These Highlights Do Not Include All The Information Needed To Use Lidocaine Hydrochloride Injection Safely And Effectively. See Full Prescribing Information For Lidocaine Hydrochloride Injections.
4643042c-ec95-3bad-e063-6294a90a8250
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine hydrochloride injection is indicated in adult and pediatric patients for the production of local or regional anesthesia or analgesia for surgery, dental, and oral surgery procedures, diagnostic and therapeutic procedures, and for obstetrical procedures. Specific concentrations and presentations of lidocaine hydrochloride injection is recommended for each type of block indicated to produce local or regional anesthesia or analgesia [see Dosage and Administration ( 2.2 )] .
Indications and Usage
Lidocaine hydrochloride injection is indicated in adult and pediatric patients for the production of local or regional anesthesia or analgesia for surgery, dental, and oral surgery procedures, diagnostic and therapeutic procedures, and for obstetrical procedures. Specific concentrations and presentations of lidocaine hydrochloride injection is recommended for each type of block indicated to produce local or regional anesthesia or analgesia [see Dosage and Administration ( 2.2 )] .
Dosage and Administration
See Full Prescribing Information for recommended dosages and administration information for adult and pediatric patients.
Warnings and Precautions
Dose-Related Toxicity : Monitor cardiovascular and respiratory vital signs and patient's state of consciousness after injection of lidocaine hydrochloride injection. ( 5.1 ) Methemoglobinemia : Cases of methemoglobinemia have been reported in association with local anesthetics use. See full prescribing information for more details on managing these risks. ( 5.2 ) Chondrolysis with Intra-Articular Infusion : Avoid Intra-articular infusions as there have been post-marketing reports of chondrolysis in patients receiving such infusion. ( 5.4 ) Allergic-Type Reactions to Sulfites in Lidocaine Hydrochloride Injection and Anaphylactic Reactions: Lidocaine hydrochloride injection without epinephrine does not contain sodium metabisulfite. ( 5.6 ) Risk of Systemic Toxicities with Unintended Intravascular or Intrathecal Injection : Unintended intravascular or intrathecal injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progression ultimately to respiratory arrest. Aspirate for blood or cerebrospinal fluid (where applicable) prior to each dose and consider using a test dose of lidocaine hydrochloride injection. ( 5.7 )
Contraindications
Lidocaine hydrochloride injection is contraindicated in patients with a known hypersensitivity to lidocaine or to any local anesthetics of the amide type or to other components of lidocaine hydrochloride injection.
Adverse Reactions
Small doses of local anesthetics (e.g., lidocaine hydrochloride injection) injected into the head and neck area, including retrobulbar, dental and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses. The injection procedures require the utmost care. Confusion, convulsions, respiratory depression and/or respiratory arrest, and cardiovascular stimulation or depression have been reported. These reactions may be due to intra-arterial injection of the local anesthetic with retrograde flow to the cerebral circulation. They may also be due to puncture of the dural sheath of the optic nerve during retrobulbar block with diffusion of any local anesthetic along the subdural space to the midbrain. Monitor circulation and respiration and constantly observe patients receiving lidocaine hydrochloride injection blocks. Resuscitative equipment and drugs, and personnel for treating adverse reactions should be immediately available. Dosage recommendations should not be exceeded [see Dosage and Administration ( 2.2 )].
Drug Interactions
Local Anesthetics : The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects when additional local anesthetics are administered. ( 7.1 ) Monoamine Oxidase Inhibitors and Tricyclic Antidepressants : Administration of lidocaine hydrochloride injection to patients receiving monoamine oxidase inhibitors or tricyclic antidepressants may produce severe, prolonged hypertension Concurrent use of these agents should generally be avoided. ( 5.5 , 7.2 ) Ergot-type Oxytocic Drugs : Concurrent administration of lidocaine hydrochloride injection and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents. ( 5.5 , 7.3 ) Nonselective Beta-Adrenergic Antagonists : Administration of lidocaine hydrochloride injection in patients receiving nonselective beta-adrenergic antagonist may cause severe hypertension and bradycardia. Concurrent use of these agents should generally be avoided. ( 5.5 , 7.4 ) Drugs Associated with Methemoglobinemia : Patients are at increased risk of developing methemoglobinemia when concurrently exposed to nitrates, nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants and other drugs. ( 7.5 ). Geriatric Use : Elderly patients should be given reduced doses commensurate with their age and physical condition. ( 8.5 ) Hepatic Impairment : consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment. ( 8.6 )
Storage and Handling
Lidocaine hydrochloride injection, USP is a clear, colorless solution supplied as follows: Lidocaine Hydrochloride Injection, USP (1%) NDC (10 mg per mL) Package Factor 83634-651-50 500 mg per 50 mL Multiple-Dose Vial 25 vials per carton
How Supplied
Lidocaine hydrochloride injection, USP is a clear, colorless solution supplied as follows: Lidocaine Hydrochloride Injection, USP (1%) NDC (10 mg per mL) Package Factor 83634-651-50 500 mg per 50 mL Multiple-Dose Vial 25 vials per carton
Medication Information
Warnings and Precautions
Dose-Related Toxicity : Monitor cardiovascular and respiratory vital signs and patient's state of consciousness after injection of lidocaine hydrochloride injection. ( 5.1 ) Methemoglobinemia : Cases of methemoglobinemia have been reported in association with local anesthetics use. See full prescribing information for more details on managing these risks. ( 5.2 ) Chondrolysis with Intra-Articular Infusion : Avoid Intra-articular infusions as there have been post-marketing reports of chondrolysis in patients receiving such infusion. ( 5.4 ) Allergic-Type Reactions to Sulfites in Lidocaine Hydrochloride Injection and Anaphylactic Reactions: Lidocaine hydrochloride injection without epinephrine does not contain sodium metabisulfite. ( 5.6 ) Risk of Systemic Toxicities with Unintended Intravascular or Intrathecal Injection : Unintended intravascular or intrathecal injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progression ultimately to respiratory arrest. Aspirate for blood or cerebrospinal fluid (where applicable) prior to each dose and consider using a test dose of lidocaine hydrochloride injection. ( 5.7 )
Indications and Usage
Lidocaine hydrochloride injection is indicated in adult and pediatric patients for the production of local or regional anesthesia or analgesia for surgery, dental, and oral surgery procedures, diagnostic and therapeutic procedures, and for obstetrical procedures. Specific concentrations and presentations of lidocaine hydrochloride injection is recommended for each type of block indicated to produce local or regional anesthesia or analgesia [see Dosage and Administration ( 2.2 )] .
Dosage and Administration
See Full Prescribing Information for recommended dosages and administration information for adult and pediatric patients.
Contraindications
Lidocaine hydrochloride injection is contraindicated in patients with a known hypersensitivity to lidocaine or to any local anesthetics of the amide type or to other components of lidocaine hydrochloride injection.
Adverse Reactions
Small doses of local anesthetics (e.g., lidocaine hydrochloride injection) injected into the head and neck area, including retrobulbar, dental and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses. The injection procedures require the utmost care. Confusion, convulsions, respiratory depression and/or respiratory arrest, and cardiovascular stimulation or depression have been reported. These reactions may be due to intra-arterial injection of the local anesthetic with retrograde flow to the cerebral circulation. They may also be due to puncture of the dural sheath of the optic nerve during retrobulbar block with diffusion of any local anesthetic along the subdural space to the midbrain. Monitor circulation and respiration and constantly observe patients receiving lidocaine hydrochloride injection blocks. Resuscitative equipment and drugs, and personnel for treating adverse reactions should be immediately available. Dosage recommendations should not be exceeded [see Dosage and Administration ( 2.2 )].
Drug Interactions
Local Anesthetics : The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects when additional local anesthetics are administered. ( 7.1 ) Monoamine Oxidase Inhibitors and Tricyclic Antidepressants : Administration of lidocaine hydrochloride injection to patients receiving monoamine oxidase inhibitors or tricyclic antidepressants may produce severe, prolonged hypertension Concurrent use of these agents should generally be avoided. ( 5.5 , 7.2 ) Ergot-type Oxytocic Drugs : Concurrent administration of lidocaine hydrochloride injection and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents. ( 5.5 , 7.3 ) Nonselective Beta-Adrenergic Antagonists : Administration of lidocaine hydrochloride injection in patients receiving nonselective beta-adrenergic antagonist may cause severe hypertension and bradycardia. Concurrent use of these agents should generally be avoided. ( 5.5 , 7.4 ) Drugs Associated with Methemoglobinemia : Patients are at increased risk of developing methemoglobinemia when concurrently exposed to nitrates, nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants and other drugs. ( 7.5 ). Geriatric Use : Elderly patients should be given reduced doses commensurate with their age and physical condition. ( 8.5 ) Hepatic Impairment : consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment. ( 8.6 )
Storage and Handling
Lidocaine hydrochloride injection, USP is a clear, colorless solution supplied as follows: Lidocaine Hydrochloride Injection, USP (1%) NDC (10 mg per mL) Package Factor 83634-651-50 500 mg per 50 mL Multiple-Dose Vial 25 vials per carton
How Supplied
Lidocaine hydrochloride injection, USP is a clear, colorless solution supplied as follows: Lidocaine Hydrochloride Injection, USP (1%) NDC (10 mg per mL) Package Factor 83634-651-50 500 mg per 50 mL Multiple-Dose Vial 25 vials per carton
Description
Lidocaine hydrochloride injection is indicated in adult and pediatric patients for the production of local or regional anesthesia or analgesia for surgery, dental, and oral surgery procedures, diagnostic and therapeutic procedures, and for obstetrical procedures. Specific concentrations and presentations of lidocaine hydrochloride injection is recommended for each type of block indicated to produce local or regional anesthesia or analgesia [see Dosage and Administration ( 2.2 )] .
Section 42229-5
Adults
For normal healthy adults, the maximum individual dose should not exceed 4.5 mg/kg of body weight, and in general it is recommended that the maximum total dose does not exceed 300 mg.
The maximum recommended dose per 90 minute period of lidocaine hydrochloride for paracervical block in obstetrical patients and non-obstetrical patients is 200 mg total. One half of the total dose is usually administered to each side. Inject slowly, five minutes between sides [see Pregnancy ( 8.1)].
Section 44425-7
Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.]
Protect from light.
Sterile, Nonpyrogenic.
The container closure is not made with natural rubber latex.
11 Description
Lidocaine hydrochloride injection, USP contains lidocaine hydrochloride, an amide local anesthetic, as the active pharmaceutical ingredient. The route of administration for Lidocaine hydrochloride injection, USP is by injection, for infiltration and nerve block. Multiple dose vials contain methylparaben and they should not be used for caudal and lumbar epidural blocks.
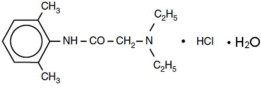
Lidocaine hydrochloride, is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride monohydrate and has the molecular weight of 288.8 g/mol. Lidocaine hydrochloride molecular formula is C 14H 22N 2O • HCl•H 2O, and has the following structural formula:
Lidocaine hydrochloride injection, USP in multiple dose vials is a sterile, nonpyrogenic, isotonic, clear, colorless solution containing lidocaine hydrochloride and sodium chloride. Each mL contains 1 mg methylparaben as an antiseptic preservative. The pH of these solutions is adjusted to approximately 6.5 (5.0 to 7.0) with sodium hydroxide and hydrochloric acid.
|
£Quantity is equivalent to 7 mg per mL Lidocaine Hydrochloride, USP (Monohydrate). |
|
| Ingredients | Strength |
| 1% | |
| Amount (Per mL) | |
| Lidocaine Hydrochloride (Anhydrous) | 10 mg £ |
| Sodium Chloride | 7 mg |
| Methylparaben | 1 mg |
| Sodium Hydroxide | Added for pH Adjustment to approximately
6.5 (5.0 to 7.0) |
| Hydrochloric Acid |
8.4 Pediatric Use
Dosages in children should be reduced, commensurate with age, body weight and physical condition [see Dosage and Administration ( 2.6)].
8.5 Geriatric Use
Elderly patients should be given reduced doses commensurate with their age and physical condition [see Dosage and Administration ( 2.6)].
4 Contraindications
Lidocaine hydrochloride injection is contraindicated in patients with a known hypersensitivity to lidocaine or to any local anesthetics of the amide type or to other components of lidocaine hydrochloride injection.
6 Adverse Reactions
The following clinically significant adverse reactions have been reported and described in the Warnings and Precautions section of the labeling:
- Dose-Related Toxicity [see Warnings and Precautions ( 5.1)]
- Methemoglobinemia [see Warnings and Precautions ( 5.2)]
- Chondrolysis with Intra-Articular Infusion [see Warnings and Precautions ( 5.4)]
- Severe, Persistent Hypertension, Cerebrovascular Accidents, and Bradycardia Due to Drug Interactions [see Warnings and Precautions ( 5.5)]
- Allergic-Type Reactions [see Warnings and Precautions ( 5.6)]
- Systemic Toxicities with Unintended Intravascular or Intrathecal Injection [see Warnings and Precautions ( 5.7)]
- Respiratory Arrest Following Retrobulbar Block [see Warnings and Precautions ( 5.14)]
The following adverse reactions from voluntary reports or clinical studies have been reported with lidocaine or lidocaine and epinephrine. Because many of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions to lidocaine hydrochloride injection are characteristic of those associated with other amide-type local anesthetic. A major cause of adverse reactions to this group of drugs is excessive plasma levels, which may be due to overdosage, unintentional intravascular injection, or slow metabolic degradation.
The most commonly encountered acute adverse reactions that demand immediate counter measures were related to the CNS and the cardiovascular system. These adverse reactions were generally dose-related and due to high plasma levels which may have resulted from overdosage, rapid absorption from the injection site, diminished tolerance, or from unintentional intravascular injection of the local anesthetic solution. In addition to systemic does-related toxicity, unintentional intrathecal injection of drug during the intended performance of caudal or lumbar epidural block or nerve blocks near the vertebral column (especially in the head and neck region) has resulted in underventilation or apnea (“Total or High Spinal”). Also, hypertension due to loss of sympathetic tone and respiratory paralysis or underventilation due to cephalad extension of the motor level of anesthesia have occurred. This has led to secondary cardiac arrest when untreated.
When used for dental injections, paresthesia of the lips, tongue, and oral tissues have been reported. Persistent paresthesia lasting weeks to months and, in some instances, lasting greater than one year, have also been reported.
7 Drug Interactions
- Local Anesthetics: The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects when additional local anesthetics are administered. ( 7.1)
- Monoamine Oxidase Inhibitors and Tricyclic Antidepressants: Administration of lidocaine hydrochloride injection to patients receiving monoamine oxidase inhibitors or tricyclic antidepressants may produce severe, prolonged hypertension Concurrent use of these agents should generally be avoided. ( 5.5, 7.2)
- Ergot-type Oxytocic Drugs: Concurrent administration of lidocaine hydrochloride injection and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents. ( 5.5, 7.3)
- Nonselective Beta-Adrenergic Antagonists: Administration of lidocaine hydrochloride injection in patients receiving nonselective beta-adrenergic antagonist may cause severe hypertension and bradycardia. Concurrent use of these agents should generally be avoided. ( 5.5, 7.4)
- Drugs Associated with Methemoglobinemia: Patients are at increased risk of developing methemoglobinemia when concurrently exposed to nitrates, nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants and other drugs. ( 7.5).
- Geriatric Use: Elderly patients should be given reduced doses commensurate with their age and physical condition. ( 8.5)
- Hepatic Impairment: consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment. ( 8.6)
12.2 Pharmacodynamics
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. With central neural blockade these changes may be attributable to block of autonomic fibers, a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system, and/or the beta-adrenergic receptor stimulating action of epinephrine when present. The net effect is normally a modest hypotension when the recommended dosages are not exceeded.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine hydrochloride required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 mcg free base per mL.
12.3 Pharmacokinetics
Systemic plasma levels of lidocaine following lidocaine hydrochloride injection do not correlate with local efficacy.
5.2 Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition [see Drug Interactions ( 7.5)] . If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure and are characterized by a cyanotic skin discoloration and abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue lidocaine hydrochloride injection and any other oxidizing agents. Depending on the severity of the symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. More severe symptoms may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
7.1 Local Anesthetics
The toxic effects of local anesthetics are additive. If coadministration of other local anesthetics with lidocaine hydrochloride injection cannot be avoided, monitor patients for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Warnings and Precautions ( 5.1)].
17.3 Methemoglobinemia
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to stop use and seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue [see Warnings and Precautions ( 5.2)].
AVENACY
Mfd. for Avenacy
Schaumburg, IL 60173 (USA)
Made in India
©2025 Avenacy
March 2025
8.6 Hepatic Impairment
Amide-type local anesthetics such as lidocaine are metabolized by the liver. Patients with severe hepatic impairment, because of their inability to metabolize local anesthetics normally, are at greater risk of developing toxic plasma concentrations and potentially local anesthetic systemic toxicity. Therefore, consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment treated with lidocaine hydrochloride injection, especially with repeat doses [see Warnings and Precautions ( 5.8)].
1 Indications and Usage
Lidocaine hydrochloride injection is indicated in adult and pediatric patients for the production of local or regional anesthesia or analgesia for surgery, dental, and oral surgery procedures, diagnostic and therapeutic procedures, and for obstetrical procedures. Specific concentrations and presentations of lidocaine hydrochloride injection is recommended for each type of block indicated to produce local or regional anesthesia or analgesia [see Dosage and Administration ( 2.2)] .
12.1 Mechanism of Action
Lidocaine hydrochloride stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses thereby effecting local anesthetic action.
5.1 Dose Related Toxicity
The safety and effectiveness of lidocaine hydrochloride injection depends on proper dosage, correct technique, adequate precautions, and readiness for emergencies. Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient's state of consciousness should be performed after injection of lidocaine hydrochloride solutions.
Possible early warning signs of central nervous system (CNS) toxicity are restlessness, anxiety, incoherent speech, lightheadedness, numbness and tingling of the mouth and lips, metallic taste, tinnitus, dizziness, blurred vision, tremors, twitching, CNS depression, or drowsiness. Delay in proper management of dose-related toxicity, underventilation from any cause, and/or altered sensitivity may lead to the development of acidosis, cardiac arrest, and, possibly, death.
During major regional nerve blocks, such as those of the brachial plexus or lower extremity, the patient should have an indwelling intravenous catheter to assure adequate intravenous access. Use the lowest dosage of lidocaine hydrochloride injection that results in effective anesthesia to avoid high plasma levels and serious adverse effects. Avoid rapid injection of a large volume of lidocaine hydrochloride injection solution and administer fractional (incremental) doses when feasible.
Injection of repeated doses of lidocaine hydrochloride injection may cause significant increases in plasma levels with each repeated dose due to slow accumulation of the drug or its metabolites, or to slow metabolic degradation. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients and acutely ill patients should be given reduced doses commensurate with their age and physical status.
5 Warnings and Precautions
- Dose-Related Toxicity: Monitor cardiovascular and respiratory vital signs and patient's state of consciousness after injection of lidocaine hydrochloride injection. ( 5.1)
- Methemoglobinemia: Cases of methemoglobinemia have been reported in association with local anesthetics use. See full prescribing information for more details on managing these risks. ( 5.2)
- Chondrolysis with Intra-Articular Infusion: Avoid Intra-articular infusions as there have been post-marketing reports of chondrolysis in patients receiving such infusion. ( 5.4)
- Allergic-Type Reactions to Sulfites in Lidocaine Hydrochloride Injection and Anaphylactic Reactions:Lidocaine hydrochloride injection without epinephrine does not contain sodium metabisulfite. ( 5.6)
- Risk of Systemic Toxicities with Unintended Intravascular or Intrathecal Injection: Unintended intravascular or intrathecal injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progression ultimately to respiratory arrest. Aspirate for blood or cerebrospinal fluid (where applicable) prior to each dose and consider using a test dose of lidocaine hydrochloride injection. ( 5.7)
2 Dosage and Administration
- See Full Prescribing Information for recommended dosages and administration information for adult and pediatric patients.
17.1 Allergic Type Reactions
3 Dosage Forms and Strengths
- Lidocaine hydrochloride injection, USP: 1%
7.3 Ergot Type Oxytocic Drugs
Concurrent administration of vasopressor drugs (for the treatment of hypotension related to obstetric blocks) and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents. Avoid use of lidocaine hydrochloride injection concomitantly with ergot-type oxytocic drugs [see Warnings and Precautions ( 5.5)].
16 How Supplied/storage and Handling
Lidocaine hydrochloride injection, USP is a clear, colorless solution supplied as follows:
| Lidocaine Hydrochloride Injection, USP (1%) | ||
| NDC | (10 mg per mL) | Package Factor |
| 83634-651-50 | 500 mg per 50 mL Multiple-Dose Vial | 25 vials per carton |
5.13 Familial Malignant Hyperthermia
Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for the management of malignant hyperthermia should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspect triggering agent(s) and institution of treatment, including oxygen therapy, indicated supportive measures and dantrolene (consult dantrolene sodium intravenous package insert before using).
Principal Display Panel Vial Label
Lidocaine HCl Injection, USP - 1%
500 mg per 50 mL (10 mg per mL)
For Infiltration and Nerve Block
Not for Caudal or Epidural Use
50 mL Multiple-Dose Vial
Rx only
5.16 Drug/laboratory Test Interactions
The intramuscular injection of lidocaine HCl may result in an increase in creatine phosphokinase levels. Thus, the use of this enzyme determination, without isoenzyme separation, as a diagnostic test for the presence of acute myocardial infarction may be compromised by the intramuscular injection of lidocaine HCl.
Principal Display Panel Outer Package
NDC 71872-7362-1
1% Lidocaine HCl Injection, USP
500 mg/50 mL (10 mg/mL)
1 x 50 mL Muliple-Dose Vial
Rx only
7.5 Drugs Associated With Methemoglobinemia
Patients that are administered local anesthetics may be at increased risk of developing methemoglobinemia when concurrently exposed to the following oxidizing agents:
| Class | Examples |
| Nitrates/Nitrites | nitroglycerin, nitroprusside, nitric oxide, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic agents | cyclophosphamide, flutamide, rasburicase, ifosfamide, hydroxyurea |
| Antibiotics | dapsone, sulfonamides, nitrofurantoin, para-aminosalicylic acid |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | phenytoin, sodium valproate, phenobarbital |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
7.4 Nonselective Beta Adrenergic Antagonists
Administration of lidocaine hydrochloride injection in patients receiving nonselective beta-adrenergic antagonists may cause severe hypertension and bradycardia. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful monitoring of the patient's blood pressure and heart rate is essential [see Warnings and Precautions ( 5.5)].
5.4 Chondrolysis With Intra Articular Infusion
Intra-articular infusions of local anesthetics following arthroscopic and other surgical procedures is an unapproved use, and there have been post-marketing reports of chondrolysis in patients receiving such infusions. The majority of reported cases of chondrolysis have involved the shoulder joint; cases of gleno-humeral chondrolysis have been described in pediatric and adult patients following intra-articular infusions of local anesthetics with and without epinephrine for periods of 48 to 72 hours. There is insufficient information to determine whether shorter infusion periods are not associated with these findings. The time of onset of symptoms, such as joint pain, stiffness and loss of motion can be variable, but may begin as early as the 2 ndmonth after surgery. Currently, there is no effective treatment for chondrolysis; patients who experienced chondrolysis have required additional diagnostic and therapeutic procedures and some required arthroplasty or shoulder replacement.
2.1 Important Dosage and Administration Information
- Lidocaine hydrochloride injection is not recommended for intrathecal use.
- Avoid use of lidocaine hydrochloride injection solutions containing antimicrobial preservatives (i.e., multiple-dose vials) for epidural or caudal anesthesia [see Warnings and Precautions ( 5.3)] .
- Visually inspect this product for particulate matter and discoloration prior to administration whenever solution and container permit. Lidocaine hydrochloride injection is a clear, colorless solutions. Do not administer solutions which are discolored or contain particulate matter.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever the solution and container permit. Solutions which are discolored (e.g., pinkish or darker than slightly yellow) or which contain particulate matter or precipitate should not be administered.
- Mixing or the prior or intercurrent use of any other local anesthetic with lidocaine hydrochloride injection is not recommended because of insufficient data on the clinical use of such mixtures.
Administration Precautions
- Lidocaine hydrochloride injection is to be administered in carefully adjusted dosages by or under the supervision of experienced clinicians who are well versed in the diagnosis and management of dose-related toxicity and other acute emergencies which might arise from the block to be employed.
- Use lidocaine hydrochloride injection only if the following are immediately available: oxygen, cardiopulmonary resuscitative equipment and drugs, and the personnel resources needed for proper management of toxic reactions and related emergencies [see Warnings and Precautions ( 5.1), Adverse Reactions ( 6), and Overdosage ( 10)].
- The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects related to local anesthetic systemic toxicity when additional local anesthetics are administered with lidocaine hydrochloride injection [see Warnings and Precautions ( 5.1), Drug Interactions ( 7.1), and Overdosage ( 10)].
- Aspirate for blood or cerebrospinal fluid (where applicable) prior to injecting lidocaine hydrochloride injection, both the initial dose and all subsequent doses, to avoid intravascular or intrathecal injection. However, a negative aspiration for blood or cerebrospinal fluid does not ensure against an intravascular or intrathecal injection [see Warnings and Precautions ( 5.7)] .
- Avoid rapid injection of a large volume of lidocaine hydrochloride injection and use fractional (incremental) doses when feasible.
- During major regional nerve blocks, such as those of the brachial plexus or lower extremity, the patient should have an indwelling intravenous catheter to assure adequate intravenous access. The lowest dosage of lidocaine hydrochloride injection that results in effective anesthesia should be used to avoid high plasma levels and serious adverse reactions.
- Perform careful and constant monitoring of cardiovascular and respiratory (adequacy of oxygenation and ventilation) vital signs and the patient's level of consciousness after each local anesthetic injection.
- Use lidocaine hydrochloride injection in carefully restricted quantities in areas of the body supplied by end arteries or having otherwise compromised blood supply such as digits, nose, external ear, or penis [see Warnings and Precautions (5.10)].
5.3 Antimicrobial Preservatives in Multiple Dose Vials
Avoid use of lidocaine hydrochloride injection solutions containing antimicrobial preservatives (i.e., those supplied in multiple-dose vials) for epidural or caudal anesthesia because safety has not been established with such use.
5.8 Risk of Toxicity in Patients With Hepatic Impairment
Because amide local anesthetics such as lidocaine are metabolized by the liver, consider reduced dosing and increased monitoring for lidocaine systemic toxicity in patients with moderate to severe hepatic impairment who are treated with lidocaine hydrochloride injection, especially with repeat doses [see Use in Specific Populations ( 8.6)] .
5.14 Risk of Respiratory Arrest With Use in Ophthalmic Surgery
Clinicians who perform retrobulbar blocks should be aware that there have been reports of respiratory arrest following local anesthetic injection. Prior to retrobulbar block (e.g., with lidocaine hydrochloride injection), as with all other regional procedures, resuscitative equipment and drugs, and personnel to manage respiratory arrest or depression, convulsions, and cardiac stimulation or depression should be immediately available [see Warnings and Precautions ( 5.14)] . As with other anesthetic procedures, patients should be constantly monitored following ophthalmic blocks for signs of these adverse reactions, which may occur following relatively low total doses.
7.2 Monoamine Oxidase Inhibitors and Tricyclic Antidepressants
The administration of lidocaine hydrochloride injection to patients receiving monoamine oxidase inhibitors or tricyclic antidepressants may produce severe, prolonged hypertension. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful monitoring of the patient's hemodynamic status is essential [see Warnings and Precautions ( 5.5)].
5.12 Risk of Adverse Reactions With Use in the Head and Neck Area
Small doses of local anesthetics (e.g., lidocaine hydrochloride injection) injected into the head and neck area, including retrobulbar, dental and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses. The injection procedures require the utmost care. Confusion, convulsions, respiratory depression and/or respiratory arrest, and cardiovascular stimulation or depression have been reported. These reactions may be due to intra-arterial injection of the local anesthetic with retrograde flow to the cerebral circulation. They may also be due to puncture of the dural sheath of the optic nerve during retrobulbar block with diffusion of any local anesthetic along the subdural space to the midbrain. Monitor circulation and respiration and constantly observe patients receiving lidocaine hydrochloride injection blocks. Resuscitative equipment and drugs, and personnel for treating adverse reactions should be immediately available. Dosage recommendations should not be exceeded [see Dosage and Administration ( 2.2)].
5.9 Risk of Use in Patients With Impaired Cardiovascular Function
Lidocaine hydrochloride injection should also be given in reduced doses in patients with impaired cardiovascular function since they may be less able to compensate for functional changes associated with the prolongation of A-V conduction produced by these drugs. Monitor patients closely for blood pressure, heart rate, and ECG changes.
5.7 Risk of Systemic Toxicities With Unintended Intravascular Or Intrathecal Injection
Unintended intravascular or intrathecal injection of lidocaine hydrochloride injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progressing ultimately to respiratory arrest. Unintentional intrathecal injection during the intended performance of nerve blocks near the vertebral column has resulted in underventilation or apnea ("Total or High Spinal"). A high spinal has been characterized by paralysis of the legs, loss of consciousness, respiratory paralysis, and bradycardia [see Adverse Reactions ( 6)] .
Aspirate for blood or cerebrospinal fluid (where applicable) before injecting lidocaine hydrochloride injection, both the initial dose and all subsequent doses, to avoid intravascular or intrathecal injection. However, a negative aspiration for blood or cerebrospinal fluid does not ensure against an intravascular or intrathecal injection.
2.2 Recommended Concentrations and Dosages of Lidocaine Hydrochloride Injection in Adults
The dosage of lidocaine hydrochloride injection administered varies with the anesthetic procedure, the area to be anesthetized, the vascularity of the tissues, the number of neuronal segments to be blocked, the depth of anesthesia and degree of muscle relaxation required, the duration of anesthesia desired, individual tolerance, and the physical condition of the patient. Administer the smallest dosage and concentration required to produce the desired result.
The types of block and recommended lidocaine hydrochloride injection concentrations are shown in Table 1. The dosages suggested in this table are for normal healthy adults and refer to the use of epinephrine-free solutions. Consider administration of solutions containing epinephrine when large volumes are required.
| Procedure | Lidocaine Hydrochloride Injection | ||
| Conc (%) | Vol (mL) | Total Dose (mg) | |
| Infiltration | |||
| Percutaneous | 0.5 or 1 | 1 to 60 | 5 to 300 |
| Peripheral Nerve Blocks, e.g., | |||
| Brachial | 1.5 | 15 to 20 | 225 to 300 |
| Dental | 2 | 1 to 5 | 20 to 100 |
| Intercostal | 1 | 3 | 30 |
| Paravertebral | 1 | 3 to 5 | 30 to 50 |
| Pudendal (each side) | 1 | 10 | 100 |
| Paracervical | |||
| Obstetrical analgesia (each side) | 1 | 10 | 100 |
| Sympathetic Nerve Blocks, e.g., | |||
| Cervical (stellate ganglion) | 1 | 5 | 50 |
| Lumbar | 1 | 5 to 10 | 50 to 100 |
The above suggested concentrations and volumes serve only as a guide. Other volumes and concentrations may be used provided the total maximum recommended dose is not exceeded [see Dosage and Administration (2.5)].
These recommended doses serve only as a guide to the amount of local anesthetic required for most indicated procedures. The actual volumes and concentrations to be used depend on a number of factors such as type and extent of surgical procedure, depth of anesthesia and degree of muscular relaxation required, duration of anesthesia required, and the physical condition of the patient. In all cases, the lowest concentration and smallest dose that will produce the desired result should be given. The maximum dosage limit within the recommended dosage range must be individualized in each case after evaluating the size and physical status of the patient, as well as the anticipated rate of systemic absorption from a particular injection site.
5.6 Allergic Type Reactions to Sulfites in Lidocaine Hydrochloride Injection and Anaphylactic Reactions
Lidocaine hydrochloride injection without epinephrine does not contain sodium metabisulfite.
Anaphylactic reactions may occur following administration of lidocaine hydrochloride [see Adverse Reactions ( 6)]. Lidocaine hydrochloride should be used with caution in persons with known drug sensitivities. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross-sensitivity to lidocaine hydrochloride.
Structured Label Content
Section 42229-5 (42229-5)
Adults
For normal healthy adults, the maximum individual dose should not exceed 4.5 mg/kg of body weight, and in general it is recommended that the maximum total dose does not exceed 300 mg.
The maximum recommended dose per 90 minute period of lidocaine hydrochloride for paracervical block in obstetrical patients and non-obstetrical patients is 200 mg total. One half of the total dose is usually administered to each side. Inject slowly, five minutes between sides [see Pregnancy ( 8.1)].
Section 44425-7 (44425-7)
Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.]
Protect from light.
Sterile, Nonpyrogenic.
The container closure is not made with natural rubber latex.
11 Description (11 DESCRIPTION)
Lidocaine hydrochloride injection, USP contains lidocaine hydrochloride, an amide local anesthetic, as the active pharmaceutical ingredient. The route of administration for Lidocaine hydrochloride injection, USP is by injection, for infiltration and nerve block. Multiple dose vials contain methylparaben and they should not be used for caudal and lumbar epidural blocks.
Lidocaine hydrochloride, is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride monohydrate and has the molecular weight of 288.8 g/mol. Lidocaine hydrochloride molecular formula is C 14H 22N 2O • HCl•H 2O, and has the following structural formula:
Lidocaine hydrochloride injection, USP in multiple dose vials is a sterile, nonpyrogenic, isotonic, clear, colorless solution containing lidocaine hydrochloride and sodium chloride. Each mL contains 1 mg methylparaben as an antiseptic preservative. The pH of these solutions is adjusted to approximately 6.5 (5.0 to 7.0) with sodium hydroxide and hydrochloric acid.
|
£Quantity is equivalent to 7 mg per mL Lidocaine Hydrochloride, USP (Monohydrate). |
|
| Ingredients | Strength |
| 1% | |
| Amount (Per mL) | |
| Lidocaine Hydrochloride (Anhydrous) | 10 mg £ |
| Sodium Chloride | 7 mg |
| Methylparaben | 1 mg |
| Sodium Hydroxide | Added for pH Adjustment to approximately
6.5 (5.0 to 7.0) |
| Hydrochloric Acid |
8.4 Pediatric Use
Dosages in children should be reduced, commensurate with age, body weight and physical condition [see Dosage and Administration ( 2.6)].
8.5 Geriatric Use
Elderly patients should be given reduced doses commensurate with their age and physical condition [see Dosage and Administration ( 2.6)].
4 Contraindications (4 CONTRAINDICATIONS)
Lidocaine hydrochloride injection is contraindicated in patients with a known hypersensitivity to lidocaine or to any local anesthetics of the amide type or to other components of lidocaine hydrochloride injection.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions have been reported and described in the Warnings and Precautions section of the labeling:
- Dose-Related Toxicity [see Warnings and Precautions ( 5.1)]
- Methemoglobinemia [see Warnings and Precautions ( 5.2)]
- Chondrolysis with Intra-Articular Infusion [see Warnings and Precautions ( 5.4)]
- Severe, Persistent Hypertension, Cerebrovascular Accidents, and Bradycardia Due to Drug Interactions [see Warnings and Precautions ( 5.5)]
- Allergic-Type Reactions [see Warnings and Precautions ( 5.6)]
- Systemic Toxicities with Unintended Intravascular or Intrathecal Injection [see Warnings and Precautions ( 5.7)]
- Respiratory Arrest Following Retrobulbar Block [see Warnings and Precautions ( 5.14)]
The following adverse reactions from voluntary reports or clinical studies have been reported with lidocaine or lidocaine and epinephrine. Because many of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions to lidocaine hydrochloride injection are characteristic of those associated with other amide-type local anesthetic. A major cause of adverse reactions to this group of drugs is excessive plasma levels, which may be due to overdosage, unintentional intravascular injection, or slow metabolic degradation.
The most commonly encountered acute adverse reactions that demand immediate counter measures were related to the CNS and the cardiovascular system. These adverse reactions were generally dose-related and due to high plasma levels which may have resulted from overdosage, rapid absorption from the injection site, diminished tolerance, or from unintentional intravascular injection of the local anesthetic solution. In addition to systemic does-related toxicity, unintentional intrathecal injection of drug during the intended performance of caudal or lumbar epidural block or nerve blocks near the vertebral column (especially in the head and neck region) has resulted in underventilation or apnea (“Total or High Spinal”). Also, hypertension due to loss of sympathetic tone and respiratory paralysis or underventilation due to cephalad extension of the motor level of anesthesia have occurred. This has led to secondary cardiac arrest when untreated.
When used for dental injections, paresthesia of the lips, tongue, and oral tissues have been reported. Persistent paresthesia lasting weeks to months and, in some instances, lasting greater than one year, have also been reported.
7 Drug Interactions (7 DRUG INTERACTIONS)
- Local Anesthetics: The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects when additional local anesthetics are administered. ( 7.1)
- Monoamine Oxidase Inhibitors and Tricyclic Antidepressants: Administration of lidocaine hydrochloride injection to patients receiving monoamine oxidase inhibitors or tricyclic antidepressants may produce severe, prolonged hypertension Concurrent use of these agents should generally be avoided. ( 5.5, 7.2)
- Ergot-type Oxytocic Drugs: Concurrent administration of lidocaine hydrochloride injection and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents. ( 5.5, 7.3)
- Nonselective Beta-Adrenergic Antagonists: Administration of lidocaine hydrochloride injection in patients receiving nonselective beta-adrenergic antagonist may cause severe hypertension and bradycardia. Concurrent use of these agents should generally be avoided. ( 5.5, 7.4)
- Drugs Associated with Methemoglobinemia: Patients are at increased risk of developing methemoglobinemia when concurrently exposed to nitrates, nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants and other drugs. ( 7.5).
- Geriatric Use: Elderly patients should be given reduced doses commensurate with their age and physical condition. ( 8.5)
- Hepatic Impairment: consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment. ( 8.6)
12.2 Pharmacodynamics
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. With central neural blockade these changes may be attributable to block of autonomic fibers, a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system, and/or the beta-adrenergic receptor stimulating action of epinephrine when present. The net effect is normally a modest hypotension when the recommended dosages are not exceeded.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine hydrochloride required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 mcg free base per mL.
12.3 Pharmacokinetics
Systemic plasma levels of lidocaine following lidocaine hydrochloride injection do not correlate with local efficacy.
5.2 Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition [see Drug Interactions ( 7.5)] . If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure and are characterized by a cyanotic skin discoloration and abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue lidocaine hydrochloride injection and any other oxidizing agents. Depending on the severity of the symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. More severe symptoms may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
7.1 Local Anesthetics
The toxic effects of local anesthetics are additive. If coadministration of other local anesthetics with lidocaine hydrochloride injection cannot be avoided, monitor patients for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Warnings and Precautions ( 5.1)].
17.3 Methemoglobinemia
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to stop use and seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue [see Warnings and Precautions ( 5.2)].
AVENACY
Mfd. for Avenacy
Schaumburg, IL 60173 (USA)
Made in India
©2025 Avenacy
March 2025
8.6 Hepatic Impairment
Amide-type local anesthetics such as lidocaine are metabolized by the liver. Patients with severe hepatic impairment, because of their inability to metabolize local anesthetics normally, are at greater risk of developing toxic plasma concentrations and potentially local anesthetic systemic toxicity. Therefore, consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with hepatic impairment treated with lidocaine hydrochloride injection, especially with repeat doses [see Warnings and Precautions ( 5.8)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Lidocaine hydrochloride injection is indicated in adult and pediatric patients for the production of local or regional anesthesia or analgesia for surgery, dental, and oral surgery procedures, diagnostic and therapeutic procedures, and for obstetrical procedures. Specific concentrations and presentations of lidocaine hydrochloride injection is recommended for each type of block indicated to produce local or regional anesthesia or analgesia [see Dosage and Administration ( 2.2)] .
12.1 Mechanism of Action
Lidocaine hydrochloride stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses thereby effecting local anesthetic action.
5.1 Dose Related Toxicity (5.1 Dose-Related Toxicity)
The safety and effectiveness of lidocaine hydrochloride injection depends on proper dosage, correct technique, adequate precautions, and readiness for emergencies. Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient's state of consciousness should be performed after injection of lidocaine hydrochloride solutions.
Possible early warning signs of central nervous system (CNS) toxicity are restlessness, anxiety, incoherent speech, lightheadedness, numbness and tingling of the mouth and lips, metallic taste, tinnitus, dizziness, blurred vision, tremors, twitching, CNS depression, or drowsiness. Delay in proper management of dose-related toxicity, underventilation from any cause, and/or altered sensitivity may lead to the development of acidosis, cardiac arrest, and, possibly, death.
During major regional nerve blocks, such as those of the brachial plexus or lower extremity, the patient should have an indwelling intravenous catheter to assure adequate intravenous access. Use the lowest dosage of lidocaine hydrochloride injection that results in effective anesthesia to avoid high plasma levels and serious adverse effects. Avoid rapid injection of a large volume of lidocaine hydrochloride injection solution and administer fractional (incremental) doses when feasible.
Injection of repeated doses of lidocaine hydrochloride injection may cause significant increases in plasma levels with each repeated dose due to slow accumulation of the drug or its metabolites, or to slow metabolic degradation. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients and acutely ill patients should be given reduced doses commensurate with their age and physical status.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Dose-Related Toxicity: Monitor cardiovascular and respiratory vital signs and patient's state of consciousness after injection of lidocaine hydrochloride injection. ( 5.1)
- Methemoglobinemia: Cases of methemoglobinemia have been reported in association with local anesthetics use. See full prescribing information for more details on managing these risks. ( 5.2)
- Chondrolysis with Intra-Articular Infusion: Avoid Intra-articular infusions as there have been post-marketing reports of chondrolysis in patients receiving such infusion. ( 5.4)
- Allergic-Type Reactions to Sulfites in Lidocaine Hydrochloride Injection and Anaphylactic Reactions:Lidocaine hydrochloride injection without epinephrine does not contain sodium metabisulfite. ( 5.6)
- Risk of Systemic Toxicities with Unintended Intravascular or Intrathecal Injection: Unintended intravascular or intrathecal injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progression ultimately to respiratory arrest. Aspirate for blood or cerebrospinal fluid (where applicable) prior to each dose and consider using a test dose of lidocaine hydrochloride injection. ( 5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- See Full Prescribing Information for recommended dosages and administration information for adult and pediatric patients.
17.1 Allergic Type Reactions (17.1 Allergic-Type Reactions)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Lidocaine hydrochloride injection, USP: 1%
7.3 Ergot Type Oxytocic Drugs (7.3 Ergot-Type Oxytocic Drugs)
Concurrent administration of vasopressor drugs (for the treatment of hypotension related to obstetric blocks) and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents. Avoid use of lidocaine hydrochloride injection concomitantly with ergot-type oxytocic drugs [see Warnings and Precautions ( 5.5)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Lidocaine hydrochloride injection, USP is a clear, colorless solution supplied as follows:
| Lidocaine Hydrochloride Injection, USP (1%) | ||
| NDC | (10 mg per mL) | Package Factor |
| 83634-651-50 | 500 mg per 50 mL Multiple-Dose Vial | 25 vials per carton |
5.13 Familial Malignant Hyperthermia
Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for the management of malignant hyperthermia should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspect triggering agent(s) and institution of treatment, including oxygen therapy, indicated supportive measures and dantrolene (consult dantrolene sodium intravenous package insert before using).
Principal Display Panel Vial Label (PRINCIPAL DISPLAY PANEL - VIAL LABEL)
Lidocaine HCl Injection, USP - 1%
500 mg per 50 mL (10 mg per mL)
For Infiltration and Nerve Block
Not for Caudal or Epidural Use
50 mL Multiple-Dose Vial
Rx only
5.16 Drug/laboratory Test Interactions (5.16 Drug/Laboratory Test Interactions)
The intramuscular injection of lidocaine HCl may result in an increase in creatine phosphokinase levels. Thus, the use of this enzyme determination, without isoenzyme separation, as a diagnostic test for the presence of acute myocardial infarction may be compromised by the intramuscular injection of lidocaine HCl.
Principal Display Panel Outer Package (PRINCIPAL DISPLAY PANEL - OUTER PACKAGE)
NDC 71872-7362-1
1% Lidocaine HCl Injection, USP
500 mg/50 mL (10 mg/mL)
1 x 50 mL Muliple-Dose Vial
Rx only
7.5 Drugs Associated With Methemoglobinemia (7.5 Drugs Associated with Methemoglobinemia)
Patients that are administered local anesthetics may be at increased risk of developing methemoglobinemia when concurrently exposed to the following oxidizing agents:
| Class | Examples |
| Nitrates/Nitrites | nitroglycerin, nitroprusside, nitric oxide, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic agents | cyclophosphamide, flutamide, rasburicase, ifosfamide, hydroxyurea |
| Antibiotics | dapsone, sulfonamides, nitrofurantoin, para-aminosalicylic acid |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | phenytoin, sodium valproate, phenobarbital |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
7.4 Nonselective Beta Adrenergic Antagonists (7.4 Nonselective Beta-Adrenergic Antagonists)
Administration of lidocaine hydrochloride injection in patients receiving nonselective beta-adrenergic antagonists may cause severe hypertension and bradycardia. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful monitoring of the patient's blood pressure and heart rate is essential [see Warnings and Precautions ( 5.5)].
5.4 Chondrolysis With Intra Articular Infusion (5.4 Chondrolysis with Intra-Articular Infusion)
Intra-articular infusions of local anesthetics following arthroscopic and other surgical procedures is an unapproved use, and there have been post-marketing reports of chondrolysis in patients receiving such infusions. The majority of reported cases of chondrolysis have involved the shoulder joint; cases of gleno-humeral chondrolysis have been described in pediatric and adult patients following intra-articular infusions of local anesthetics with and without epinephrine for periods of 48 to 72 hours. There is insufficient information to determine whether shorter infusion periods are not associated with these findings. The time of onset of symptoms, such as joint pain, stiffness and loss of motion can be variable, but may begin as early as the 2 ndmonth after surgery. Currently, there is no effective treatment for chondrolysis; patients who experienced chondrolysis have required additional diagnostic and therapeutic procedures and some required arthroplasty or shoulder replacement.
2.1 Important Dosage and Administration Information
- Lidocaine hydrochloride injection is not recommended for intrathecal use.
- Avoid use of lidocaine hydrochloride injection solutions containing antimicrobial preservatives (i.e., multiple-dose vials) for epidural or caudal anesthesia [see Warnings and Precautions ( 5.3)] .
- Visually inspect this product for particulate matter and discoloration prior to administration whenever solution and container permit. Lidocaine hydrochloride injection is a clear, colorless solutions. Do not administer solutions which are discolored or contain particulate matter.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever the solution and container permit. Solutions which are discolored (e.g., pinkish or darker than slightly yellow) or which contain particulate matter or precipitate should not be administered.
- Mixing or the prior or intercurrent use of any other local anesthetic with lidocaine hydrochloride injection is not recommended because of insufficient data on the clinical use of such mixtures.
Administration Precautions
- Lidocaine hydrochloride injection is to be administered in carefully adjusted dosages by or under the supervision of experienced clinicians who are well versed in the diagnosis and management of dose-related toxicity and other acute emergencies which might arise from the block to be employed.
- Use lidocaine hydrochloride injection only if the following are immediately available: oxygen, cardiopulmonary resuscitative equipment and drugs, and the personnel resources needed for proper management of toxic reactions and related emergencies [see Warnings and Precautions ( 5.1), Adverse Reactions ( 6), and Overdosage ( 10)].
- The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects related to local anesthetic systemic toxicity when additional local anesthetics are administered with lidocaine hydrochloride injection [see Warnings and Precautions ( 5.1), Drug Interactions ( 7.1), and Overdosage ( 10)].
- Aspirate for blood or cerebrospinal fluid (where applicable) prior to injecting lidocaine hydrochloride injection, both the initial dose and all subsequent doses, to avoid intravascular or intrathecal injection. However, a negative aspiration for blood or cerebrospinal fluid does not ensure against an intravascular or intrathecal injection [see Warnings and Precautions ( 5.7)] .
- Avoid rapid injection of a large volume of lidocaine hydrochloride injection and use fractional (incremental) doses when feasible.
- During major regional nerve blocks, such as those of the brachial plexus or lower extremity, the patient should have an indwelling intravenous catheter to assure adequate intravenous access. The lowest dosage of lidocaine hydrochloride injection that results in effective anesthesia should be used to avoid high plasma levels and serious adverse reactions.
- Perform careful and constant monitoring of cardiovascular and respiratory (adequacy of oxygenation and ventilation) vital signs and the patient's level of consciousness after each local anesthetic injection.
- Use lidocaine hydrochloride injection in carefully restricted quantities in areas of the body supplied by end arteries or having otherwise compromised blood supply such as digits, nose, external ear, or penis [see Warnings and Precautions (5.10)].
5.3 Antimicrobial Preservatives in Multiple Dose Vials (5.3 Antimicrobial Preservatives in Multiple-Dose Vials)
Avoid use of lidocaine hydrochloride injection solutions containing antimicrobial preservatives (i.e., those supplied in multiple-dose vials) for epidural or caudal anesthesia because safety has not been established with such use.
5.8 Risk of Toxicity in Patients With Hepatic Impairment (5.8 Risk of Toxicity in Patients with Hepatic Impairment)
Because amide local anesthetics such as lidocaine are metabolized by the liver, consider reduced dosing and increased monitoring for lidocaine systemic toxicity in patients with moderate to severe hepatic impairment who are treated with lidocaine hydrochloride injection, especially with repeat doses [see Use in Specific Populations ( 8.6)] .
5.14 Risk of Respiratory Arrest With Use in Ophthalmic Surgery (5.14 Risk of Respiratory Arrest with Use in Ophthalmic Surgery)
Clinicians who perform retrobulbar blocks should be aware that there have been reports of respiratory arrest following local anesthetic injection. Prior to retrobulbar block (e.g., with lidocaine hydrochloride injection), as with all other regional procedures, resuscitative equipment and drugs, and personnel to manage respiratory arrest or depression, convulsions, and cardiac stimulation or depression should be immediately available [see Warnings and Precautions ( 5.14)] . As with other anesthetic procedures, patients should be constantly monitored following ophthalmic blocks for signs of these adverse reactions, which may occur following relatively low total doses.
7.2 Monoamine Oxidase Inhibitors and Tricyclic Antidepressants
The administration of lidocaine hydrochloride injection to patients receiving monoamine oxidase inhibitors or tricyclic antidepressants may produce severe, prolonged hypertension. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful monitoring of the patient's hemodynamic status is essential [see Warnings and Precautions ( 5.5)].
5.12 Risk of Adverse Reactions With Use in the Head and Neck Area (5.12 Risk of Adverse Reactions with Use in the Head and Neck Area)
Small doses of local anesthetics (e.g., lidocaine hydrochloride injection) injected into the head and neck area, including retrobulbar, dental and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses. The injection procedures require the utmost care. Confusion, convulsions, respiratory depression and/or respiratory arrest, and cardiovascular stimulation or depression have been reported. These reactions may be due to intra-arterial injection of the local anesthetic with retrograde flow to the cerebral circulation. They may also be due to puncture of the dural sheath of the optic nerve during retrobulbar block with diffusion of any local anesthetic along the subdural space to the midbrain. Monitor circulation and respiration and constantly observe patients receiving lidocaine hydrochloride injection blocks. Resuscitative equipment and drugs, and personnel for treating adverse reactions should be immediately available. Dosage recommendations should not be exceeded [see Dosage and Administration ( 2.2)].
5.9 Risk of Use in Patients With Impaired Cardiovascular Function (5.9 Risk of Use in Patients with Impaired Cardiovascular Function)
Lidocaine hydrochloride injection should also be given in reduced doses in patients with impaired cardiovascular function since they may be less able to compensate for functional changes associated with the prolongation of A-V conduction produced by these drugs. Monitor patients closely for blood pressure, heart rate, and ECG changes.
5.7 Risk of Systemic Toxicities With Unintended Intravascular Or Intrathecal Injection (5.7 Risk of Systemic Toxicities with Unintended Intravascular or Intrathecal Injection)
Unintended intravascular or intrathecal injection of lidocaine hydrochloride injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progressing ultimately to respiratory arrest. Unintentional intrathecal injection during the intended performance of nerve blocks near the vertebral column has resulted in underventilation or apnea ("Total or High Spinal"). A high spinal has been characterized by paralysis of the legs, loss of consciousness, respiratory paralysis, and bradycardia [see Adverse Reactions ( 6)] .
Aspirate for blood or cerebrospinal fluid (where applicable) before injecting lidocaine hydrochloride injection, both the initial dose and all subsequent doses, to avoid intravascular or intrathecal injection. However, a negative aspiration for blood or cerebrospinal fluid does not ensure against an intravascular or intrathecal injection.
2.2 Recommended Concentrations and Dosages of Lidocaine Hydrochloride Injection in Adults
The dosage of lidocaine hydrochloride injection administered varies with the anesthetic procedure, the area to be anesthetized, the vascularity of the tissues, the number of neuronal segments to be blocked, the depth of anesthesia and degree of muscle relaxation required, the duration of anesthesia desired, individual tolerance, and the physical condition of the patient. Administer the smallest dosage and concentration required to produce the desired result.
The types of block and recommended lidocaine hydrochloride injection concentrations are shown in Table 1. The dosages suggested in this table are for normal healthy adults and refer to the use of epinephrine-free solutions. Consider administration of solutions containing epinephrine when large volumes are required.
| Procedure | Lidocaine Hydrochloride Injection | ||
| Conc (%) | Vol (mL) | Total Dose (mg) | |
| Infiltration | |||
| Percutaneous | 0.5 or 1 | 1 to 60 | 5 to 300 |
| Peripheral Nerve Blocks, e.g., | |||
| Brachial | 1.5 | 15 to 20 | 225 to 300 |
| Dental | 2 | 1 to 5 | 20 to 100 |
| Intercostal | 1 | 3 | 30 |
| Paravertebral | 1 | 3 to 5 | 30 to 50 |
| Pudendal (each side) | 1 | 10 | 100 |
| Paracervical | |||
| Obstetrical analgesia (each side) | 1 | 10 | 100 |
| Sympathetic Nerve Blocks, e.g., | |||
| Cervical (stellate ganglion) | 1 | 5 | 50 |
| Lumbar | 1 | 5 to 10 | 50 to 100 |
The above suggested concentrations and volumes serve only as a guide. Other volumes and concentrations may be used provided the total maximum recommended dose is not exceeded [see Dosage and Administration (2.5)].
These recommended doses serve only as a guide to the amount of local anesthetic required for most indicated procedures. The actual volumes and concentrations to be used depend on a number of factors such as type and extent of surgical procedure, depth of anesthesia and degree of muscular relaxation required, duration of anesthesia required, and the physical condition of the patient. In all cases, the lowest concentration and smallest dose that will produce the desired result should be given. The maximum dosage limit within the recommended dosage range must be individualized in each case after evaluating the size and physical status of the patient, as well as the anticipated rate of systemic absorption from a particular injection site.
5.6 Allergic Type Reactions to Sulfites in Lidocaine Hydrochloride Injection and Anaphylactic Reactions (5.6 Allergic-Type Reactions to Sulfites in Lidocaine Hydrochloride Injection and Anaphylactic Reactions)
Lidocaine hydrochloride injection without epinephrine does not contain sodium metabisulfite.
Anaphylactic reactions may occur following administration of lidocaine hydrochloride [see Adverse Reactions ( 6)]. Lidocaine hydrochloride should be used with caution in persons with known drug sensitivities. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross-sensitivity to lidocaine hydrochloride.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:25.814170 · Updated: 2026-03-14T22:40:22.461215