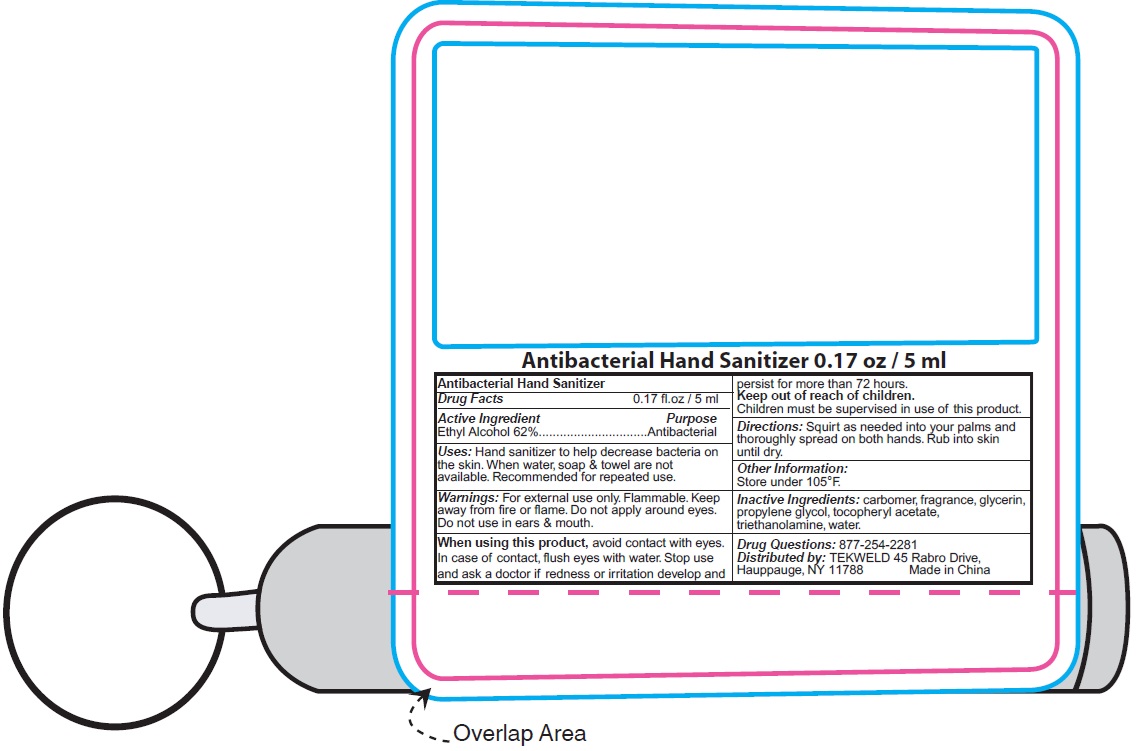
Antibacterial Hand Sanitizer
45af86a4-a413-0309-e054-00144ff88e88
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyl Alcohol 62%
Purpose
Antibacterial
Medication Information
Purpose
Antibacterial
Description
Ethyl Alcohol 62%
Uses:
For hand washing to decrease bacteria on the skin.
Warnings:
Flammable.Keep away from fire or flame. For external use only.
Drug Facts
Directions:
Wet hands thoroughly with product and allow to dry without wiping.
Active Ingredients
Ethyl Alcohol 62%
Other Information:
Store under 105°F.
Inactive Ingredients:
Carbomer, Fragrance, Glycerin, Propylene Glycol, Tocopheryl Acetate, Triethanolamine, Water.
Questions Or Comments?
www.tekweld.com
When Using This Product
Keep out of eyes. In case of contact with eyes, rinse with water.
Stop Use and Ask A Doctor
if irritation and redness develops and persists.
Package Labeling:71160 189 05
Package Labeling:71160 189 10
Keep Out of Reach of Children.
If swallowed, get medical help promptly and contact Poison Control Center.
Package Labeling: 70412 189 20
Structured Label Content
Uses:
For hand washing to decrease bacteria on the skin.
Purpose
Antibacterial
Warnings:
Flammable.Keep away from fire or flame. For external use only.
Drug Facts
Directions:
Wet hands thoroughly with product and allow to dry without wiping.
Active Ingredients
Ethyl Alcohol 62%
Other Information:
Store under 105°F.
Inactive Ingredients:
Carbomer, Fragrance, Glycerin, Propylene Glycol, Tocopheryl Acetate, Triethanolamine, Water.
Questions Or Comments? (Questions or Comments?)
www.tekweld.com
When Using This Product (When using this product)
Keep out of eyes. In case of contact with eyes, rinse with water.
Stop Use and Ask A Doctor (Stop use and ask a doctor)
if irritation and redness develops and persists.
Package Labeling:71160 189 05 (Package Labeling:71160-189-05)
Package Labeling:71160 189 10 (Package Labeling:71160-189-10)
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help promptly and contact Poison Control Center.
Package Labeling: 70412 189 20 (Package Labeling: 70412-189-20)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:46.056323 · Updated: 2026-03-14T23:02:29.359860