These Highlights Do Not Include All The Information Needed To Use Gloperba ®
467cd83f-d695-4040-9434-10e80b4271da
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
GLOPERBA ® (colchicine) Oral Solution is indicated for prophylaxis of gout flares in adults.
Indications and Usage
GLOPERBA ® (colchicine) Oral Solution is indicated for prophylaxis of gout flares in adults.
Dosage and Administration
0.6 mg (5 mL) once or twice daily. Maximum dose 1.2 mg/day. ( 2.1 ) GLOPERBA is administered orally, without regard to meals ( 2.1 )
Warnings and Precautions
Fatal overdoses have been reported with colchicine in adults and children. Keep GLOPERBA out of the reach of children ( 5.1 , 10 ). Blood dyscrasias: myelosuppression, leukopenia, granulocytopenia, thrombocytopenia and aplastic anemia have been reported ( 5.2 ). Monitor for toxicity and, if present, consider lowering the dose, temporary interruption or discontinuation of colchicine ( 5.2 , 5.3 , 5.4 , 6 , 10 ). Drug interaction with CYP3A4 and P-gp inhibitors: Co-administration of colchicine with dual CYP3A4 and P-gp inhibitors has resulted in life threatening interactions and death ( 5.3 , 7 ). Neuromuscular toxicity: Myotoxicity including rhabdomyolysis may occur, especially in combination with other drugs known to cause this effect. Consider lowering the dose, temporary interruption or discontinuation of GLOPERBA ( 5.4 , 6 ).
Contraindications
Patients with renal or hepatic impairment should not be given GLOPERBA in conjunction with drugs that inhibit both CYP3A4 and P-gp [see Drug Interactions (7) ]. Combining these dual inhibitors with colchicine in patients with renal or hepatic impairment has resulted in life threatening or fatal colchicine toxicity. Patients with both renal and hepatic impairment should not be given GLOPERBA.
Adverse Reactions
Gastrointestinal disorders are the most common adverse reactions with colchicine. These disorders are often the first signs of toxicity and may indicate that the colchicine dose needs to be reduced or therapy stopped. These disorders include diarrhea, nausea, vomiting, and abdominal pain. Colchicine has been reported to cause neuromuscular toxicity, which may present as muscle pain or weakness [see Warnings and Precautions (5.4) ] . Toxic manifestations associated with colchicine include myelosuppression, disseminated intravascular coagulation and injury to cells in the renal, hepatic, circulatory and central nervous systems. These toxicities most often occur with excessive accumulation or overdosage [see Overdosage (10) ] . The following adverse reactions have been reported with colchicine. These adverse reactions have been generally reversible upon interrupting treatment or lowering the dose of colchicine. Neurological: sensory motor neuropathy Dermatological: alopecia, maculopapular rash, purpura, rash Digestive: abdominal cramping, abdominal pain, diarrhea, lactose intolerance, nausea, vomiting Hematological: leukopenia, granulocytopenia, thrombocytopenia, pancytopenia, aplastic anemia Hepatobiliary: elevated AST, elevated ALT Musculoskeletal: myopathy, elevated CPK, myotonia, muscle weakness, muscle pain, rhabdomyolysis Reproductive: azoospermia, oligospermia
Drug Interactions
Because colchicine is a substrate for both the CYP3A4 metabolizing enzyme and the P-gp efflux transporter, inhibition of either of these pathways may lead to colchicine related toxicity. Inhibition of both CYP3A4 and P-gp by dual inhibitors (i.e., clarithromycin) has been reported to produce life threatening or fatal colchicine toxicity due to significant increases in systemic colchicine levels. Therefore, concomitant use of GLOPERBA with inhibitors of both CYP3A4 and P-gp should be avoided. If treatment with colchicine is necessary, a reduced daily dose should be considered and the patient should be closely monitored for colchicine toxicity [see Drug Interactions (7) ]. Use of GLOPERBA in conjunction with drugs that inhibit both CYP3A4 and P-gp is contraindicated in patients with renal or hepatic impairment [see Contraindications (4) ].
How Supplied
GLOPERBA (colchicine) Oral Solution is a slightly hazy, red liquid with a cherry odor and the strength of 0.6 mg/5 mL. GLOPERBA (150 mL) is provided in a white, oblong, high density polyethylene bottle with a child-resistant cap. 150 mL: NDC 69557-222-01
Medication Information
Warnings and Precautions
Fatal overdoses have been reported with colchicine in adults and children. Keep GLOPERBA out of the reach of children ( 5.1 , 10 ). Blood dyscrasias: myelosuppression, leukopenia, granulocytopenia, thrombocytopenia and aplastic anemia have been reported ( 5.2 ). Monitor for toxicity and, if present, consider lowering the dose, temporary interruption or discontinuation of colchicine ( 5.2 , 5.3 , 5.4 , 6 , 10 ). Drug interaction with CYP3A4 and P-gp inhibitors: Co-administration of colchicine with dual CYP3A4 and P-gp inhibitors has resulted in life threatening interactions and death ( 5.3 , 7 ). Neuromuscular toxicity: Myotoxicity including rhabdomyolysis may occur, especially in combination with other drugs known to cause this effect. Consider lowering the dose, temporary interruption or discontinuation of GLOPERBA ( 5.4 , 6 ).
Indications and Usage
GLOPERBA ® (colchicine) Oral Solution is indicated for prophylaxis of gout flares in adults.
Dosage and Administration
0.6 mg (5 mL) once or twice daily. Maximum dose 1.2 mg/day. ( 2.1 ) GLOPERBA is administered orally, without regard to meals ( 2.1 )
Contraindications
Patients with renal or hepatic impairment should not be given GLOPERBA in conjunction with drugs that inhibit both CYP3A4 and P-gp [see Drug Interactions (7) ]. Combining these dual inhibitors with colchicine in patients with renal or hepatic impairment has resulted in life threatening or fatal colchicine toxicity. Patients with both renal and hepatic impairment should not be given GLOPERBA.
Adverse Reactions
Gastrointestinal disorders are the most common adverse reactions with colchicine. These disorders are often the first signs of toxicity and may indicate that the colchicine dose needs to be reduced or therapy stopped. These disorders include diarrhea, nausea, vomiting, and abdominal pain. Colchicine has been reported to cause neuromuscular toxicity, which may present as muscle pain or weakness [see Warnings and Precautions (5.4) ] . Toxic manifestations associated with colchicine include myelosuppression, disseminated intravascular coagulation and injury to cells in the renal, hepatic, circulatory and central nervous systems. These toxicities most often occur with excessive accumulation or overdosage [see Overdosage (10) ] . The following adverse reactions have been reported with colchicine. These adverse reactions have been generally reversible upon interrupting treatment or lowering the dose of colchicine. Neurological: sensory motor neuropathy Dermatological: alopecia, maculopapular rash, purpura, rash Digestive: abdominal cramping, abdominal pain, diarrhea, lactose intolerance, nausea, vomiting Hematological: leukopenia, granulocytopenia, thrombocytopenia, pancytopenia, aplastic anemia Hepatobiliary: elevated AST, elevated ALT Musculoskeletal: myopathy, elevated CPK, myotonia, muscle weakness, muscle pain, rhabdomyolysis Reproductive: azoospermia, oligospermia
Drug Interactions
Because colchicine is a substrate for both the CYP3A4 metabolizing enzyme and the P-gp efflux transporter, inhibition of either of these pathways may lead to colchicine related toxicity. Inhibition of both CYP3A4 and P-gp by dual inhibitors (i.e., clarithromycin) has been reported to produce life threatening or fatal colchicine toxicity due to significant increases in systemic colchicine levels. Therefore, concomitant use of GLOPERBA with inhibitors of both CYP3A4 and P-gp should be avoided. If treatment with colchicine is necessary, a reduced daily dose should be considered and the patient should be closely monitored for colchicine toxicity [see Drug Interactions (7) ]. Use of GLOPERBA in conjunction with drugs that inhibit both CYP3A4 and P-gp is contraindicated in patients with renal or hepatic impairment [see Contraindications (4) ].
How Supplied
GLOPERBA (colchicine) Oral Solution is a slightly hazy, red liquid with a cherry odor and the strength of 0.6 mg/5 mL. GLOPERBA (150 mL) is provided in a white, oblong, high density polyethylene bottle with a child-resistant cap. 150 mL: NDC 69557-222-01
Description
GLOPERBA ® (colchicine) Oral Solution is indicated for prophylaxis of gout flares in adults.
Section 42229-5
Limitations of use: The safety and effectiveness of GLOPERBA for acute treatment of gout flares during prophylaxis has not been studied.
GLOPERBA is not an analgesic medication and should not be used to treat pain from other causes.
Section 42231-1
| MEDICATION GUIDE | ||||
|---|---|---|---|---|
| GLOPERBA
®(Glow per' bah)
(colchicine) Oral Solution |
||||
| What is the most important information I should know about GLOPERBA? | ||||
GLOPERBA can cause serious side effects or death if levels of GLOPERBA are too high in your body.
|
||||
| What is GLOPERBA? | ||||
| GLOPERBA is a prescription medicine used to prevent gout flares in adults. | ||||
| It is not known if GLOPERBA is safe and effective for the treatment of sudden (acute) gout flares. | ||||
| GLOPERBA is not a pain medicine, and it should not be taken to treat pain related to other medical conditions unless specifically prescribed for those conditions. | ||||
| It is not known if GLOPERBA is safe and effective in children. | ||||
Do not take GLOPERBA if you:
|
||||
| See " What is the most important information that I should know about GLOPERBA?" | ||||
Before taking GLOPERBA, tell your healthcare provider about all of your medical conditions, including if you:
|
||||
Tell your healthcare provider about all the medicines you take, including prescription, over-the-counter medicines, vitamins, and herbal supplements.
|
||||
| Ask your healthcare provider or pharmacist if you are not sure if you take any of the medicines listed above. This is not a complete list of all the medicines that can affect GLOPERBA. | ||||
How should I take GLOPERBA?
|
||||
| What should I avoid while taking GLOPERBA? | ||||
| Avoid eating grapefruit or drinking grapefruit juice while taking GLOPERBA. It can increase your chances of having serious side effects. | ||||
| What are the possible side effects of GLOPERBA? | ||||
| GLOPERBA can cause serious side effects, including: | ||||
See
"
What is the most important information that I should know about GLOPERBA?"
|
||||
|
|
|||
|
||||
|
|
|||
| The most common side effects of GLOPERBA include: | ||||
|
|
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. | ||||
| These are not all of the possible side effects of GLOPERBA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
How should I store GLOPERBA?
|
||||
| Keep GLOPERBA and all medicines out of the reach of children. | ||||
| General information about the safe and effective use of GLOPERBA. | ||||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use GLOPERBA for a condition for which it was not prescribed. Do not give GLOPERBA to other people, even if they have the same symptoms that you have. It may harm them. | ||||
| If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about GLOPERBA that is written for healthcare professionals. | ||||
| What are the ingredients in GLOPERBA? | ||||
| Active Ingredient:colchicine | ||||
| Inactive Ingredients:benzyl alcohol, FD&C Red No. 40, artificial cherry flavor, anhydrous citric acid, dibasic sodium phosphate, glycerin, propylene glycol, sucralose, xanthan gum and purified water. | ||||
| Rx ONLY | ||||
|
Manufactured for:
SCILEX Pharmaceuticals, Inc. Palo Alto, California For more information, visit scilexholding.com or call 1-866-SCILEX3. |
||||
| This Medication Guide has been approved by the U.S Food and Drug Administration | Issued: 7/2019 |
7.1 Cyp3a4
The concomitant use of GLOPERBA and CYP3A4 inhibitors (e.g., clarithromycin, ketoconazole, grapefruit juice, erythromycin, verapamil, etc.) should be avoided due to the potential for serious and life threatening toxicity [see Warnings and Precautions (5.3)and Clinical Pharmacology (12.3)] .
If co-administration of GLOPERBA and a CYP3A4 inhibitor is necessary, the dose of GLOPERBA should be adjusted by either reducing the daily dose or reducing the dose frequency, and the patient should be monitored carefully for colchicine toxicity [see Clinical Pharmacology (12.3) ].
16.2 Storage
Store at 20°-25°C (68° - 77°F); excursions permitted to 15°-30°C (59°-86°F). [See USP Controlled Room Temperature.]
10 Overdosage
The dose of colchicine that would induce significant toxicity for an individual is unknown. Fatalities have occurred after ingestion of a dose as low as 7 mg over a four day period, while other patients have survived after ingesting more than 60 mg. A review of 150 patients who overdosed on colchicine found that those who ingested less than 0.5 mg/kg survived and tended to have milder adverse reactions such as gastrointestinal symptoms, whereas those who took 0.5 to 0.8 mg/kg had more severe adverse reactions, including myelosuppression. There was 100% mortality in those who ingested more than 0.8 mg/kg.
- The first stage of acute colchicine toxicity typically begins within 24 hours of ingestion and includes gastrointestinal symptoms such as abdominal pain, nausea, vomiting, diarrhea and significant fluid loss, leading to volume depletion. Peripheral leukocytosis may also be seen.
- Life threatening complications occur during the second stage, which occurs 24 to 72 hours after drug administration, attributed to multiorgan failure and its consequences. Death is usually a result of respiratory depression and cardiovascular collapse. If the patient survives, recovery of multiorgan injury may be accompanied by rebound leukocytosis and alopecia starting about one week after the initial ingestion.
- Treatment of colchicine poisoning should begin with gastric lavage and measures to prevent shock. Otherwise, treatment is symptomatic and supportive. No specific antidote is known. Colchicine is not effectively removed by hemodialysis [see Clinical Pharmacology (12.3)] .
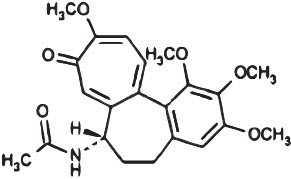
11 Description
Colchicine is an alkaloid obtained from various species of Colchicum. The chemical name for colchicine is (S)- N-(5,6,7,9-tetrahydro- 1,2,3,10-tetramethoxy-9-oxobenzo[ a]heptalen-7-yl) acetamide with a molecular formula of C 22H 25NO 6and a molecular weight of 399.4.
The structural formula of colchicine is provided in Figure 1.
Figure 1: Colchicine Structural Formula
Colchicine consists of pale yellow scales or powder; it darkens on exposure to light. Colchicine is soluble in water, freely soluble in alcohol, and slightly soluble in ether.
GLOPERBA is supplied for oral administration as a slightly hazy, red liquid with a cherry odor, containing 0.6 mg/5 mL of the active ingredient colchicine USP. Inactive ingredients: benzyl alcohol, FD&C Red No. 40, artificial cherry flavor, anhydrous citric acid, dibasic sodium phosphate, glycerin, propylene glycol, sucralose, xanthan gum and purified water.
16.1 How Supplied
GLOPERBA (colchicine) Oral Solution is a slightly hazy, red liquid with a cherry odor and the strength of 0.6 mg/5 mL. GLOPERBA (150 mL) is provided in a white, oblong, high density polyethylene bottle with a child-resistant cap.
150 mL: NDC 69557-222-01
8.4 Pediatric Use
Gout is rare in pediatric patients; safety and effectiveness of GLOPERBA in pediatric patients has not been established.
8.5 Geriatric Use
Because of the increased incidence of decreased renal function in the elderly population, and the higher incidence of other co-morbid conditions in the elderly population requiring the use of other medications, reducing the dosage of colchicine when elderly patients are treated with colchicine should be carefully considered [see Clinical Pharmacology (12.3)] .
5.1 Fatal Overdose
Fatal overdoses, both accidental and intentional, have been reported in adults and children who have ingested colchicine [see Overdosage (10)]. GLOPERBA should be kept out of the reach of children.
7.2 P Glycoprotein
The concomitant use of GLOPERBA and inhibitors of P-glycoprotein (e.g. clarithromycin, ketoconazole, cyclosporine, etc.) should be avoided due to the potential for serious and life threatening toxicity [see Warnings and Precautions (5.3)and Clinical Pharmacology (12.3)] .
If co-administration of GLOPERBA and a P-gp inhibitor is necessary, the dose of GLOPERBA should be adjusted by either reducing the daily dose or reducing the dose frequency, and the patient should be monitored carefully for colchicine toxicity [see Clinical Pharmacology (12.3)] .
14 Clinical Studies
The evidence for the efficacy of colchicine in patients with chronic gout is derived from the published literature. Two randomized clinical trials assessed the efficacy of colchicine 0.6 mg twice a day for the prophylaxis of gout flares in patients with gout initiating treatment with urate-lowering therapy. In both trials, treatment with colchicine decreased the frequency of gout flares.
4 Contraindications
Patients with renal or hepatic impairment should not be given GLOPERBA in conjunction with drugs that inhibit both CYP3A4 and P-gp [see Drug Interactions (7)]. Combining these dual inhibitors with colchicine in patients with renal or hepatic impairment has resulted in life threatening or fatal colchicine toxicity.
Patients with both renal and hepatic impairment should not be given GLOPERBA.
6 Adverse Reactions
Gastrointestinal disorders are the most common adverse reactions with colchicine. These disorders are often the first signs of toxicity and may indicate that the colchicine dose needs to be reduced or therapy stopped. These disorders include diarrhea, nausea, vomiting, and abdominal pain.
Colchicine has been reported to cause neuromuscular toxicity, which may present as muscle pain or weakness [see Warnings and Precautions (5.4)] .
Toxic manifestations associated with colchicine include myelosuppression, disseminated intravascular coagulation and injury to cells in the renal, hepatic, circulatory and central nervous systems. These toxicities most often occur with excessive accumulation or overdosage [see Overdosage (10)] .
The following adverse reactions have been reported with colchicine. These adverse reactions have been generally reversible upon interrupting treatment or lowering the dose of colchicine.
Neurological: sensory motor neuropathy
Dermatological: alopecia, maculopapular rash, purpura, rash
Digestive: abdominal cramping, abdominal pain, diarrhea, lactose intolerance, nausea, vomiting
Hematological: leukopenia, granulocytopenia, thrombocytopenia, pancytopenia, aplastic anemia
Hepatobiliary: elevated AST, elevated ALT
Musculoskeletal: myopathy, elevated CPK, myotonia, muscle weakness, muscle pain, rhabdomyolysis
Reproductive: azoospermia, oligospermia
7 Drug Interactions
Colchicine is a substrate of the CYP3A4 metabolizing enzyme and the P-glycoprotein (P-gp) efflux transporter. Fatal drug interactions have been reported when colchicine is administered with clarithromycin, a dual inhibitor of CYP3A4 and P-glycoprotein. Toxicities have also been reported when colchicine is administered with inhibitors of CYP3A4 that may not be potent inhibitors of P-gp (e.g., grapefruit juice, erythromycin, verapamil), or inhibitors of P-gp that may not be potent inhibitors of CYP3A4 (e.g., cyclosporine).
Patients with renal or hepatic impairment should not be given GLOPERBA with drugs that inhibit both CYP3A4 and P-glycoprotein [see Contraindications (4)] . Combining these dual inhibitors with GLOPERBA in patients with renal and hepatic impairment has resulted in life threatening or fatal colchicine toxicity.
Physicians should ensure that patients are suitable candidates for treatment with GLOPERBA and remain alert for signs and symptoms of toxic reactions associated with increased colchicine exposure due to drug interactions. Signs and symptoms of colchicine toxicity should be evaluated promptly and, if toxicity is suspected, consider lowering the dose, interruption or discontinuation of GLOPERBA.
2.1 Gout Prophylaxis
For prophylaxis of gout flares, the recommended dosage of GLOPERBA is 0.6 mg (5 mL) once or twice daily. The maximum dose is 1.2 mg/day.
GLOPERBA is administered orally, without regard to meals.
5.2 Blood Dyscrasias
Myelosuppression, leukopenia, granulocytopenia, thrombocytopenia, pancytopenia and aplastic anemia have been reported with colchicine used in therapeutic doses.
8.6 Renal Impairment
No dedicated pharmacokinetic study has been conducted using GLOPERBA in patients with varying degrees of renal impairment. Colchicine is known to be excreted in urine in humans and the presence of severe renal impairment has been associated with colchicine toxicity. Urinary clearance of colchicine and its metabolites may be decreased in patients with impaired renal function. Dose reduction or alternatives should be considered for the prophylaxis of gout flares in patients with severe renal impairment. Colchicine is not effectively removed by hemodialysis. Patients who are undergoing hemodialysis should be monitored carefully for colchicine toxicity.
12.2 Pharmacodynamics
The pharmacodynamics of colchicine are unknown.
5.3 Drug Interactions
Because colchicine is a substrate for both the CYP3A4 metabolizing enzyme and the P-gp efflux transporter, inhibition of either of these pathways may lead to colchicine related toxicity. Inhibition of both CYP3A4 and P-gp by dual inhibitors (i.e., clarithromycin) has been reported to produce life threatening or fatal colchicine toxicity due to significant increases in systemic colchicine levels. Therefore, concomitant use of GLOPERBA with inhibitors of both CYP3A4 and P-gp should be avoided. If treatment with colchicine is necessary, a reduced daily dose should be considered and the patient should be closely monitored for colchicine toxicity [see Drug Interactions (7)].
Use of GLOPERBA in conjunction with drugs that inhibit both CYP3A4 and P-gp is contraindicated in patients with renal or hepatic impairment [see Contraindications (4)].
8.7 Hepatic Impairment
No dedicated pharmacokinetic study using GLOPERBA has been conducted in patients with varying degrees of hepatic impairment. Colchicine is known to be metabolized in humans and the presence of severe hepatic impairment has been associated with colchicine toxicity. Hepatic clearance of colchicine may be significantly reduced and plasma half life prolonged in patients with chronic hepatic impairment.
Dose reduction or alternatives should be considered for the prophylaxis of gout flares in patients with severe hepatic impairment.
1 Indications and Usage
GLOPERBA ®(colchicine) Oral Solution is indicated for prophylaxis of gout flares in adults.
12.1 Mechanism of Action
Colchicine's effectiveness as a prophylactic treatment for gout has been postulated to be due to its ability to block neutrophil mediated inflammatory responses induced by monosodium urate crystals in synovial fluid. Colchicine disrupts the polymerization of β-tubulin into microtubules, thereby preventing the activation, degranulation and migration of neutrophils to sites of inflammation. Colchicine also interferes with the inflammasome complex found in neutrophils and monocytes that mediates interleukin-1β (IL-1β) activation.
5 Warnings and Precautions
- Fatal overdoses have been reported with colchicine in adults and children. Keep GLOPERBA out of the reach of children ( 5.1, 10).
- Blood dyscrasias: myelosuppression, leukopenia, granulocytopenia, thrombocytopenia and aplastic anemia have been reported ( 5.2).
- Monitor for toxicity and, if present, consider lowering the dose, temporary interruption or discontinuation of colchicine ( 5.2, 5.3, 5.4, 6, 10).
- Drug interaction with CYP3A4 and P-gp inhibitors: Co-administration of colchicine with dual CYP3A4 and P-gp inhibitors has resulted in life threatening interactions and death ( 5.3, 7).
- Neuromuscular toxicity: Myotoxicity including rhabdomyolysis may occur, especially in combination with other drugs known to cause this effect. Consider lowering the dose, temporary interruption or discontinuation of GLOPERBA ( 5.4, 6).
5.4 Neuromuscular Toxicity
Colchicine induced neuromuscular toxicity and rhabdomyolysis have been reported with chronic treatment in therapeutic doses, especially in combination with other drugs known to cause this effect. Patients with impaired renal function and elderly patients, even those with normal renal and hepatic function, are at increased risk. Once colchicine treatment is stopped, the symptoms generally resolve within one week to several months.
2 Dosage and Administration
3 Dosage Forms and Strengths
Ready to use solution for oral administration containing 0.6 mg/5 mL of colchicine. The oral solution is a slightly hazy, red liquid with a cherry odor.
7.4 Drug Interaction Studies
Two pharmacokinetic studies evaluated the effects of co-administration of posaconazole (300 mg QD), ciprofloxacin (500 mg BID), amlodipine (5 to 10 mg QD), and carvedilol (20 to 40 mg QD) on the systemic levels of colchicine.
GLOPERBA can be administered with amlodipine, carvedilol, and ciprofloxacin at the tested doses without a need for dose adjustment. However, the results should not be extrapolated to other co-administered drugs. Colchicine plasma levels were markedly elevated when GLOPERBA was co-administered with posaconazole. The recommended dose of GLOPERBA when co-administered with posaconazole is 0.24 mg (2 mL).
8 Use in Specific Populations
- In the presence of renal or hepatic impairment, patients should be monitored closely and dose adjustment should be considered as necessary ( 8.6, 8.7).
- Females and Males of Reproductive Potential: Advise males that GLOPERBA may rarely and transiently impair fertility ( 8.3).
- Geriatric Use: The recommended dose of colchicine should be based on renal and hepatic function ( 8.5, 12.3).
17 Patient Counseling Information
Advise the patient to read the FDA approved patient labeling (Medication Guide).
7.3 Hmg Coa Reductase Inhibitors and Fibrates
Some drugs such as HMG-CoA reductase inhibitors and fibrates may increase the risk of myopathy when combined with GLOPERBA. Complaints of muscle pain or weakness could be an indication to check serum creatinine kinase levels for signs of myopathy.
Principal Display Panel 150 Ml Bottle Carton
NDC 69557-222-01
GLOPERBA
®
(colchicine) oral solution
0.6 mg/5 mL
Rx Only
150 mL
Structured Label Content
Section 42229-5 (42229-5)
Limitations of use: The safety and effectiveness of GLOPERBA for acute treatment of gout flares during prophylaxis has not been studied.
GLOPERBA is not an analgesic medication and should not be used to treat pain from other causes.
Section 42231-1 (42231-1)
| MEDICATION GUIDE | ||||
|---|---|---|---|---|
| GLOPERBA
®(Glow per' bah)
(colchicine) Oral Solution |
||||
| What is the most important information I should know about GLOPERBA? | ||||
GLOPERBA can cause serious side effects or death if levels of GLOPERBA are too high in your body.
|
||||
| What is GLOPERBA? | ||||
| GLOPERBA is a prescription medicine used to prevent gout flares in adults. | ||||
| It is not known if GLOPERBA is safe and effective for the treatment of sudden (acute) gout flares. | ||||
| GLOPERBA is not a pain medicine, and it should not be taken to treat pain related to other medical conditions unless specifically prescribed for those conditions. | ||||
| It is not known if GLOPERBA is safe and effective in children. | ||||
Do not take GLOPERBA if you:
|
||||
| See " What is the most important information that I should know about GLOPERBA?" | ||||
Before taking GLOPERBA, tell your healthcare provider about all of your medical conditions, including if you:
|
||||
Tell your healthcare provider about all the medicines you take, including prescription, over-the-counter medicines, vitamins, and herbal supplements.
|
||||
| Ask your healthcare provider or pharmacist if you are not sure if you take any of the medicines listed above. This is not a complete list of all the medicines that can affect GLOPERBA. | ||||
How should I take GLOPERBA?
|
||||
| What should I avoid while taking GLOPERBA? | ||||
| Avoid eating grapefruit or drinking grapefruit juice while taking GLOPERBA. It can increase your chances of having serious side effects. | ||||
| What are the possible side effects of GLOPERBA? | ||||
| GLOPERBA can cause serious side effects, including: | ||||
See
"
What is the most important information that I should know about GLOPERBA?"
|
||||
|
|
|||
|
||||
|
|
|||
| The most common side effects of GLOPERBA include: | ||||
|
|
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. | ||||
| These are not all of the possible side effects of GLOPERBA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
How should I store GLOPERBA?
|
||||
| Keep GLOPERBA and all medicines out of the reach of children. | ||||
| General information about the safe and effective use of GLOPERBA. | ||||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use GLOPERBA for a condition for which it was not prescribed. Do not give GLOPERBA to other people, even if they have the same symptoms that you have. It may harm them. | ||||
| If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about GLOPERBA that is written for healthcare professionals. | ||||
| What are the ingredients in GLOPERBA? | ||||
| Active Ingredient:colchicine | ||||
| Inactive Ingredients:benzyl alcohol, FD&C Red No. 40, artificial cherry flavor, anhydrous citric acid, dibasic sodium phosphate, glycerin, propylene glycol, sucralose, xanthan gum and purified water. | ||||
| Rx ONLY | ||||
|
Manufactured for:
SCILEX Pharmaceuticals, Inc. Palo Alto, California For more information, visit scilexholding.com or call 1-866-SCILEX3. |
||||
| This Medication Guide has been approved by the U.S Food and Drug Administration | Issued: 7/2019 |
7.1 Cyp3a4 (7.1 CYP3A4)
The concomitant use of GLOPERBA and CYP3A4 inhibitors (e.g., clarithromycin, ketoconazole, grapefruit juice, erythromycin, verapamil, etc.) should be avoided due to the potential for serious and life threatening toxicity [see Warnings and Precautions (5.3)and Clinical Pharmacology (12.3)] .
If co-administration of GLOPERBA and a CYP3A4 inhibitor is necessary, the dose of GLOPERBA should be adjusted by either reducing the daily dose or reducing the dose frequency, and the patient should be monitored carefully for colchicine toxicity [see Clinical Pharmacology (12.3) ].
16.2 Storage
Store at 20°-25°C (68° - 77°F); excursions permitted to 15°-30°C (59°-86°F). [See USP Controlled Room Temperature.]
10 Overdosage (10 OVERDOSAGE)
The dose of colchicine that would induce significant toxicity for an individual is unknown. Fatalities have occurred after ingestion of a dose as low as 7 mg over a four day period, while other patients have survived after ingesting more than 60 mg. A review of 150 patients who overdosed on colchicine found that those who ingested less than 0.5 mg/kg survived and tended to have milder adverse reactions such as gastrointestinal symptoms, whereas those who took 0.5 to 0.8 mg/kg had more severe adverse reactions, including myelosuppression. There was 100% mortality in those who ingested more than 0.8 mg/kg.
- The first stage of acute colchicine toxicity typically begins within 24 hours of ingestion and includes gastrointestinal symptoms such as abdominal pain, nausea, vomiting, diarrhea and significant fluid loss, leading to volume depletion. Peripheral leukocytosis may also be seen.
- Life threatening complications occur during the second stage, which occurs 24 to 72 hours after drug administration, attributed to multiorgan failure and its consequences. Death is usually a result of respiratory depression and cardiovascular collapse. If the patient survives, recovery of multiorgan injury may be accompanied by rebound leukocytosis and alopecia starting about one week after the initial ingestion.
- Treatment of colchicine poisoning should begin with gastric lavage and measures to prevent shock. Otherwise, treatment is symptomatic and supportive. No specific antidote is known. Colchicine is not effectively removed by hemodialysis [see Clinical Pharmacology (12.3)] .
11 Description (11 DESCRIPTION)
Colchicine is an alkaloid obtained from various species of Colchicum. The chemical name for colchicine is (S)- N-(5,6,7,9-tetrahydro- 1,2,3,10-tetramethoxy-9-oxobenzo[ a]heptalen-7-yl) acetamide with a molecular formula of C 22H 25NO 6and a molecular weight of 399.4.
The structural formula of colchicine is provided in Figure 1.
Figure 1: Colchicine Structural Formula
Colchicine consists of pale yellow scales or powder; it darkens on exposure to light. Colchicine is soluble in water, freely soluble in alcohol, and slightly soluble in ether.
GLOPERBA is supplied for oral administration as a slightly hazy, red liquid with a cherry odor, containing 0.6 mg/5 mL of the active ingredient colchicine USP. Inactive ingredients: benzyl alcohol, FD&C Red No. 40, artificial cherry flavor, anhydrous citric acid, dibasic sodium phosphate, glycerin, propylene glycol, sucralose, xanthan gum and purified water.
16.1 How Supplied
GLOPERBA (colchicine) Oral Solution is a slightly hazy, red liquid with a cherry odor and the strength of 0.6 mg/5 mL. GLOPERBA (150 mL) is provided in a white, oblong, high density polyethylene bottle with a child-resistant cap.
150 mL: NDC 69557-222-01
8.4 Pediatric Use
Gout is rare in pediatric patients; safety and effectiveness of GLOPERBA in pediatric patients has not been established.
8.5 Geriatric Use
Because of the increased incidence of decreased renal function in the elderly population, and the higher incidence of other co-morbid conditions in the elderly population requiring the use of other medications, reducing the dosage of colchicine when elderly patients are treated with colchicine should be carefully considered [see Clinical Pharmacology (12.3)] .
5.1 Fatal Overdose
Fatal overdoses, both accidental and intentional, have been reported in adults and children who have ingested colchicine [see Overdosage (10)]. GLOPERBA should be kept out of the reach of children.
7.2 P Glycoprotein (7.2 P-Glycoprotein)
The concomitant use of GLOPERBA and inhibitors of P-glycoprotein (e.g. clarithromycin, ketoconazole, cyclosporine, etc.) should be avoided due to the potential for serious and life threatening toxicity [see Warnings and Precautions (5.3)and Clinical Pharmacology (12.3)] .
If co-administration of GLOPERBA and a P-gp inhibitor is necessary, the dose of GLOPERBA should be adjusted by either reducing the daily dose or reducing the dose frequency, and the patient should be monitored carefully for colchicine toxicity [see Clinical Pharmacology (12.3)] .
14 Clinical Studies (14 CLINICAL STUDIES)
The evidence for the efficacy of colchicine in patients with chronic gout is derived from the published literature. Two randomized clinical trials assessed the efficacy of colchicine 0.6 mg twice a day for the prophylaxis of gout flares in patients with gout initiating treatment with urate-lowering therapy. In both trials, treatment with colchicine decreased the frequency of gout flares.
4 Contraindications (4 CONTRAINDICATIONS)
Patients with renal or hepatic impairment should not be given GLOPERBA in conjunction with drugs that inhibit both CYP3A4 and P-gp [see Drug Interactions (7)]. Combining these dual inhibitors with colchicine in patients with renal or hepatic impairment has resulted in life threatening or fatal colchicine toxicity.
Patients with both renal and hepatic impairment should not be given GLOPERBA.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Gastrointestinal disorders are the most common adverse reactions with colchicine. These disorders are often the first signs of toxicity and may indicate that the colchicine dose needs to be reduced or therapy stopped. These disorders include diarrhea, nausea, vomiting, and abdominal pain.
Colchicine has been reported to cause neuromuscular toxicity, which may present as muscle pain or weakness [see Warnings and Precautions (5.4)] .
Toxic manifestations associated with colchicine include myelosuppression, disseminated intravascular coagulation and injury to cells in the renal, hepatic, circulatory and central nervous systems. These toxicities most often occur with excessive accumulation or overdosage [see Overdosage (10)] .
The following adverse reactions have been reported with colchicine. These adverse reactions have been generally reversible upon interrupting treatment or lowering the dose of colchicine.
Neurological: sensory motor neuropathy
Dermatological: alopecia, maculopapular rash, purpura, rash
Digestive: abdominal cramping, abdominal pain, diarrhea, lactose intolerance, nausea, vomiting
Hematological: leukopenia, granulocytopenia, thrombocytopenia, pancytopenia, aplastic anemia
Hepatobiliary: elevated AST, elevated ALT
Musculoskeletal: myopathy, elevated CPK, myotonia, muscle weakness, muscle pain, rhabdomyolysis
Reproductive: azoospermia, oligospermia
7 Drug Interactions (7 DRUG INTERACTIONS)
Colchicine is a substrate of the CYP3A4 metabolizing enzyme and the P-glycoprotein (P-gp) efflux transporter. Fatal drug interactions have been reported when colchicine is administered with clarithromycin, a dual inhibitor of CYP3A4 and P-glycoprotein. Toxicities have also been reported when colchicine is administered with inhibitors of CYP3A4 that may not be potent inhibitors of P-gp (e.g., grapefruit juice, erythromycin, verapamil), or inhibitors of P-gp that may not be potent inhibitors of CYP3A4 (e.g., cyclosporine).
Patients with renal or hepatic impairment should not be given GLOPERBA with drugs that inhibit both CYP3A4 and P-glycoprotein [see Contraindications (4)] . Combining these dual inhibitors with GLOPERBA in patients with renal and hepatic impairment has resulted in life threatening or fatal colchicine toxicity.
Physicians should ensure that patients are suitable candidates for treatment with GLOPERBA and remain alert for signs and symptoms of toxic reactions associated with increased colchicine exposure due to drug interactions. Signs and symptoms of colchicine toxicity should be evaluated promptly and, if toxicity is suspected, consider lowering the dose, interruption or discontinuation of GLOPERBA.
2.1 Gout Prophylaxis
For prophylaxis of gout flares, the recommended dosage of GLOPERBA is 0.6 mg (5 mL) once or twice daily. The maximum dose is 1.2 mg/day.
GLOPERBA is administered orally, without regard to meals.
5.2 Blood Dyscrasias
Myelosuppression, leukopenia, granulocytopenia, thrombocytopenia, pancytopenia and aplastic anemia have been reported with colchicine used in therapeutic doses.
8.6 Renal Impairment
No dedicated pharmacokinetic study has been conducted using GLOPERBA in patients with varying degrees of renal impairment. Colchicine is known to be excreted in urine in humans and the presence of severe renal impairment has been associated with colchicine toxicity. Urinary clearance of colchicine and its metabolites may be decreased in patients with impaired renal function. Dose reduction or alternatives should be considered for the prophylaxis of gout flares in patients with severe renal impairment. Colchicine is not effectively removed by hemodialysis. Patients who are undergoing hemodialysis should be monitored carefully for colchicine toxicity.
12.2 Pharmacodynamics
The pharmacodynamics of colchicine are unknown.
5.3 Drug Interactions
Because colchicine is a substrate for both the CYP3A4 metabolizing enzyme and the P-gp efflux transporter, inhibition of either of these pathways may lead to colchicine related toxicity. Inhibition of both CYP3A4 and P-gp by dual inhibitors (i.e., clarithromycin) has been reported to produce life threatening or fatal colchicine toxicity due to significant increases in systemic colchicine levels. Therefore, concomitant use of GLOPERBA with inhibitors of both CYP3A4 and P-gp should be avoided. If treatment with colchicine is necessary, a reduced daily dose should be considered and the patient should be closely monitored for colchicine toxicity [see Drug Interactions (7)].
Use of GLOPERBA in conjunction with drugs that inhibit both CYP3A4 and P-gp is contraindicated in patients with renal or hepatic impairment [see Contraindications (4)].
8.7 Hepatic Impairment
No dedicated pharmacokinetic study using GLOPERBA has been conducted in patients with varying degrees of hepatic impairment. Colchicine is known to be metabolized in humans and the presence of severe hepatic impairment has been associated with colchicine toxicity. Hepatic clearance of colchicine may be significantly reduced and plasma half life prolonged in patients with chronic hepatic impairment.
Dose reduction or alternatives should be considered for the prophylaxis of gout flares in patients with severe hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
GLOPERBA ®(colchicine) Oral Solution is indicated for prophylaxis of gout flares in adults.
12.1 Mechanism of Action
Colchicine's effectiveness as a prophylactic treatment for gout has been postulated to be due to its ability to block neutrophil mediated inflammatory responses induced by monosodium urate crystals in synovial fluid. Colchicine disrupts the polymerization of β-tubulin into microtubules, thereby preventing the activation, degranulation and migration of neutrophils to sites of inflammation. Colchicine also interferes with the inflammasome complex found in neutrophils and monocytes that mediates interleukin-1β (IL-1β) activation.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Fatal overdoses have been reported with colchicine in adults and children. Keep GLOPERBA out of the reach of children ( 5.1, 10).
- Blood dyscrasias: myelosuppression, leukopenia, granulocytopenia, thrombocytopenia and aplastic anemia have been reported ( 5.2).
- Monitor for toxicity and, if present, consider lowering the dose, temporary interruption or discontinuation of colchicine ( 5.2, 5.3, 5.4, 6, 10).
- Drug interaction with CYP3A4 and P-gp inhibitors: Co-administration of colchicine with dual CYP3A4 and P-gp inhibitors has resulted in life threatening interactions and death ( 5.3, 7).
- Neuromuscular toxicity: Myotoxicity including rhabdomyolysis may occur, especially in combination with other drugs known to cause this effect. Consider lowering the dose, temporary interruption or discontinuation of GLOPERBA ( 5.4, 6).
5.4 Neuromuscular Toxicity
Colchicine induced neuromuscular toxicity and rhabdomyolysis have been reported with chronic treatment in therapeutic doses, especially in combination with other drugs known to cause this effect. Patients with impaired renal function and elderly patients, even those with normal renal and hepatic function, are at increased risk. Once colchicine treatment is stopped, the symptoms generally resolve within one week to several months.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Ready to use solution for oral administration containing 0.6 mg/5 mL of colchicine. The oral solution is a slightly hazy, red liquid with a cherry odor.
7.4 Drug Interaction Studies
Two pharmacokinetic studies evaluated the effects of co-administration of posaconazole (300 mg QD), ciprofloxacin (500 mg BID), amlodipine (5 to 10 mg QD), and carvedilol (20 to 40 mg QD) on the systemic levels of colchicine.
GLOPERBA can be administered with amlodipine, carvedilol, and ciprofloxacin at the tested doses without a need for dose adjustment. However, the results should not be extrapolated to other co-administered drugs. Colchicine plasma levels were markedly elevated when GLOPERBA was co-administered with posaconazole. The recommended dose of GLOPERBA when co-administered with posaconazole is 0.24 mg (2 mL).
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- In the presence of renal or hepatic impairment, patients should be monitored closely and dose adjustment should be considered as necessary ( 8.6, 8.7).
- Females and Males of Reproductive Potential: Advise males that GLOPERBA may rarely and transiently impair fertility ( 8.3).
- Geriatric Use: The recommended dose of colchicine should be based on renal and hepatic function ( 8.5, 12.3).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA approved patient labeling (Medication Guide).
7.3 Hmg Coa Reductase Inhibitors and Fibrates (7.3 HMG-CoA Reductase Inhibitors and Fibrates)
Some drugs such as HMG-CoA reductase inhibitors and fibrates may increase the risk of myopathy when combined with GLOPERBA. Complaints of muscle pain or weakness could be an indication to check serum creatinine kinase levels for signs of myopathy.
Principal Display Panel 150 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 150 mL Bottle Carton)
NDC 69557-222-01
GLOPERBA
®
(colchicine) oral solution
0.6 mg/5 mL
Rx Only
150 mL
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:36.541746 · Updated: 2026-03-14T22:16:35.361266