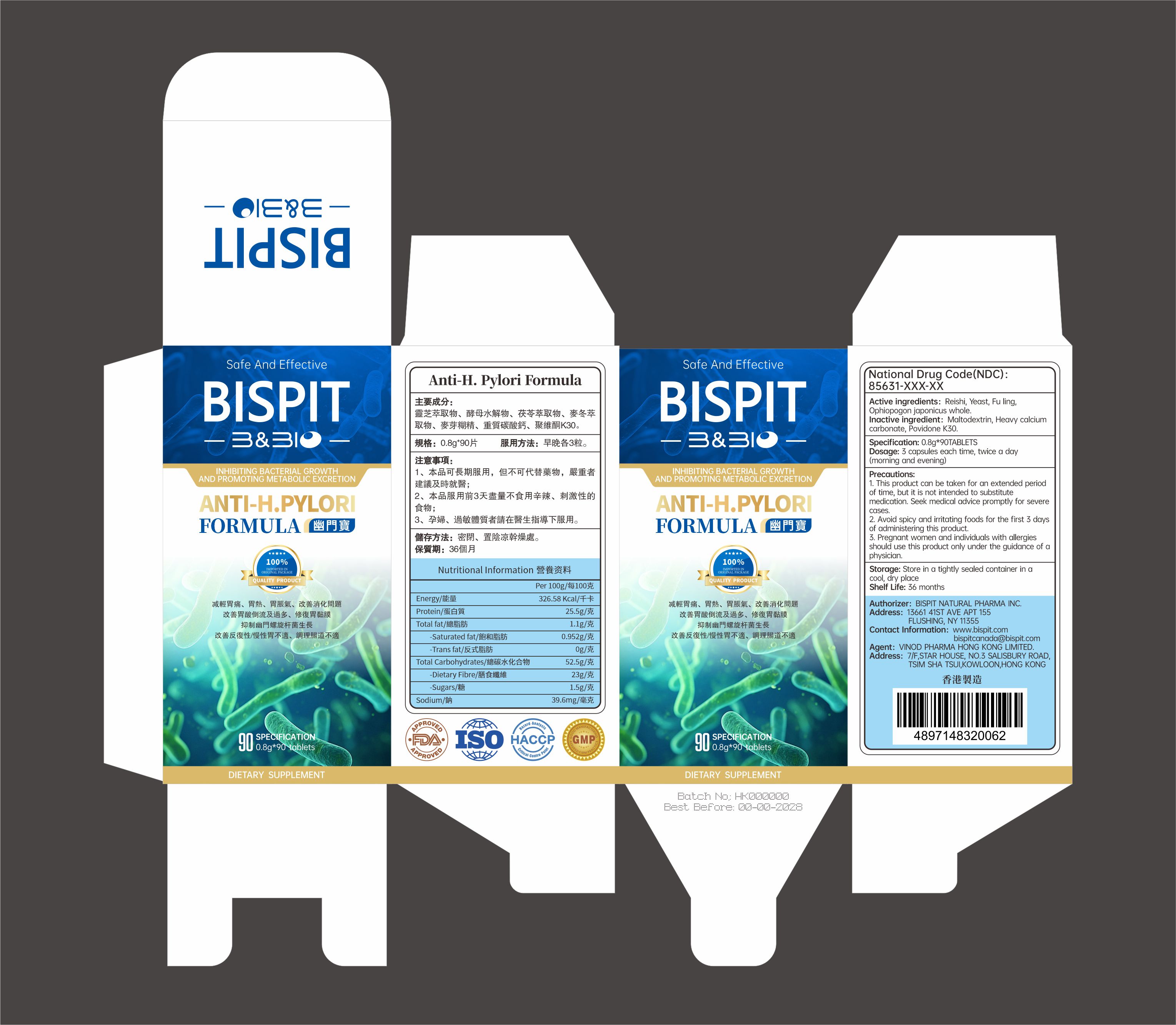
85631-016-01anti-h.pylori Formula
468a15c7-c6f8-ed82-e063-6294a90a5520
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients: Reishi, Yeast, Fu lingOphiopogon japonicus whole.
Medication Information
Warnings and Precautions
1. This product can be taken for an extended periodof time, but it is not intended to substitutemedication. Seek medical advice promptly for severecases.
2. Avoid spicy and irritating foods for the first 3 daysof administering this product.3. Pregnant women and individuals with allergiesshould use this product only under the guidance of aphysician.
Indications and Usage
Avoid spicy and irritating foods for the first 3 daysf administering this product.
Dosage and Administration
Dosage: 3 capsules each time, twice a day(morning and evening)
Description
Active ingredients: Reishi, Yeast, Fu lingOphiopogon japonicus whole.
Section 50565-1
Storage: Store in a tightly sealed container in acool, dry place,Keep out of children
Section 51727-6
Inactive ingredient: Maltodextrin, Heavy calciumcarbonate, Povidone K30.
Section 51945-4
Section 55105-1
INHIBITING BACTERIAL GROWTH
AND PROMOTING METABOLIC EXCRETION
Section 55106-9
Active ingredients: Reishi, Yeast, Fu lingOphiopogon japonicus whole.
Structured Label Content
Indications and Usage (34067-9)
Avoid spicy and irritating foods for the first 3 daysf administering this product.
Dosage and Administration (34068-7)
Dosage: 3 capsules each time, twice a day(morning and evening)
Warnings and Precautions (34071-1)
1. This product can be taken for an extended periodof time, but it is not intended to substitutemedication. Seek medical advice promptly for severecases.
2. Avoid spicy and irritating foods for the first 3 daysof administering this product.3. Pregnant women and individuals with allergiesshould use this product only under the guidance of aphysician.
Section 50565-1 (50565-1)
Storage: Store in a tightly sealed container in acool, dry place,Keep out of children
Section 51727-6 (51727-6)
Inactive ingredient: Maltodextrin, Heavy calciumcarbonate, Povidone K30.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
INHIBITING BACTERIAL GROWTH
AND PROMOTING METABOLIC EXCRETION
Section 55106-9 (55106-9)
Active ingredients: Reishi, Yeast, Fu lingOphiopogon japonicus whole.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:51.473059 · Updated: 2026-03-14T23:11:47.306350