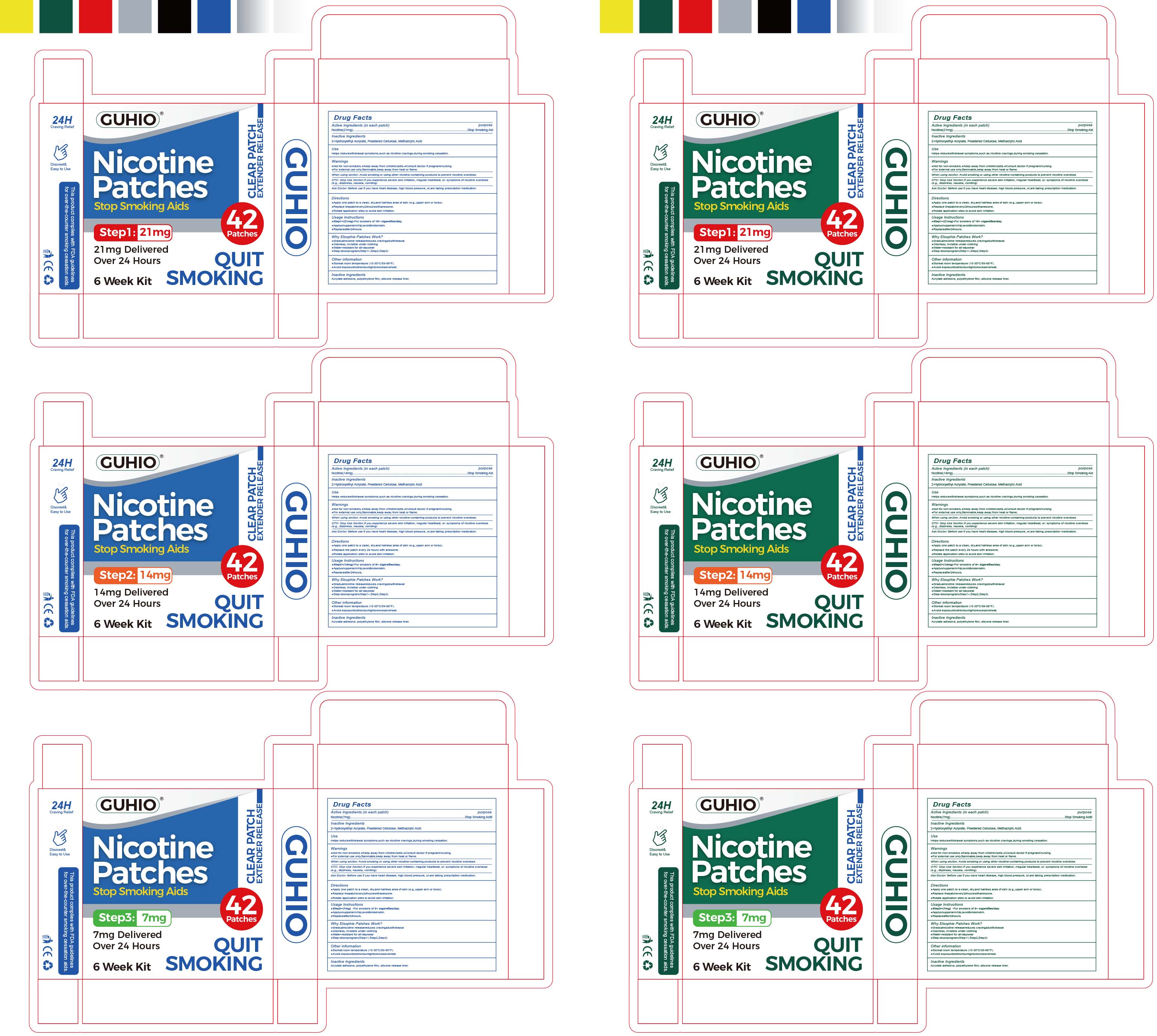
87278-001 Guhio Nicotine Patch
45a53cd6-3e3f-697a-e063-6394a90a6c83
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Nicotine
Medication Information
Warnings and Precautions
Not for non-smokers. Keep away from children/pets. Consult doctor if pregnant/nursing
For external use only.flammable,keep away from heat or flame.
Indications and Usage
Helps reducewithdrawal symptoms,such as nicotine cravings,during smoking cessation.
Dosage and Administration
Apply one patch to a clean, dry,and hairless area of skin (e.g.,upper arm or torso)
Replace thepatchevery24hourswithanewone
Rotate application sites to avoid skin irritation
Description
Nicotine
Section 50565-1
Keep away from children/pets. Consult doctor if pregnant/nursing
Section 50566-9
OTC- Stop Use Section.lf you experience severe skin iritation, rregular heartbeat, or: symptoms of nicotine overdose (e.g.,dizziness, nausea, vomiting)
Section 50567-7
When using section. Avoid smoking or using other nicotine-containing products to prevent nicotine overdose
Ask Doctor. Before use if you have heart disease, high blood pressure, or.are taking prescription medication.
Section 50570-1
Not for non-smokers.
Section 51727-6
2-Hydroxyethyl Acrylate, Powdered Cellulose, Methacrylic Acid
Section 51945-4
Section 55105-1
Stop Smoking Aid
Section 55106-9
Nicotine
Structured Label Content
Indications and Usage (34067-9)
Helps reducewithdrawal symptoms,such as nicotine cravings,during smoking cessation.
Dosage and Administration (34068-7)
Apply one patch to a clean, dry,and hairless area of skin (e.g.,upper arm or torso)
Replace thepatchevery24hourswithanewone
Rotate application sites to avoid skin irritation
Warnings and Precautions (34071-1)
Not for non-smokers. Keep away from children/pets. Consult doctor if pregnant/nursing
For external use only.flammable,keep away from heat or flame.
Section 50565-1 (50565-1)
Keep away from children/pets. Consult doctor if pregnant/nursing
Section 50566-9 (50566-9)
OTC- Stop Use Section.lf you experience severe skin iritation, rregular heartbeat, or: symptoms of nicotine overdose (e.g.,dizziness, nausea, vomiting)
Section 50567-7 (50567-7)
When using section. Avoid smoking or using other nicotine-containing products to prevent nicotine overdose
Ask Doctor. Before use if you have heart disease, high blood pressure, or.are taking prescription medication.
Section 50570-1 (50570-1)
Not for non-smokers.
Section 51727-6 (51727-6)
2-Hydroxyethyl Acrylate, Powdered Cellulose, Methacrylic Acid
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Stop Smoking Aid
Section 55106-9 (55106-9)
Nicotine
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:36.780940 · Updated: 2026-03-14T23:11:16.641314