84291-005
45944b40-d43b-a354-e063-6394a90ad3c5
34390-5
HUMAN OTC DRUG LABEL
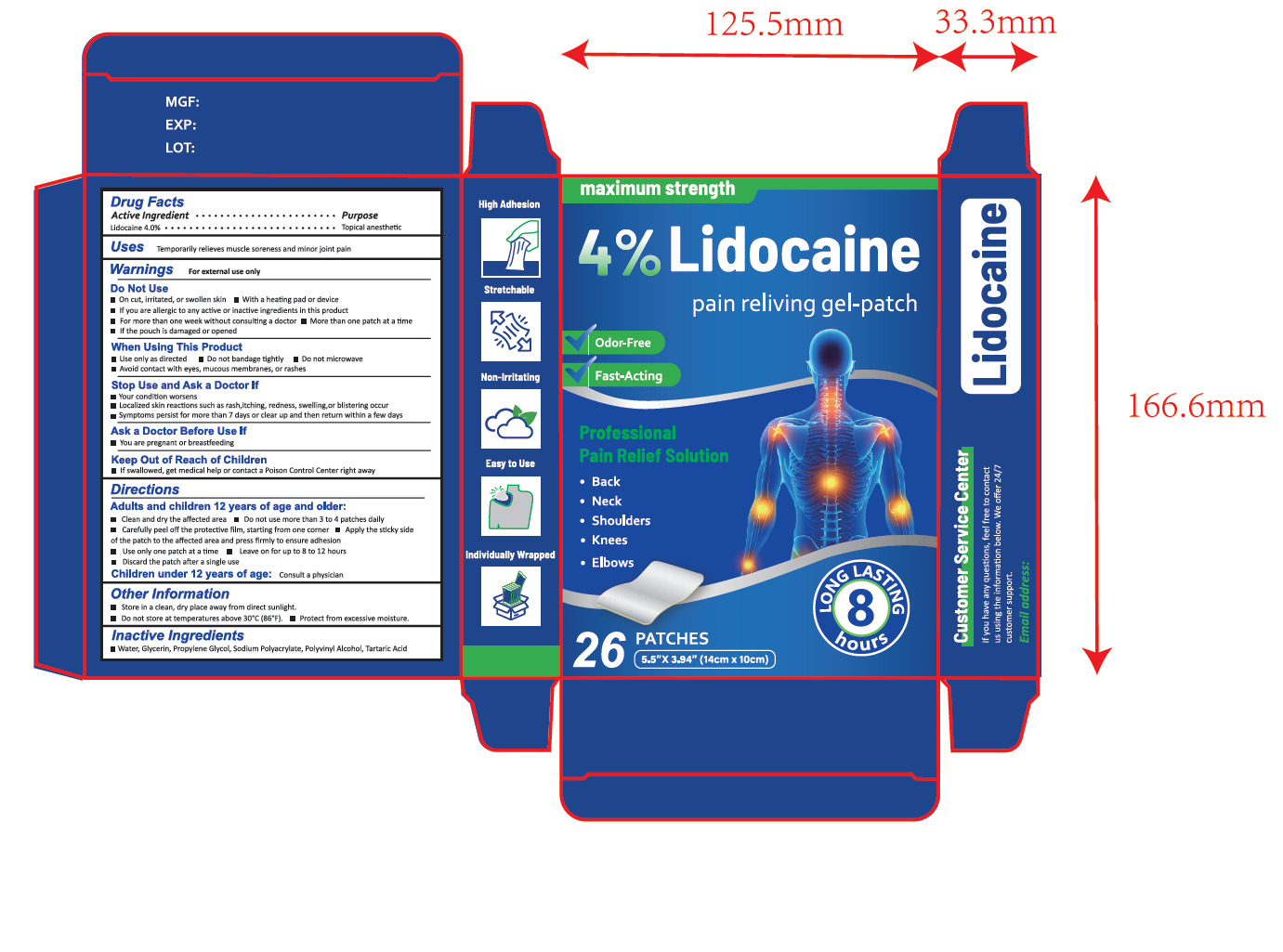
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4.0%
Purpose
Topical anesthetic
Medication Information
Purpose
Topical anesthetic
Description
Lidocaine 4.0%
Use
Temporarily relieves muscle soreness and minor joint pain
Section 50565-1
If swallowed, get medical help or contact a Poison Control Center right away
Section 50566-9
Your condition worsens
Localized skin reactions such as rash,itching, redness, swelling,or blistering occur
Symptoms persist for more than 7 days or clear up and then return within a few days
Section 50567-7
Use only as directed
Do not bandage tightly
Do not microwave
Avoid contact with eyes, mucous membranes, or rashes
Warnings
For external use only
Directions
Adults and children 12 years of age and older:
■ Clean and dry the affected area
■ Do not use more than 3 to 4 patches daily
■ Carefully peel off the protective film, starting from one corner
■ Apply the sticky side of the patch to the affected area and press firmly to ensure adhesion
■ Use only one patch at a time
■ Leave on for up to 8 to 12 hours
■ Discard the patch after a single use
Children under 12 years of age:Consult a physician
Do Not Use
■On cut, irritated, or swollen skin ■ With a heating pad or device
■If you are allergic to any active or inactive ingredients in this product
■For more than one week without consulting a doctor ■More than one patch at a time
■If the pouch is damaged or opened
Other Information
Store in a clean, dry place away from direct sunlight.
Do not store at temperatures above 30°C (86°F).
Protect from excessive moisture..
Active Ingredient(s)
Lidocaine 4.0%
Inactive Ingredients
Water, Glycerin, Propylene Glycol, Sodium Polyacrylate, Polyvinyl Alcohol, Tartaric Acid
Package Label Principal Display Panel
Structured Label Content
Use
Temporarily relieves muscle soreness and minor joint pain
Section 50565-1 (50565-1)
If swallowed, get medical help or contact a Poison Control Center right away
Section 50566-9 (50566-9)
Your condition worsens
Localized skin reactions such as rash,itching, redness, swelling,or blistering occur
Symptoms persist for more than 7 days or clear up and then return within a few days
Section 50567-7 (50567-7)
Use only as directed
Do not bandage tightly
Do not microwave
Avoid contact with eyes, mucous membranes, or rashes
Purpose
Topical anesthetic
Warnings
For external use only
Directions
Adults and children 12 years of age and older:
■ Clean and dry the affected area
■ Do not use more than 3 to 4 patches daily
■ Carefully peel off the protective film, starting from one corner
■ Apply the sticky side of the patch to the affected area and press firmly to ensure adhesion
■ Use only one patch at a time
■ Leave on for up to 8 to 12 hours
■ Discard the patch after a single use
Children under 12 years of age:Consult a physician
Do Not Use (Do not use)
■On cut, irritated, or swollen skin ■ With a heating pad or device
■If you are allergic to any active or inactive ingredients in this product
■For more than one week without consulting a doctor ■More than one patch at a time
■If the pouch is damaged or opened
Other Information (Other information)
Store in a clean, dry place away from direct sunlight.
Do not store at temperatures above 30°C (86°F).
Protect from excessive moisture..
Active Ingredient(s)
Lidocaine 4.0%
Inactive Ingredients (Inactive ingredients)
Water, Glycerin, Propylene Glycol, Sodium Polyacrylate, Polyvinyl Alcohol, Tartaric Acid
Package Label Principal Display Panel (Package Label - Principal Display Panel)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:36.775335 · Updated: 2026-03-14T23:11:28.912883