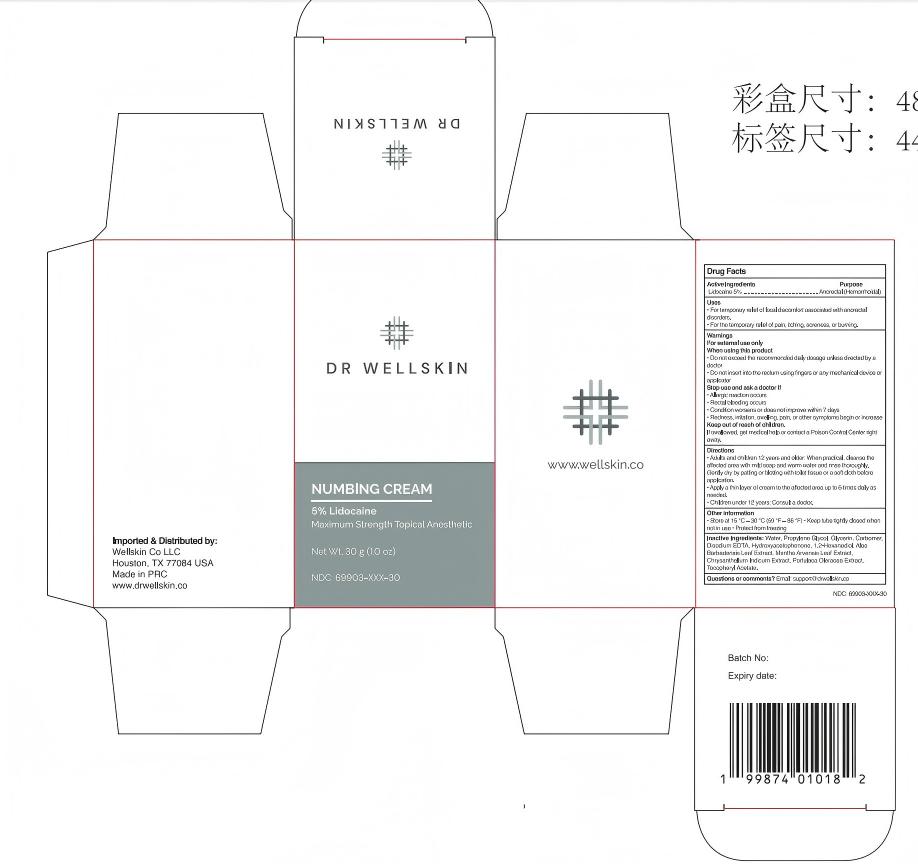
dr wellskin numbing cream
482f25cc-dee3-c3b6-e063-6294a90a8674
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
For external use only.
Medication Information
Warnings and Precautions
For external use only.
Active Ingredient
4% Lidocaine
Indications and Usage
Adults and children 12 years and older: When practical, cleanse theaffected area with mild soap and warm water and rinse thoroughly.Gently dry by patting or blotting with toilet tissue or a soft cloth before application.
Apply a thin layer of cream to the affected area up to 6 times daily as needed.
Children under 12 years: Consult a doctor.
Dosage and Administration
Adults and children 12
years and older: Apply a
thin layer to the affected
area 1-3 times daily or
as directed by a
healthcare provider.
Description
For external use only.
Section 50565-1
If swallowed, get medical help or contact a Poison Control Center right
away.
Section 50566-9
Allergic reaction occurs.
Rectal bleeding occurs.
Condition worsens or does not improve within 7 days.
Redness, irritation, sweling, pain, or other symptoms begin or increase.
Section 50567-7
Do not exceed the recommended daily dosage unless directed by adoctor
Do not insert into the rectum using fingers or any mechanical device orapplicator
Section 51727-6
Water, Propylene Glycol, Glycerin, Carbomer, Disodium EDTA, Hydroxyacetophenone, 1,2-Hexanediol, Aloe Barbadensis Leaf Extract, Mentha Arvensis Leaf Extract, Chrysanthellum Indicum Extract, Portulaca Oleracea Extract,Tocopheryl Acetate.
Section 51945-4
Section 55105-1
Topical Anesthetic
Structured Label Content
Warnings and Precautions (34071-1)
For external use only.
Active Ingredient (55106-9)
4% Lidocaine
Indications and Usage (34067-9)
Adults and children 12 years and older: When practical, cleanse theaffected area with mild soap and warm water and rinse thoroughly.Gently dry by patting or blotting with toilet tissue or a soft cloth before application.
Apply a thin layer of cream to the affected area up to 6 times daily as needed.
Children under 12 years: Consult a doctor.
Dosage and Administration (34068-7)
Adults and children 12
years and older: Apply a
thin layer to the affected
area 1-3 times daily or
as directed by a
healthcare provider.
Section 50565-1 (50565-1)
If swallowed, get medical help or contact a Poison Control Center right
away.
Section 50566-9 (50566-9)
Allergic reaction occurs.
Rectal bleeding occurs.
Condition worsens or does not improve within 7 days.
Redness, irritation, sweling, pain, or other symptoms begin or increase.
Section 50567-7 (50567-7)
Do not exceed the recommended daily dosage unless directed by adoctor
Do not insert into the rectum using fingers or any mechanical device orapplicator
Section 51727-6 (51727-6)
Water, Propylene Glycol, Glycerin, Carbomer, Disodium EDTA, Hydroxyacetophenone, 1,2-Hexanediol, Aloe Barbadensis Leaf Extract, Mentha Arvensis Leaf Extract, Chrysanthellum Indicum Extract, Portulaca Oleracea Extract,Tocopheryl Acetate.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Topical Anesthetic
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:11.527046 · Updated: 2026-03-14T23:12:21.703136