions (5.3)]
- Cardiovascular Adverse Reactions [see Warnings and Precautions (5.4)]
- Infusion-Associated Reactions [see Warnings and Precautions (5.5)]
12.2 Pharmacodynamics
No formal pharmacodynamic studies have been conducted with cerliponase alfa.
1 Indications and Usage
BRINEURA is indicated to slow the loss of ambulation in pediatric patients with neuronal ceroid lipofuscinosis type 2 (CLN2 disease), also known as tripeptidyl peptidase 1 (TPP1) deficiency.
12.1 Mechanism of Action
CLN2 disease is a neurodegenerative disease caused by deficiency of the lysosomal enzyme tripeptidyl peptidase-1 (TPP1), which catabolizes polypeptides in the CNS. TPP1 has no known substrate specificity. Deficiency in TPP1 activity results in the accumulation of lysosomal storage materials normally metabolized by this enzyme in the central nervous system (CNS), leading to progressive decline in motor function.
Cerliponase alfa (rhTTP1), a proenzyme, is taken up by target cells in the CNS and is translocated to the lysosomes through the Cation Independent Mannose-6-Phosphate Receptor (CI-MPR, also known as M6P/IGF2 receptor). Cerliponase alfa is activated in the lysosome and the activated proteolytic form of rhTPP1 cleaves tripeptides from the N-terminus of proteins.
5 Warnings and Precautions
- Meningitis and Other Intraventricular Access Device-Related Infections: Monitor the device insertion site for signs of infection. (4, 5.2)
- Intraventricular Access Device-Related Complications: Consult a neurosurgeon for any complications with the implanted device. In case of device-related complication, discontinue the infusion and refer to the device labeling for further instructions. (4, 5.3)
- Cardiovascular Adverse Reactions: Monitor vital signs before, during, and post-infusion. Monitor Electrocardiogram (ECG) in patients with a history of bradycardia, conduction disorder, or with structural heart disease, during the infusion. In patients without cardiac abnormalities, perform regular 12-lead ECG evaluations every 6 months. (2.5, 5.4)
- Infusion-Associated Reactions (IARs): If an IAR occurs, decreasing the infusion rate, temporarily stopping the infusion, and/or administering antihistamines and/or antipyretics may ameliorate the symptoms. (5.5)
2 Dosage and Administration
- Administration of BRINEURA should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis. (2.1)
- BRINEURA should be administered by or under the supervision of a physician experienced in intraventricular administration. (2.1)
- Premedication of patients with antihistamines with or without antipyretics or corticosteroids is recommended. (2.1)
- Prior to each infusion, inspect the scalp for signs of intraventricular access device reservoir leakage, failure, or potential infection. (2.1)
- Recommended BRINEURA dosage is 300 mg administered once every other week as an intraventricular infusion. In patients less than 2 years of age, lower doses are recommended. (2.2)
- Dosing is not recommended in patients less than 37 weeks post-menstrual age or those weighing less than 2.5 kg. (2.2)
- See the full prescribing information for dosage modifications due to hypersensitivity reactions. (2.3)
- See the full prescribing information for preparation and administration instructions. (2.4, 2.6)
3 Dosage Forms and Strengths
Injection: BRINEURA 150 mg/5 mL (30 mg/mL) solution, two single-dose vials per carton co-packaged with Intraventricular Electrolytes Injection 5 mL in a single-dose vial. BRINEURA is a clear to slightly opalescent and colorless to pale yellow solution. Intraventricular Electrolytes is a clear to colorless solution [see How Supplied/Storage and Handling (16)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of BRINEURA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Immune system disorders: Anaphylactic reaction characterized by acute pyrexia, respiratory distress (bronchospasm, hypoxemia, perioral cyanosis), tachycardia, hypotension, diarrhea, and rash [see Warnings and Precautions (5.1)].
- Infections and infestations: Bacterial meningitis [see Warnings and Precautions (5.2)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
- Hypersensitivity Reactions Including Anaphylaxis and Infusion-Associated Reactions (IARs)
- Advise patients and caregivers that life-threatening hypersensitivity reactions, including anaphylaxis, and IARs may occur with BRINEURA treatment.
- Advise patients and caregivers that anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy.
- Inform patients and caregivers of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis, and IARs and to seek immediate medical care should symptoms occur [see Warnings and Precautions (5.1, 5.5)].
- Intraventricular Access Device-Related Infections
Advise patients and caregivers of the risk of device-related infections, including meningitis. Familiarize them with the signs and symptoms of these infections and instruct them to immediately contact their healthcare provider if an infection is suspected [see Warnings and Precautions (5.2)]. - Cardiovascular Adverse Reactions
Advise patients and caregivers that hypotension and/or bradycardia may occur during and following the infusion of BRINEURA. Instruct patients immediately to contact their healthcare provider if these reactions occur [see Warnings and Precautions (5.4)].
5.5 Infusion Associated Reactions
Infusion-associated reactions (IARs) such as vomiting, seizure, rash, pyrexia, hypersensitivity, and anaphylactic reaction have been observed in patients treated with BRINEURA [see Adverse Reactions (6.1)].
Premedication of patients with antihistamines with or without antipyretics or corticosteroids is recommended 30 to 60 minutes prior to the start of infusion. If an IAR occurs, decreasing the infusion rate, temporarily stopping the infusion, and/or administering antihistamines and/or antipyretics may ameliorate the symptoms. Closely monitor patients who have experienced IARs when re-administering BRINEURA.
In Trial 3, infusion-associated reactions were reported in 8 of 8 patients less than 3 years of age at baseline as compared to 4 of 6 patients ≥ 3 years of age at baseline.
16 How Supplied/storage and Handling
BRINEURA is supplied as a sterile, clear to slightly opalescent and colorless to pale yellow solution for intraventricular infusion and Intraventricular Electrolytes Injection is supplied as a clear to colorless solution for intraventricular infusion; both are included in package 1 of 2. The Administration Kit for use with BRINEURA is supplied separately as package 2 of 2 [see Dosage and Administration (2.4)].
5.4 Cardiovascular Adverse Reactions
Monitor vital signs (blood pressure, heart rate) before infusion starts, periodically during infusion, and post-infusion in a healthcare setting [see Dosage and Administration (2.6)]. Upon completion of the infusion, clinically assess the patient status. Continued observation may be necessary if clinically indicated.
Perform electrocardiogram (ECG) monitoring during infusion in patients with a history of bradycardia, conduction disorder, or with structural heart disease, as some patients with CLN2 disease may develop conduction disorders or heart disease. In patients without cardiac abnormalities, regular 12-lead ECG evaluations should be performed every 6 months.
In clinical Trial 1 and Trial 2 to 96 weeks, hypotension was reported in 2 of 24 (8%) patients, which occurred during or up to eight hours after BRINEURA infusion. Patients did not require alteration in treatment, and reactions resolved spontaneously or after intravenous fluid administration [see Adverse Reactions (6.1)].
2.2 Recommended Dosage and Administration
The recommended dosage of BRINEURA in pediatric patients is provided in Table 1. The dose is administered once every other week by intraventricular infusion.
BRINEURA is not recommended in patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age) or those weighing less than 2.5 kg [see Use in Specific Populations (8.4)].
Administer BRINEURA first followed by infusion of the Intraventricular Electrolytes. The complete BRINEURA infusion, including the required infusion of Intraventricular Electrolytes, is approximately 2 to 4.5 hours, depending on the dose and volume administered. See Table 1 for the appropriate volume and infusion rate. For volumes that are not whole numbers, see Dosage and Administration (2.6) .
| Age groups | BRINEURA dose administered every other week | Volume of BRINEURA solution | Infusion rate |
|---|---|---|---|
| Birth to < 6 months | 100 mg | 3.3 mL | 1.25 mL/hr |
| 6 months to < 1 year | 150 mg | 5 mL | 2.5 mL/hr |
| 1 year to < 2 years | 200 mg (first 4 doses) | 6.7 mL (first 4 doses) | 2.5 mL/hr |
| 300 mg (subsequent doses) | 10 mL (subsequent doses) | ||
| 2 years and older | 300 mg | 10 mL | 2.5 mL/hr |
Principal Display Panel 5 Ml Vial Label
Intraventricular
Electrolytes
Injection
Use after Brineura®
(cerliponase alfa)
5 mL
For Intraventricular
Infusion Only
2.1 Recommendations Prior to Brineura Treatment
- Administration of BRINEURA should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis.
- Initiate BRINEURA in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment [see Warnings and Precautions (5.1)].
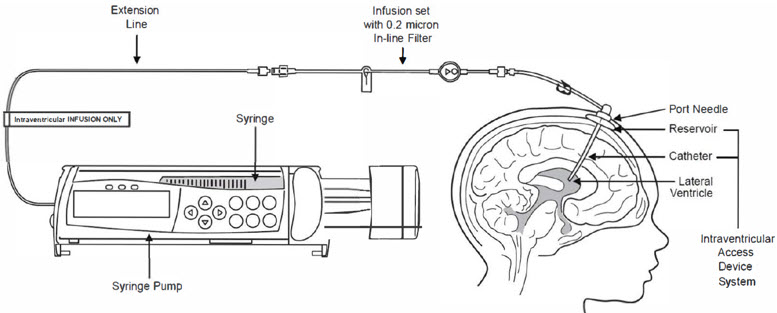
- BRINEURA should be administered by or under the supervision of a physician experienced in intraventricular administration via a surgically implanted intraventricular access device system which consists of the reservoir and catheter components [see Dosage and Administration (2.6)].
- Premedication of patients with antihistamines with or without antipyretics or corticosteroids is recommended 30 to 60 minutes prior to the start of infusion [see Warnings and Precautions (5.1, 5.5)].
- Aseptic technique must be strictly observed during preparation and administration [see Dosage and Administration (2.6)].
- Prior to each infusion of BRINEURA, inspect the scalp for signs of intraventricular access device reservoir leakage, failure or potential infection [see Warnings and Precautions (5.2, 5.3)].
- Prior to each infusion of BRINEURA and when clinically indicated, obtain a sample of CSF for cell count and culture [see Warnings and Precautions (5.2)].
- Replace the intraventricular access device reservoir prior to 4 years of single-puncture administrations [see Warnings and Precautions (5.3)].
Principal Display Panel 150 Mg/5 Ml Vial Label
Brineura®
(cerliponase alfa)
Injection
150 mg/5 mL
(30 mg/mL)
For Intraventricular Infusion Only
Use before Intraventricular
Electrolytes
Principal Display Panel Kit Carton Package 1
NDC 68135-811-02
Brineura®
(cerliponase alfa)
Injection
150 mg/5 mL (30 mg/mL)
For Intraventricular Infusion Only
Each vial of Brineura® contains 150 mg cerliponase alfa
in 5 mL of solution (30 mg/mL)
No preservatives
Single-Dose Vial. Discard unused portion.
Brineura® and Intraventricular Electrolytes must be
administered with the Administration Kit provided.
Each carton contains:
2 vials, each containing Brineura®
(cerliponase alfa) Injection, 150 mg/5 mL
1 vial of Intraventricular Electrolytes
Injection, 5 mL
Package 1 of 2
Administration Kit is supplied
and stored separately as
package 2 of 2.
Rx Only
BIOMARIN®
Principal Display Panel Kit Carton Package 2
Administration Kit for use with Brineura® (cerliponase alfa)
Intraventricular infusion only
Recommended Dosage: See Prescribing Information
Contents of Administration Kit:
- Two single-use 20-mL syringes
- Two single-use syringe needles, 21 G, 25.4 mm
- One single-use extension line
- One single-use infusion set with 0.2 micron inline filter
- One single-use port needle, 22 G, 16 mm
Package 2 of 2
Brineura® and Intraventricular Electrolytes Injection are separately supplied and
stored frozen as package 1 of 2
Not for resale
Only for use with Brineura®
Caution:
-
-Do not use if the seals or package are damaged.
-
-Do not resterilize.
-
-Single use only. Do not reuse.
-
-Do not freeze.
-
-Store in original package separately from Brineura®.
NOTE:
-
-This Administration Kit has no components made of natural latex rubber.
-
-Individual devices are provided in separate sterile packaging.
-
-Expiration dates may vary for each device included.
Manufactured for
BioMarin Pharmaceutical Inc.
Novato, CA 94949
www.bmrn.com
Rx Only
BIOMARIN®
5.1 Hypersensitivity Reactions Including Anaphylaxis
Life-threatening hypersensitivity reactions including anaphylaxis have been reported in patients treated with enzyme replacement therapies, including BRINEURA. BRINEURA-treated patients have had these reactions occur in clinical studies and postmarketing use [see Adverse Reactions (6)]. In clinical Trial 1 and Trial 2 to 96 weeks, a total of 11 of 24 (46%) patients experienced hypersensitivity reactions during the infusion or within 24 hours of completion of the infusion. Patients in clinical trials were routinely pre-medicated with antihistamines with or without antipyretics or corticosteroids, prior to infusion of BRINEURA. During postmarketing use, anaphylactic reactions occurred during or within several hours of BRINEURA infusion. Epinephrine was administered in these patients, and they received subsequent BRINEURA infusions without recurrence of anaphylaxis.
In Trial 3, hypersensitivity reactions were reported in 5 of 8 (63 %) patients less than 3 years of age at baseline as compared to 0 of 6 patients ≥ 3 years of age at baseline [see Adverse Reactions (6.1)]. Of the reported hypersensitivity reactions, a single anaphylactic reaction occurred in a subject < 3 years of age.
Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy. Administration of BRINEURA should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis. Initiate BRINERUA in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. Premedication of patients with antihistamines with or without antipyretics or corticosteroids is recommended 30 to 60 minutes prior to the start of infusion. Observe patients closely during and after the infusion. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue BRINEURA and immediately initiate appropriate medical treatment including use of epinephrine. Inform patients and caregivers of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and to seek immediate medical care should symptoms occur.
The management of hypersensitivity reactions should be based on the severity of the reaction and may include temporarily interrupting the infusion, and/or treatment with antihistamines, antipyretics, and/or corticosteroids. Consider the risks and benefits of readministration of BRINEURA following an anaphylactic reaction. If the decision is made to readminister BRINEURA after the occurrence of anaphylaxis, ensure appropriately trained personnel and equipment for emergency resuscitation (including epinephrine and other emergency medicines) are readily available during infusion. Initiate subsequent infusion at approximately one-half the initial infusion rate at which the anaphylactic reaction occurred.
5.3 Intraventricular Access Device Related Complications
During the clinical trials and in postmarketing reports, intraventricular access device-related complications were reported (e.g., device leakage, device failure extravasation of CSF fluid, or bulging of the scalp around or above the intraventricular access device) [see Adverse Reactions (6.1)]. For any complications with the implanted intraventricular access device, consult with a neurosurgeon to confirm the integrity or performance of the device. In case of intraventricular access device-related complications, discontinue the BRINEURA infusion and refer to the device manufacturer's labeling for further instructions [see Contraindications (4)].
Material degradation of the intraventricular access device reservoir was reported after approximately 4 years of administration, which may impact the effective and safe use of the device. During benchtop testing such material degradation was recognized after approximately 105 perforations of the intraventricular access device. The intraventricular access device should be replaced prior to 4 years of single-puncture administrations, which equates to approximately 105 administrations of BRINEURA.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity, genotoxicity, and fertility studies have not been performed with cerliponase alfa. Based on the mechanism of action, cerliponase alfa is not expected to be tumorigenic.
Warning: Hypersensitivity Reactions Including Anaphylaxis
Patients treated with enzyme replacement therapies have experienced life-threatening hypersensitivity reactions, including anaphylaxis. Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy.
Initiate BRINEURA in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue BRINEURA and immediately initiate appropriate medical treatment, including use of epinephrine. Inform patients of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and to seek immediate medical care should symptoms occur [see Warnings and Precautions (5.1)].
5.2 Meningitis and Other Intraventricular Access Device Related Infections
Bacterial meningitis requiring antibiotic treatment and removal of the device was reported during postmarketing use of BRINEURA. Additionally, in clinical trials and during postmarketing use there were reports of other device-related clinical infections which were confirmed by positive CSF cultures, treated with antibiotics and removal of the entire intraventricular access device, and in which patients resumed treatment with BRINEURA after the device was replaced [see Adverse Reactions (6.1)]. In all cases, antibiotics were administered, the intraventricular access devices were removed and replaced, and patients resumed treatment with BRINEURA.
The signs and symptoms of infections may not be readily apparent in patients with CLN2 disease. To reduce the risk of infectious complications, BRINEURA should be administered by, or under the supervision of, a physician experienced in intraventricular administration. Prior to each infusion of BRINEURA and when clinically indicated, obtain a sample of CSF for cell count and culture. Do not administer BRINEURA if there are localized signs of infection on or around the device insertion site, such as erythema, tenderness, or discharge or suspected or confirmed CNS infection (e.g., cloudy CSF or positive CSF gram stain, or meningitis) [see Contraindications (4)].
Healthcare providers should be vigilant for the development of signs and symptoms of infection, including meningitis, during treatment with BRINEURA and monitor the device insertion site for signs of infection.
2.4 Preparation Instructions for Intraventricular Administration of the Product
BRINEURA and the Intraventricular Electrolytes must only be administered by the intraventricular route, using the provided Administration Kit for use with BRINEURA. Each vial of BRINEURA and Intraventricular Electrolytes is intended for a single dose only.
BRINEURA is administered into the cerebrospinal fluid (CSF) by infusion via a surgically implanted reservoir and catheter (the "intraventricular access device system"). BRINEURA is intended to be administered via the Codman® HOLTER RICKHAM Reservoirs (Part Numbers: 82-1625, 82-1621, 82-1616) with the Codman® Ventricular Catheter (Part Number: 82-1650). The intraventricular access device reservoir and catheter must be implanted prior to the first infusion. It is recommended that the first dose be administered at least 5 to 7 days after device implantation.
BRINEURA is intended to be administered with the B Braun Perfusor® Space Infusion Pump System (Product Code: 8713030U). If an alternative pump must be used, the essential performance requirements for this syringe pump used to deliver BRINEURA are as follows:
- Delivery rate of 1.25 or 2.5 mL per hr with delivery accuracy of +/- 1 mL per hr
- Compatible with 20 mL syringes provided in the Administration Kit for use with BRINEURA
- Occlusion alarm setting to ≤ 281 mm Hg
- Cleared for intraventricular route of administration
Administer BRINEURA and the Intraventricular Electrolytes using the provided Administration Kit for use with BRINEURA components [see How Supplied/Storage and Handling (16)].
Each infusion consists of 3.3 to 10 mL of BRINEURA followed by 2 mL of Intraventricular Electrolytes. The complete infusion must be administered using an infusion set with a 0.2 micron inline filter. The Intraventricular Electrolytes are used to flush the infusion line, port needle, and intraventricular access device in order to fully administer BRINEURA and to maintain patency of the intraventricular access device.
Supplies for Infusion Preparation
- BRINEURA and Intraventricular Electrolytes Injection vials (package 1 of 2) [see How Supplied/Storage and Handling (16)]
- Administration Kit for use with BRINEURA (package 2 of 2) [see How Supplied/Storage and Handling (16)]
- Syringe pump (not supplied)
- Empty sterile single-use luer lock syringe, no larger than 3 mL (not supplied)
Inspect the Administration Kit infusion components to ensure the components are in the individual packages and have not been compromised.
Thaw BRINEURA and Intraventricular Electrolytes Injection vials at room temperature 20°C to 25°C (68°F to 77°F) for approximately 60 minutes. Do not thaw or warm vials any other way. Do not shake vials. Condensation will occur during thawing period. Do not re-freeze vials or freeze syringes containing BRINEURA or Intraventricular Electrolytes.
Inspect fully thawed BRINEURA and Intraventricular Electrolytes Injection vials. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. BRINEURA is a clear to slightly opalescent and colorless to pale yellow solution. Intraventricular Electrolytes is a clear to colorless solution. Do not use if the solutions are discolored or if there is other foreign particulate matter in the solutions. BRINEURA vials may occasionally contain thin translucent fibers or opaque particles. These naturally occurring particles are cerliponase alfa. These particles are removed via the 0.2 micron inline filter without having a detectable effect on the purity or strength of BRINEURA. Intraventricular Electrolytes may contain particles, which appear during the thaw period; however, these dissolve when the solution reaches room temperature.
2.3 Administration Modifications Due to Hypersensitivity Reactions Including Anaphylaxis
In the event of a severe hypersensitivity reaction (e.g., anaphylaxis), immediately discontinue BRINEURA administration and initiate appropriate medical treatment. If the decision is made to readminister BRINEURA after the occurrence of anaphylaxis, initiate the subsequent infusion at approximately one-half the infusion rate at which the anaphylactic reaction occurred with appropriate premedication. For additional recommendations in the event of a hypersensitivity reaction, see Warnings and Precautions (5.1) .
Structured Label Content
Section 42229-5 (42229-5)
Missed Dose
If one or more doses are missed, restart BRINEURA treatment as soon as possible, maintaining the 2-week interval between infusions thereafter.
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage
BRINEURA (cerliponase alfa) Injection and Intraventricular Electrolytes Injection:
Store upright in a freezer (-25°C to -15°C) in original carton to protect from light.
Administration Kit for use with BRINEURA:
Store in original carton separately from BRINEURA. Do not freeze.
11 Description (11 DESCRIPTION)
Cerliponase alfa is a purified human enzyme produced by recombinant DNA technology in a Chinese hamster ovary cell line. The active substance is a recombinant human tripeptidyl peptidase-1 (rhTPP1), a lysosomal exopeptidase. The primary activity of the mature enzyme is the cleavage of N-terminal tripeptides from a broad range of protein substrates.
Cerliponase alfa contains 544 amino acids with an average molecular mass of 59 kDa. The mature enzyme is 368 amino acids in length. There are 5 consensus N-glycosylation sites on rhTPP1 that contain high mannose, phosphorylated high mannose and complex glycosylation structures.
BRINEURA (cerliponase alfa) Injection and Intraventricular Electrolytes Injection are administered by intraventricular infusion. The solutions are sterile, nonpyrogenic, and free of foreign particulates. BRINEURA is a clear to slightly opalescent and colorless to pale yellow solution. Intraventricular Electrolytes is a clear to colorless solution.
BRINEURA and Intraventricular Electrolytes Injection are packaged in 10 mL clear Type 1 single-dose glass vials [see How Supplied/Storage and Handling (16)]. Each vial of BRINEURA provides 5 mL of solution containing 150 mg cerliponase alfa. Each vial of Intraventricular Electrolytes Injection provides 5 mL of solution. Both BRINEURA and Intraventricular Electrolytes Injection are formulated with the following excipients: calcium chloride dihydrate (1.05 mg); magnesium chloride hexahydrate (0.8 mg); potassium chloride (1.1 mg); sodium chloride (43.85 mg); sodium phosphate, dibasic, heptahydrate (0.55 mg); sodium phosphate, monobasic, monohydrate (0.4 mg); and Water for Injection, USP. The pH of the solution is between 6.2 to 6.8 for BRINEURA, and between 6.0 to 7.0 for Intraventricular Electrolytes Injection.
Each vial contains: sodium: 0.76 mEq, and potassium: 0.015 mEq.
8.4 Pediatric Use
The safety and effectiveness of BRINEURA have been established to slow the loss of ambulation in pediatric patients with CLN2 disease and the information on this use is discussed throughout the labeling.
BRINEURA is not recommended for use in patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age) or those weighing less than 2.5 kg due to physiologic immaturity which may increase risk of serious and clinically significant adverse reactions observed with BRINEURA [see Warnings and Precautions (5)].
Patients less than 3 years of age may be at increased risk for developing hypersensitivity reactions with BRINEURA use compared to patients 3 years of age and older [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies including cerliponase alfa.
In Trials 1 and 2 [see Clinical Studies (14)], 19 of 24 (79%) and 10 of 24 (42%) patients treated with BRINEURA developed anti-drug antibodies (ADAs) in serum and CSF, respectively. Drug-specific neutralizing antibodies (NAb) capable of inhibiting receptor-mediated cellular uptake of cerliponase alfa were detected in the CSF of 3 of 24 (13%) patients at a single visit and were undetectable in all other CSF samples tested in ADA positive patients. In Trial 3 [see Clinical Studies (14)], 14 of 14 (100%) and 3 of 14 (21%, all of the 3 patients were < 3 years of age) patients treated with BRINEURA developed ADAs in serum and CSF, respectively. NAb responses were not detected in the CSF of any ADA positive patients. Overall, patients younger than 3 years of age had higher ADA titers compared to patients 3 years of age and older.
In Trials 1 and 2, patients who experienced moderate to severe hypersensitivity adverse reactions were tested for drug-specific IgE and found to be negative. No association was found between serum ADA titers and incidence or severity of hypersensitivity. In Trial 3, hypersensitivity occurred in higher percentage in BRINEURA-treated patients < 3 years of age at baseline (amongst these patients, one patient experienced anaphylaxis), and higher ADA titers were also observed in this age group compared to patients ≥ 3 years of age at baseline. There was no identified clinically significant effect of ADA on pharmacokinetics or efficacy of BRINEURA. There is insufficient information to characterize the effects of ADA on safety.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of BRINEURA was assessed over 96 weeks in a non-randomized single-arm dose escalation clinical study with extension in symptomatic pediatric patients with late infantile neuronal ceroid lipofuscinosis type 2 (CLN2) disease, confirmed by TPP1 deficiency. BRINEURA-treated patients were compared to untreated patients from a natural history cohort. The Motor domain of a CLN2 Clinical Rating Scale was used to assess disease progression. Scores ranged from 3 (grossly normal) to 0 (profoundly impaired) with unit decrements representing milestone events in the loss of motor function (ability to walk or crawl). Due to the inability to establish comparability for the CLN2 Language domain ratings between the clinical study with extension and the natural history cohort, efficacy of BRINEURA for the Language domain cannot be established.
Twenty-four patients, aged 3 to 8 years were enrolled in the BRINEURA single-arm clinical study (Trial 1, NCT01907087). Sixty-three percent of patients were female and 37% were male. Ninety-six percent of patients were White and 4% were Asian; for ethnicity, 4% identified as Hispanic/Latino, 96% as non-Hispanic/Latino. One patient withdrew after week 1 due to inability to continue with study procedures; 23 patients were treated with BRINEURA 300 mg every other week by intraventricular infusion for 48 weeks, and continued treatment during the 240-week extension period, Trial 2 (NCT02485899), for a total duration of 288 weeks, plus a 24-week safety follow-up.
In the clinical study with extension, patients were assessed for decline in the Motor domain of the CLN2 Clinical Rating Scale at 48, 72 and 96 weeks. Decline was defined as having an unreversed (sustained) 2-category decline or an unreversed score of 0 in the Motor domain of the CLN2 Clinical Rating Scale. Patients' responses to BRINEURA treatment were evaluated if at screening a combined Motor plus Language CLN2 score of less than 6 was recorded. Two patients with a combined Motor plus Language CLN2 score of 6 were excluded from the analyses; they maintained that score throughout the study period. The patient who terminated early was analyzed as having a decline at the time of termination. Data used in the analyses from the natural history cohort began at 36 months of age or greater and at the first time a Motor plus Language CLN2 score less than 6 was recorded.
Motor scores of the 22 BRINEURA-treated patients in the clinical study with extension were compared to scores of the independent natural history cohort that included 42 untreated patients who satisfied inclusion criteria for the clinical study. The results of logistic modeling with covariates (screening age, screening motor score, genotype: 0 key mutations (yes/no)), demonstrated the odds of BRINEURA-treated patients not having a decline by 96 weeks were 13 times the odds of natural history cohort patients not having a decline (Odds Ratio (95% CI): 13.1 (1.2, 146.9)).
4 Contraindications (4 CONTRAINDICATIONS)
BRINEURA is contraindicated in patients with:
- any sign or symptom of acute, unresolved localized infection on or around the device insertion site (e.g., cellulitis or abscess); or suspected or confirmed CNS infection (e.g., cloudy CSF or positive CSF gram stain, or meningitis) [see Warnings and Precautions (5.2)].
- any acute intraventricular access device-related complication (e.g., leakage, extravasation of fluid, or device failure) [see Warnings and Precautions (5.3)].
- ventriculoperitoneal shunts.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described below and elsewhere in the labeling:
- Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.1)]
- Meningitis and Other Intraventricular Access Device-Related Infections [see Warnings and Precautions (5.2)]
- Intraventricular Access Device-Related Complications [see Warnings and Precautions (5.3)]
- Cardiovascular Adverse Reactions [see Warnings and Precautions (5.4)]
- Infusion-Associated Reactions [see Warnings and Precautions (5.5)]
12.2 Pharmacodynamics
No formal pharmacodynamic studies have been conducted with cerliponase alfa.
1 Indications and Usage (1 INDICATIONS AND USAGE)
BRINEURA is indicated to slow the loss of ambulation in pediatric patients with neuronal ceroid lipofuscinosis type 2 (CLN2 disease), also known as tripeptidyl peptidase 1 (TPP1) deficiency.
12.1 Mechanism of Action
CLN2 disease is a neurodegenerative disease caused by deficiency of the lysosomal enzyme tripeptidyl peptidase-1 (TPP1), which catabolizes polypeptides in the CNS. TPP1 has no known substrate specificity. Deficiency in TPP1 activity results in the accumulation of lysosomal storage materials normally metabolized by this enzyme in the central nervous system (CNS), leading to progressive decline in motor function.
Cerliponase alfa (rhTTP1), a proenzyme, is taken up by target cells in the CNS and is translocated to the lysosomes through the Cation Independent Mannose-6-Phosphate Receptor (CI-MPR, also known as M6P/IGF2 receptor). Cerliponase alfa is activated in the lysosome and the activated proteolytic form of rhTPP1 cleaves tripeptides from the N-terminus of proteins.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Meningitis and Other Intraventricular Access Device-Related Infections: Monitor the device insertion site for signs of infection. (4, 5.2)
- Intraventricular Access Device-Related Complications: Consult a neurosurgeon for any complications with the implanted device. In case of device-related complication, discontinue the infusion and refer to the device labeling for further instructions. (4, 5.3)
- Cardiovascular Adverse Reactions: Monitor vital signs before, during, and post-infusion. Monitor Electrocardiogram (ECG) in patients with a history of bradycardia, conduction disorder, or with structural heart disease, during the infusion. In patients without cardiac abnormalities, perform regular 12-lead ECG evaluations every 6 months. (2.5, 5.4)
- Infusion-Associated Reactions (IARs): If an IAR occurs, decreasing the infusion rate, temporarily stopping the infusion, and/or administering antihistamines and/or antipyretics may ameliorate the symptoms. (5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Administration of BRINEURA should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis. (2.1)
- BRINEURA should be administered by or under the supervision of a physician experienced in intraventricular administration. (2.1)
- Premedication of patients with antihistamines with or without antipyretics or corticosteroids is recommended. (2.1)
- Prior to each infusion, inspect the scalp for signs of intraventricular access device reservoir leakage, failure, or potential infection. (2.1)
- Recommended BRINEURA dosage is 300 mg administered once every other week as an intraventricular infusion. In patients less than 2 years of age, lower doses are recommended. (2.2)
- Dosing is not recommended in patients less than 37 weeks post-menstrual age or those weighing less than 2.5 kg. (2.2)
- See the full prescribing information for dosage modifications due to hypersensitivity reactions. (2.3)
- See the full prescribing information for preparation and administration instructions. (2.4, 2.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: BRINEURA 150 mg/5 mL (30 mg/mL) solution, two single-dose vials per carton co-packaged with Intraventricular Electrolytes Injection 5 mL in a single-dose vial. BRINEURA is a clear to slightly opalescent and colorless to pale yellow solution. Intraventricular Electrolytes is a clear to colorless solution [see How Supplied/Storage and Handling (16)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of BRINEURA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Immune system disorders: Anaphylactic reaction characterized by acute pyrexia, respiratory distress (bronchospasm, hypoxemia, perioral cyanosis), tachycardia, hypotension, diarrhea, and rash [see Warnings and Precautions (5.1)].
- Infections and infestations: Bacterial meningitis [see Warnings and Precautions (5.2)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
- Hypersensitivity Reactions Including Anaphylaxis and Infusion-Associated Reactions (IARs)
- Advise patients and caregivers that life-threatening hypersensitivity reactions, including anaphylaxis, and IARs may occur with BRINEURA treatment.
- Advise patients and caregivers that anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy.
- Inform patients and caregivers of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis, and IARs and to seek immediate medical care should symptoms occur [see Warnings and Precautions (5.1, 5.5)].
- Intraventricular Access Device-Related Infections
Advise patients and caregivers of the risk of device-related infections, including meningitis. Familiarize them with the signs and symptoms of these infections and instruct them to immediately contact their healthcare provider if an infection is suspected [see Warnings and Precautions (5.2)]. - Cardiovascular Adverse Reactions
Advise patients and caregivers that hypotension and/or bradycardia may occur during and following the infusion of BRINEURA. Instruct patients immediately to contact their healthcare provider if these reactions occur [see Warnings and Precautions (5.4)].
5.5 Infusion Associated Reactions (5.5 Infusion-Associated Reactions)
Infusion-associated reactions (IARs) such as vomiting, seizure, rash, pyrexia, hypersensitivity, and anaphylactic reaction have been observed in patients treated with BRINEURA [see Adverse Reactions (6.1)].
Premedication of patients with antihistamines with or without antipyretics or corticosteroids is recommended 30 to 60 minutes prior to the start of infusion. If an IAR occurs, decreasing the infusion rate, temporarily stopping the infusion, and/or administering antihistamines and/or antipyretics may ameliorate the symptoms. Closely monitor patients who have experienced IARs when re-administering BRINEURA.
In Trial 3, infusion-associated reactions were reported in 8 of 8 patients less than 3 years of age at baseline as compared to 4 of 6 patients ≥ 3 years of age at baseline.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
BRINEURA is supplied as a sterile, clear to slightly opalescent and colorless to pale yellow solution for intraventricular infusion and Intraventricular Electrolytes Injection is supplied as a clear to colorless solution for intraventricular infusion; both are included in package 1 of 2. The Administration Kit for use with BRINEURA is supplied separately as package 2 of 2 [see Dosage and Administration (2.4)].
5.4 Cardiovascular Adverse Reactions
Monitor vital signs (blood pressure, heart rate) before infusion starts, periodically during infusion, and post-infusion in a healthcare setting [see Dosage and Administration (2.6)]. Upon completion of the infusion, clinically assess the patient status. Continued observation may be necessary if clinically indicated.
Perform electrocardiogram (ECG) monitoring during infusion in patients with a history of bradycardia, conduction disorder, or with structural heart disease, as some patients with CLN2 disease may develop conduction disorders or heart disease. In patients without cardiac abnormalities, regular 12-lead ECG evaluations should be performed every 6 months.
In clinical Trial 1 and Trial 2 to 96 weeks, hypotension was reported in 2 of 24 (8%) patients, which occurred during or up to eight hours after BRINEURA infusion. Patients did not require alteration in treatment, and reactions resolved spontaneously or after intravenous fluid administration [see Adverse Reactions (6.1)].
2.2 Recommended Dosage and Administration
The recommended dosage of BRINEURA in pediatric patients is provided in Table 1. The dose is administered once every other week by intraventricular infusion.
BRINEURA is not recommended in patients less than 37 weeks post-menstrual age (gestational age at birth plus post-natal age) or those weighing less than 2.5 kg [see Use in Specific Populations (8.4)].
Administer BRINEURA first followed by infusion of the Intraventricular Electrolytes. The complete BRINEURA infusion, including the required infusion of Intraventricular Electrolytes, is approximately 2 to 4.5 hours, depending on the dose and volume administered. See Table 1 for the appropriate volume and infusion rate. For volumes that are not whole numbers, see Dosage and Administration (2.6) .
| Age groups | BRINEURA dose administered every other week | Volume of BRINEURA solution | Infusion rate |
|---|---|---|---|
| Birth to < 6 months | 100 mg | 3.3 mL | 1.25 mL/hr |
| 6 months to < 1 year | 150 mg | 5 mL | 2.5 mL/hr |
| 1 year to < 2 years | 200 mg (first 4 doses) | 6.7 mL (first 4 doses) | 2.5 mL/hr |
| 300 mg (subsequent doses) | 10 mL (subsequent doses) | ||
| 2 years and older | 300 mg | 10 mL | 2.5 mL/hr |
Principal Display Panel 5 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 5 mL Vial Label)
Intraventricular
Electrolytes
Injection
Use after Brineura®
(cerliponase alfa)
5 mL
For Intraventricular
Infusion Only
2.1 Recommendations Prior to Brineura Treatment (2.1 Recommendations Prior to BRINEURA Treatment)
- Administration of BRINEURA should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis.
- Initiate BRINEURA in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment [see Warnings and Precautions (5.1)].
- BRINEURA should be administered by or under the supervision of a physician experienced in intraventricular administration via a surgically implanted intraventricular access device system which consists of the reservoir and catheter components [see Dosage and Administration (2.6)].
- Premedication of patients with antihistamines with or without antipyretics or corticosteroids is recommended 30 to 60 minutes prior to the start of infusion [see Warnings and Precautions (5.1, 5.5)].
- Aseptic technique must be strictly observed during preparation and administration [see Dosage and Administration (2.6)].
- Prior to each infusion of BRINEURA, inspect the scalp for signs of intraventricular access device reservoir leakage, failure or potential infection [see Warnings and Precautions (5.2, 5.3)].
- Prior to each infusion of BRINEURA and when clinically indicated, obtain a sample of CSF for cell count and culture [see Warnings and Precautions (5.2)].
- Replace the intraventricular access device reservoir prior to 4 years of single-puncture administrations [see Warnings and Precautions (5.3)].
Principal Display Panel 150 Mg/5 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 150 mg/5 mL Vial Label)
Brineura®
(cerliponase alfa)
Injection
150 mg/5 mL
(30 mg/mL)
For Intraventricular Infusion Only
Use before Intraventricular
Electrolytes
Principal Display Panel Kit Carton Package 1 (PRINCIPAL DISPLAY PANEL - Kit Carton - Package 1)
NDC 68135-811-02
Brineura®
(cerliponase alfa)
Injection
150 mg/5 mL (30 mg/mL)
For Intraventricular Infusion Only
Each vial of Brineura® contains 150 mg cerliponase alfa
in 5 mL of solution (30 mg/mL)
No preservatives
Single-Dose Vial. Discard unused portion.
Brineura® and Intraventricular Electrolytes must be
administered with the Administration Kit provided.
Each carton contains:
2 vials, each containing Brineura®
(cerliponase alfa) Injection, 150 mg/5 mL
1 vial of Intraventricular Electrolytes
Injection, 5 mL
Package 1 of 2
Administration Kit is supplied
and stored separately as
package 2 of 2.
Rx Only
BIOMARIN®
Principal Display Panel Kit Carton Package 2 (PRINCIPAL DISPLAY PANEL - Kit Carton - Package 2)
Administration Kit for use with Brineura® (cerliponase alfa)
Intraventricular infusion only
Recommended Dosage: See Prescribing Information
Contents of Administration Kit:
- Two single-use 20-mL syringes
- Two single-use syringe needles, 21 G, 25.4 mm
- One single-use extension line
- One single-use infusion set with 0.2 micron inline filter
- One single-use port needle, 22 G, 16 mm
Package 2 of 2
Brineura® and Intraventricular Electrolytes Injection are separately supplied and
stored frozen as package 1 of 2
Not for resale
Only for use with Brineura®
Caution:
-
-Do not use if the seals or package are damaged.
-
-Do not resterilize.
-
-Single use only. Do not reuse.
-
-Do not freeze.
-
-Store in original package separately from Brineura®.
NOTE:
-
-This Administration Kit has no components made of natural latex rubber.
-
-Individual devices are provided in separate sterile packaging.
-
-Expiration dates may vary for each device included.
Manufactured for
BioMarin Pharmaceutical Inc.
Novato, CA 94949
www.bmrn.com
Rx Only
BIOMARIN®
5.1 Hypersensitivity Reactions Including Anaphylaxis
Life-threatening hypersensitivity reactions including anaphylaxis have been reported in patients treated with enzyme replacement therapies, including BRINEURA. BRINEURA-treated patients have had these reactions occur in clinical studies and postmarketing use [see Adverse Reactions (6)]. In clinical Trial 1 and Trial 2 to 96 weeks, a total of 11 of 24 (46%) patients experienced hypersensitivity reactions during the infusion or within 24 hours of completion of the infusion. Patients in clinical trials were routinely pre-medicated with antihistamines with or without antipyretics or corticosteroids, prior to infusion of BRINEURA. During postmarketing use, anaphylactic reactions occurred during or within several hours of BRINEURA infusion. Epinephrine was administered in these patients, and they received subsequent BRINEURA infusions without recurrence of anaphylaxis.
In Trial 3, hypersensitivity reactions were reported in 5 of 8 (63 %) patients less than 3 years of age at baseline as compared to 0 of 6 patients ≥ 3 years of age at baseline [see Adverse Reactions (6.1)]. Of the reported hypersensitivity reactions, a single anaphylactic reaction occurred in a subject < 3 years of age.
Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy. Administration of BRINEURA should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis. Initiate BRINERUA in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. Premedication of patients with antihistamines with or without antipyretics or corticosteroids is recommended 30 to 60 minutes prior to the start of infusion. Observe patients closely during and after the infusion. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue BRINEURA and immediately initiate appropriate medical treatment including use of epinephrine. Inform patients and caregivers of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and to seek immediate medical care should symptoms occur.
The management of hypersensitivity reactions should be based on the severity of the reaction and may include temporarily interrupting the infusion, and/or treatment with antihistamines, antipyretics, and/or corticosteroids. Consider the risks and benefits of readministration of BRINEURA following an anaphylactic reaction. If the decision is made to readminister BRINEURA after the occurrence of anaphylaxis, ensure appropriately trained personnel and equipment for emergency resuscitation (including epinephrine and other emergency medicines) are readily available during infusion. Initiate subsequent infusion at approximately one-half the initial infusion rate at which the anaphylactic reaction occurred.
5.3 Intraventricular Access Device Related Complications (5.3 Intraventricular Access Device-Related Complications)
During the clinical trials and in postmarketing reports, intraventricular access device-related complications were reported (e.g., device leakage, device failure extravasation of CSF fluid, or bulging of the scalp around or above the intraventricular access device) [see Adverse Reactions (6.1)]. For any complications with the implanted intraventricular access device, consult with a neurosurgeon to confirm the integrity or performance of the device. In case of intraventricular access device-related complications, discontinue the BRINEURA infusion and refer to the device manufacturer's labeling for further instructions [see Contraindications (4)].
Material degradation of the intraventricular access device reservoir was reported after approximately 4 years of administration, which may impact the effective and safe use of the device. During benchtop testing such material degradation was recognized after approximately 105 perforations of the intraventricular access device. The intraventricular access device should be replaced prior to 4 years of single-puncture administrations, which equates to approximately 105 administrations of BRINEURA.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity, genotoxicity, and fertility studies have not been performed with cerliponase alfa. Based on the mechanism of action, cerliponase alfa is not expected to be tumorigenic.
Warning: Hypersensitivity Reactions Including Anaphylaxis (WARNING: HYPERSENSITIVITY REACTIONS INCLUDING ANAPHYLAXIS)
Patients treated with enzyme replacement therapies have experienced life-threatening hypersensitivity reactions, including anaphylaxis. Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy.
Initiate BRINEURA in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue BRINEURA and immediately initiate appropriate medical treatment, including use of epinephrine. Inform patients of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and to seek immediate medical care should symptoms occur [see Warnings and Precautions (5.1)].
5.2 Meningitis and Other Intraventricular Access Device Related Infections (5.2 Meningitis and Other Intraventricular Access Device-Related Infections)
Bacterial meningitis requiring antibiotic treatment and removal of the device was reported during postmarketing use of BRINEURA. Additionally, in clinical trials and during postmarketing use there were reports of other device-related clinical infections which were confirmed by positive CSF cultures, treated with antibiotics and removal of the entire intraventricular access device, and in which patients resumed treatment with BRINEURA after the device was replaced [see Adverse Reactions (6.1)]. In all cases, antibiotics were administered, the intraventricular access devices were removed and replaced, and patients resumed treatment with BRINEURA.
The signs and symptoms of infections may not be readily apparent in patients with CLN2 disease. To reduce the risk of infectious complications, BRINEURA should be administered by, or under the supervision of, a physician experienced in intraventricular administration. Prior to each infusion of BRINEURA and when clinically indicated, obtain a sample of CSF for cell count and culture. Do not administer BRINEURA if there are localized signs of infection on or around the device insertion site, such as erythema, tenderness, or discharge or suspected or confirmed CNS infection (e.g., cloudy CSF or positive CSF gram stain, or meningitis) [see Contraindications (4)].
Healthcare providers should be vigilant for the development of signs and symptoms of infection, including meningitis, during treatment with BRINEURA and monitor the device insertion site for signs of infection.
2.4 Preparation Instructions for Intraventricular Administration of the Product
BRINEURA and the Intraventricular Electrolytes must only be administered by the intraventricular route, using the provided Administration Kit for use with BRINEURA. Each vial of BRINEURA and Intraventricular Electrolytes is intended for a single dose only.
BRINEURA is administered into the cerebrospinal fluid (CSF) by infusion via a surgically implanted reservoir and catheter (the "intraventricular access device system"). BRINEURA is intended to be administered via the Codman® HOLTER RICKHAM Reservoirs (Part Numbers: 82-1625, 82-1621, 82-1616) with the Codman® Ventricular Catheter (Part Number: 82-1650). The intraventricular access device reservoir and catheter must be implanted prior to the first infusion. It is recommended that the first dose be administered at least 5 to 7 days after device implantation.
BRINEURA is intended to be administered with the B Braun Perfusor® Space Infusion Pump System (Product Code: 8713030U). If an alternative pump must be used, the essential performance requirements for this syringe pump used to deliver BRINEURA are as follows:
- Delivery rate of 1.25 or 2.5 mL per hr with delivery accuracy of +/- 1 mL per hr
- Compatible with 20 mL syringes provided in the Administration Kit for use with BRINEURA
- Occlusion alarm setting to ≤ 281 mm Hg
- Cleared for intraventricular route of administration
Administer BRINEURA and the Intraventricular Electrolytes using the provided Administration Kit for use with BRINEURA components [see How Supplied/Storage and Handling (16)].
Each infusion consists of 3.3 to 10 mL of BRINEURA followed by 2 mL of Intraventricular Electrolytes. The complete infusion must be administered using an infusion set with a 0.2 micron inline filter. The Intraventricular Electrolytes are used to flush the infusion line, port needle, and intraventricular access device in order to fully administer BRINEURA and to maintain patency of the intraventricular access device.
Supplies for Infusion Preparation
- BRINEURA and Intraventricular Electrolytes Injection vials (package 1 of 2) [see How Supplied/Storage and Handling (16)]
- Administration Kit for use with BRINEURA (package 2 of 2) [see How Supplied/Storage and Handling (16)]
- Syringe pump (not supplied)
- Empty sterile single-use luer lock syringe, no larger than 3 mL (not supplied)
Inspect the Administration Kit infusion components to ensure the components are in the individual packages and have not been compromised.
Thaw BRINEURA and Intraventricular Electrolytes Injection vials at room temperature 20°C to 25°C (68°F to 77°F) for approximately 60 minutes. Do not thaw or warm vials any other way. Do not shake vials. Condensation will occur during thawing period. Do not re-freeze vials or freeze syringes containing BRINEURA or Intraventricular Electrolytes.
Inspect fully thawed BRINEURA and Intraventricular Electrolytes Injection vials. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. BRINEURA is a clear to slightly opalescent and colorless to pale yellow solution. Intraventricular Electrolytes is a clear to colorless solution. Do not use if the solutions are discolored or if there is other foreign particulate matter in the solutions. BRINEURA vials may occasionally contain thin translucent fibers or opaque particles. These naturally occurring particles are cerliponase alfa. These particles are removed via the 0.2 micron inline filter without having a detectable effect on the purity or strength of BRINEURA. Intraventricular Electrolytes may contain particles, which appear during the thaw period; however, these dissolve when the solution reaches room temperature.
2.3 Administration Modifications Due to Hypersensitivity Reactions Including Anaphylaxis (2.3 Administration Modifications due to Hypersensitivity Reactions Including Anaphylaxis)
In the event of a severe hypersensitivity reaction (e.g., anaphylaxis), immediately discontinue BRINEURA administration and initiate appropriate medical treatment. If the decision is made to readminister BRINEURA after the occurrence of anaphylaxis, initiate the subsequent infusion at approximately one-half the infusion rate at which the anaphylactic reaction occurred with appropriate premedication. For additional recommendations in the event of a hypersensitivity reaction, see Warnings and Precautions (5.1) .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:42:17.704524 · Updated: 2026-03-14T22:08:27.804807