These Highlights Do Not Include All The Information Needed To Use Acetadote Safely And Effectively. See Full Prescribing Information For Acetadote.
472f158a-5ab9-4308-8e49-1116e6ea3d39
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 ) 11/2024
Indications and Usage
ACETADOTE is indicated to prevent or lessen hepatic injury after ingestion of a potentially hepatotoxic quantity of acetaminophen in adults and pediatric patients who weigh 5 kg or greater with acute ingestion or from repeated supratherapeutic ingestion (RSI).
Dosage and Administration
Pre-Treatment Assessment Following Acute Ingestion ( 2.1 ) : Prior to initiating treatment with ACETADOTE, decide whether the three-bag or two-bag regimen will be used. Obtain a plasma or serum sample to assay for acetaminophen concentration at least 4 hours after ingestion. If the time of acetaminophen ingestion is unknown: Administer a loading dose of ACETADOTE immediately. Obtain an acetaminophen concentration to determine need for continued treatment. If the acetaminophen concentration cannot be obtained (or is unavailable or uninterpretable) within the 8-hour time interval after acetaminophen ingestion or there is clinical evidence of acetaminophen toxicity: Administer a loading dose of ACETADOTE immediately and continue treatment for a total of two doses over 20 hours or three doses over 21 hours ( 2.5 ). If the patient presents more than 8 hours after ingestion and the time of acute acetaminophen ingestion is known: Administer a loading dose of ACETADOTE immediately Obtain acetaminophen concentration to determine need for continued treatment If the patient presents less than 8 hours after ingestion and the time of acute acetaminophen ingestion is known and the acetaminophen concentration is known: Use the revised Rumack-Matthew nomogram ( Figure 1 ) to determine whether or not to initiate treatment with ACETADOTE ( 2.2 ) Nomogram for Estimating Potential for Hepatotoxicity from Acute Acetaminophen Ingestion ( 2.2 ): See Full Prescribing Information for instructions on how to use the nomogram to determine the need for dosing. Preparation and Storage of Diluted Solution Prior to Administration ( 2.3 ) : Calculate the dose (mg) based on the patient's weight in kg; multiple vials of ACETADOTE may be required. ACETADOTE is hyperosmolar (2,600 mOsmol/L), therefore ACETADOTE must be diluted in the recommended volume of sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water injection prior to intravenous administration. In general, 0.45% normal saline is the preferred diluent because it provides a more consistent osmolarity profile, reduces the amount of free water delivered to the patient, and better approximates physiologic fluids. See Full Prescribing Information for examples of osmolarity depending on the type of solution and ACETADOTE concentration. General Considerations for Selecting the Three-Bag or Two-Bag Regimen ( 2.4 ): It is not known whether the two-bag regimen is comparable to the three-bag regimen in preventing hepatotoxicity. Patients 40 kg or less should receive the three-bag regimen. For patients weighing 41 kg or greater, the three-bag regimen may be preferred for those with early signs of severe liver injury or a large acetaminophen ingestion. Recommended Dosage for Acute Acetaminophen Ingestion ( 2.5 ): ACETADOTE is for intravenous administration only Total dosage of ACETADOTE is 300 mg/kg given intravenously as either: 3 separate doses infused over a total of 21 hours OR 2 separate doses infused over a total of 20 hours. See Full Prescribing Information for weight-based dosage and weight-based dilution ( 2.5 ) See Full Prescribing Information for recommendations for continuing ACETADOTE treatment after 21 hours ( 2.2 ). Repeated Supratherapeutic Acetaminophen Ingestion ( 2.6 ): Obtain acetaminophen concentration and other laboratory tests to guide treatment; revised Rumack-Matthew nomogram does not apply.
Warnings and Precautions
Hypersensitivity Reactions: Fatal or life-threatening anaphylaxis, rash, hypotension, wheezing, shortness of breath, and/or bronchospasm have been observed. Observe patients during and after the infusion; immediately discontinue infusion if a serious reaction occurs and initiate appropriate treatment. ACETADOTE infusion may be carefully restarted after treatment of hypersensitivity has been initiated and acute symptoms have resolved ( 5.1 ). Fluid Overload: Total volume administered should be reduced for patients weighing less than 40 kg and for those requiring fluid restriction ( 5.2 ).
Contraindications
ACETADOTE is contraindicated in patients with a previous hypersensitivity reaction to acetylcysteine [see Warnings and Precautions ( 5.1 )] .
Adverse Reactions
Most common adverse reactions (> 2%) are rash, urticaria/facial flushing and pruritus ( 6.1 ). To report SUSPECTED ADVERSE REACTIONS, contact Cumberland Pharmaceuticals Inc. at 1-877-484-2700 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Storage and Handling
ACETADOTE (acetylcysteine) injection is available as a 20% solution (200 mg/mL) in 30 mL single-dose glass vials. Each single dose vial contains 6 g/30 mL (200 mg/mL) of ACETADOTE injection. ACETADOTE is sterile and can be used for intravenous administration. It is available as follows: 30 mL vials, carton of 4 (NDC 66220-207-30) Do not use previously opened vials for intravenous administration. Note: The color of ACETADOTE may turn from essentially colorless to a slight pink or purple once the stopper is punctured. The color change does not affect the quality of the product. The stopper in the ACETADOTE vial is formulated with a synthetic base-polymer and does not contain natural rubber latex, dry natural rubber, or blends of natural rubber. Store unopened vials at controlled room temperature, 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]
How Supplied
ACETADOTE (acetylcysteine) injection is available as a 20% solution (200 mg/mL) in 30 mL single-dose glass vials. Each single dose vial contains 6 g/30 mL (200 mg/mL) of ACETADOTE injection. ACETADOTE is sterile and can be used for intravenous administration. It is available as follows: 30 mL vials, carton of 4 (NDC 66220-207-30) Do not use previously opened vials for intravenous administration. Note: The color of ACETADOTE may turn from essentially colorless to a slight pink or purple once the stopper is punctured. The color change does not affect the quality of the product. The stopper in the ACETADOTE vial is formulated with a synthetic base-polymer and does not contain natural rubber latex, dry natural rubber, or blends of natural rubber. Store unopened vials at controlled room temperature, 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]
Medication Information
Warnings and Precautions
Hypersensitivity Reactions: Fatal or life-threatening anaphylaxis, rash, hypotension, wheezing, shortness of breath, and/or bronchospasm have been observed. Observe patients during and after the infusion; immediately discontinue infusion if a serious reaction occurs and initiate appropriate treatment. ACETADOTE infusion may be carefully restarted after treatment of hypersensitivity has been initiated and acute symptoms have resolved ( 5.1 ). Fluid Overload: Total volume administered should be reduced for patients weighing less than 40 kg and for those requiring fluid restriction ( 5.2 ).
Indications and Usage
ACETADOTE is indicated to prevent or lessen hepatic injury after ingestion of a potentially hepatotoxic quantity of acetaminophen in adults and pediatric patients who weigh 5 kg or greater with acute ingestion or from repeated supratherapeutic ingestion (RSI).
Dosage and Administration
Pre-Treatment Assessment Following Acute Ingestion ( 2.1 ) : Prior to initiating treatment with ACETADOTE, decide whether the three-bag or two-bag regimen will be used. Obtain a plasma or serum sample to assay for acetaminophen concentration at least 4 hours after ingestion. If the time of acetaminophen ingestion is unknown: Administer a loading dose of ACETADOTE immediately. Obtain an acetaminophen concentration to determine need for continued treatment. If the acetaminophen concentration cannot be obtained (or is unavailable or uninterpretable) within the 8-hour time interval after acetaminophen ingestion or there is clinical evidence of acetaminophen toxicity: Administer a loading dose of ACETADOTE immediately and continue treatment for a total of two doses over 20 hours or three doses over 21 hours ( 2.5 ). If the patient presents more than 8 hours after ingestion and the time of acute acetaminophen ingestion is known: Administer a loading dose of ACETADOTE immediately Obtain acetaminophen concentration to determine need for continued treatment If the patient presents less than 8 hours after ingestion and the time of acute acetaminophen ingestion is known and the acetaminophen concentration is known: Use the revised Rumack-Matthew nomogram ( Figure 1 ) to determine whether or not to initiate treatment with ACETADOTE ( 2.2 ) Nomogram for Estimating Potential for Hepatotoxicity from Acute Acetaminophen Ingestion ( 2.2 ): See Full Prescribing Information for instructions on how to use the nomogram to determine the need for dosing. Preparation and Storage of Diluted Solution Prior to Administration ( 2.3 ) : Calculate the dose (mg) based on the patient's weight in kg; multiple vials of ACETADOTE may be required. ACETADOTE is hyperosmolar (2,600 mOsmol/L), therefore ACETADOTE must be diluted in the recommended volume of sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water injection prior to intravenous administration. In general, 0.45% normal saline is the preferred diluent because it provides a more consistent osmolarity profile, reduces the amount of free water delivered to the patient, and better approximates physiologic fluids. See Full Prescribing Information for examples of osmolarity depending on the type of solution and ACETADOTE concentration. General Considerations for Selecting the Three-Bag or Two-Bag Regimen ( 2.4 ): It is not known whether the two-bag regimen is comparable to the three-bag regimen in preventing hepatotoxicity. Patients 40 kg or less should receive the three-bag regimen. For patients weighing 41 kg or greater, the three-bag regimen may be preferred for those with early signs of severe liver injury or a large acetaminophen ingestion. Recommended Dosage for Acute Acetaminophen Ingestion ( 2.5 ): ACETADOTE is for intravenous administration only Total dosage of ACETADOTE is 300 mg/kg given intravenously as either: 3 separate doses infused over a total of 21 hours OR 2 separate doses infused over a total of 20 hours. See Full Prescribing Information for weight-based dosage and weight-based dilution ( 2.5 ) See Full Prescribing Information for recommendations for continuing ACETADOTE treatment after 21 hours ( 2.2 ). Repeated Supratherapeutic Acetaminophen Ingestion ( 2.6 ): Obtain acetaminophen concentration and other laboratory tests to guide treatment; revised Rumack-Matthew nomogram does not apply.
Contraindications
ACETADOTE is contraindicated in patients with a previous hypersensitivity reaction to acetylcysteine [see Warnings and Precautions ( 5.1 )] .
Adverse Reactions
Most common adverse reactions (> 2%) are rash, urticaria/facial flushing and pruritus ( 6.1 ). To report SUSPECTED ADVERSE REACTIONS, contact Cumberland Pharmaceuticals Inc. at 1-877-484-2700 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Storage and Handling
ACETADOTE (acetylcysteine) injection is available as a 20% solution (200 mg/mL) in 30 mL single-dose glass vials. Each single dose vial contains 6 g/30 mL (200 mg/mL) of ACETADOTE injection. ACETADOTE is sterile and can be used for intravenous administration. It is available as follows: 30 mL vials, carton of 4 (NDC 66220-207-30) Do not use previously opened vials for intravenous administration. Note: The color of ACETADOTE may turn from essentially colorless to a slight pink or purple once the stopper is punctured. The color change does not affect the quality of the product. The stopper in the ACETADOTE vial is formulated with a synthetic base-polymer and does not contain natural rubber latex, dry natural rubber, or blends of natural rubber. Store unopened vials at controlled room temperature, 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]
How Supplied
ACETADOTE (acetylcysteine) injection is available as a 20% solution (200 mg/mL) in 30 mL single-dose glass vials. Each single dose vial contains 6 g/30 mL (200 mg/mL) of ACETADOTE injection. ACETADOTE is sterile and can be used for intravenous administration. It is available as follows: 30 mL vials, carton of 4 (NDC 66220-207-30) Do not use previously opened vials for intravenous administration. Note: The color of ACETADOTE may turn from essentially colorless to a slight pink or purple once the stopper is punctured. The color change does not affect the quality of the product. The stopper in the ACETADOTE vial is formulated with a synthetic base-polymer and does not contain natural rubber latex, dry natural rubber, or blends of natural rubber. Store unopened vials at controlled room temperature, 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]
Description
Dosage and Administration ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 ) 11/2024
Section 42229-5
Loading dose
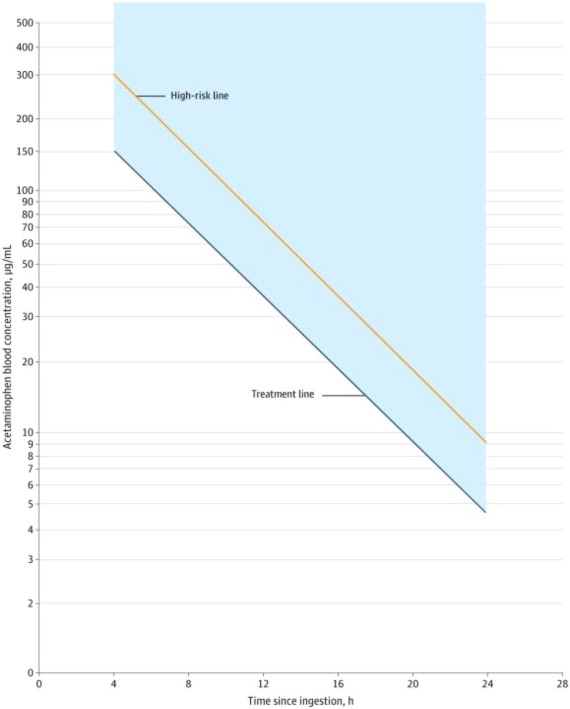
For patients whose acetaminophen concentrations are at or above the treatment line (see Figure 1):
- Administer a loading dose of ACETADOTE [see Dosage and Administration (2.5)].
For patients with an acute overdose from an extended-release acetaminophen, if the acetaminophen concentration at 4 hours post ingestion is below the treatment line (see Figure 1) then obtain a second sample for acetaminophen concentration 8 to 10 hours after the acute ingestion. If the second value is at or above the treatment line:
- Administer a loading dose of ACETADOTE [see Dosage and Administration (2.5)].
For patients whose values are below the treatment line (see Figure 1), but time of ingestion was unknown or sample was obtained less than 4 hours after ingestion:
- Administer a loading dose of ACETADOTE [see Dosage and Administration (2.5)].
For patients whose values are below the treatment line (see Figure 1) and time of ingestion is known and the sample was obtained more than 4 hours after ingestion, do not administer ACETADOTE because there is minimal risk of hepatotoxicity.
Figure 1. Revised Rumack-Matthew Nomogram for Estimating Risk of Hepatoxicity After Acute Ingestion of Acetaminophen
Section 44425-7
Refer to Table 2 and Table 3 to calculate the dose (mg) based on the patient's weight in kg; multiple vials of ACETADOTE may be required [see Dosage and Administration (2.5)]. Discard any unused portion left in the vial.
Because ACETADOTE is hyperosmolar (2,600 mOsmol/L), ACETADOTE must be diluted in the recommended volume of sterile water for injection, 0.45% sodium chloride injection (1/2 normal saline), or 5% dextrose in water prior to intravenous administration. The total injection volume will vary based on the patient's weight and chosen dosage regimen (i.e., three-bag or two-bag) [see Dosage and Administration (2.5), Warnings and Precautions (5.2)].
The choice of diluent should be based on the individual patient's clinical status, concurrent medical conditions, and institutional protocols. The treating clinician should assess each case individually and consult with their pharmacy if there are any concerns about the appropriate diluent choice. In general, 0.45% normal saline is the preferred diluent because it provides a more consistent osmolarity profile, reduces the amount of free water delivered to the patient, and better approximates physiologic fluids than 5% dextrose in water or sterile water for injection. However, consider 5% dextrose in water or sterile water for injection if sodium load is a concern for the patient.
Dilution of ACETADOTE in each of these three solutions results in different osmolarity of the acetylcysteine solution for intravenous administration (see Table 1 for examples of different osmolarity of the solution depending on the type of solution and the ACETADOTE concentration).
|
* Adjust osmolarity to a physiologically safe level (generally not less than 150 mOsmol/L in pediatric patients). |
|||
| Acetylcysteine Concentration | Osmolarity | ||
| Sterile Water for Injection | 0.45% Sodium Chloride Injection | 5% Dextrose in Water (D5W) |
|
| 4 mg/mL (lowest concentration 3-bag protocol) | 52 mOsmol/L* | 194 mOsmol/L | 311 mOsmol/L |
| 54.5 mg/mL (highest concentration 3-bag protocol) | 744 mOsmol/L | 855 mOsmol/L | 957 mOsmol/L |
| 7.9 mg/mL (lowest concentration 2-bag protocol) | 105 mOsmol/L | 241 mOsmol/L | 360 mOsmol/L |
| 18.2 mg/mL (highest concentration 2-bag protocol) | 239 mOsmol/L | 368 mOsmol/L | 487 mOsmol/L |
The choice of diluent should be based on the individual patient's clinical status, concurrent medical conditions, and institutional protocols. The treating clinician should assess each case individually and consult with their pharmacy if there are any concerns about the appropriate diluent choice. In general, 0.45% normal saline is the preferred diluent, as it provides a more consistent osmolarity profile, reduces the amount of free water delivered to the patient, and better approximates physiologic fluids than 5% dextrose in water or sterile water for injection.
Visually inspect for particular matter and discoloration prior to administration. The color of the diluted solution ranges from colorless to a slight pink or purple once the stopper is punctured (the color change does not affect the quality of the product). The diluted solution can be stored for 24 hours at room temperature. Discard the unused portion. If a vial was previously opened, do not use for intravenous administration.
Section 51945-4
PRINCIPAL DISPLAY PANEL – Vial Label
30 mL NDC 66220-207-30
Sterile
Acetadote
®
(acetylcysteine) Injection
6 g/30 mL
(200 mg/mL)
MUST BE FURTHER DILUTED
PRIOR TO INTRAVENOUS USE
CUMBERLAND ®
PHARMACEUTICALS
10 Overdosage
Fatal and life-threatening adverse events have been reported following acetylcysteine overdosage, including anaphylaxis, cerebral edema, and hemolytic-uremic syndrome (HUS). Stop acetylcysteine administration in the setting of suspected acetylcysteine overdosage and manage as clinically indicated.
Anaphylaxis, including cases with a fatal outcome, has been reported following acetylcysteine overdosage. Patients who experienced anaphylaxis following acetylcysteine overdosage often became symptomatic during the loading dose and experienced hypotension, rash, angioedema, bronchospasm, or respiratory distress. Cases of anaphylaxis with acetylcysteine overdosage also described coagulopathy, renal failure, or respiratory failure.
Cerebral edema, including cases with a fatal outcome, has been reported following acetylcysteine overdosage. Patients who experienced cerebral edema following acetylcysteine overdose presented with altered mental status, abnormal pupillary responses, seizures. Some cases of cerebral edema with acetylcysteine overdosage described brain herniation.
HUS has been reported following acetylcysteine overdosage. Patients who experienced HUS presented with microangiopathic hemolytic anemia, thrombocytopenia, and acute kidney injury.
Removal of acetylcysteine via hemodialysis has been reported in the literature outside of the context of acetylcysteine overdosage. Studies of hemodialysis in acetaminophen overdose report significant extracorporeal removal of acetylcysteine during hemodialysis. Contact the Poison Center (1-800-222-1222) for overdosage management recommendations for ACETADOTE including considerations for hemodialysis.
11 Description
Acetylcysteine injection is an intravenous antidote for the treatment of acetaminophen overdose. Acetylcysteine is the nonproprietary name for the N-acetyl derivative of the naturally occurring amino acid, L-cysteine (N-acetyl-L-cysteine,). The compound is a white crystalline powder, which melts in the range of 104° to 110°C and has a very slight odor.
The molecular formula of the compound is C5H9NO3S, and its molecular weight is 163.2. Acetylcysteine has the following structural formula:
ACETADOTE is supplied as a sterile solution in vials containing 20% w/v (200 mg/mL) acetylcysteine. The pH of the solution ranges from 6.0 to 7.5. ACETADOTE contains the following inactive ingredients: sodium hydroxide (used for pH adjustment), and Water for Injection, USP.
The amount of sodium in ACETADOTE is approximately 30 mg/mL. Because ACETADOTE is administered based on a patient's weight, the amount of sodium administered in a course of treatment will vary from approximately 225 mg to 4500 mg. The use of ½ normal saline will contribute approximately an additional 1770 mg of sodium per liter of diluent.
8.4 Pediatric Use
Safety and effectiveness of ACETADOTE in pediatric patients have not been established by adequate and well-controlled studies. Use of ACETADOTE in pediatric patients 5 kg and greater is based on clinical practice [see Dosage and Administration (2.5)].
5.2 Fluid Overload
The total volume of ACETADOTE administered should be adjusted for patients less than 40 kg and for those requiring fluid restriction.
Intravenous administration of ACETADOTE can cause fluid overload, potentially resulting in hyponatremia, seizure, and death. To avoid fluid overload, use the recommended dilutions [see Dosage and Administration (2.5)].
4 Contraindications
ACETADOTE is contraindicated in patients with a previous hypersensitivity reaction to acetylcysteine [see Warnings and Precautions (5.1)].
6 Adverse Reactions
Most common adverse reactions (> 2%) are rash, urticaria/facial flushing
and pruritus (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Cumberland Pharmaceuticals Inc. at 1-877-484-2700 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
12.3 Pharmacokinetics
After a single intravenous dose of acetylcysteine, the plasma concentration of total acetylcysteine declined in a poly-exponential decay manner with a mean terminal half-life (T1/2) of 5.6 hours. The mean clearance (CL) for acetylcysteine was 0.11 liter/hr/kg and renal CL constituted about 30% of the total CL.
1 Indications and Usage
ACETADOTE is indicated to prevent or lessen hepatic injury after ingestion of a potentially hepatotoxic quantity of acetaminophen in adults and pediatric patients who weigh 5 kg or greater with acute ingestion or from repeated supratherapeutic ingestion (RSI).
12.1 Mechanism of Action
Acetylcysteine has been shown to reduce the extent of liver injury following acetaminophen overdose. Acetaminophen doses of 150 mg/kg or greater have been associated with hepatotoxicity. Acetylcysteine probably protects the liver by maintaining or restoring the glutathione levels, or by acting as an alternate substrate for conjugation with, and thus detoxification of, the reactive metabolite of acetaminophen.
5 Warnings and Precautions
- Hypersensitivity Reactions: Fatal or life-threatening anaphylaxis, rash, hypotension, wheezing, shortness of breath, and/or bronchospasm have been observed. Observe patients during and after the infusion; immediately discontinue infusion if a serious reaction occurs and initiate appropriate treatment. ACETADOTE infusion may be carefully restarted after treatment of hypersensitivity has been initiated and acute symptoms have resolved (5.1).
- Fluid Overload: Total volume administered should be reduced for patients weighing less than 40 kg and for those requiring fluid restriction (5.2).
2 Dosage and Administration
Pre-Treatment Assessment Following Acute Ingestion (2.1):
Prior to initiating treatment with ACETADOTE, decide whether the three-bag or two-bag regimen will be used.
Obtain a plasma or serum sample to assay for acetaminophen concentration at least 4 hours after ingestion.
- If the time of acetaminophen ingestion is unknown:
- Administer a loading dose of ACETADOTE immediately.
- Obtain an acetaminophen concentration to determine need for continued treatment.
- If the acetaminophen concentration cannot be obtained (or is unavailable or uninterpretable) within the 8-hour time interval after acetaminophen ingestion or there is clinical evidence of acetaminophen toxicity:
- Administer a loading dose of ACETADOTE immediately and continue treatment for a total of two doses over 20 hours or three doses over 21 hours (2.5).
- If the patient presents more than 8 hours after ingestion and the time of acute acetaminophen ingestion is known:
- Administer a loading dose of ACETADOTE immediately
- Obtain acetaminophen concentration to determine need for continued treatment
- If the patient presents less than 8 hours after ingestion and the time of acute acetaminophen ingestion is known and the acetaminophen concentration is known:
Nomogram for Estimating Potential for Hepatotoxicity from Acute Acetaminophen Ingestion (2.2):
See Full Prescribing Information for instructions on how to use the nomogram to determine the need for dosing.
Preparation and Storage of Diluted Solution Prior to Administration (2.3):
- Calculate the dose (mg) based on the patient's weight in kg; multiple vials of ACETADOTE may be required.
- ACETADOTE is hyperosmolar (2,600 mOsmol/L), therefore ACETADOTE must be diluted in the recommended volume of sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water injection prior to intravenous administration. In general, 0.45% normal saline is the preferred diluent because it provides a more consistent osmolarity profile, reduces the amount of free water delivered to the patient, and better approximates physiologic fluids.
See Full Prescribing Information for examples of osmolarity depending on the type of solution and ACETADOTE concentration.
General Considerations for Selecting the Three-Bag or Two-Bag Regimen (2.4):
- It is not known whether the two-bag regimen is comparable to the three-bag regimen in preventing hepatotoxicity.
- Patients 40 kg or less should receive the three-bag regimen.
- For patients weighing 41 kg or greater, the three-bag regimen may be preferred for those with early signs of severe liver injury or a large acetaminophen ingestion.
Recommended Dosage for Acute Acetaminophen Ingestion (2.5):
- ACETADOTE is for intravenous administration only
- Total dosage of ACETADOTE is 300 mg/kg given intravenously as either:
- 3 separate doses infused over a total of 21 hours
OR - 2 separate doses infused over a total of 20 hours.
- 3 separate doses infused over a total of 21 hours
- See Full Prescribing Information for weight-based dosage and weight-based dilution (2.5)
See Full Prescribing Information for recommendations for continuing ACETADOTE treatment after 21 hours (2.2).
Repeated Supratherapeutic Acetaminophen Ingestion (2.6):
- Obtain acetaminophen concentration and other laboratory tests to guide treatment; revised Rumack-Matthew nomogram does not apply.
3 Dosage Forms and Strengths
Injection: 6000 mg/30 mL (200 mg/mL) of acetylcysteine in a single-dose vial.
5.1 Hypersensitivity Reactions
Serious acute hypersensitivity reactions; including fatal or life-threatening anaphylaxis, rash, hypotension, wheezing, and/or shortness of breath; have been observed in patients receiving intravenous acetylcysteine for acetaminophen overdose and occurred soon after initiation of the infusion [see Adverse Reactions (6)]. If a severe hypersensitivity reaction occurs, immediately stop the infusion of ACETADOTE and initiate appropriate treatment.
Patients with asthma should be closely monitored during initiation of ACETADOTE therapy and throughout ACETADOTE therapy.
Acute flushing and erythema of the skin may occur in patients receiving acetylcysteine intravenously. These reactions usually occur 30 to 60 minutes after initiating the infusion and often resolve spontaneously despite continued infusion of acetylcysteine. If a reaction to acetylcysteine involves more than simply flushing and erythema of the skin, it should be treated as a hypersensitivity reaction.
Management of less severe hypersensitivity reactions should be based upon the severity of the reaction and include temporary interruption of the infusion and/or administration of antihistaminic drugs. The ACETADOTE infusion may be carefully restarted after treatment of the hypersensitivity symptoms has been initiated and acute symptoms have resolved; however, if the hypersensitivity reaction returns upon re-initiation of treatment or increases in severity, ACETADOTE should be discontinued and alternative patient management should be considered.
6.1 Clinical Studies Experience
The following clinically significant adverse reactions are described elsewhere in labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Fluid Overload [see Warnings and Precautions (5.2)]
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In the literature, the most frequently reported adverse reactions attributed to intravenous acetylcysteine administration were rash, urticaria and pruritus. The frequency of adverse reactions has been reported to be between 0.2% and 21%, and they most commonly occur during the initial loading dose of acetylcysteine.
16 How Supplied/storage and Handling
ACETADOTE (acetylcysteine) injection is available as a 20% solution (200 mg/mL) in 30 mL single-dose glass vials. Each single dose vial contains 6 g/30 mL (200 mg/mL) of ACETADOTE injection. ACETADOTE is sterile and can be used for intravenous administration. It is available as follows:
- 30 mL vials, carton of 4 (NDC 66220-207-30)
Do not use previously opened vials for intravenous administration.
Note: The color of ACETADOTE may turn from essentially colorless to a slight pink or purple once the stopper is punctured. The color change does not affect the quality of the product.
The stopper in the ACETADOTE vial is formulated with a synthetic base-polymer and does not contain natural rubber latex, dry natural rubber, or blends of natural rubber.
Store unopened vials at controlled room temperature, 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]
2.6 Recommendations for Repeated Supratherapeutic Acetaminophen Ingestion
Repeated supratherapeutic acetaminophen ingestion (RSI) is an ingestion of acetaminophen at dosages higher than those recommended for extended periods of time. The risk of hepatotoxicity and the recommendations for treatment of acute acetaminophen ingestion (i.e., the revised Rumack-Matthew nomogram) do not apply to patients with RSI. Therefore, obtain the following information to guide ACETADOTE treatment for RSI:
Repeated supratherapeutic acetaminophen ingestion (RSI) is an ingestion of acetaminophen at dosages higher than those recommended for extended periods of time. The risk of hepatotoxicity and the recommendations for treatment of acute acetaminophen ingestion (i.e., the revised Rumack-Matthew nomogram) do not apply to patients with RSI. Therefore, obtain the following information to guide ACETADOTE treatment for RSI:
- Acetaminophen serum or plasma concentrations. A reported history of the quantity of acetaminophen ingested is often inaccurate and is not a reliable guide to therapy.
- Laboratory tests to monitor hepatic and renal function and electrolyte and fluid balance: AST, ALT, bilirubin, INR, creatinine, BUN, blood glucose, and electrolytes.
For specific ACETADOTE dosage and administration information in patients with RSI, consider contacting your regional poison center at 1-800-222-1222, or alternatively, a special health professional assistance line for acetaminophen overdose at 1-800-525-6115.
2.1 Pre Treatment Assessment and Testing Following Acute Acetaminophen Ingestion
Prior to initiating treatment with ACETADOTE, decide whether the three-bag or two-bag regimen will be used [see Dosage and Administration (2.4)].
The following recommendations are related to acute acetaminophen ingestion. For recommendations related to repeated supratherapeutic exposure [see Dosage and Administration (2.6)].
- Assess the history and timing of acetaminophen ingestion as an overdose.
- The reported history of the quantity of acetaminophen ingested as an overdose is often inaccurate and is not a reliable guide to therapy.
- Obtain the following laboratory tests to monitor hepatic and renal function and electrolyte and fluid balance: aspartate aminotransferase (AST), alanine aminotransferase (ALT), bilirubin, international normalized ratio (INR), creatinine, blood urea nitrogen (BUN), blood glucose, and electrolytes.
- Obtain a plasma or serum sample to assay for acetaminophen concentration at least 4 hours after ingestion. Acetaminophen concentrations obtained earlier than 4 hours post-ingestion may be misleading as they may not represent maximum acetaminophen concentrations.
- If the time of acute acetaminophen ingestion is unknown:
- If the acetaminophen concentration cannot be obtained (or is unavailable or uninterpretable) within the 8-hour time interval after acetaminophen ingestion or there is clinical evidence of acetaminophen toxicity:
- Administer a loading dose of ACETADOTE immediately and continue treatment for a total of two doses over 20 hours or three doses over 21 hours [see Dosage and Administration (2.5)].
- If the patient presents more than 8 hours after ingestion and the time of acute acetaminophen ingestion is known:
- If the patient presents less than 8 hours after ingestion and the time of acute acetaminophen ingestion is known and the acetaminophen concentration is known:
2.2 Nomogram for Estimating Potential for Hepatoxicity From Acute Acetaminophen Ingestion and Need for Acetadote Treatment
ACETADOTE is an antidote for acetaminophen overdose. The critical ingestion-treatment interval for maximal protection against severe hepatic injury is between 0 and 8 hours. Efficacy diminishes progressively after 8 hours and treatment initiation between 15 and 24 hours post-ingestion of acetaminophen yields limited efficacy. However, it does not appear to worsen the condition of patients and it should not be withheld, since the reported time of ingestion may not be correct.
If the timing of the acute acetaminophen ingestion is known and the results of the acetaminophen assay are available within 8 hours:
- Refer to the revised Rumack-Matthew nomogram (see Figure 1) to determine whether or not to initiate treatment with ACETADOTE.
- Initiation of ACETADOTE depends on the plasma or serum acetaminophen concentration and also the clinical presentation of the patient.
The nomogram may underestimate the hepatotoxicity risk in patients with chronic alcoholism, malnutrition, or CYP2E1 enzyme inducing drugs (e.g., isoniazid), and consideration should be given to treating these patients even if the acetaminophen concentrations are in the nontoxic range.
Structured Label Content
Section 42229-5 (42229-5)
Loading dose
For patients whose acetaminophen concentrations are at or above the treatment line (see Figure 1):
- Administer a loading dose of ACETADOTE [see Dosage and Administration (2.5)].
For patients with an acute overdose from an extended-release acetaminophen, if the acetaminophen concentration at 4 hours post ingestion is below the treatment line (see Figure 1) then obtain a second sample for acetaminophen concentration 8 to 10 hours after the acute ingestion. If the second value is at or above the treatment line:
- Administer a loading dose of ACETADOTE [see Dosage and Administration (2.5)].
For patients whose values are below the treatment line (see Figure 1), but time of ingestion was unknown or sample was obtained less than 4 hours after ingestion:
- Administer a loading dose of ACETADOTE [see Dosage and Administration (2.5)].
For patients whose values are below the treatment line (see Figure 1) and time of ingestion is known and the sample was obtained more than 4 hours after ingestion, do not administer ACETADOTE because there is minimal risk of hepatotoxicity.
Figure 1. Revised Rumack-Matthew Nomogram for Estimating Risk of Hepatoxicity After Acute Ingestion of Acetaminophen
Section 44425-7 (44425-7)
Refer to Table 2 and Table 3 to calculate the dose (mg) based on the patient's weight in kg; multiple vials of ACETADOTE may be required [see Dosage and Administration (2.5)]. Discard any unused portion left in the vial.
Because ACETADOTE is hyperosmolar (2,600 mOsmol/L), ACETADOTE must be diluted in the recommended volume of sterile water for injection, 0.45% sodium chloride injection (1/2 normal saline), or 5% dextrose in water prior to intravenous administration. The total injection volume will vary based on the patient's weight and chosen dosage regimen (i.e., three-bag or two-bag) [see Dosage and Administration (2.5), Warnings and Precautions (5.2)].
The choice of diluent should be based on the individual patient's clinical status, concurrent medical conditions, and institutional protocols. The treating clinician should assess each case individually and consult with their pharmacy if there are any concerns about the appropriate diluent choice. In general, 0.45% normal saline is the preferred diluent because it provides a more consistent osmolarity profile, reduces the amount of free water delivered to the patient, and better approximates physiologic fluids than 5% dextrose in water or sterile water for injection. However, consider 5% dextrose in water or sterile water for injection if sodium load is a concern for the patient.
Dilution of ACETADOTE in each of these three solutions results in different osmolarity of the acetylcysteine solution for intravenous administration (see Table 1 for examples of different osmolarity of the solution depending on the type of solution and the ACETADOTE concentration).
|
* Adjust osmolarity to a physiologically safe level (generally not less than 150 mOsmol/L in pediatric patients). |
|||
| Acetylcysteine Concentration | Osmolarity | ||
| Sterile Water for Injection | 0.45% Sodium Chloride Injection | 5% Dextrose in Water (D5W) |
|
| 4 mg/mL (lowest concentration 3-bag protocol) | 52 mOsmol/L* | 194 mOsmol/L | 311 mOsmol/L |
| 54.5 mg/mL (highest concentration 3-bag protocol) | 744 mOsmol/L | 855 mOsmol/L | 957 mOsmol/L |
| 7.9 mg/mL (lowest concentration 2-bag protocol) | 105 mOsmol/L | 241 mOsmol/L | 360 mOsmol/L |
| 18.2 mg/mL (highest concentration 2-bag protocol) | 239 mOsmol/L | 368 mOsmol/L | 487 mOsmol/L |
The choice of diluent should be based on the individual patient's clinical status, concurrent medical conditions, and institutional protocols. The treating clinician should assess each case individually and consult with their pharmacy if there are any concerns about the appropriate diluent choice. In general, 0.45% normal saline is the preferred diluent, as it provides a more consistent osmolarity profile, reduces the amount of free water delivered to the patient, and better approximates physiologic fluids than 5% dextrose in water or sterile water for injection.
Visually inspect for particular matter and discoloration prior to administration. The color of the diluted solution ranges from colorless to a slight pink or purple once the stopper is punctured (the color change does not affect the quality of the product). The diluted solution can be stored for 24 hours at room temperature. Discard the unused portion. If a vial was previously opened, do not use for intravenous administration.
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL – Vial Label
30 mL NDC 66220-207-30
Sterile
Acetadote
®
(acetylcysteine) Injection
6 g/30 mL
(200 mg/mL)
MUST BE FURTHER DILUTED
PRIOR TO INTRAVENOUS USE
CUMBERLAND ®
PHARMACEUTICALS
10 Overdosage (10 OVERDOSAGE)
Fatal and life-threatening adverse events have been reported following acetylcysteine overdosage, including anaphylaxis, cerebral edema, and hemolytic-uremic syndrome (HUS). Stop acetylcysteine administration in the setting of suspected acetylcysteine overdosage and manage as clinically indicated.
Anaphylaxis, including cases with a fatal outcome, has been reported following acetylcysteine overdosage. Patients who experienced anaphylaxis following acetylcysteine overdosage often became symptomatic during the loading dose and experienced hypotension, rash, angioedema, bronchospasm, or respiratory distress. Cases of anaphylaxis with acetylcysteine overdosage also described coagulopathy, renal failure, or respiratory failure.
Cerebral edema, including cases with a fatal outcome, has been reported following acetylcysteine overdosage. Patients who experienced cerebral edema following acetylcysteine overdose presented with altered mental status, abnormal pupillary responses, seizures. Some cases of cerebral edema with acetylcysteine overdosage described brain herniation.
HUS has been reported following acetylcysteine overdosage. Patients who experienced HUS presented with microangiopathic hemolytic anemia, thrombocytopenia, and acute kidney injury.
Removal of acetylcysteine via hemodialysis has been reported in the literature outside of the context of acetylcysteine overdosage. Studies of hemodialysis in acetaminophen overdose report significant extracorporeal removal of acetylcysteine during hemodialysis. Contact the Poison Center (1-800-222-1222) for overdosage management recommendations for ACETADOTE including considerations for hemodialysis.
11 Description (11 DESCRIPTION)
Acetylcysteine injection is an intravenous antidote for the treatment of acetaminophen overdose. Acetylcysteine is the nonproprietary name for the N-acetyl derivative of the naturally occurring amino acid, L-cysteine (N-acetyl-L-cysteine,). The compound is a white crystalline powder, which melts in the range of 104° to 110°C and has a very slight odor.
The molecular formula of the compound is C5H9NO3S, and its molecular weight is 163.2. Acetylcysteine has the following structural formula:
ACETADOTE is supplied as a sterile solution in vials containing 20% w/v (200 mg/mL) acetylcysteine. The pH of the solution ranges from 6.0 to 7.5. ACETADOTE contains the following inactive ingredients: sodium hydroxide (used for pH adjustment), and Water for Injection, USP.
The amount of sodium in ACETADOTE is approximately 30 mg/mL. Because ACETADOTE is administered based on a patient's weight, the amount of sodium administered in a course of treatment will vary from approximately 225 mg to 4500 mg. The use of ½ normal saline will contribute approximately an additional 1770 mg of sodium per liter of diluent.
8.4 Pediatric Use
Safety and effectiveness of ACETADOTE in pediatric patients have not been established by adequate and well-controlled studies. Use of ACETADOTE in pediatric patients 5 kg and greater is based on clinical practice [see Dosage and Administration (2.5)].
5.2 Fluid Overload
The total volume of ACETADOTE administered should be adjusted for patients less than 40 kg and for those requiring fluid restriction.
Intravenous administration of ACETADOTE can cause fluid overload, potentially resulting in hyponatremia, seizure, and death. To avoid fluid overload, use the recommended dilutions [see Dosage and Administration (2.5)].
4 Contraindications (4 CONTRAINDICATIONS)
ACETADOTE is contraindicated in patients with a previous hypersensitivity reaction to acetylcysteine [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (> 2%) are rash, urticaria/facial flushing
and pruritus (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Cumberland Pharmaceuticals Inc. at 1-877-484-2700 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
12.3 Pharmacokinetics
After a single intravenous dose of acetylcysteine, the plasma concentration of total acetylcysteine declined in a poly-exponential decay manner with a mean terminal half-life (T1/2) of 5.6 hours. The mean clearance (CL) for acetylcysteine was 0.11 liter/hr/kg and renal CL constituted about 30% of the total CL.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ACETADOTE is indicated to prevent or lessen hepatic injury after ingestion of a potentially hepatotoxic quantity of acetaminophen in adults and pediatric patients who weigh 5 kg or greater with acute ingestion or from repeated supratherapeutic ingestion (RSI).
12.1 Mechanism of Action
Acetylcysteine has been shown to reduce the extent of liver injury following acetaminophen overdose. Acetaminophen doses of 150 mg/kg or greater have been associated with hepatotoxicity. Acetylcysteine probably protects the liver by maintaining or restoring the glutathione levels, or by acting as an alternate substrate for conjugation with, and thus detoxification of, the reactive metabolite of acetaminophen.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions: Fatal or life-threatening anaphylaxis, rash, hypotension, wheezing, shortness of breath, and/or bronchospasm have been observed. Observe patients during and after the infusion; immediately discontinue infusion if a serious reaction occurs and initiate appropriate treatment. ACETADOTE infusion may be carefully restarted after treatment of hypersensitivity has been initiated and acute symptoms have resolved (5.1).
- Fluid Overload: Total volume administered should be reduced for patients weighing less than 40 kg and for those requiring fluid restriction (5.2).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Pre-Treatment Assessment Following Acute Ingestion (2.1):
Prior to initiating treatment with ACETADOTE, decide whether the three-bag or two-bag regimen will be used.
Obtain a plasma or serum sample to assay for acetaminophen concentration at least 4 hours after ingestion.
- If the time of acetaminophen ingestion is unknown:
- Administer a loading dose of ACETADOTE immediately.
- Obtain an acetaminophen concentration to determine need for continued treatment.
- If the acetaminophen concentration cannot be obtained (or is unavailable or uninterpretable) within the 8-hour time interval after acetaminophen ingestion or there is clinical evidence of acetaminophen toxicity:
- Administer a loading dose of ACETADOTE immediately and continue treatment for a total of two doses over 20 hours or three doses over 21 hours (2.5).
- If the patient presents more than 8 hours after ingestion and the time of acute acetaminophen ingestion is known:
- Administer a loading dose of ACETADOTE immediately
- Obtain acetaminophen concentration to determine need for continued treatment
- If the patient presents less than 8 hours after ingestion and the time of acute acetaminophen ingestion is known and the acetaminophen concentration is known:
Nomogram for Estimating Potential for Hepatotoxicity from Acute Acetaminophen Ingestion (2.2):
See Full Prescribing Information for instructions on how to use the nomogram to determine the need for dosing.
Preparation and Storage of Diluted Solution Prior to Administration (2.3):
- Calculate the dose (mg) based on the patient's weight in kg; multiple vials of ACETADOTE may be required.
- ACETADOTE is hyperosmolar (2,600 mOsmol/L), therefore ACETADOTE must be diluted in the recommended volume of sterile water for injection, 0.45% sodium chloride injection, or 5% dextrose in water injection prior to intravenous administration. In general, 0.45% normal saline is the preferred diluent because it provides a more consistent osmolarity profile, reduces the amount of free water delivered to the patient, and better approximates physiologic fluids.
See Full Prescribing Information for examples of osmolarity depending on the type of solution and ACETADOTE concentration.
General Considerations for Selecting the Three-Bag or Two-Bag Regimen (2.4):
- It is not known whether the two-bag regimen is comparable to the three-bag regimen in preventing hepatotoxicity.
- Patients 40 kg or less should receive the three-bag regimen.
- For patients weighing 41 kg or greater, the three-bag regimen may be preferred for those with early signs of severe liver injury or a large acetaminophen ingestion.
Recommended Dosage for Acute Acetaminophen Ingestion (2.5):
- ACETADOTE is for intravenous administration only
- Total dosage of ACETADOTE is 300 mg/kg given intravenously as either:
- 3 separate doses infused over a total of 21 hours
OR - 2 separate doses infused over a total of 20 hours.
- 3 separate doses infused over a total of 21 hours
- See Full Prescribing Information for weight-based dosage and weight-based dilution (2.5)
See Full Prescribing Information for recommendations for continuing ACETADOTE treatment after 21 hours (2.2).
Repeated Supratherapeutic Acetaminophen Ingestion (2.6):
- Obtain acetaminophen concentration and other laboratory tests to guide treatment; revised Rumack-Matthew nomogram does not apply.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 6000 mg/30 mL (200 mg/mL) of acetylcysteine in a single-dose vial.
5.1 Hypersensitivity Reactions
Serious acute hypersensitivity reactions; including fatal or life-threatening anaphylaxis, rash, hypotension, wheezing, and/or shortness of breath; have been observed in patients receiving intravenous acetylcysteine for acetaminophen overdose and occurred soon after initiation of the infusion [see Adverse Reactions (6)]. If a severe hypersensitivity reaction occurs, immediately stop the infusion of ACETADOTE and initiate appropriate treatment.
Patients with asthma should be closely monitored during initiation of ACETADOTE therapy and throughout ACETADOTE therapy.
Acute flushing and erythema of the skin may occur in patients receiving acetylcysteine intravenously. These reactions usually occur 30 to 60 minutes after initiating the infusion and often resolve spontaneously despite continued infusion of acetylcysteine. If a reaction to acetylcysteine involves more than simply flushing and erythema of the skin, it should be treated as a hypersensitivity reaction.
Management of less severe hypersensitivity reactions should be based upon the severity of the reaction and include temporary interruption of the infusion and/or administration of antihistaminic drugs. The ACETADOTE infusion may be carefully restarted after treatment of the hypersensitivity symptoms has been initiated and acute symptoms have resolved; however, if the hypersensitivity reaction returns upon re-initiation of treatment or increases in severity, ACETADOTE should be discontinued and alternative patient management should be considered.
6.1 Clinical Studies Experience
The following clinically significant adverse reactions are described elsewhere in labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Fluid Overload [see Warnings and Precautions (5.2)]
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In the literature, the most frequently reported adverse reactions attributed to intravenous acetylcysteine administration were rash, urticaria and pruritus. The frequency of adverse reactions has been reported to be between 0.2% and 21%, and they most commonly occur during the initial loading dose of acetylcysteine.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ACETADOTE (acetylcysteine) injection is available as a 20% solution (200 mg/mL) in 30 mL single-dose glass vials. Each single dose vial contains 6 g/30 mL (200 mg/mL) of ACETADOTE injection. ACETADOTE is sterile and can be used for intravenous administration. It is available as follows:
- 30 mL vials, carton of 4 (NDC 66220-207-30)
Do not use previously opened vials for intravenous administration.
Note: The color of ACETADOTE may turn from essentially colorless to a slight pink or purple once the stopper is punctured. The color change does not affect the quality of the product.
The stopper in the ACETADOTE vial is formulated with a synthetic base-polymer and does not contain natural rubber latex, dry natural rubber, or blends of natural rubber.
Store unopened vials at controlled room temperature, 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]
2.6 Recommendations for Repeated Supratherapeutic Acetaminophen Ingestion
Repeated supratherapeutic acetaminophen ingestion (RSI) is an ingestion of acetaminophen at dosages higher than those recommended for extended periods of time. The risk of hepatotoxicity and the recommendations for treatment of acute acetaminophen ingestion (i.e., the revised Rumack-Matthew nomogram) do not apply to patients with RSI. Therefore, obtain the following information to guide ACETADOTE treatment for RSI:
Repeated supratherapeutic acetaminophen ingestion (RSI) is an ingestion of acetaminophen at dosages higher than those recommended for extended periods of time. The risk of hepatotoxicity and the recommendations for treatment of acute acetaminophen ingestion (i.e., the revised Rumack-Matthew nomogram) do not apply to patients with RSI. Therefore, obtain the following information to guide ACETADOTE treatment for RSI:
- Acetaminophen serum or plasma concentrations. A reported history of the quantity of acetaminophen ingested is often inaccurate and is not a reliable guide to therapy.
- Laboratory tests to monitor hepatic and renal function and electrolyte and fluid balance: AST, ALT, bilirubin, INR, creatinine, BUN, blood glucose, and electrolytes.
For specific ACETADOTE dosage and administration information in patients with RSI, consider contacting your regional poison center at 1-800-222-1222, or alternatively, a special health professional assistance line for acetaminophen overdose at 1-800-525-6115.
2.1 Pre Treatment Assessment and Testing Following Acute Acetaminophen Ingestion (2.1 Pre-Treatment Assessment and Testing Following Acute Acetaminophen Ingestion)
Prior to initiating treatment with ACETADOTE, decide whether the three-bag or two-bag regimen will be used [see Dosage and Administration (2.4)].
The following recommendations are related to acute acetaminophen ingestion. For recommendations related to repeated supratherapeutic exposure [see Dosage and Administration (2.6)].
- Assess the history and timing of acetaminophen ingestion as an overdose.
- The reported history of the quantity of acetaminophen ingested as an overdose is often inaccurate and is not a reliable guide to therapy.
- Obtain the following laboratory tests to monitor hepatic and renal function and electrolyte and fluid balance: aspartate aminotransferase (AST), alanine aminotransferase (ALT), bilirubin, international normalized ratio (INR), creatinine, blood urea nitrogen (BUN), blood glucose, and electrolytes.
- Obtain a plasma or serum sample to assay for acetaminophen concentration at least 4 hours after ingestion. Acetaminophen concentrations obtained earlier than 4 hours post-ingestion may be misleading as they may not represent maximum acetaminophen concentrations.
- If the time of acute acetaminophen ingestion is unknown:
- If the acetaminophen concentration cannot be obtained (or is unavailable or uninterpretable) within the 8-hour time interval after acetaminophen ingestion or there is clinical evidence of acetaminophen toxicity:
- Administer a loading dose of ACETADOTE immediately and continue treatment for a total of two doses over 20 hours or three doses over 21 hours [see Dosage and Administration (2.5)].
- If the patient presents more than 8 hours after ingestion and the time of acute acetaminophen ingestion is known:
- If the patient presents less than 8 hours after ingestion and the time of acute acetaminophen ingestion is known and the acetaminophen concentration is known:
2.2 Nomogram for Estimating Potential for Hepatoxicity From Acute Acetaminophen Ingestion and Need for Acetadote Treatment (2.2 Nomogram for Estimating Potential for Hepatoxicity from Acute Acetaminophen Ingestion and Need for ACETADOTE Treatment)
ACETADOTE is an antidote for acetaminophen overdose. The critical ingestion-treatment interval for maximal protection against severe hepatic injury is between 0 and 8 hours. Efficacy diminishes progressively after 8 hours and treatment initiation between 15 and 24 hours post-ingestion of acetaminophen yields limited efficacy. However, it does not appear to worsen the condition of patients and it should not be withheld, since the reported time of ingestion may not be correct.
If the timing of the acute acetaminophen ingestion is known and the results of the acetaminophen assay are available within 8 hours:
- Refer to the revised Rumack-Matthew nomogram (see Figure 1) to determine whether or not to initiate treatment with ACETADOTE.
- Initiation of ACETADOTE depends on the plasma or serum acetaminophen concentration and also the clinical presentation of the patient.
The nomogram may underestimate the hepatotoxicity risk in patients with chronic alcoholism, malnutrition, or CYP2E1 enzyme inducing drugs (e.g., isoniazid), and consideration should be given to treating these patients even if the acetaminophen concentrations are in the nontoxic range.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:30.708969 · Updated: 2026-03-14T22:30:10.662825