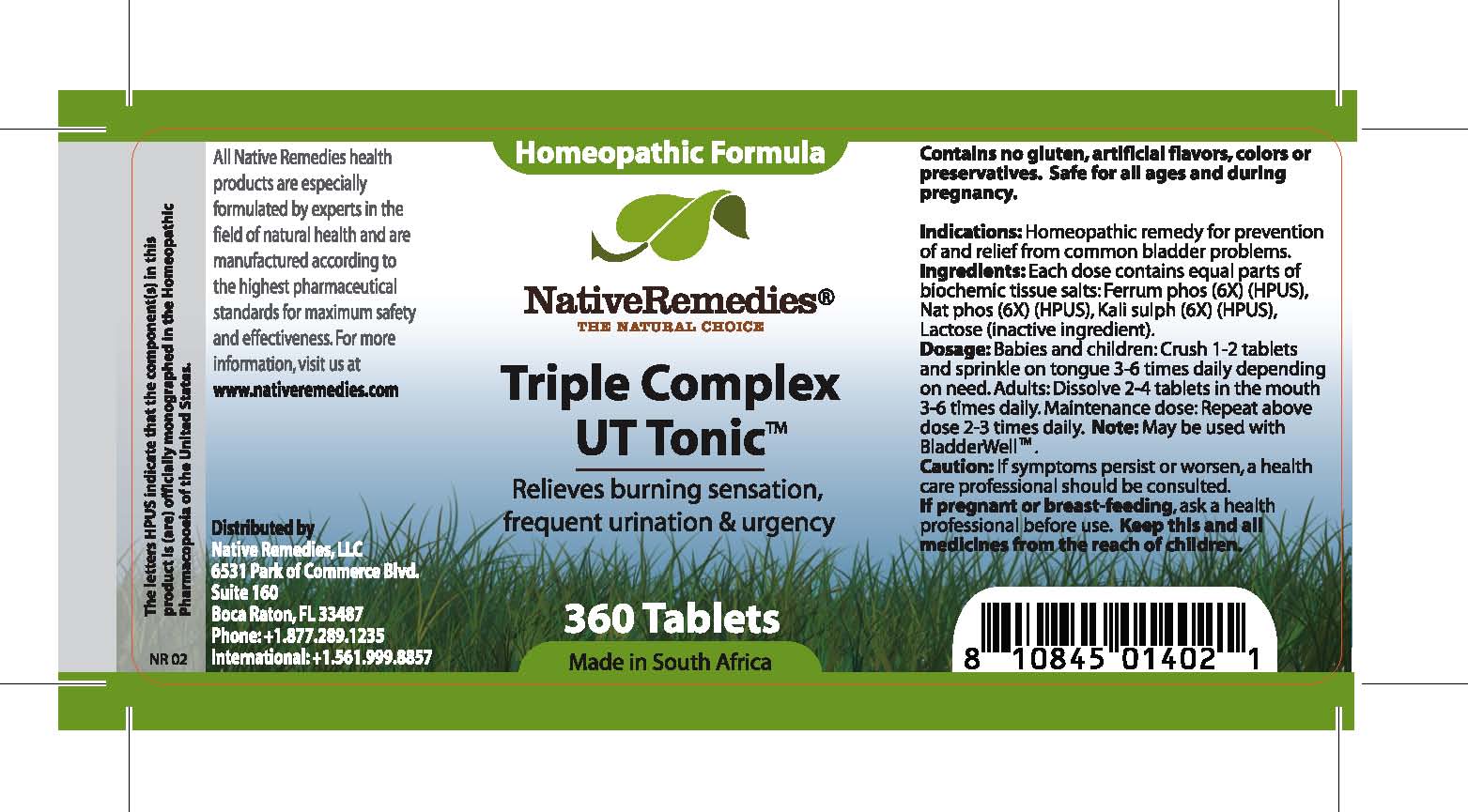
Triple Complex Ut Tonic
45228646-e25a-42c1-9288-b3213dd72dce
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Relieves burning sensation, frequent urination and urgency
Medication Information
Warnings and Precautions
Contains no gluten, artificial flavors, colors or preservatives. Safe for all ages and during pregnancy.
Indications and Usage
Indications: Homeopathic remedy for prevention of and relief from common bladder problems.
Dosage and Administration
Dosage: Babies and children: Crush 1-2 tablets and sprinkle on tongue 3-6 times daily depending on need. Adults: Dissolve 2-4 tablets in the mouth 3-6 times daily. Maintenance dose: Repeat above dose 2-3 times daily. May be used with BladderWell .
Description
Relieves burning sensation, frequent urination and urgency
Section 34072-9
Dosage: Babies and children: Crush 1-2 tablets and sprinkle on tongue 3-6 times daily depending on need. Adults: Dissolve 2-4 tablets in the mouth 3-6 times daily. Maintenance dose: Repeat above dose 2-3 times daily. May be used with BladderWell .
Section 34076-0
All Native Remedies health products are especially formulated by experts in the field of natural health and are manufactured according to the highest pharmaceutical standards for maximum safety and effectiveness. For more information, visit us at www.nativeremedies.com
Distributed by
Native Remedies, LLC
6531 Park of Commerce Blvd.
Suite 160
Boca Raton, FL 33487
Phone: 1.877.289.1235
International: + 1.561.999.8857
The letters HPUS indicate that the component(s) in this product is (are) officially monographed in the Homeopathic Pharmacopoeia of the United States.
Section 42228-7
If pregnant or breastfeeding, ask a health professional before use.
Section 50565-1
Keep this and all medicines from the reach of children.
Section 51727-6
Lactose (inactive ingredient).
Section 51945-4
Section 55105-1
Relieves burning sensation, frequent urination and urgency
Section 55106-9
Ingredients: Each dose contains equal parts of biochemic tissue salts: Ferrum phos (6X) (HPUS), Nat phos (6X) (HPUS), Kali sulph (6X) (HPUS)
Structured Label Content
Indications and Usage (34067-9)
Indications: Homeopathic remedy for prevention of and relief from common bladder problems.
Dosage and Administration (34068-7)
Dosage: Babies and children: Crush 1-2 tablets and sprinkle on tongue 3-6 times daily depending on need. Adults: Dissolve 2-4 tablets in the mouth 3-6 times daily. Maintenance dose: Repeat above dose 2-3 times daily. May be used with BladderWell .
Warnings and Precautions (34071-1)
Contains no gluten, artificial flavors, colors or preservatives. Safe for all ages and during pregnancy.
Section 34072-9 (34072-9)
Dosage: Babies and children: Crush 1-2 tablets and sprinkle on tongue 3-6 times daily depending on need. Adults: Dissolve 2-4 tablets in the mouth 3-6 times daily. Maintenance dose: Repeat above dose 2-3 times daily. May be used with BladderWell .
Section 34076-0 (34076-0)
All Native Remedies health products are especially formulated by experts in the field of natural health and are manufactured according to the highest pharmaceutical standards for maximum safety and effectiveness. For more information, visit us at www.nativeremedies.com
Distributed by
Native Remedies, LLC
6531 Park of Commerce Blvd.
Suite 160
Boca Raton, FL 33487
Phone: 1.877.289.1235
International: + 1.561.999.8857
The letters HPUS indicate that the component(s) in this product is (are) officially monographed in the Homeopathic Pharmacopoeia of the United States.
Section 42228-7 (42228-7)
If pregnant or breastfeeding, ask a health professional before use.
Section 50565-1 (50565-1)
Keep this and all medicines from the reach of children.
Section 51727-6 (51727-6)
Lactose (inactive ingredient).
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Relieves burning sensation, frequent urination and urgency
Section 55106-9 (55106-9)
Ingredients: Each dose contains equal parts of biochemic tissue salts: Ferrum phos (6X) (HPUS), Nat phos (6X) (HPUS), Kali sulph (6X) (HPUS)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:09:07.668006 · Updated: 2026-03-14T23:14:16.282691