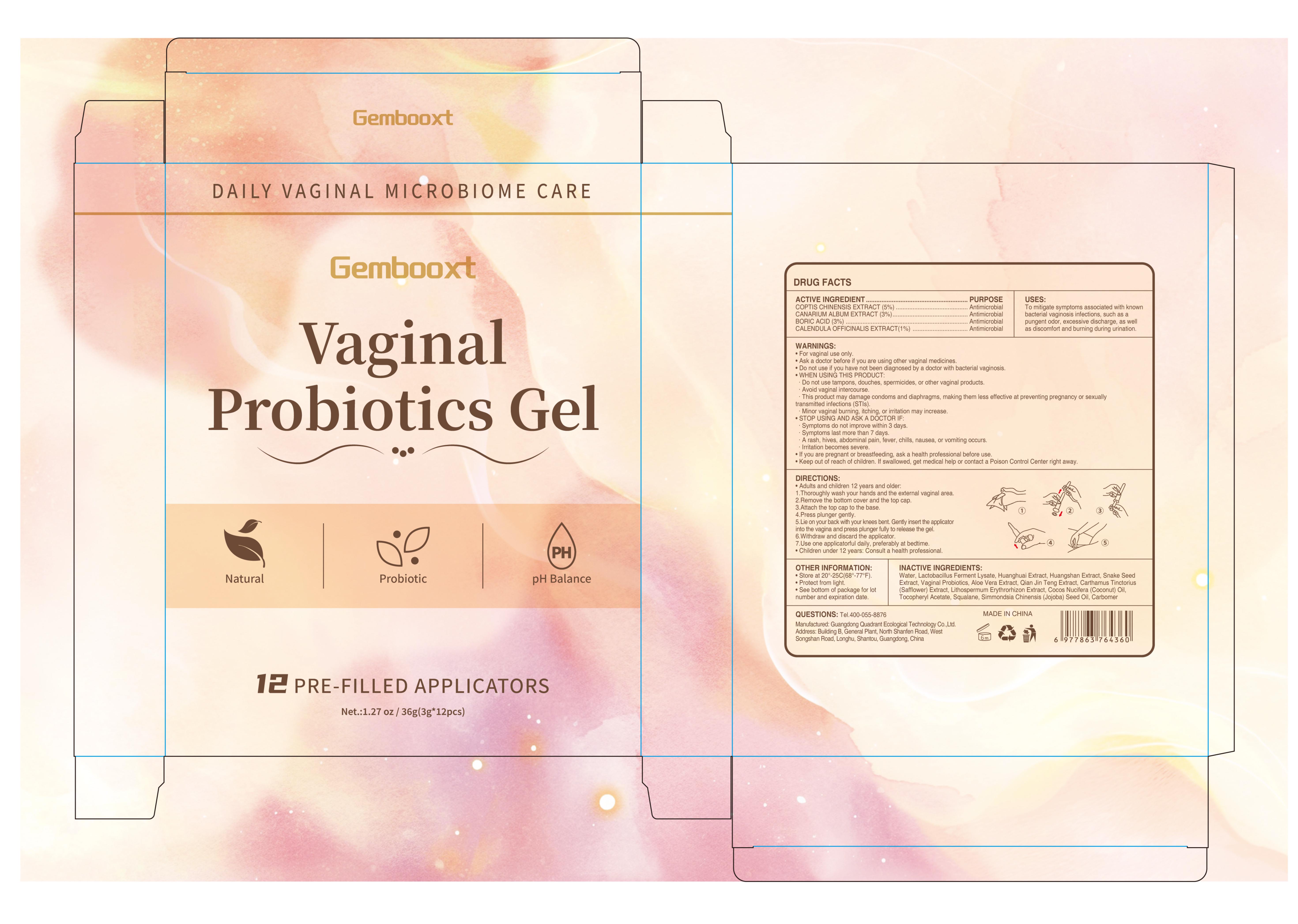
76986-021 gembooxt vaginal probiotics gel
464b0e4a-6d92-9a03-e063-6294a90a6ff4
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
COPTIS CHINENSIS EXTRACT 5% CANARIUM ALBUM EXTRACT 3% BORIC ACID 3 CALENDULA OFFICINALIS EXTRACT 1%
Medication Information
Warnings and Precautions
For vaginal use only.
Active Ingredient
COPTIS CHINENSIS EXTRACT 5%
CANARIUM ALBUM EXTRACT 3%
BORIC ACID 3
CALENDULA OFFICINALIS EXTRACT 1%
Indications and Usage
To mitigate symptoms associated with known bacterial vaginosis infections, such as a pungent odor, excessive discharge, as well as discomfort and burning during urination.
Dosage and Administration
Adults and children 12 years and older:
1.Thoroughly wash your hands and the external vaginal area.
2.Remove the bottom cover and the top cap.
3.Attach the top cap to the base.
4.Press plunger gently.
5.Lie on your back with your knees bent. Gently insert the applicator into the vagina and press plunger fully to release the gel.
6.Withdraw and discard the applicator.
7.Use one applicatorful daily, preferably at bedtime.
Children under 12 years: Consult a health professional
Description
COPTIS CHINENSIS EXTRACT 5% CANARIUM ALBUM EXTRACT 3% BORIC ACID 3 CALENDULA OFFICINALIS EXTRACT 1%
Section 50565-1
Keep out of reach of children. lf swallowed, get medical help or contact a Poison Control Center riaht away.
Section 50566-9
STOP USING AND ASK A DOCTOR IF:
Symptoms do not improve within 3 days.
Symptoms last more than 7 days
A rash, hives, abdominal pain, fever, chills, nausea, or vomiting occurs
lrritation becomes severe.
Section 50567-7
Ask a doctor before if you are using other vaginal medicines.
WHEN USING THIS PRODUCT:
Do not use tampons, douches, spermicides, or other vaginal products.
Avoid vaginal intercourse.
This product may damage condoms and diaphragms, making them less efective at preventing pregnancy or sexually transmitted infections (STls)
Minor vaginal burning, itching, or irritation may increase.
If you are pregnant or breastfeeding, ask a health professional before use.
Section 50570-1
Do not use if you have not been diagnosed by a doctor with bacterial vaginosis.
Section 51727-6
WATER
LACTOBACILLUS FERMENT LYSATE
SNAKE SEED EXTRACT
ALOE VERA EXTRACT
QIAN JIN TENG EXTRACT
CARTHAMUS TINCTORIUS (SAFFLOWER) EXTRACT
LITHOSPERMUM ERYTHRORHIZON EXTRACT
COCOS NUCIFERA (COCONUT) OIL
TOCOPHERYL ACETATE
SQUALANE
SIMMONDSIA CHINENSIS (JOJOBA) SEED OIL
CARBOMER
Section 51945-4
Section 55105-1
Antimicrobial
Structured Label Content
Warnings and Precautions (34071-1)
For vaginal use only.
Active Ingredient (55106-9)
COPTIS CHINENSIS EXTRACT 5%
CANARIUM ALBUM EXTRACT 3%
BORIC ACID 3
CALENDULA OFFICINALIS EXTRACT 1%
Indications and Usage (34067-9)
To mitigate symptoms associated with known bacterial vaginosis infections, such as a pungent odor, excessive discharge, as well as discomfort and burning during urination.
Dosage and Administration (34068-7)
Adults and children 12 years and older:
1.Thoroughly wash your hands and the external vaginal area.
2.Remove the bottom cover and the top cap.
3.Attach the top cap to the base.
4.Press plunger gently.
5.Lie on your back with your knees bent. Gently insert the applicator into the vagina and press plunger fully to release the gel.
6.Withdraw and discard the applicator.
7.Use one applicatorful daily, preferably at bedtime.
Children under 12 years: Consult a health professional
Section 50565-1 (50565-1)
Keep out of reach of children. lf swallowed, get medical help or contact a Poison Control Center riaht away.
Section 50566-9 (50566-9)
STOP USING AND ASK A DOCTOR IF:
Symptoms do not improve within 3 days.
Symptoms last more than 7 days
A rash, hives, abdominal pain, fever, chills, nausea, or vomiting occurs
lrritation becomes severe.
Section 50567-7 (50567-7)
Ask a doctor before if you are using other vaginal medicines.
WHEN USING THIS PRODUCT:
Do not use tampons, douches, spermicides, or other vaginal products.
Avoid vaginal intercourse.
This product may damage condoms and diaphragms, making them less efective at preventing pregnancy or sexually transmitted infections (STls)
Minor vaginal burning, itching, or irritation may increase.
If you are pregnant or breastfeeding, ask a health professional before use.
Section 50570-1 (50570-1)
Do not use if you have not been diagnosed by a doctor with bacterial vaginosis.
Section 51727-6 (51727-6)
WATER
LACTOBACILLUS FERMENT LYSATE
SNAKE SEED EXTRACT
ALOE VERA EXTRACT
QIAN JIN TENG EXTRACT
CARTHAMUS TINCTORIUS (SAFFLOWER) EXTRACT
LITHOSPERMUM ERYTHRORHIZON EXTRACT
COCOS NUCIFERA (COCONUT) OIL
TOCOPHERYL ACETATE
SQUALANE
SIMMONDSIA CHINENSIS (JOJOBA) SEED OIL
CARBOMER
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Antimicrobial
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:47.683886 · Updated: 2026-03-14T23:11:35.907821