These Highlights Do Not Include All The Information Needed To Use Tazarotene Cream Safely And Effectively. See Full Prescribing Information For Tazarotene Cream.
478ff059-9d7b-438e-a64e-c853dfac2872
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Tazarotene cream 0.1% is a retinoid indicated for the topical treatment of plaque psoriasis. ( 1.1 ) Tazarotene cream 0.1% is indicated for the topical treatment of acne vulgaris. ( 1.2 )
Indications and Usage
Tazarotene cream 0.1% is a retinoid indicated for the topical treatment of plaque psoriasis. ( 1.1 ) Tazarotene cream 0.1% is indicated for the topical treatment of acne vulgaris. ( 1.2 )
Dosage and Administration
Apply a thin layer of tazarotene cream only to the affected area once daily in the evening. ( 2.1 , 2.2 ) Not for ophthalmic, oral, or intravaginal use. ( 2.2 ) If contact with eyes occurs, rinse thoroughly with water. ( 2.2 )
Warnings and Precautions
Embryofetal Toxicity: Tazarotene cream contains tazarotene, which is a teratogenic substance. Tazarotene cream is contraindicated in pregnancy. Females of child-bearing potential should have a negative pregnancy test within 2 weeks prior to initiating treatment and use an effective method of contraception during treatment. ( 5.1 ) Local Irritation: Some individuals may experience excessive pruritus, burning, skin redness or peeling. If these effects occur, discontinue until the integrity of the skin has been restored, or reduce dosing interval, or in the case of psoriasis, may switch to the lower concentration. Tazarotene cream should not be used on eczematous skin, as it may cause severe irritation. ( 5.2 ) Photosensitivity and Risk for Sunburn: Avoid exposure to sunlight, sunlamps, and weather extremes. Wear sunscreen daily. Tazarotene cream should be administered with caution if the patient is also taking drugs known to be photosensitizers. ( 5.3 )
Contraindications
Tazarotene cream is contraindicated in: Pregnancy. Retinoids may cause fetal harm when administered to a pregnant female [see Warnings and Precautions (5.1) , Use in Specific Populations (8.1 , 8.3) ] . Individuals who have known hypersensitivity to any of its components [see Warnings and Precautions (5.2) ].
Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of the labeling: Embryofetal toxicity [see Warnings and Precautions (5.1) ] Photosensitivity and Risk of Sunburn [see Warnings and Precautions (5.3) ]
Drug Interactions
No formal drug-drug interaction studies were conducted with tazarotene cream. In a trial of 27 healthy female subjects between the ages of 20 to 55 years receiving a combination oral contraceptive tablet containing 1 mg norethindrone and 35 mcg ethinyl estradiol, concomitant use of tazarotene administered as 1.1 mg orally (mean ± SD C max and AUC 0-24 of tazarotenic acid were 28.9 ± 9.4 ng/mL and 120.6 ± 28.5 ng∙hr/mL) did not affect the pharmacokinetics of norethindrone and ethinyl estradiol over a complete cycle. The impact of tazarotene on the pharmacokinetics of progestin only oral contraceptives (i.e., minipills) has not been evaluated.
Storage and Handling
Tazarotene cream is a white cream available in a concentration of 0.1%. It is supplied in a collapsible aluminum tube with a tamper-evident aluminum membrane over the opening and a white polypropylene screw cap, in 30 g and 60 g sizes. Tazarotene Cream, 0.1% 30 gram NDC 51672-1373-2 60 gram NDC 51672-1373-3
How Supplied
Tazarotene cream is a white cream available in a concentration of 0.1%. It is supplied in a collapsible aluminum tube with a tamper-evident aluminum membrane over the opening and a white polypropylene screw cap, in 30 g and 60 g sizes. Tazarotene Cream, 0.1% 30 gram NDC 51672-1373-2 60 gram NDC 51672-1373-3
Medication Information
Warnings and Precautions
Embryofetal Toxicity: Tazarotene cream contains tazarotene, which is a teratogenic substance. Tazarotene cream is contraindicated in pregnancy. Females of child-bearing potential should have a negative pregnancy test within 2 weeks prior to initiating treatment and use an effective method of contraception during treatment. ( 5.1 ) Local Irritation: Some individuals may experience excessive pruritus, burning, skin redness or peeling. If these effects occur, discontinue until the integrity of the skin has been restored, or reduce dosing interval, or in the case of psoriasis, may switch to the lower concentration. Tazarotene cream should not be used on eczematous skin, as it may cause severe irritation. ( 5.2 ) Photosensitivity and Risk for Sunburn: Avoid exposure to sunlight, sunlamps, and weather extremes. Wear sunscreen daily. Tazarotene cream should be administered with caution if the patient is also taking drugs known to be photosensitizers. ( 5.3 )
Indications and Usage
Tazarotene cream 0.1% is a retinoid indicated for the topical treatment of plaque psoriasis. ( 1.1 ) Tazarotene cream 0.1% is indicated for the topical treatment of acne vulgaris. ( 1.2 )
Dosage and Administration
Apply a thin layer of tazarotene cream only to the affected area once daily in the evening. ( 2.1 , 2.2 ) Not for ophthalmic, oral, or intravaginal use. ( 2.2 ) If contact with eyes occurs, rinse thoroughly with water. ( 2.2 )
Contraindications
Tazarotene cream is contraindicated in: Pregnancy. Retinoids may cause fetal harm when administered to a pregnant female [see Warnings and Precautions (5.1) , Use in Specific Populations (8.1 , 8.3) ] . Individuals who have known hypersensitivity to any of its components [see Warnings and Precautions (5.2) ].
Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of the labeling: Embryofetal toxicity [see Warnings and Precautions (5.1) ] Photosensitivity and Risk of Sunburn [see Warnings and Precautions (5.3) ]
Drug Interactions
No formal drug-drug interaction studies were conducted with tazarotene cream. In a trial of 27 healthy female subjects between the ages of 20 to 55 years receiving a combination oral contraceptive tablet containing 1 mg norethindrone and 35 mcg ethinyl estradiol, concomitant use of tazarotene administered as 1.1 mg orally (mean ± SD C max and AUC 0-24 of tazarotenic acid were 28.9 ± 9.4 ng/mL and 120.6 ± 28.5 ng∙hr/mL) did not affect the pharmacokinetics of norethindrone and ethinyl estradiol over a complete cycle. The impact of tazarotene on the pharmacokinetics of progestin only oral contraceptives (i.e., minipills) has not been evaluated.
Storage and Handling
Tazarotene cream is a white cream available in a concentration of 0.1%. It is supplied in a collapsible aluminum tube with a tamper-evident aluminum membrane over the opening and a white polypropylene screw cap, in 30 g and 60 g sizes. Tazarotene Cream, 0.1% 30 gram NDC 51672-1373-2 60 gram NDC 51672-1373-3
How Supplied
Tazarotene cream is a white cream available in a concentration of 0.1%. It is supplied in a collapsible aluminum tube with a tamper-evident aluminum membrane over the opening and a white polypropylene screw cap, in 30 g and 60 g sizes. Tazarotene Cream, 0.1% 30 gram NDC 51672-1373-2 60 gram NDC 51672-1373-3
Description
Tazarotene cream 0.1% is a retinoid indicated for the topical treatment of plaque psoriasis. ( 1.1 ) Tazarotene cream 0.1% is indicated for the topical treatment of acne vulgaris. ( 1.2 )
Section 42229-5
Females of Child-bearing Potential
Females of child-bearing potential should be warned of the potential risk and use adequate birth-control measures when tazarotene cream is used. The possibility that a female of child-bearing potential is pregnant at the time of institution of therapy should be considered.
A negative result for pregnancy test should be obtained within 2 weeks prior to tazarotene cream therapy. Tazarotene cream therapy should begin during a menstrual period [see Use in Specific Populations (8.1)] .
Section 42230-3
| Patient Information
Tazarotene (taz-AR-oh-teen) Cream, 0.1% |
|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. |
| Revised: August 2025 5262058-0825-01 75 |
| Important information: Tazarotene cream is for use on skin only. Do not use tazarotene cream in your eyes, mouth, or vagina. |
|
What is the most important information I should know about tazarotene cream?
Tazarotene cream may cause birth defects if used during pregnancy.
|
What is tazarotene cream?
|
|
Who should not use tazarotene cream?
Do not use tazarotene cream if you:
|
|
What should I tell my doctor before using tazarotene cream?
Before you use tazarotene cream, tell your doctor about all of your medical conditions, including if you:
Certain medicines, vitamins, or supplements may make your skin more sensitive to sunlight. Also, tell your doctor about any cosmetics you use, including moisturizers, creams, lotions, or products that can dry out your skin. |
How should I use tazarotene cream?
|
Follow these instructions for applying tazarotene cream:
|
What should I avoid while using tazarotene cream?
|
|
What are the possible side effects of tazarotene cream?
Tazarotene cream may cause serious side effects, including:
The most common side effects of tazarotene cream in people with acne includepeeling, dry skin, redness and burning. These are not all the possible side effects of tazarotene cream. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
How should I store tazarotene cream?
|
|
General information about the safe and effective use of tazarotene cream.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use tazarotene cream for a condition for which it was not prescribed. Do not give tazarotene cream to other people, even if they have the same symptoms you have. It may harm them. You can ask your doctor or pharmacist for information about tazarotene cream that is written for health professionals. |
|
What are the ingredients in tazarotene cream?
Active ingredient: tazarotene Inactive ingredients:benzyl alcohol 1%, carbomer copolymer type B, carbomer homopolymer type B, edetate disodium, medium-chain triglycerides, mineral oil, purified water, sodium hydroxide, sodium thiosulfate, and sorbitan monooleate. |
| Manufactured by: Sun Pharma Canada Inc., Brampton, Ontario, Canada L6T 1C1
Distributed by: Sun Pharmaceutical Industries, Inc., Cranbury, NJ 08512 |
Section 44425-7
Store at 20° to 25°C (68° to 77°F)[see USP Controlled Room Temperature].
2.3 Acne
Cleanse the face gently. After the skin is dry, apply a thin layer (2 mg/cm 2) of tazarotene cream 0.1% once per day, in the evening, to the skin areas where acne lesions appear. Use enough to cover the entire affected area.
Use effective sunscreens and wear protective clothing while using tazarotene cream [see Warnings and Precautions (5.3)].
10 Overdosage
Excessive topical use of tazarotene cream, 0.1% may lead to marked redness, peeling, or discomfort [see Warnings and Precautions (5.2)] .
Tazarotene cream, 0.1% is not for oral use. Oral ingestion of the drug may lead to the same adverse effects as those associated with excessive oral intake of Vitamin A (hypervitaminosis A) or other retinoids. If oral ingestion occurs, the patient should be monitored, and appropriate supportive measures should be administered as necessary.
2.2 Psoriasis
It is recommended that treatment starts with tazarotene cream, 0.05%, with strength increased to 0.1% if tolerated and medically indicated. Apply a thin film (2 mg/cm 2) of tazarotene cream once per day, in the evening, to cover only the psoriatic lesions. If a bath or shower is taken prior to application, the skin should be dry before applying the cream. If emollients are used, they should be applied at least an hour before application of tazarotene cream. Because unaffected skin may be more susceptible to irritation, application of tazarotene cream to these areas should be carefully avoided.
11 Description
Tazarotene cream, 0.1% is for topical use and contains the active ingredient, tazarotene. Each gram of tazarotene cream, 0.1% contains 1 mg of tazarotene in a white cream base.
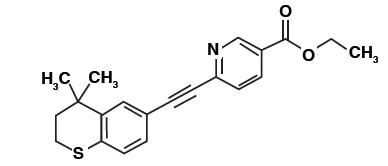
Tazarotene is a member of the acetylenic class of retinoids. Chemically, tazarotene is ethyl 6-[(4,4-dimethylthiochroman-6-yl)ethynyl]nicotinate. The compound has an empirical formula of C 21H 21NO 2S and molecular weight of 351.46. The structural formula is shown below:
Tazarotene cream contains the following inactive ingredients: benzyl alcohol 1%, carbomer copolymer type B, carbomer homopolymer type B, edetate disodium, medium-chain triglycerides, mineral oil, purified water, sodium hydroxide (to adjust pH), sodium thiosulfate, and sorbitan monooleate.
1.2 Acne Vulgaris
Tazarotene cream, 0.1% is also indicated for the topical treatment of patients with acne vulgaris.
8.4 Pediatric Use
The safety and efficacy of tazarotene cream have not been established in patients with psoriasis under the age of 18 years, or in patients with acne under the age of 12 years.
8.5 Geriatric Use
Tazarotene cream for the treatment of acne has not been clinically tested in persons 65 years of age or older.
Of the total number of subjects in clinical trials of tazarotene cream for plaque psoriasis, 120 were over the age of 65. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Currently there is no other clinical experience on the differences in responses between the elderly and younger subjects, but greater sensitivity of some older individuals cannot be ruled out.
14 Clinical Studies
In two 12-week vehicle-controlled clinical trials, tazarotene cream, 0.05% and 0.1% was significantly more effective than vehicle in reducing the severity of stable plaque psoriasis. Tazarotene cream, 0.1% and 0.05% demonstrated superiority over vehicle cream as early as 1 week and 2 weeks, respectively, after starting treatment.
In these trials, the primary efficacy endpoint was "clinical success," defined as the proportion of subjects with none, minimal, or mild overall lesional assessment at Week 12, and shown in Table 1. "Clinical success" was also significantly greater with tazarotene cream, 0.05% and 0.1% versus vehicle at most follow-up visits.
| Tazarotene Cream, 0.05% | Tazarotene Cream, 0.1% | Vehicle Cream | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Trial 1
N=218 |
Trial 2
N=210 |
Trial 1
N=221 |
Trial 2
N=211 |
Trial 1
N=229 |
Trial 2
N=214 |
||||||||||
| Score | BL | Wk 12 | Wk 24 | BL | Wk 12 | BL | Wk 12 | Wk 24 | BL | Wk 12 | BL | Wk 12 | Wk 24 | BL | Wk 12 |
|
|||||||||||||||
| None
(0) |
0 | 1
(0.5%) |
1
(0.5%) |
0 | 2
(1%) |
0 | 0 | 0 | 0 | 6
(3%) |
0 | 0 | 1
(0.4%) |
0 | 1
(0.5%) |
| Minimal
(1) |
0 | 11
(5%) |
12
(6%) |
0 | 7
(3%) |
0 | 12
(5%) |
14
(6%) |
0 | 11
(5%) |
0 | 7
(3%) |
6
(3%) |
0 | 1
(0.5%) |
| Mild
(2) |
0 | 79
(36%) |
60
(28%) |
0 | 76
(36%) |
0 | 75
(34%) |
53
(24%) |
0 | 90
(43%) |
0 | 49
(21%) |
43
(19%) |
0 | 54
(25%) |
| Moderate
(3) |
141
(65%) |
86
(39%) |
90
(41%) |
100
(48%) |
74
(35%) |
122
(55%) |
97
(44%) |
107
(48%) |
96
(45%) |
62
(29%) |
139
(61%) |
119
(52%) |
114
(50%) |
97
(45%) |
99
(46%) |
| Severe
(4) |
69
(32%) |
39
(18%) |
51
(23%) |
80
(38%) |
36
(17%) |
91
(41%) |
36
(16%) |
46
(21%) |
86
(41%) |
29
(14%) |
81
(35%) |
51
(22%) |
61
(27%) |
93
(44%) |
47
(22%) |
| Very Severe
(5) |
8
(4%) |
2
(0.9%) |
4
(2%) |
30
(14%) |
15
(7%) |
8
(4%) |
1
(0.5%) |
1
(0.5%) |
29
(14%) |
13
(6%) |
9
(4%) |
3
(1%) |
4
(2%) |
24
(11%) |
12
(6%) |
| "Clinical Success" | 0 | 91
(42% Denotes statistically significant difference for "Clinical Success" compared with vehicle. )
|
73
(33% ) |
0 | 85
(40% ) |
0 | 87
(39% ) |
67
(30% ) |
0 | 107
(51% ) |
0 | 56
(24%) |
50
(22%) |
0 | 56
(26%) |
At the end of 12 weeks of treatment, tazarotene cream, 0.05% and 0.1% was consistently superior to vehicle in reducing the plaque thickness of psoriasis. Improvements in erythema and scaling were generally significantly greater with tazarotene cream, 0.05% and 0.1% than with vehicle. Tazarotene Cream, 0.1% was also generally more effective than tazarotene cream, 0.05% in reducing the severity of the individual signs of disease. However, tazarotene cream, 0.1% was associated with a greater degree of local irritation than tazarotene cream, 0.05%.
| Tazarotene Cream, 0.05% | Tazarotene Cream, 0.1% | Vehicle Cream | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lesion | Trunk/Arm/Leg lesions | Knee/Elbow lesions | All Treated | Trunk/Arm/Leg lesions | Knee/Elbow lesions | All Treated | Trunk/Arm/Leg lesions | Knee/Elbow lesions | All Treated | ||||||||||
| Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | ||
| N=218 | N=210 | N=218 | N=210 | N=218 | N=210 | N=221 | N=211 | N=221 | N=211 | N=221 | N=211 | N=229 | N=214 | N=229 | N=214 | N=229 | N=214 | ||
| Plaque elevation, scaling and erythema scored on a 0-4 scale with 0=none, 1=mild, 2=moderate, 3=severe and 4=very severe. | |||||||||||||||||||
| B#=Mean Baseline Severity; | |||||||||||||||||||
| C-12=Mean Change from Baseline at end of 12 weeks of therapy; | |||||||||||||||||||
| C-24=Mean Change from Baseline at week 24 (12 weeks after the end of therapy). | |||||||||||||||||||
| Plaque elevation | B# | 2.29 | 2.50 | 2.40 | 2.52 | 2.28 | 2.51 | 2.34 | 2.52 | 2.35 | 2.49 | 2.32 | 2.51 | 2.28 | 2.51 | 2.35 | 2.51 | 2.29 | 2.51 |
| C-12 | -0.83
Denotes statistically significant difference compared with vehicle.
|
-0.98 | -0.91 | -1.04 | -0.75 | -0.90 | -1.08 | -1.25 | -0.96 | -1.21 | -0.83 | -1.08 | -0.59 | -0.69 | -0.57 | -0.68 | -0.48 | -0.61 | |
| C-24 | -0.75 | -0.73 | -0.60 | -0.87 | -0.73 | -0.63 | -0.57 | -0.49 | -0.42 | ||||||||||
| Scaling | B# | 2.26 | 2.45 | 2.47 | 2.60 | 2.32 | 2.47 | 2.37 | 2.45 | 2.40 | 2.57 | 2.36 | 2.53 | 2.34 | 2.46 | 2.45 | 2.61 | 2.31 | 2.53 |
| C-12 | -0.75 | -0.90 | -0.78 | -0.98 | -0.67 | -0.80 | -0.84 | -1.06 | -0.76 | -1.13 | -0.73 | -1.03 | -0.66 | -0.79 | -0.62 | -0.76 | -0.46 | -0.70 | |
| C-24 | -0.68 | -0.62 | -0.51 | -0.79 | -0.61 | -0.59 | -0.56 | -0.45 | -0.34 | ||||||||||
| Erythema | B# | 2.26 | 2.51 | 2.17 | 2.40 | 2.23 | 2.48 | 2.25 | 2.53 | 2.17 | 2.42 | 2.21 | 2.51 | 2.24 | 2.47 | 2.17 | 2.34 | 2.24 | 2.47 |
| C-12 | -0.49 | -0.65 | -0.44 | -0.66 | -0.40 | -0.62 | -0.49 | -0.82 | -0.57 | -0.82 | -0.42 | -0.78 | -0.42 | -0.46 | -0.38 | -0.44 | -0.37 | -0.47 | |
| C-24 | -0.52 | -0.44 | -0.41 | -0.55 | -0.52 | -0.39 | -0.43 | -0.34 | -0.33 |
4 Contraindications
Tazarotene cream is contraindicated in:
- Pregnancy. Retinoids may cause fetal harm when administered to a pregnant female [see Warnings and Precautions (5.1), Use in Specific Populations (8.1, 8.3)] .
- Individuals who have known hypersensitivity to any of its components [see Warnings and Precautions (5.2)].
6 Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of the labeling:
- Embryofetal toxicity [see Warnings and Precautions (5.1)]
- Photosensitivity and Risk of Sunburn [see Warnings and Precautions (5.3)]
7 Drug Interactions
No formal drug-drug interaction studies were conducted with tazarotene cream.
In a trial of 27 healthy female subjects between the ages of 20 to 55 years receiving a combination oral contraceptive tablet containing 1 mg norethindrone and 35 mcg ethinyl estradiol, concomitant use of tazarotene administered as 1.1 mg orally (mean ± SD C maxand AUC 0-24of tazarotenic acid were 28.9 ± 9.4 ng/mL and 120.6 ± 28.5 ng∙hr/mL) did not affect the pharmacokinetics of norethindrone and ethinyl estradiol over a complete cycle.
The impact of tazarotene on the pharmacokinetics of progestin only oral contraceptives (i.e., minipills) has not been evaluated.
1.1 Plaque Psoriasis
Tazarotene cream, 0.1% is indicated for the topical treatment of patients with plaque psoriasis.
12.3 Pharmacokinetics
Following topical application, tazarotene undergoes esterase hydrolysis to form its active metabolite, tazarotenic acid. Little parent compound could be detected in the plasma. Tazarotenic acid was highly bound to plasma proteins (greater than 99%). Tazarotene and tazarotenic acid were metabolized to sulfoxides, sulfones and other polar metabolites which were eliminated through urinary and fecal pathways. The half-life of tazarotenic acid was approximately 18 hours, following topical application of tazarotene to normal, acne or psoriatic skin.
In a multiple dose trial with a once daily dose for 14 consecutive days in 9 psoriatic subjects (male=5; female=4), measured doses of tazarotene cream, 0.1% were applied by medical staff to involved skin without occlusion (5 to 35% of total body surface area: mean ± SD: 14 ± 11%). The C maxof tazarotenic acid was 2.31 ± 2.78 ng/mL occurring 8 hours after the final dose, and the AUC 0-24hwas 31.2 ± 35.2 ng∙hr/mL on day 15 in the five subjects who were administered clinical doses of 2 mg cream/cm 2.
During clinical trials with tazarotene cream, 0.05% or 0.1% treatment for plaque psoriasis, three out of 139 subjects with their systemic exposure monitored had detectable plasma tazarotene concentrations, with the highest value at 0.09 ng/mL. Tazarotenic acid was detected in 78 out of 139 subjects (LLOQ = 0.05 ng/mL). Three subjects using tazarotene cream 0.1% had plasma tazarotenic acid concentrations greater than 1 ng/mL. The highest value was 2.4 ng/mL. However, because of the variations in the time of blood sampling, the area of psoriasis involvement, and the dose of tazarotene applied, actual maximal plasma levels are unknown.
Tazarotene cream 0.1% was applied once daily to either the face (N=8) or to 15% of body surface area (N=10) of female subjects with moderate to severe acne vulgaris. The mean C maxand AUC values of tazarotenic acid peaked at day 15 for both dosing groups during a 29 day treatment period. Mean C maxand AUC 0-24hvalues of tazarotenic acid from subjects in the 15% body surface area dosing group were more than 10 times higher than those from subjects in the face-only dosing group. The single highest C maxthroughout the trial period was 1.91 ng/mL on day 15 in the exaggerated dosing group. In the face-only group, the mean ± SD values of C maxand AUC 0-24hof tazarotenic acid on day 15 were 0.10 ± 0.06 ng/mL and 1.54 ± 1.01 ng∙hr/mL, respectively, whereas in the 15% body surface area dosing group, the mean ± SD values of C maxand AUC 0-24hof tazarotenic acid on day 15 were 1.20 ± 0.41 ng/mL and 17.01 ± 6.15 ng∙hr/mL, respectively. The steady state pharmacokinetics of tazarotenic acid had been reached by day 8 in the face-only and by day 15 in the 15% body surface area dosing groups.
In a Phase 3 clinical trial, tazarotene cream, 0.1% was applied once daily for 12 weeks to each of 48 subjects (22 females and 26 males) with facial acne vulgaris. The mean ± SD values of plasma tazarotenic acid at weeks 4 and 8 were 0.078 ± 0.073 ng/mL (N=47) and 0.052 ± 0.037 ng/mL (N=42), respectively. The highest observed individual plasma tazarotenic acid concentration was 0.41 ng/mL at week 4 from a female subject. The magnitude of plasma tazarotenic acid concentrations appears to be independent of gender, age, and body weight.
1 Indications and Usage
12.1 Mechanism of Action
Tazarotene is a retinoid prodrug which is converted to its active form, the carboxylic acid of tazarotene, by deesterification. Tazarotenic acid binds to all three members of the retinoic acid receptor (RAR) family: RARα, RARβ, and RARγ, but shows relative selectivity for RARβ, and RARγ and may modify gene expression. The clinical significance of these findings is unknown.
5.1 Embryofetal Toxicity
Systemic exposure to tazarotenic acid is dependent upon the extent of the body surface area treated. In patients treated topically over sufficient body surface area, exposure could be in the same order of magnitude as in orally treated animals. Although there may be less systemic exposure in the treatment of acne of the face alone due to less surface area for application, tazarotene is a teratogenic substance, and it is not known what level of exposure is required for teratogenicity in humans [see Clinical Pharmacology (12.3)] .
There were thirteen reported pregnancies in subjects who participated in the clinical trials for topical tazarotene. Nine of the subjects were found to have been treated with topical tazarotene, and the other four had been treated with vehicle. One of the subjects who was treated with tazarotene cream elected to terminate the pregnancy for non-medical reasons unrelated to treatment. The other eight pregnant women who were inadvertently exposed to topical tazarotene during clinical trials subsequently delivered apparently healthy babies. As the exact timing and extent of exposure in relation to the gestation times are not certain, the significance of these findings is unknown.
5 Warnings and Precautions
- Embryofetal Toxicity: Tazarotene cream contains tazarotene, which is a teratogenic substance. Tazarotene cream is contraindicated in pregnancy. Females of child-bearing potential should have a negative pregnancy test within 2 weeks prior to initiating treatment and use an effective method of contraception during treatment. ( 5.1)
- Local Irritation: Some individuals may experience excessive pruritus, burning, skin redness or peeling. If these effects occur, discontinue until the integrity of the skin has been restored, or reduce dosing interval, or in the case of psoriasis, may switch to the lower concentration. Tazarotene cream should not be used on eczematous skin, as it may cause severe irritation. ( 5.2)
- Photosensitivity and Risk for Sunburn: Avoid exposure to sunlight, sunlamps, and weather extremes. Wear sunscreen daily. Tazarotene cream should be administered with caution if the patient is also taking drugs known to be photosensitizers. ( 5.3)
2 Dosage and Administration
3 Dosage Forms and Strengths
Cream, 0.1%. Each gram of tazarotene cream, 0.1% contains 1 mg of tazarotene in a white cream base.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of tazarotene. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and subcutaneous tissue disorders:blister, dermatitis, urticaria, skin exfoliation, skin discoloration (including skin hyperpigmentation or skin hypopigmentation), swelling at or near application sites, and pain.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice. In human dermal safety trials, tazarotene cream, 0.05% and 0.1% did not induce allergic contact sensitization, phototoxicity, or photoallergy.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
Tazarotene cream is a white cream available in a concentration of 0.1%. It is supplied in a collapsible aluminum tube with a tamper-evident aluminum membrane over the opening and a white polypropylene screw cap, in 30 g and 60 g sizes.
Tazarotene Cream, 0.1%
30 gram NDC 51672-1373-2
60 gram NDC 51672-1373-3
2.1 Important Administration Instructions
Tazarotene cream is for topical use only. Tazarotene cream is not for ophthalmic, oral, or intravaginal use. If contact with mucous membranes occurs, rinse thoroughly with water [see Warnings and Precautions (5.2)] . Wash hands thoroughly after application.
5.3 Photosensitivity and Risk for Sunburn
Because of heightened burning susceptibility, exposure to sunlight (including sunlamps) should be avoided unless deemed medically necessary, and in such cases, exposure should be minimized during the use of tazarotene cream. Patients must be warned to use sunscreens and protective clothing when using tazarotene cream. Patients with sunburn should be advised not to use tazarotene cream until fully recovered. Patients who may have considerable sun exposure due to their occupation and those patients with inherent sensitivity to sunlight should exercise particular caution when using tazarotene cream.
Tazarotene cream should be administered with caution if the patient is also taking drugs known to be photosensitizers (e.g., thiazides, tetracyclines, fluoroquinolones, phenothiazines, sulfonamides) because of the increased possibility of augmented photosensitivity.
Principal Display Panel 30 G Tube Carton
NDC 51672-1373-2
Tazarotene
Cream 0.1%
FOR DERMATOLOGICAL USE ONLY.
NOT FOR OPHTHALMIC USE.
Keep this and all medications out of the reach of children.
30 g
Rx only
5.2 Local Irritation and Hypersensitivity Reactions
Local tolerability reactions (including blistering and skin desquamation) and hypersensitivity adverse reactions (including urticaria) have been observed with topical tazarotene. Application of tazarotene cream may cause excessive irritation in the skin of certain sensitive individuals. Some individuals may experience excessive pruritus, burning, skin redness or peeling. If these effects occur, the medication should either be discontinued until the integrity of the skin is restored, or the dosing should be reduced to an interval the patient can tolerate. However, efficacy at reduced frequency of application has not been established. Alternatively, patients with psoriasis who are being treated with the 0.1% concentration can be switched to the lower concentration. Frequency of application should be closely monitored by careful observation of the clinical therapeutic response and skin tolerance. Therapy can be resumed, or the drug concentration or frequency of application can be increased as the patient becomes able to tolerate treatment.
Concomitant topical medications and cosmetics that have a strong drying effect should be avoided. It is also advisable to "rest" a patient's skin until the effects of such preparations subside before use of tazarotene cream is begun.
Tazarotene cream should not be used on eczematous skin, as it may cause severe irritation.
Weather extremes, such as wind or cold, may be more irritating to patients using tazarotene cream.
Structured Label Content
Section 42229-5 (42229-5)
Females of Child-bearing Potential
Females of child-bearing potential should be warned of the potential risk and use adequate birth-control measures when tazarotene cream is used. The possibility that a female of child-bearing potential is pregnant at the time of institution of therapy should be considered.
A negative result for pregnancy test should be obtained within 2 weeks prior to tazarotene cream therapy. Tazarotene cream therapy should begin during a menstrual period [see Use in Specific Populations (8.1)] .
Section 42230-3 (42230-3)
| Patient Information
Tazarotene (taz-AR-oh-teen) Cream, 0.1% |
|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. |
| Revised: August 2025 5262058-0825-01 75 |
| Important information: Tazarotene cream is for use on skin only. Do not use tazarotene cream in your eyes, mouth, or vagina. |
|
What is the most important information I should know about tazarotene cream?
Tazarotene cream may cause birth defects if used during pregnancy.
|
What is tazarotene cream?
|
|
Who should not use tazarotene cream?
Do not use tazarotene cream if you:
|
|
What should I tell my doctor before using tazarotene cream?
Before you use tazarotene cream, tell your doctor about all of your medical conditions, including if you:
Certain medicines, vitamins, or supplements may make your skin more sensitive to sunlight. Also, tell your doctor about any cosmetics you use, including moisturizers, creams, lotions, or products that can dry out your skin. |
How should I use tazarotene cream?
|
Follow these instructions for applying tazarotene cream:
|
What should I avoid while using tazarotene cream?
|
|
What are the possible side effects of tazarotene cream?
Tazarotene cream may cause serious side effects, including:
The most common side effects of tazarotene cream in people with acne includepeeling, dry skin, redness and burning. These are not all the possible side effects of tazarotene cream. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
How should I store tazarotene cream?
|
|
General information about the safe and effective use of tazarotene cream.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use tazarotene cream for a condition for which it was not prescribed. Do not give tazarotene cream to other people, even if they have the same symptoms you have. It may harm them. You can ask your doctor or pharmacist for information about tazarotene cream that is written for health professionals. |
|
What are the ingredients in tazarotene cream?
Active ingredient: tazarotene Inactive ingredients:benzyl alcohol 1%, carbomer copolymer type B, carbomer homopolymer type B, edetate disodium, medium-chain triglycerides, mineral oil, purified water, sodium hydroxide, sodium thiosulfate, and sorbitan monooleate. |
| Manufactured by: Sun Pharma Canada Inc., Brampton, Ontario, Canada L6T 1C1
Distributed by: Sun Pharmaceutical Industries, Inc., Cranbury, NJ 08512 |
Section 44425-7 (44425-7)
Store at 20° to 25°C (68° to 77°F)[see USP Controlled Room Temperature].
2.3 Acne
Cleanse the face gently. After the skin is dry, apply a thin layer (2 mg/cm 2) of tazarotene cream 0.1% once per day, in the evening, to the skin areas where acne lesions appear. Use enough to cover the entire affected area.
Use effective sunscreens and wear protective clothing while using tazarotene cream [see Warnings and Precautions (5.3)].
10 Overdosage (10 OVERDOSAGE)
Excessive topical use of tazarotene cream, 0.1% may lead to marked redness, peeling, or discomfort [see Warnings and Precautions (5.2)] .
Tazarotene cream, 0.1% is not for oral use. Oral ingestion of the drug may lead to the same adverse effects as those associated with excessive oral intake of Vitamin A (hypervitaminosis A) or other retinoids. If oral ingestion occurs, the patient should be monitored, and appropriate supportive measures should be administered as necessary.
2.2 Psoriasis
It is recommended that treatment starts with tazarotene cream, 0.05%, with strength increased to 0.1% if tolerated and medically indicated. Apply a thin film (2 mg/cm 2) of tazarotene cream once per day, in the evening, to cover only the psoriatic lesions. If a bath or shower is taken prior to application, the skin should be dry before applying the cream. If emollients are used, they should be applied at least an hour before application of tazarotene cream. Because unaffected skin may be more susceptible to irritation, application of tazarotene cream to these areas should be carefully avoided.
11 Description (11 DESCRIPTION)
Tazarotene cream, 0.1% is for topical use and contains the active ingredient, tazarotene. Each gram of tazarotene cream, 0.1% contains 1 mg of tazarotene in a white cream base.
Tazarotene is a member of the acetylenic class of retinoids. Chemically, tazarotene is ethyl 6-[(4,4-dimethylthiochroman-6-yl)ethynyl]nicotinate. The compound has an empirical formula of C 21H 21NO 2S and molecular weight of 351.46. The structural formula is shown below:
Tazarotene cream contains the following inactive ingredients: benzyl alcohol 1%, carbomer copolymer type B, carbomer homopolymer type B, edetate disodium, medium-chain triglycerides, mineral oil, purified water, sodium hydroxide (to adjust pH), sodium thiosulfate, and sorbitan monooleate.
1.2 Acne Vulgaris
Tazarotene cream, 0.1% is also indicated for the topical treatment of patients with acne vulgaris.
8.4 Pediatric Use
The safety and efficacy of tazarotene cream have not been established in patients with psoriasis under the age of 18 years, or in patients with acne under the age of 12 years.
8.5 Geriatric Use
Tazarotene cream for the treatment of acne has not been clinically tested in persons 65 years of age or older.
Of the total number of subjects in clinical trials of tazarotene cream for plaque psoriasis, 120 were over the age of 65. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Currently there is no other clinical experience on the differences in responses between the elderly and younger subjects, but greater sensitivity of some older individuals cannot be ruled out.
14 Clinical Studies (14 CLINICAL STUDIES)
In two 12-week vehicle-controlled clinical trials, tazarotene cream, 0.05% and 0.1% was significantly more effective than vehicle in reducing the severity of stable plaque psoriasis. Tazarotene cream, 0.1% and 0.05% demonstrated superiority over vehicle cream as early as 1 week and 2 weeks, respectively, after starting treatment.
In these trials, the primary efficacy endpoint was "clinical success," defined as the proportion of subjects with none, minimal, or mild overall lesional assessment at Week 12, and shown in Table 1. "Clinical success" was also significantly greater with tazarotene cream, 0.05% and 0.1% versus vehicle at most follow-up visits.
| Tazarotene Cream, 0.05% | Tazarotene Cream, 0.1% | Vehicle Cream | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Trial 1
N=218 |
Trial 2
N=210 |
Trial 1
N=221 |
Trial 2
N=211 |
Trial 1
N=229 |
Trial 2
N=214 |
||||||||||
| Score | BL | Wk 12 | Wk 24 | BL | Wk 12 | BL | Wk 12 | Wk 24 | BL | Wk 12 | BL | Wk 12 | Wk 24 | BL | Wk 12 |
|
|||||||||||||||
| None
(0) |
0 | 1
(0.5%) |
1
(0.5%) |
0 | 2
(1%) |
0 | 0 | 0 | 0 | 6
(3%) |
0 | 0 | 1
(0.4%) |
0 | 1
(0.5%) |
| Minimal
(1) |
0 | 11
(5%) |
12
(6%) |
0 | 7
(3%) |
0 | 12
(5%) |
14
(6%) |
0 | 11
(5%) |
0 | 7
(3%) |
6
(3%) |
0 | 1
(0.5%) |
| Mild
(2) |
0 | 79
(36%) |
60
(28%) |
0 | 76
(36%) |
0 | 75
(34%) |
53
(24%) |
0 | 90
(43%) |
0 | 49
(21%) |
43
(19%) |
0 | 54
(25%) |
| Moderate
(3) |
141
(65%) |
86
(39%) |
90
(41%) |
100
(48%) |
74
(35%) |
122
(55%) |
97
(44%) |
107
(48%) |
96
(45%) |
62
(29%) |
139
(61%) |
119
(52%) |
114
(50%) |
97
(45%) |
99
(46%) |
| Severe
(4) |
69
(32%) |
39
(18%) |
51
(23%) |
80
(38%) |
36
(17%) |
91
(41%) |
36
(16%) |
46
(21%) |
86
(41%) |
29
(14%) |
81
(35%) |
51
(22%) |
61
(27%) |
93
(44%) |
47
(22%) |
| Very Severe
(5) |
8
(4%) |
2
(0.9%) |
4
(2%) |
30
(14%) |
15
(7%) |
8
(4%) |
1
(0.5%) |
1
(0.5%) |
29
(14%) |
13
(6%) |
9
(4%) |
3
(1%) |
4
(2%) |
24
(11%) |
12
(6%) |
| "Clinical Success" | 0 | 91
(42% Denotes statistically significant difference for "Clinical Success" compared with vehicle. )
|
73
(33% ) |
0 | 85
(40% ) |
0 | 87
(39% ) |
67
(30% ) |
0 | 107
(51% ) |
0 | 56
(24%) |
50
(22%) |
0 | 56
(26%) |
At the end of 12 weeks of treatment, tazarotene cream, 0.05% and 0.1% was consistently superior to vehicle in reducing the plaque thickness of psoriasis. Improvements in erythema and scaling were generally significantly greater with tazarotene cream, 0.05% and 0.1% than with vehicle. Tazarotene Cream, 0.1% was also generally more effective than tazarotene cream, 0.05% in reducing the severity of the individual signs of disease. However, tazarotene cream, 0.1% was associated with a greater degree of local irritation than tazarotene cream, 0.05%.
| Tazarotene Cream, 0.05% | Tazarotene Cream, 0.1% | Vehicle Cream | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lesion | Trunk/Arm/Leg lesions | Knee/Elbow lesions | All Treated | Trunk/Arm/Leg lesions | Knee/Elbow lesions | All Treated | Trunk/Arm/Leg lesions | Knee/Elbow lesions | All Treated | ||||||||||
| Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | Trial 1 | Trial 2 | ||
| N=218 | N=210 | N=218 | N=210 | N=218 | N=210 | N=221 | N=211 | N=221 | N=211 | N=221 | N=211 | N=229 | N=214 | N=229 | N=214 | N=229 | N=214 | ||
| Plaque elevation, scaling and erythema scored on a 0-4 scale with 0=none, 1=mild, 2=moderate, 3=severe and 4=very severe. | |||||||||||||||||||
| B#=Mean Baseline Severity; | |||||||||||||||||||
| C-12=Mean Change from Baseline at end of 12 weeks of therapy; | |||||||||||||||||||
| C-24=Mean Change from Baseline at week 24 (12 weeks after the end of therapy). | |||||||||||||||||||
| Plaque elevation | B# | 2.29 | 2.50 | 2.40 | 2.52 | 2.28 | 2.51 | 2.34 | 2.52 | 2.35 | 2.49 | 2.32 | 2.51 | 2.28 | 2.51 | 2.35 | 2.51 | 2.29 | 2.51 |
| C-12 | -0.83
Denotes statistically significant difference compared with vehicle.
|
-0.98 | -0.91 | -1.04 | -0.75 | -0.90 | -1.08 | -1.25 | -0.96 | -1.21 | -0.83 | -1.08 | -0.59 | -0.69 | -0.57 | -0.68 | -0.48 | -0.61 | |
| C-24 | -0.75 | -0.73 | -0.60 | -0.87 | -0.73 | -0.63 | -0.57 | -0.49 | -0.42 | ||||||||||
| Scaling | B# | 2.26 | 2.45 | 2.47 | 2.60 | 2.32 | 2.47 | 2.37 | 2.45 | 2.40 | 2.57 | 2.36 | 2.53 | 2.34 | 2.46 | 2.45 | 2.61 | 2.31 | 2.53 |
| C-12 | -0.75 | -0.90 | -0.78 | -0.98 | -0.67 | -0.80 | -0.84 | -1.06 | -0.76 | -1.13 | -0.73 | -1.03 | -0.66 | -0.79 | -0.62 | -0.76 | -0.46 | -0.70 | |
| C-24 | -0.68 | -0.62 | -0.51 | -0.79 | -0.61 | -0.59 | -0.56 | -0.45 | -0.34 | ||||||||||
| Erythema | B# | 2.26 | 2.51 | 2.17 | 2.40 | 2.23 | 2.48 | 2.25 | 2.53 | 2.17 | 2.42 | 2.21 | 2.51 | 2.24 | 2.47 | 2.17 | 2.34 | 2.24 | 2.47 |
| C-12 | -0.49 | -0.65 | -0.44 | -0.66 | -0.40 | -0.62 | -0.49 | -0.82 | -0.57 | -0.82 | -0.42 | -0.78 | -0.42 | -0.46 | -0.38 | -0.44 | -0.37 | -0.47 | |
| C-24 | -0.52 | -0.44 | -0.41 | -0.55 | -0.52 | -0.39 | -0.43 | -0.34 | -0.33 |
4 Contraindications (4 CONTRAINDICATIONS)
Tazarotene cream is contraindicated in:
- Pregnancy. Retinoids may cause fetal harm when administered to a pregnant female [see Warnings and Precautions (5.1), Use in Specific Populations (8.1, 8.3)] .
- Individuals who have known hypersensitivity to any of its components [see Warnings and Precautions (5.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in more detail in other sections of the labeling:
- Embryofetal toxicity [see Warnings and Precautions (5.1)]
- Photosensitivity and Risk of Sunburn [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
No formal drug-drug interaction studies were conducted with tazarotene cream.
In a trial of 27 healthy female subjects between the ages of 20 to 55 years receiving a combination oral contraceptive tablet containing 1 mg norethindrone and 35 mcg ethinyl estradiol, concomitant use of tazarotene administered as 1.1 mg orally (mean ± SD C maxand AUC 0-24of tazarotenic acid were 28.9 ± 9.4 ng/mL and 120.6 ± 28.5 ng∙hr/mL) did not affect the pharmacokinetics of norethindrone and ethinyl estradiol over a complete cycle.
The impact of tazarotene on the pharmacokinetics of progestin only oral contraceptives (i.e., minipills) has not been evaluated.
1.1 Plaque Psoriasis
Tazarotene cream, 0.1% is indicated for the topical treatment of patients with plaque psoriasis.
12.3 Pharmacokinetics
Following topical application, tazarotene undergoes esterase hydrolysis to form its active metabolite, tazarotenic acid. Little parent compound could be detected in the plasma. Tazarotenic acid was highly bound to plasma proteins (greater than 99%). Tazarotene and tazarotenic acid were metabolized to sulfoxides, sulfones and other polar metabolites which were eliminated through urinary and fecal pathways. The half-life of tazarotenic acid was approximately 18 hours, following topical application of tazarotene to normal, acne or psoriatic skin.
In a multiple dose trial with a once daily dose for 14 consecutive days in 9 psoriatic subjects (male=5; female=4), measured doses of tazarotene cream, 0.1% were applied by medical staff to involved skin without occlusion (5 to 35% of total body surface area: mean ± SD: 14 ± 11%). The C maxof tazarotenic acid was 2.31 ± 2.78 ng/mL occurring 8 hours after the final dose, and the AUC 0-24hwas 31.2 ± 35.2 ng∙hr/mL on day 15 in the five subjects who were administered clinical doses of 2 mg cream/cm 2.
During clinical trials with tazarotene cream, 0.05% or 0.1% treatment for plaque psoriasis, three out of 139 subjects with their systemic exposure monitored had detectable plasma tazarotene concentrations, with the highest value at 0.09 ng/mL. Tazarotenic acid was detected in 78 out of 139 subjects (LLOQ = 0.05 ng/mL). Three subjects using tazarotene cream 0.1% had plasma tazarotenic acid concentrations greater than 1 ng/mL. The highest value was 2.4 ng/mL. However, because of the variations in the time of blood sampling, the area of psoriasis involvement, and the dose of tazarotene applied, actual maximal plasma levels are unknown.
Tazarotene cream 0.1% was applied once daily to either the face (N=8) or to 15% of body surface area (N=10) of female subjects with moderate to severe acne vulgaris. The mean C maxand AUC values of tazarotenic acid peaked at day 15 for both dosing groups during a 29 day treatment period. Mean C maxand AUC 0-24hvalues of tazarotenic acid from subjects in the 15% body surface area dosing group were more than 10 times higher than those from subjects in the face-only dosing group. The single highest C maxthroughout the trial period was 1.91 ng/mL on day 15 in the exaggerated dosing group. In the face-only group, the mean ± SD values of C maxand AUC 0-24hof tazarotenic acid on day 15 were 0.10 ± 0.06 ng/mL and 1.54 ± 1.01 ng∙hr/mL, respectively, whereas in the 15% body surface area dosing group, the mean ± SD values of C maxand AUC 0-24hof tazarotenic acid on day 15 were 1.20 ± 0.41 ng/mL and 17.01 ± 6.15 ng∙hr/mL, respectively. The steady state pharmacokinetics of tazarotenic acid had been reached by day 8 in the face-only and by day 15 in the 15% body surface area dosing groups.
In a Phase 3 clinical trial, tazarotene cream, 0.1% was applied once daily for 12 weeks to each of 48 subjects (22 females and 26 males) with facial acne vulgaris. The mean ± SD values of plasma tazarotenic acid at weeks 4 and 8 were 0.078 ± 0.073 ng/mL (N=47) and 0.052 ± 0.037 ng/mL (N=42), respectively. The highest observed individual plasma tazarotenic acid concentration was 0.41 ng/mL at week 4 from a female subject. The magnitude of plasma tazarotenic acid concentrations appears to be independent of gender, age, and body weight.
1 Indications and Usage (1 INDICATIONS AND USAGE)
12.1 Mechanism of Action
Tazarotene is a retinoid prodrug which is converted to its active form, the carboxylic acid of tazarotene, by deesterification. Tazarotenic acid binds to all three members of the retinoic acid receptor (RAR) family: RARα, RARβ, and RARγ, but shows relative selectivity for RARβ, and RARγ and may modify gene expression. The clinical significance of these findings is unknown.
5.1 Embryofetal Toxicity
Systemic exposure to tazarotenic acid is dependent upon the extent of the body surface area treated. In patients treated topically over sufficient body surface area, exposure could be in the same order of magnitude as in orally treated animals. Although there may be less systemic exposure in the treatment of acne of the face alone due to less surface area for application, tazarotene is a teratogenic substance, and it is not known what level of exposure is required for teratogenicity in humans [see Clinical Pharmacology (12.3)] .
There were thirteen reported pregnancies in subjects who participated in the clinical trials for topical tazarotene. Nine of the subjects were found to have been treated with topical tazarotene, and the other four had been treated with vehicle. One of the subjects who was treated with tazarotene cream elected to terminate the pregnancy for non-medical reasons unrelated to treatment. The other eight pregnant women who were inadvertently exposed to topical tazarotene during clinical trials subsequently delivered apparently healthy babies. As the exact timing and extent of exposure in relation to the gestation times are not certain, the significance of these findings is unknown.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Embryofetal Toxicity: Tazarotene cream contains tazarotene, which is a teratogenic substance. Tazarotene cream is contraindicated in pregnancy. Females of child-bearing potential should have a negative pregnancy test within 2 weeks prior to initiating treatment and use an effective method of contraception during treatment. ( 5.1)
- Local Irritation: Some individuals may experience excessive pruritus, burning, skin redness or peeling. If these effects occur, discontinue until the integrity of the skin has been restored, or reduce dosing interval, or in the case of psoriasis, may switch to the lower concentration. Tazarotene cream should not be used on eczematous skin, as it may cause severe irritation. ( 5.2)
- Photosensitivity and Risk for Sunburn: Avoid exposure to sunlight, sunlamps, and weather extremes. Wear sunscreen daily. Tazarotene cream should be administered with caution if the patient is also taking drugs known to be photosensitizers. ( 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Cream, 0.1%. Each gram of tazarotene cream, 0.1% contains 1 mg of tazarotene in a white cream base.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of tazarotene. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and subcutaneous tissue disorders:blister, dermatitis, urticaria, skin exfoliation, skin discoloration (including skin hyperpigmentation or skin hypopigmentation), swelling at or near application sites, and pain.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice. In human dermal safety trials, tazarotene cream, 0.05% and 0.1% did not induce allergic contact sensitization, phototoxicity, or photoallergy.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Tazarotene cream is a white cream available in a concentration of 0.1%. It is supplied in a collapsible aluminum tube with a tamper-evident aluminum membrane over the opening and a white polypropylene screw cap, in 30 g and 60 g sizes.
Tazarotene Cream, 0.1%
30 gram NDC 51672-1373-2
60 gram NDC 51672-1373-3
2.1 Important Administration Instructions
Tazarotene cream is for topical use only. Tazarotene cream is not for ophthalmic, oral, or intravaginal use. If contact with mucous membranes occurs, rinse thoroughly with water [see Warnings and Precautions (5.2)] . Wash hands thoroughly after application.
5.3 Photosensitivity and Risk for Sunburn
Because of heightened burning susceptibility, exposure to sunlight (including sunlamps) should be avoided unless deemed medically necessary, and in such cases, exposure should be minimized during the use of tazarotene cream. Patients must be warned to use sunscreens and protective clothing when using tazarotene cream. Patients with sunburn should be advised not to use tazarotene cream until fully recovered. Patients who may have considerable sun exposure due to their occupation and those patients with inherent sensitivity to sunlight should exercise particular caution when using tazarotene cream.
Tazarotene cream should be administered with caution if the patient is also taking drugs known to be photosensitizers (e.g., thiazides, tetracyclines, fluoroquinolones, phenothiazines, sulfonamides) because of the increased possibility of augmented photosensitivity.
Principal Display Panel 30 G Tube Carton (PRINCIPAL DISPLAY PANEL - 30 g Tube Carton)
NDC 51672-1373-2
Tazarotene
Cream 0.1%
FOR DERMATOLOGICAL USE ONLY.
NOT FOR OPHTHALMIC USE.
Keep this and all medications out of the reach of children.
30 g
Rx only
5.2 Local Irritation and Hypersensitivity Reactions
Local tolerability reactions (including blistering and skin desquamation) and hypersensitivity adverse reactions (including urticaria) have been observed with topical tazarotene. Application of tazarotene cream may cause excessive irritation in the skin of certain sensitive individuals. Some individuals may experience excessive pruritus, burning, skin redness or peeling. If these effects occur, the medication should either be discontinued until the integrity of the skin is restored, or the dosing should be reduced to an interval the patient can tolerate. However, efficacy at reduced frequency of application has not been established. Alternatively, patients with psoriasis who are being treated with the 0.1% concentration can be switched to the lower concentration. Frequency of application should be closely monitored by careful observation of the clinical therapeutic response and skin tolerance. Therapy can be resumed, or the drug concentration or frequency of application can be increased as the patient becomes able to tolerate treatment.
Concomitant topical medications and cosmetics that have a strong drying effect should be avoided. It is also advisable to "rest" a patient's skin until the effects of such preparations subside before use of tazarotene cream is begun.
Tazarotene cream should not be used on eczematous skin, as it may cause severe irritation.
Weather extremes, such as wind or cold, may be more irritating to patients using tazarotene cream.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:28.280294 · Updated: 2026-03-14T22:40:31.369897