4526bacf-6451-b297-e063-6294a90af71a
4526bacf-6451-b297-e063-6294a90af71a
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Apply generously and evenly 15 minutes before sun exposure. Children under 6 months of age: Ask a doctor. Reapply at least every 2 hours. Use a water-resistant sunscreen if swimming or sweating.
Medication Information
Warnings and Precautions
- For external use only.
- Do not use on damaged or broken skin.
- When using this product, keep out of eyes. Rinse with water to remove.
- Stop use and ask a doctor if rash occurs.
- Keep out of reach of children.
Active Ingredient
Homosalate 14.00%
Octocrylene 10.00%
Octisalate 4.50%
Avobenzone 2.80%
Indications and Usage
- Helps prevent sunburn
- When used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun.
Dosage and Administration
- Apply generously and evenly 15 minutes before sun exposure.
- Children under 6 months of age: Ask a doctor.
- Reapply at least every 2 hours.
- Use a water-resistant sunscreen if swimming or sweating.
Description
Apply generously and evenly 15 minutes before sun exposure. Children under 6 months of age: Ask a doctor. Reapply at least every 2 hours. Use a water-resistant sunscreen if swimming or sweating.
Section 50565-1
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50570-1
No “Do not use” statements apply for this product when used as directed.
Section 51727-6
Water (Aqua)
Glycerin
Butyloctyl Salicylate
Propanediol
Dibutyl Adipate
Polymethylsilsesquioxane
Cetearyl Olivate
Caprylyl Methicone
1,2-Hexanediol
Pentylene Glycol
VP/Eicosene Copolymer
Sorbitan Olivate
Potassium Cetyl Phosphate
Panthenol
Glyceryl Stearate
Butylene Glycol
Cetearyl Alcohol
Polyacrylate Crosspolymer-6
Methylpropanediol
Ammonium Acryloyldimethyltaurate/VP Copolymer
Xanthan Gum
Ethylhexylglycerin
Undaria Pinnatifida Extract
Tromethamine
Spirulina Platensis Extract
Plankton Extract
Ecklonia Cava Extract
Codium Fragile Extract
Hyaluronic Acid
Polyether-1
Portulaca Oleracea Extract
Beta-Glucan
T-Butyl Alcohol
Tocopherol
Biosaccharide Gum-1
Section 51945-4
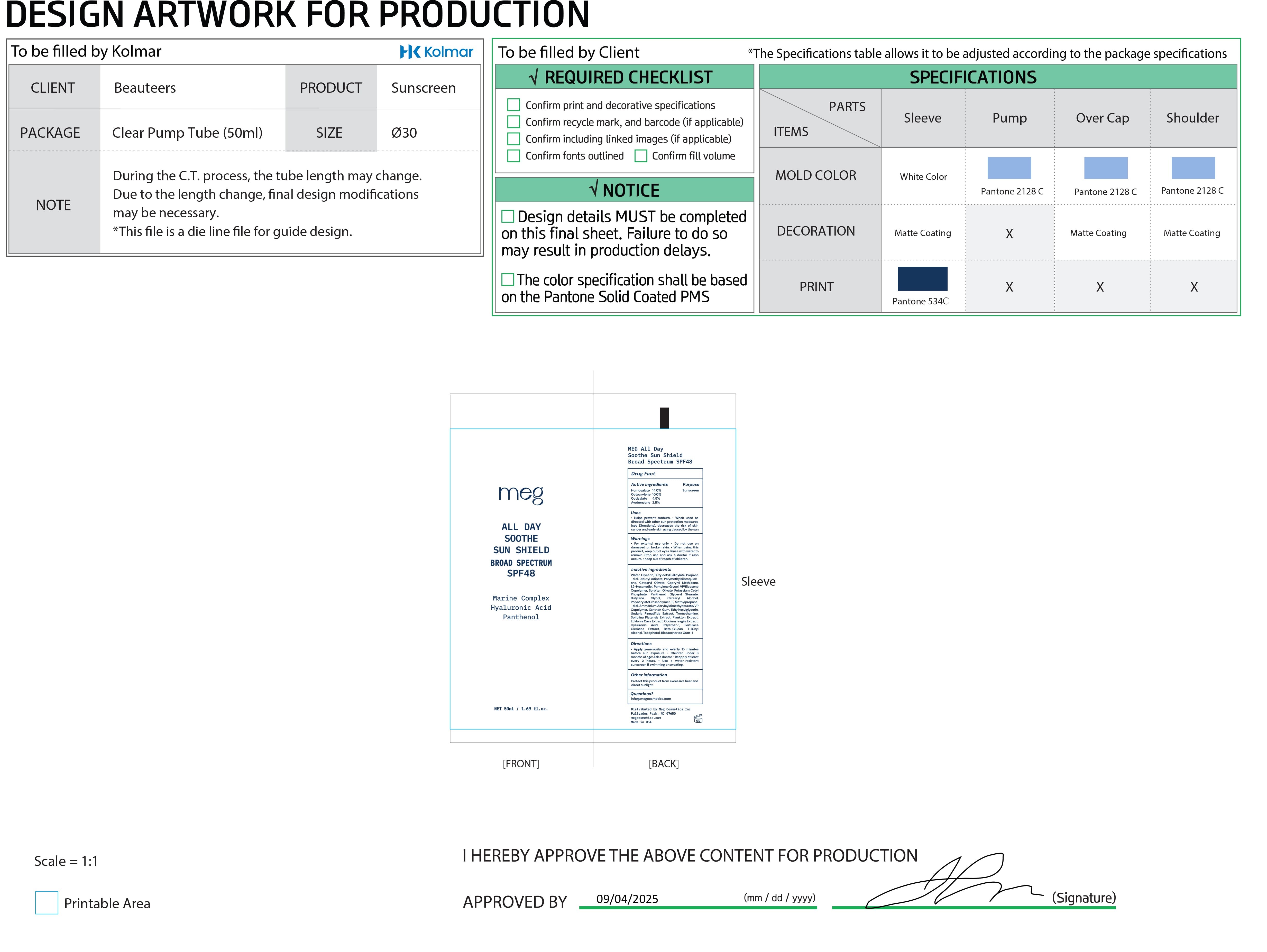
Image of immediate container labeling (inner tube), 50 mL, NDC 87225-001-01
Section 55105-1
Sunscreen
Structured Label Content
Warnings and Precautions (34071-1)
- For external use only.
- Do not use on damaged or broken skin.
- When using this product, keep out of eyes. Rinse with water to remove.
- Stop use and ask a doctor if rash occurs.
- Keep out of reach of children.
Active Ingredient (55106-9)
Homosalate 14.00%
Octocrylene 10.00%
Octisalate 4.50%
Avobenzone 2.80%
Indications and Usage (34067-9)
- Helps prevent sunburn
- When used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun.
Dosage and Administration (34068-7)
- Apply generously and evenly 15 minutes before sun exposure.
- Children under 6 months of age: Ask a doctor.
- Reapply at least every 2 hours.
- Use a water-resistant sunscreen if swimming or sweating.
Section 50565-1 (50565-1)
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50570-1 (50570-1)
No “Do not use” statements apply for this product when used as directed.
Section 51727-6 (51727-6)
Water (Aqua)
Glycerin
Butyloctyl Salicylate
Propanediol
Dibutyl Adipate
Polymethylsilsesquioxane
Cetearyl Olivate
Caprylyl Methicone
1,2-Hexanediol
Pentylene Glycol
VP/Eicosene Copolymer
Sorbitan Olivate
Potassium Cetyl Phosphate
Panthenol
Glyceryl Stearate
Butylene Glycol
Cetearyl Alcohol
Polyacrylate Crosspolymer-6
Methylpropanediol
Ammonium Acryloyldimethyltaurate/VP Copolymer
Xanthan Gum
Ethylhexylglycerin
Undaria Pinnatifida Extract
Tromethamine
Spirulina Platensis Extract
Plankton Extract
Ecklonia Cava Extract
Codium Fragile Extract
Hyaluronic Acid
Polyether-1
Portulaca Oleracea Extract
Beta-Glucan
T-Butyl Alcohol
Tocopherol
Biosaccharide Gum-1
Section 51945-4 (51945-4)
Image of immediate container labeling (inner tube), 50 mL, NDC 87225-001-01
Section 55105-1 (55105-1)
Sunscreen
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:53.442352 · Updated: 2026-03-14T23:11:51.346073