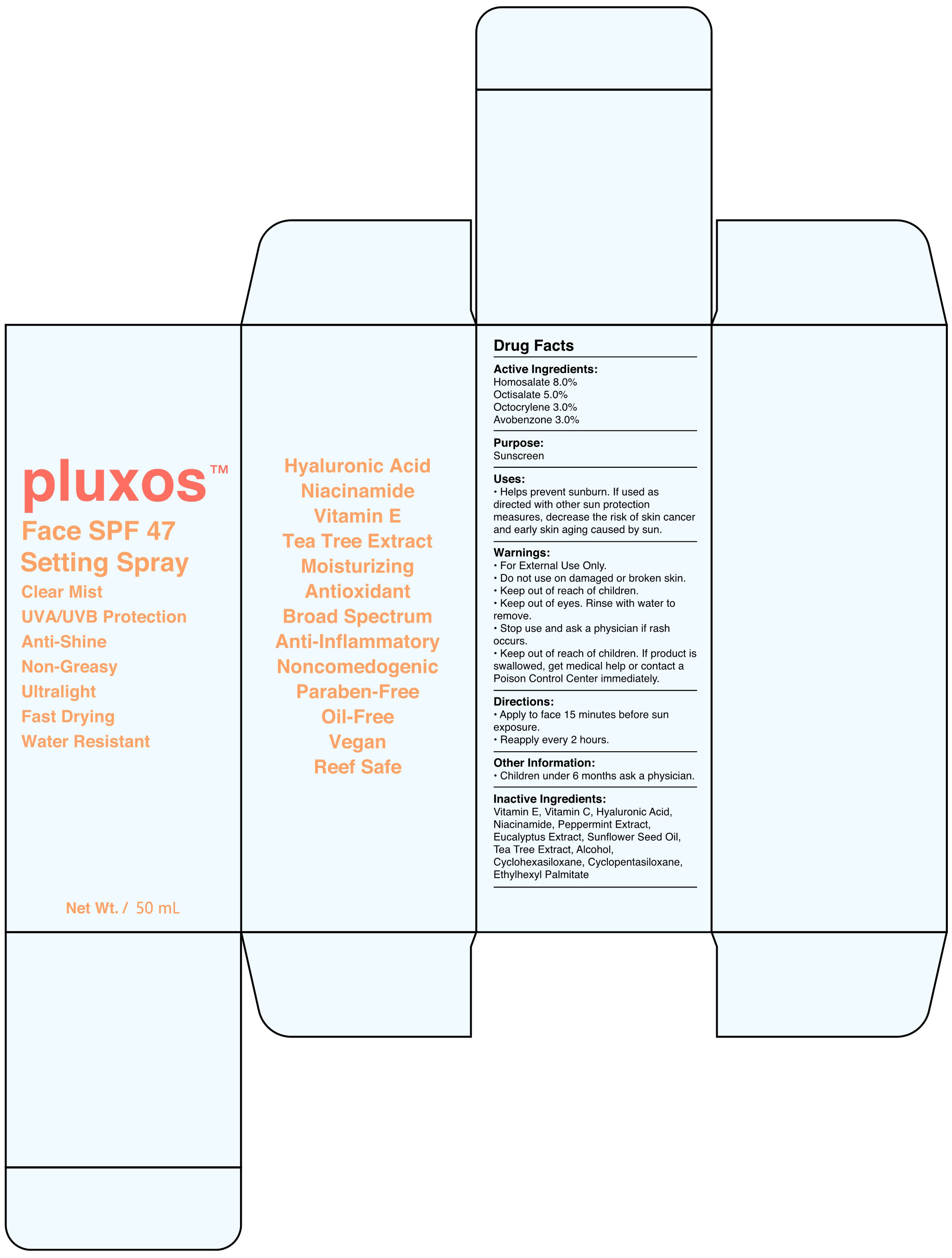
87283-001-01 Face Spf 47 Setting
47db9a54-c02f-d2ea-e063-6394a90ab973
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Homosalate 8.0% Octisalate 5.0% Octocrylene 3.0% Avobenzone 3.0%
Medication Information
Warnings and Precautions
For External Use Only.
Do not use on damaged or broken skin.
Keep out of reach of children.
Keep out of eyes. Rinse with water to remove.
Stop use and ask a physician if rash occurs.
Keep out of reach of children . If product is swallowed, get medical help or contact a Poison Control Center immediately.
Indications and Usage
Helps prevent sunburn. If used as directed with other sun protection measures, decrease the risk of skin cancer and early skin aging caused by sun.
Dosage and Administration
•Apply to face 15 minutes before sun exposure.
Reapply every 2 hours.
Description
Homosalate 8.0% Octisalate 5.0% Octocrylene 3.0% Avobenzone 3.0%
Section 50565-1
Keep out of reach of children.
Section 51727-6
Vitamin E, Vitamin C, Hyaluronic Acid, Niacinamide, Peppermint Extract, Eucalyptus Extract, Sunflower Seed Oil, Tea Tree Extract, Alcohol, Cyclohexasiloxane, Cyclopentasiloxane, Ethylhexyl Palmitate
Section 51945-4
Section 55105-1
Sunscreen
Section 55106-9
Homosalate 8.0%
Octisalate 5.0%
Octocrylene 3.0%
Avobenzone 3.0%
Structured Label Content
Indications and Usage (34067-9)
Helps prevent sunburn. If used as directed with other sun protection measures, decrease the risk of skin cancer and early skin aging caused by sun.
Dosage and Administration (34068-7)
•Apply to face 15 minutes before sun exposure.
Reapply every 2 hours.
Warnings and Precautions (34071-1)
For External Use Only.
Do not use on damaged or broken skin.
Keep out of reach of children.
Keep out of eyes. Rinse with water to remove.
Stop use and ask a physician if rash occurs.
Keep out of reach of children . If product is swallowed, get medical help or contact a Poison Control Center immediately.
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 51727-6 (51727-6)
Vitamin E, Vitamin C, Hyaluronic Acid, Niacinamide, Peppermint Extract, Eucalyptus Extract, Sunflower Seed Oil, Tea Tree Extract, Alcohol, Cyclohexasiloxane, Cyclopentasiloxane, Ethylhexyl Palmitate
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Sunscreen
Section 55106-9 (55106-9)
Homosalate 8.0%
Octisalate 5.0%
Octocrylene 3.0%
Avobenzone 3.0%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:09.014977 · Updated: 2026-03-14T23:12:18.638181