These Highlights Do Not Include All The Information Needed To Use Naloxone Hcl Nasal Spray Safely And Effectively. See Full Prescribing Information For Naloxone Hcl Nasal Spray.
4517afa1-4b17-4a15-b91b-65b18bd0558f
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Naloxone HCl Nasal Spray is indicated for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression. Naloxone HCl Nasal Spray is intended for immediate administration as emergency therapy in settings where opioids may be present. Naloxone HCl Nasal Spray is not a substitute for emergency medical care.
Indications and Usage
Naloxone HCl Nasal Spray is indicated for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression. Naloxone HCl Nasal Spray is intended for immediate administration as emergency therapy in settings where opioids may be present. Naloxone HCl Nasal Spray is not a substitute for emergency medical care.
Dosage and Administration
• Naloxone HCl Nasal Spray is for intranasal use only. ( 2.1 ) • Seek emergency medical care immediately after use. ( 2.1 ) • Administration of a single spray of Naloxone HCl Nasal Spray intranasally into one nostril. ( 2.2 ) • Administer additional doses of Naloxone HCl Nasal Spray, using a new nasal spray with each dose, if the patient does not respond or responds and then relapses into respiratory depression, additional doses of Naloxone HCl Nasal Spray may be given every 2 to 3 minutes until emergency medical assistance arrives. ( 2.2 ) • Additional supportive and/or resuscitative measures may be helpful while awaiting emergency medical assistance. ( 2.2 )
Warnings and Precautions
• Risk of Recurrent Respiratory and CNS Depression : Due to the duration of action of naloxone relative to the opioid, keep patient under continued surveillance and administer repeat doses of naloxone using a new nasal spray with each dose, as necessary, while awaiting emergency medical assistance. ( 5.1 ) • Risk of Limited Efficacy with Partial Agonists or Mixed Agonists/Antagonists : Reversal of respiratory depression caused by partial agonists or mixed agonists/antagonists, such as buprenorphine and pentazocine, may be incomplete. Larger or repeat doses may be required. ( 5.2 ) • Precipitation of Severe Opioid Withdrawal : Use in patients who are opioid dependent may precipitate opioid withdrawal. In neonates, opioid withdrawal may be life-threatening if not recognized and properly treated. Monitor for the development of opioid withdrawal. ( 5.3 ) • Risk of Cardiovascular (CV) Effects : Abrupt postoperative reversal of opioid depression may result in adverse CV effects. These events have primarily occurred in patients who had pre-existing CV disorders or received other drugs that may have similar adverse CV effects. Monitor these patients closely in an appropriate healthcare setting after use of naloxone hydrochloride. ( 5.3 )
Contraindications
Naloxone HCl Nasal Spray is contraindicated in patients known to be hypersensitive to naloxone hydrochloride or to any of the other ingredients.
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling: • Precipitation of Severe Opioid Withdrawal [see Warnings and Precautions (5.3) ] Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to the rates in the clinical studies of another drug and may not reflect the rates observed in practice. The following adverse reactions were observed in a Naloxone HCl Nasal Spray clinical study. In a pharmacokinetic study of 30 healthy adult volunteers exposed to one spray of Naloxone HCl Nasal Spray in one nostril or two sprays of Naloxone HCl Nasal Spray, one in each nostril, the most common adverse reactions were: increased blood pressure, constipation, toothache, muscle spasms, musculoskeletal pain, headache, nasal dryness, nasal edema, nasal congestion, nasal inflammation, rhinalgia, and xeroderma. The following adverse reactions have been identified primarily during post-approval use of naloxone hydrochloride in the post-operative setting. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: Hypotension, hypertension, ventricular tachycardia and fibrillation, dyspnea, pulmonary edema, and cardiac arrest. Death, coma, and encephalopathy have been reported as sequelae of these events. Excessive doses of naloxone hydrochloride in post-operative patients have resulted in significant reversal of analgesia, and have caused agitation. Abrupt reversal of opioid effects in persons who were physically dependent on opioids has precipitated an acute withdrawal syndrome. Signs and symptoms have included: body aches, fever, sweating, runny nose, sneezing, piloerection, yawning, weakness, shivering or trembling, nervousness, restlessness or irritability, diarrhea, nausea or vomiting, abdominal cramps, increased blood pressure, tachycardia. In some patients, there may be aggressive behavior upon abrupt reversal of an opioid overdose. In the neonate, opioid withdrawal signs and symptoms also included convulsions, excessive crying, and hyperactive reflexes.
Storage and Handling
Product: 50090-5908 NDC: 50090-5908-0 .1 mL in a VIAL, SINGLE-DOSE / 2 in a CARTON
How Supplied
Product: 50090-5908 NDC: 50090-5908-0 .1 mL in a VIAL, SINGLE-DOSE / 2 in a CARTON
Medication Information
Warnings and Precautions
• Risk of Recurrent Respiratory and CNS Depression : Due to the duration of action of naloxone relative to the opioid, keep patient under continued surveillance and administer repeat doses of naloxone using a new nasal spray with each dose, as necessary, while awaiting emergency medical assistance. ( 5.1 ) • Risk of Limited Efficacy with Partial Agonists or Mixed Agonists/Antagonists : Reversal of respiratory depression caused by partial agonists or mixed agonists/antagonists, such as buprenorphine and pentazocine, may be incomplete. Larger or repeat doses may be required. ( 5.2 ) • Precipitation of Severe Opioid Withdrawal : Use in patients who are opioid dependent may precipitate opioid withdrawal. In neonates, opioid withdrawal may be life-threatening if not recognized and properly treated. Monitor for the development of opioid withdrawal. ( 5.3 ) • Risk of Cardiovascular (CV) Effects : Abrupt postoperative reversal of opioid depression may result in adverse CV effects. These events have primarily occurred in patients who had pre-existing CV disorders or received other drugs that may have similar adverse CV effects. Monitor these patients closely in an appropriate healthcare setting after use of naloxone hydrochloride. ( 5.3 )
Indications and Usage
Naloxone HCl Nasal Spray is indicated for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression. Naloxone HCl Nasal Spray is intended for immediate administration as emergency therapy in settings where opioids may be present. Naloxone HCl Nasal Spray is not a substitute for emergency medical care.
Dosage and Administration
• Naloxone HCl Nasal Spray is for intranasal use only. ( 2.1 ) • Seek emergency medical care immediately after use. ( 2.1 ) • Administration of a single spray of Naloxone HCl Nasal Spray intranasally into one nostril. ( 2.2 ) • Administer additional doses of Naloxone HCl Nasal Spray, using a new nasal spray with each dose, if the patient does not respond or responds and then relapses into respiratory depression, additional doses of Naloxone HCl Nasal Spray may be given every 2 to 3 minutes until emergency medical assistance arrives. ( 2.2 ) • Additional supportive and/or resuscitative measures may be helpful while awaiting emergency medical assistance. ( 2.2 )
Contraindications
Naloxone HCl Nasal Spray is contraindicated in patients known to be hypersensitive to naloxone hydrochloride or to any of the other ingredients.
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling: • Precipitation of Severe Opioid Withdrawal [see Warnings and Precautions (5.3) ] Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to the rates in the clinical studies of another drug and may not reflect the rates observed in practice. The following adverse reactions were observed in a Naloxone HCl Nasal Spray clinical study. In a pharmacokinetic study of 30 healthy adult volunteers exposed to one spray of Naloxone HCl Nasal Spray in one nostril or two sprays of Naloxone HCl Nasal Spray, one in each nostril, the most common adverse reactions were: increased blood pressure, constipation, toothache, muscle spasms, musculoskeletal pain, headache, nasal dryness, nasal edema, nasal congestion, nasal inflammation, rhinalgia, and xeroderma. The following adverse reactions have been identified primarily during post-approval use of naloxone hydrochloride in the post-operative setting. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: Hypotension, hypertension, ventricular tachycardia and fibrillation, dyspnea, pulmonary edema, and cardiac arrest. Death, coma, and encephalopathy have been reported as sequelae of these events. Excessive doses of naloxone hydrochloride in post-operative patients have resulted in significant reversal of analgesia, and have caused agitation. Abrupt reversal of opioid effects in persons who were physically dependent on opioids has precipitated an acute withdrawal syndrome. Signs and symptoms have included: body aches, fever, sweating, runny nose, sneezing, piloerection, yawning, weakness, shivering or trembling, nervousness, restlessness or irritability, diarrhea, nausea or vomiting, abdominal cramps, increased blood pressure, tachycardia. In some patients, there may be aggressive behavior upon abrupt reversal of an opioid overdose. In the neonate, opioid withdrawal signs and symptoms also included convulsions, excessive crying, and hyperactive reflexes.
Storage and Handling
Product: 50090-5908 NDC: 50090-5908-0 .1 mL in a VIAL, SINGLE-DOSE / 2 in a CARTON
How Supplied
Product: 50090-5908 NDC: 50090-5908-0 .1 mL in a VIAL, SINGLE-DOSE / 2 in a CARTON
Description
Naloxone HCl Nasal Spray is indicated for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression. Naloxone HCl Nasal Spray is intended for immediate administration as emergency therapy in settings where opioids may be present. Naloxone HCl Nasal Spray is not a substitute for emergency medical care.
Section 42229-5
Initial Dosing
The recommended initial dose of Naloxone HCl Nasal Spray in adults and pediatric patients is one spray delivered by intranasal administration into one nostril.
Section 42230-3
|
PATIENT INFORMATION
Naloxone (nal-OX-own) Hydrochloride Nasal Spray |
|||
|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. Issued: 02/2021 |
|||
|
You and your family members or caregivers should read this Patient Information leaflet before an opioid emergency happens. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. |
|||
|
What is the most important information I should know about Naloxone HCl Nasal Spray?
|
|||
|
What is Naloxone HCl Nasal Spray?
|
|||
|
Who should not use Naloxone HCl Nasal Spray?
|
|||
|
What should I tell my healthcare provider before using Naloxone HCl Nasal Spray?
Tell your healthcare provider about the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
|||
|
How should I use Naloxone HCl Nasal Spray?
|
|||
|
What are the possible side effects of Naloxone HCl Nasal Spray?
|
|||
|
|
|
|
|
In infants under 4 weeks old who have been receiving opioids regularly, sudden opioid withdrawal may be life-threatening if not treated the right way. Signs and symptoms include: seizures, crying more than usual, and increased reflexes. |
|||
|
How should I store Naloxone HCl Nasal Spray?
Keep Naloxone HCl Nasal Spray and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of Naloxone HCl Nasal Spray.
|
|||
|
What are the ingredients in Naloxone HCl Nasal Spray?
|
11 Description
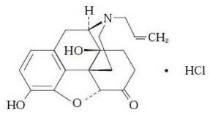
Naloxone Hydrochloride Nasal Spray is a pre-filled, single dose intranasal spray. Chemically, naloxone hydrochloride is the hydrochloride salt of 17-Allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride with the following structure:
|
C19H21NO4∙ HCl |
|
M.W. 363.84 |
Naloxone hydrochloride, an opioid antagonist, occurs as a white to slightly off-white powder, and is soluble in water, in dilute acids, and in strong alkali; slightly soluble in alcohol; practically insoluble in ether and in chloroform.
Each Naloxone HCl Nasal Spray contains a 4 mg single dose of naloxone hydrochloride (equivalent to 3.6 mg of Naloxone) in a 0.1 mL (100 microliter) aqueous solution.
Inactive ingredients include benzalkonium chloride (preservative), disodium ethylenediaminetetraacetate (stabilizer), sodium chloride, hydrochloric acid to adjust pH, and purified water. The pH range is 3.5 to 5.5.
8.4 Pediatric Use
The safety and effectiveness of Naloxone HCl Nasal Spray have been established in pediatric patients of all ages for known or suspected opioid overdose as manifested by respiratory and/or central nervous system depression. Use of naloxone hydrochloride in all pediatric patients is supported by adult bioequivalence studies coupled with evidence from the safe and effective use of other naloxone hydrochloride drug products. No pediatric studies were conducted for Naloxone HCl Nasal Spray.
Absorption of naloxone hydrochloride following intranasal administration in pediatric patients may be erratic or delayed. Even when the opiate-intoxicated pediatric patient responds appropriately to naloxone hydrochloride, he/she must be carefully monitored for at least 24 hours, as a relapse may occur as naloxone hydrochloride is metabolized.
In opioid-dependent pediatric patients, (including neonates), administration of naloxone hydrochloride may result in an abrupt and complete reversal of opioid effects, precipitating an acute opioid withdrawal syndrome. Neonatal opioid withdrawal syndrome, unlike opioid withdrawal syndrome in adults, may be life-threatening, if not recognized, and should be treated according to protocols developed by neonatology experts [see Warnings and Precautions (5.3)].
In settings such as in neonates with known or suspected exposure to maternal opioid use, where it may be preferable to avoid the abrupt precipitation of opioid withdrawal symptoms, consider use of an alternate naloxone-containing product that can be dosed according to weight and titrated to effect.
Also, in situations where the primary concern is for infants at risk for opioid overdose, consider whether the availability of alternate naloxone-containing products may be better suited than Naloxone HCl Nasal Spray.
8.5 Geriatric Use
Geriatric patients have a greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy. Therefore, the systemic exposure of naloxone hydrochloride can be higher in these patients.
Clinical studies of naloxone hydrochloride did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
4 Contraindications
Naloxone HCl Nasal Spray is contraindicated in patients known to be hypersensitive to naloxone hydrochloride or to any of the other ingredients.
6 Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling:
-
•Precipitation of Severe Opioid Withdrawal [see Warnings and Precautions (5.3)]
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to the rates in the clinical studies of another drug and may not reflect the rates observed in practice.
The following adverse reactions were observed in a Naloxone HCl Nasal Spray clinical study.
In a pharmacokinetic study of 30 healthy adult volunteers exposed to one spray of Naloxone HCl Nasal Spray in one nostril or two sprays of Naloxone HCl Nasal Spray, one in each nostril, the most common adverse reactions were: increased blood pressure, constipation, toothache, muscle spasms, musculoskeletal pain, headache, nasal dryness, nasal edema, nasal congestion, nasal inflammation, rhinalgia, and xeroderma.
The following adverse reactions have been identified primarily during post-approval use of naloxone hydrochloride in the post-operative setting. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: Hypotension, hypertension, ventricular tachycardia and fibrillation, dyspnea, pulmonary edema, and cardiac arrest. Death, coma, and encephalopathy have been reported as sequelae of these events. Excessive doses of naloxone hydrochloride in post-operative patients have resulted in significant reversal of analgesia, and have caused agitation.
Abrupt reversal of opioid effects in persons who were physically dependent on opioids has precipitated an acute withdrawal syndrome. Signs and symptoms have included: body aches, fever, sweating, runny nose, sneezing, piloerection, yawning, weakness, shivering or trembling, nervousness, restlessness or irritability, diarrhea, nausea or vomiting, abdominal cramps, increased blood pressure, tachycardia. In some patients, there may be aggressive behavior upon abrupt reversal of an opioid overdose. In the neonate, opioid withdrawal signs and symptoms also included convulsions, excessive crying, and hyperactive reflexes.
Instructions for Use
You and your family members or caregivers should read the Instructions for Use that comes with Naloxone HCl Nasal Spray before using it. Talk to your healthcare provider if you and your family members or caregivers have any questions about the use of Naloxone HCl Nasal Spray.
Use Naloxone HCl Nasal Spray for known or suspected opioid overdose in adults and children.
Important: FOR USE IN THE NOSE ONLY.
-
•Do not remove or test the Naloxone HCl Nasal Spray until ready to use.
-
•Each Naloxone HCl Nasal Spray has 1 dose and cannot be reused.
-
•You do not need to prime Naloxone HCl Nasal Spray.
How to use Naloxone HCl Nasal Spray:
|
Step 1. |
Lay the person on their back to receive a dose of Naloxone HCl Nasal Spray. |
|
|
Step 2. |
Remove Naloxone HCl Nasal Spray from the box. Peel back the tab with the circle to open the Naloxone HCl Nasal Spray. |
|
|
Note: Naloxone HCl Nasal Spray freezes at temperatures below 5°F (-15°C). If this happens, the device will not spray. Get emergency medical help right away if this happens. Do not wait for Naloxone HCl Nasal Spray to thaw. Naloxone HCl Nasal Spray may still be used if it has been thawed after being previously frozen. |
||
|
Step 3. |
Hold the Naloxone HCl Nasal Spray with your thumb on the bottom of the red plunger and your first and middle fingers on either side of the nozzle. |
|
|
Step 4. |
Tilt the person's head back and provide support under the neck with your hand. Gently insert the tip of the nozzle into one nostril until your fingers on either side of the nozzle are against the bottom of the person's nose. |
|
|
Step 5. |
Press the red plunger firmly to give the dose of Naloxone HCl Nasal Spray. |
|
|
Step 6. |
Remove the Naloxone HCl Nasal Spray from the nostril after giving the dose. |
What to do after Naloxone HCl Nasal Spray has been used:
|
Step 7. |
Get emergency medical help right away. |
|
|
||
|
||
|
Step 8. |
Put the used Naloxone HCl Nasal Spray back into its box. |
|
|
Step 9. |
Throw away (dispose of) the used Naloxone HCl Nasal Spray in a place that is away from children. |
How should I store Naloxone HCl Nasal Spray?
-
•Store below 77°F (25°C).
-
•Excursions permitted up to 104°F (40°C).
-
•Do not freeze or expose to excessive heat above 104°F (40°C).
-
•Keep Naloxone HCl Nasal Spray in the box until ready to use. Protect from light.
-
•Replace Naloxone HCl Nasal Spray before the expiration date on the box.
Keep Naloxone HCl Nasal Spray and all medicines out of the reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Distributed by Sandoz Inc., Princeton, NJ 08540.
For more information, call 1-888-511-0081.
Issued: 02/2021
12.2 Pharmacodynamics
When naloxone hydrochloride is administered intravenously, the onset of action is generally apparent within two minutes. The time to onset of action is shorter for intravenous compared to subcutaneous or intramuscular routes of administration. The duration of action is dependent upon the dose and route of administration of naloxone hydrochloride.
12.3 Pharmacokinetics
In a pharmacokinetic study in 30 healthy adult subjects, the relative bioavailability (BA) of one nasal spray in one nostril, consisting of a 2 mg total dose (0.1 mL of 20 mg/mL naloxone hydrochloride solution) and a 4 mg total dose (0.1 mL of 40 mg/mL naloxone hydrochloride solution), and two nasal sprays administered as one nasal spray in each nostril, consisting of a 4 mg total dose (0.1 mL of 20 mg/mL naloxone hydrochloride solution in each nostril) and an 8 mg total dose (0.1 mL of 40 mg/mL naloxone hydrochloride solution in each nostril), were compared to a single dose of 0.4 mg naloxone hydrochloride intramuscular injection. For intranasal administration, the subjects were instructed not to breathe through the nose during administration of the nasal spray, and remained fully supine for approximately one hour post-dose. For intramuscular administration, naloxone was administered as a single injection in the gluteus maximus muscle. The pharmacokinetic parameters obtained in the study are shown in Table 1.
| Parameter |
2 mg – One Nasal Spray in one nostril
20 mg/ml (N=29) |
4 mg – Two Nasal Sprays, one in each nostril
20 mg/ml (N=29) |
4 mg – One Nasal Spray in one nostril
40 mg/ml (N=29) |
8 mg –Two Nasal Sprays, one in each nostril
40 mg/ml (N=29) |
0.4 mg Intramuscular Injection
(N=29) |
|---|---|---|---|---|---|
|
tmax (h) tmax reported as median (minimum, maximum)
|
0.33 (0.25, 1.00) |
0.33 (0.17, 0.57) |
0.50 (0.17, 1.00) |
0.33 (0.17, 1.00) |
0.38 (0.08, 2.05) |
|
Cmax (ng/mL) |
2.91 (35) |
6.30 (34) |
4.83 (43) |
9.70 (36) |
0.88 (31) |
|
AUCt (hr.ng/mL) |
4.60 (27) |
9.64 (24) |
7.87 (37) |
15.3 (23) |
1.75 (23) |
|
AUC0-inf (h*ng/mL) |
4.66 (27) |
9.74 (24) |
7.95 (37) |
15.5 (23) |
1.79 (23) |
|
t½ (h) |
1.85 (33) |
2.19 (33) |
2.08 (30) |
2.10 (32) |
1.24 (26) |
|
Dose normalized Relative BA (%) vs. IM |
51.7 (22) |
54.0 (23) |
44.2 (31) N=28 for Relative BA.
|
43.1 (24) |
100 |
|
(a)
Figure 1 Mean ± SD Plasma Concentration of Naloxone, (a) 0-6 h and (b) 0-1h Following Intranasal Administration and Intramuscular Injection
|
|
(b)
Figure 1 Mean ± SD Plasma Concentration of Naloxone, (a) 0-6 h and (b) 0-1h Following Intranasal Administration and Intramuscular Injection
|
The median naloxone tmax after intranasal administration of Naloxone HCl Nasal Spray (one nasal spray in one nostril (2 mg or 4 mg) or two nasal sprays as one spray in each nostril (4 mg or 8 mg) was not significantly different compared to the 0.4 mg dose of naloxone hydrochloride intramuscular injection (Table 1).
The dose normalized relative bioavailability of one dose (2 mg or 4 mg) or two doses (4 mg or 8 mg) of Naloxone HCl Nasal Spray as compared to the 0.4 mg dose of naloxone hydrochloride administered by intramuscular injection was 52%, 44%, 54%, and 43%, respectively.
Naloxone Hydrochloride
1 Indications and Usage
Naloxone HCl Nasal Spray is indicated for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression.
Naloxone HCl Nasal Spray is intended for immediate administration as emergency therapy in settings where opioids may be present.
Naloxone HCl Nasal Spray is not a substitute for emergency medical care.
12.1 Mechanism of Action
Naloxone hydrochloride is an opioid antagonist that antagonizes opioid effects by competing for the same receptor sites.
Naloxone hydrochloride reverses the effects of opioids, including respiratory depression, sedation, and hypotension. It can also reverse the psychotomimetic and dysphoric effects of agonist-antagonists such as pentazocine.
5 Warnings and Precautions
-
•Risk of Recurrent Respiratory and CNS Depression: Due to the duration of action of naloxone relative to the opioid, keep patient under continued surveillance and administer repeat doses of naloxone using a new nasal spray with each dose, as necessary, while awaiting emergency medical assistance. (5.1)
-
•Risk of Limited Efficacy with Partial Agonists or Mixed Agonists/Antagonists: Reversal of respiratory depression caused by partial agonists or mixed agonists/antagonists, such as buprenorphine and pentazocine, may be incomplete. Larger or repeat doses may be required. (5.2)
-
•Precipitation of Severe Opioid Withdrawal: Use in patients who are opioid dependent may precipitate opioid withdrawal. In neonates, opioid withdrawal may be life-threatening if not recognized and properly treated. Monitor for the development of opioid withdrawal. (5.3)
-
•Risk of Cardiovascular (CV) Effects: Abrupt postoperative reversal of opioid depression may result in adverse CV effects. These events have primarily occurred in patients who had pre-existing CV disorders or received other drugs that may have similar adverse CV effects. Monitor these patients closely in an appropriate healthcare setting after use of naloxone hydrochloride. (5.3)
2 Dosage and Administration
-
•Naloxone HCl Nasal Spray is for intranasal use only. (2.1)
-
•Seek emergency medical care immediately after use. (2.1)
-
•Administration of a single spray of Naloxone HCl Nasal Spray intranasally into one nostril. (2.2)
-
•Administer additional doses of Naloxone HCl Nasal Spray, using a new nasal spray with each dose, if the patient does not respond or responds and then relapses into respiratory depression, additional doses of Naloxone HCl Nasal Spray may be given every 2 to 3 minutes until emergency medical assistance arrives. (2.2)
-
•Additional supportive and/or resuscitative measures may be helpful while awaiting emergency medical assistance. (2.2)
3 Dosage Forms and Strengths
Naloxone HCl Nasal Spray is supplied as a single-dose intranasal spray containing 4 mg of naloxone hydrochloride in 0.1 mL.
17 Patient Counseling Information
Advise the patient and family members or caregivers to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied/storage and Handling
Product: 50090-5908
NDC: 50090-5908-0 .1 mL in a VIAL, SINGLE-DOSE / 2 in a CARTON
2.1 Important Administration Instructions
Naloxone HCl Nasal Spray is for intranasal use only.
No additional device assembly is required.
Because treatment of suspected opioid overdose must be performed by someone other than the patient, instruct the prescription recipient to inform those around them about the presence of Naloxone HCl Nasal Spray and the Instructions for Use.
Instruct the patient or caregiver to read the Instructions for Use at the time they receive a prescription for Naloxone HCl Nasal Spray. Emphasize the following instructions to the patient or caregiver:
-
•Administer Naloxone HCl Nasal Spray as quickly as possible because prolonged respiratory depression may result in damage to the central nervous system or death. Since the duration of action of most opioids exceeds that of naloxone hydrochloride and the suspected opioid overdose may occur outside of supervised medical settings, seek immediate emergency medical assistance, keep the patient under continued surveillance until emergency personnel arrive, and administer repeated doses of Naloxone HCl Nasal Spray, as necessary. Always seek emergency medical assistance in the event of a suspected, potentially life-threatening opioid emergency after administration of the first dose of Naloxone HCl Nasal Spray.
-
•Additional doses of Naloxone HCl Nasal Spray may be required until emergency medical assistance becomes available.
-
•Do not attempt to reuse Naloxone HCl Nasal Spray. Each Naloxone HCl Nasal Spray contains a single dose of naloxone and cannot be reused.
-
•Re-administer Naloxone HCl Nasal Spray, using a new nasal spray, every 2 to 3 minutes if the patient does not respond or responds and then relapses into respiratory depression.
-
•Administer Naloxone HCl Nasal Spray in alternate nostrils with each dose.
-
•Administer Naloxone HCl Nasal Spray according to the printed instructions on the device label and the Instructions for Use.
-
•Place the patient in the supine position. Prior to administration, be sure the device nozzle is inserted in either nostril of the patient, and provide support to the back of the neck to allow the head to tilt back. Do not prime or test the device prior to administration.
-
•To administer the dose press firmly on the device plunger.
-
•Remove the device nozzle from the nostril after use.
-
•Turn patient on their side as shown in the Instructions for Use and call for emergency medical assistance immediately after administration of the first dose of Naloxone HCl Nasal Spray.
5.3 Precipitation of Severe Opioid Withdrawal
The use of Naloxone HCl Nasal Spray in patients who are opioid-dependent may precipitate opioid withdrawal characterized by the following signs and symptoms: body aches, diarrhea, tachycardia, fever, runny nose, sneezing, piloerection, sweating, yawning, nausea or vomiting, nervousness, restlessness or irritability, shivering or trembling, abdominal cramps, weakness, and increased blood pressure. In neonates, opioid withdrawal may be life-threatening if not recognized and properly treated and may include the following signs and symptoms: convulsions, excessive crying, and hyperactive reflexes. Monitor the patient for the development of the signs and symptoms of opioid withdrawal.
There are limited data to inform if the 2 mg dose of Naloxone HCl Nasal Spray will avoid precipitation of severe opioid withdrawal in the setting of opioid dependence. However, the 2 mg dose may not provide an adequate and timely reversal in persons who may be exposed to an overdose of a potent or very high dose of opioids.
Abrupt postoperative reversal of opioid depression after using naloxone hydrochloride may result in nausea, vomiting, sweating, tremulousness, tachycardia, hypotension, hypertension, seizures, ventricular tachycardia and fibrillation, pulmonary edema, and cardiac arrest. Death, coma, and encephalopathy have been reported as sequelae of these events. These events have primarily occurred in patients who had pre-existing cardiovascular disorders or received other drugs that may have similar adverse cardiovascular effects. Although a direct cause and effect relationship has not been established, after use of naloxone hydrochloride, monitor patients with pre-existing cardiac disease or patients who have received medications with potential adverse cardiovascular effects for hypotension, ventricular tachycardia or fibrillation, and pulmonary edema in an appropriate healthcare setting. It has been suggested that the pathogenesis of pulmonary edema associated with the use of naloxone hydrochloride is similar to neurogenic pulmonary edema, i.e., a centrally mediated massive catecholamine response leading to a dramatic shift of blood volume into the pulmonary vascular bed resulting in increased hydrostatic pressures.
There may be clinical settings, particularly the postpartum period in neonates with known or suspected exposure to maternal opioid use, where it is preferable to avoid the abrupt precipitation of opioid withdrawal symptoms. In these settings, consider use of an alternative, naloxone-containing product that can be titrated to effect and, where applicable, dosed according to weight. [see Use in Specific Populations (8.4)].
5.1 Risk of Recurrent Respiratory and Central Nervous System Depression
The duration of action of most opioids may exceed that of Naloxone HCl Nasal Spray resulting in a return of respiratory and/or central nervous system depression after an initial improvement in symptoms. Therefore, it is necessary to seek emergency medical assistance immediately after administration of the first dose of Naloxone HCl Nasal Spray and to keep the patient under continued surveillance. Administer additional doses of Naloxone HCl Nasal Spray if the patient is not adequately responding or responds and then relapses back into respiratory depression, as necessary [see Dosage and Administration (2.2)]. Additional supportive and/or resuscitative measures may be helpful while awaiting emergency medical assistance.
2.3 Dosing Modifications Due to Partial Agonists Or Mixed Agonist/antagonists
Reversal of respiratory depression by partial agonists or mixed agonist/antagonists, such as buprenorphine and pentazocine, may be incomplete and require higher doses of naloxone hydrochloride or repeated administration of Naloxone HCl Nasal Spray using a new nasal spray [see Warnings and Precautions (5.2)].
5.2 Risk of Limited Efficacy With Partial Agonists Or Mixed Agonist/antagonists
Reversal of respiratory depression by partial agonists or mixed agonist/antagonists such as buprenorphine and pentazocine, may be incomplete. Larger or repeat doses of naloxone hydrochloride may be required to antagonize buprenorphine because the latter has a long duration of action due to its slow rate of binding and subsequent slow dissociation from the opioid receptor [see Dosage and Administration (2.3)]. Buprenorphine antagonism is characterized by a gradual onset of the reversal effects and a decreased duration of action of the normally prolonged respiratory depression.
Structured Label Content
Section 42229-5 (42229-5)
Initial Dosing
The recommended initial dose of Naloxone HCl Nasal Spray in adults and pediatric patients is one spray delivered by intranasal administration into one nostril.
Section 42230-3 (42230-3)
|
PATIENT INFORMATION
Naloxone (nal-OX-own) Hydrochloride Nasal Spray |
|||
|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. Issued: 02/2021 |
|||
|
You and your family members or caregivers should read this Patient Information leaflet before an opioid emergency happens. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. |
|||
|
What is the most important information I should know about Naloxone HCl Nasal Spray?
|
|||
|
What is Naloxone HCl Nasal Spray?
|
|||
|
Who should not use Naloxone HCl Nasal Spray?
|
|||
|
What should I tell my healthcare provider before using Naloxone HCl Nasal Spray?
Tell your healthcare provider about the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
|||
|
How should I use Naloxone HCl Nasal Spray?
|
|||
|
What are the possible side effects of Naloxone HCl Nasal Spray?
|
|||
|
|
|
|
|
In infants under 4 weeks old who have been receiving opioids regularly, sudden opioid withdrawal may be life-threatening if not treated the right way. Signs and symptoms include: seizures, crying more than usual, and increased reflexes. |
|||
|
How should I store Naloxone HCl Nasal Spray?
Keep Naloxone HCl Nasal Spray and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of Naloxone HCl Nasal Spray.
|
|||
|
What are the ingredients in Naloxone HCl Nasal Spray?
|
11 Description (11 DESCRIPTION)
Naloxone Hydrochloride Nasal Spray is a pre-filled, single dose intranasal spray. Chemically, naloxone hydrochloride is the hydrochloride salt of 17-Allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride with the following structure:
|
C19H21NO4∙ HCl |
|
M.W. 363.84 |
Naloxone hydrochloride, an opioid antagonist, occurs as a white to slightly off-white powder, and is soluble in water, in dilute acids, and in strong alkali; slightly soluble in alcohol; practically insoluble in ether and in chloroform.
Each Naloxone HCl Nasal Spray contains a 4 mg single dose of naloxone hydrochloride (equivalent to 3.6 mg of Naloxone) in a 0.1 mL (100 microliter) aqueous solution.
Inactive ingredients include benzalkonium chloride (preservative), disodium ethylenediaminetetraacetate (stabilizer), sodium chloride, hydrochloric acid to adjust pH, and purified water. The pH range is 3.5 to 5.5.
8.4 Pediatric Use
The safety and effectiveness of Naloxone HCl Nasal Spray have been established in pediatric patients of all ages for known or suspected opioid overdose as manifested by respiratory and/or central nervous system depression. Use of naloxone hydrochloride in all pediatric patients is supported by adult bioequivalence studies coupled with evidence from the safe and effective use of other naloxone hydrochloride drug products. No pediatric studies were conducted for Naloxone HCl Nasal Spray.
Absorption of naloxone hydrochloride following intranasal administration in pediatric patients may be erratic or delayed. Even when the opiate-intoxicated pediatric patient responds appropriately to naloxone hydrochloride, he/she must be carefully monitored for at least 24 hours, as a relapse may occur as naloxone hydrochloride is metabolized.
In opioid-dependent pediatric patients, (including neonates), administration of naloxone hydrochloride may result in an abrupt and complete reversal of opioid effects, precipitating an acute opioid withdrawal syndrome. Neonatal opioid withdrawal syndrome, unlike opioid withdrawal syndrome in adults, may be life-threatening, if not recognized, and should be treated according to protocols developed by neonatology experts [see Warnings and Precautions (5.3)].
In settings such as in neonates with known or suspected exposure to maternal opioid use, where it may be preferable to avoid the abrupt precipitation of opioid withdrawal symptoms, consider use of an alternate naloxone-containing product that can be dosed according to weight and titrated to effect.
Also, in situations where the primary concern is for infants at risk for opioid overdose, consider whether the availability of alternate naloxone-containing products may be better suited than Naloxone HCl Nasal Spray.
8.5 Geriatric Use
Geriatric patients have a greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy. Therefore, the systemic exposure of naloxone hydrochloride can be higher in these patients.
Clinical studies of naloxone hydrochloride did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
4 Contraindications (4 CONTRAINDICATIONS)
Naloxone HCl Nasal Spray is contraindicated in patients known to be hypersensitive to naloxone hydrochloride or to any of the other ingredients.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed elsewhere in the labeling:
-
•Precipitation of Severe Opioid Withdrawal [see Warnings and Precautions (5.3)]
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to the rates in the clinical studies of another drug and may not reflect the rates observed in practice.
The following adverse reactions were observed in a Naloxone HCl Nasal Spray clinical study.
In a pharmacokinetic study of 30 healthy adult volunteers exposed to one spray of Naloxone HCl Nasal Spray in one nostril or two sprays of Naloxone HCl Nasal Spray, one in each nostril, the most common adverse reactions were: increased blood pressure, constipation, toothache, muscle spasms, musculoskeletal pain, headache, nasal dryness, nasal edema, nasal congestion, nasal inflammation, rhinalgia, and xeroderma.
The following adverse reactions have been identified primarily during post-approval use of naloxone hydrochloride in the post-operative setting. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: Hypotension, hypertension, ventricular tachycardia and fibrillation, dyspnea, pulmonary edema, and cardiac arrest. Death, coma, and encephalopathy have been reported as sequelae of these events. Excessive doses of naloxone hydrochloride in post-operative patients have resulted in significant reversal of analgesia, and have caused agitation.
Abrupt reversal of opioid effects in persons who were physically dependent on opioids has precipitated an acute withdrawal syndrome. Signs and symptoms have included: body aches, fever, sweating, runny nose, sneezing, piloerection, yawning, weakness, shivering or trembling, nervousness, restlessness or irritability, diarrhea, nausea or vomiting, abdominal cramps, increased blood pressure, tachycardia. In some patients, there may be aggressive behavior upon abrupt reversal of an opioid overdose. In the neonate, opioid withdrawal signs and symptoms also included convulsions, excessive crying, and hyperactive reflexes.
Instructions for Use
You and your family members or caregivers should read the Instructions for Use that comes with Naloxone HCl Nasal Spray before using it. Talk to your healthcare provider if you and your family members or caregivers have any questions about the use of Naloxone HCl Nasal Spray.
Use Naloxone HCl Nasal Spray for known or suspected opioid overdose in adults and children.
Important: FOR USE IN THE NOSE ONLY.
-
•Do not remove or test the Naloxone HCl Nasal Spray until ready to use.
-
•Each Naloxone HCl Nasal Spray has 1 dose and cannot be reused.
-
•You do not need to prime Naloxone HCl Nasal Spray.
How to use Naloxone HCl Nasal Spray:
|
Step 1. |
Lay the person on their back to receive a dose of Naloxone HCl Nasal Spray. |
|
|
Step 2. |
Remove Naloxone HCl Nasal Spray from the box. Peel back the tab with the circle to open the Naloxone HCl Nasal Spray. |
|
|
Note: Naloxone HCl Nasal Spray freezes at temperatures below 5°F (-15°C). If this happens, the device will not spray. Get emergency medical help right away if this happens. Do not wait for Naloxone HCl Nasal Spray to thaw. Naloxone HCl Nasal Spray may still be used if it has been thawed after being previously frozen. |
||
|
Step 3. |
Hold the Naloxone HCl Nasal Spray with your thumb on the bottom of the red plunger and your first and middle fingers on either side of the nozzle. |
|
|
Step 4. |
Tilt the person's head back and provide support under the neck with your hand. Gently insert the tip of the nozzle into one nostril until your fingers on either side of the nozzle are against the bottom of the person's nose. |
|
|
Step 5. |
Press the red plunger firmly to give the dose of Naloxone HCl Nasal Spray. |
|
|
Step 6. |
Remove the Naloxone HCl Nasal Spray from the nostril after giving the dose. |
What to do after Naloxone HCl Nasal Spray has been used:
|
Step 7. |
Get emergency medical help right away. |
|
|
||
|
||
|
Step 8. |
Put the used Naloxone HCl Nasal Spray back into its box. |
|
|
Step 9. |
Throw away (dispose of) the used Naloxone HCl Nasal Spray in a place that is away from children. |
How should I store Naloxone HCl Nasal Spray?
-
•Store below 77°F (25°C).
-
•Excursions permitted up to 104°F (40°C).
-
•Do not freeze or expose to excessive heat above 104°F (40°C).
-
•Keep Naloxone HCl Nasal Spray in the box until ready to use. Protect from light.
-
•Replace Naloxone HCl Nasal Spray before the expiration date on the box.
Keep Naloxone HCl Nasal Spray and all medicines out of the reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Distributed by Sandoz Inc., Princeton, NJ 08540.
For more information, call 1-888-511-0081.
Issued: 02/2021
12.2 Pharmacodynamics
When naloxone hydrochloride is administered intravenously, the onset of action is generally apparent within two minutes. The time to onset of action is shorter for intravenous compared to subcutaneous or intramuscular routes of administration. The duration of action is dependent upon the dose and route of administration of naloxone hydrochloride.
12.3 Pharmacokinetics
In a pharmacokinetic study in 30 healthy adult subjects, the relative bioavailability (BA) of one nasal spray in one nostril, consisting of a 2 mg total dose (0.1 mL of 20 mg/mL naloxone hydrochloride solution) and a 4 mg total dose (0.1 mL of 40 mg/mL naloxone hydrochloride solution), and two nasal sprays administered as one nasal spray in each nostril, consisting of a 4 mg total dose (0.1 mL of 20 mg/mL naloxone hydrochloride solution in each nostril) and an 8 mg total dose (0.1 mL of 40 mg/mL naloxone hydrochloride solution in each nostril), were compared to a single dose of 0.4 mg naloxone hydrochloride intramuscular injection. For intranasal administration, the subjects were instructed not to breathe through the nose during administration of the nasal spray, and remained fully supine for approximately one hour post-dose. For intramuscular administration, naloxone was administered as a single injection in the gluteus maximus muscle. The pharmacokinetic parameters obtained in the study are shown in Table 1.
| Parameter |
2 mg – One Nasal Spray in one nostril
20 mg/ml (N=29) |
4 mg – Two Nasal Sprays, one in each nostril
20 mg/ml (N=29) |
4 mg – One Nasal Spray in one nostril
40 mg/ml (N=29) |
8 mg –Two Nasal Sprays, one in each nostril
40 mg/ml (N=29) |
0.4 mg Intramuscular Injection
(N=29) |
|---|---|---|---|---|---|
|
tmax (h) tmax reported as median (minimum, maximum)
|
0.33 (0.25, 1.00) |
0.33 (0.17, 0.57) |
0.50 (0.17, 1.00) |
0.33 (0.17, 1.00) |
0.38 (0.08, 2.05) |
|
Cmax (ng/mL) |
2.91 (35) |
6.30 (34) |
4.83 (43) |
9.70 (36) |
0.88 (31) |
|
AUCt (hr.ng/mL) |
4.60 (27) |
9.64 (24) |
7.87 (37) |
15.3 (23) |
1.75 (23) |
|
AUC0-inf (h*ng/mL) |
4.66 (27) |
9.74 (24) |
7.95 (37) |
15.5 (23) |
1.79 (23) |
|
t½ (h) |
1.85 (33) |
2.19 (33) |
2.08 (30) |
2.10 (32) |
1.24 (26) |
|
Dose normalized Relative BA (%) vs. IM |
51.7 (22) |
54.0 (23) |
44.2 (31) N=28 for Relative BA.
|
43.1 (24) |
100 |
|
(a)
Figure 1 Mean ± SD Plasma Concentration of Naloxone, (a) 0-6 h and (b) 0-1h Following Intranasal Administration and Intramuscular Injection
|
|
(b)
Figure 1 Mean ± SD Plasma Concentration of Naloxone, (a) 0-6 h and (b) 0-1h Following Intranasal Administration and Intramuscular Injection
|
The median naloxone tmax after intranasal administration of Naloxone HCl Nasal Spray (one nasal spray in one nostril (2 mg or 4 mg) or two nasal sprays as one spray in each nostril (4 mg or 8 mg) was not significantly different compared to the 0.4 mg dose of naloxone hydrochloride intramuscular injection (Table 1).
The dose normalized relative bioavailability of one dose (2 mg or 4 mg) or two doses (4 mg or 8 mg) of Naloxone HCl Nasal Spray as compared to the 0.4 mg dose of naloxone hydrochloride administered by intramuscular injection was 52%, 44%, 54%, and 43%, respectively.
Naloxone Hydrochloride (NALOXONE HYDROCHLORIDE)
1 Indications and Usage (1 INDICATIONS AND USAGE)
Naloxone HCl Nasal Spray is indicated for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression.
Naloxone HCl Nasal Spray is intended for immediate administration as emergency therapy in settings where opioids may be present.
Naloxone HCl Nasal Spray is not a substitute for emergency medical care.
12.1 Mechanism of Action
Naloxone hydrochloride is an opioid antagonist that antagonizes opioid effects by competing for the same receptor sites.
Naloxone hydrochloride reverses the effects of opioids, including respiratory depression, sedation, and hypotension. It can also reverse the psychotomimetic and dysphoric effects of agonist-antagonists such as pentazocine.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Risk of Recurrent Respiratory and CNS Depression: Due to the duration of action of naloxone relative to the opioid, keep patient under continued surveillance and administer repeat doses of naloxone using a new nasal spray with each dose, as necessary, while awaiting emergency medical assistance. (5.1)
-
•Risk of Limited Efficacy with Partial Agonists or Mixed Agonists/Antagonists: Reversal of respiratory depression caused by partial agonists or mixed agonists/antagonists, such as buprenorphine and pentazocine, may be incomplete. Larger or repeat doses may be required. (5.2)
-
•Precipitation of Severe Opioid Withdrawal: Use in patients who are opioid dependent may precipitate opioid withdrawal. In neonates, opioid withdrawal may be life-threatening if not recognized and properly treated. Monitor for the development of opioid withdrawal. (5.3)
-
•Risk of Cardiovascular (CV) Effects: Abrupt postoperative reversal of opioid depression may result in adverse CV effects. These events have primarily occurred in patients who had pre-existing CV disorders or received other drugs that may have similar adverse CV effects. Monitor these patients closely in an appropriate healthcare setting after use of naloxone hydrochloride. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Naloxone HCl Nasal Spray is for intranasal use only. (2.1)
-
•Seek emergency medical care immediately after use. (2.1)
-
•Administration of a single spray of Naloxone HCl Nasal Spray intranasally into one nostril. (2.2)
-
•Administer additional doses of Naloxone HCl Nasal Spray, using a new nasal spray with each dose, if the patient does not respond or responds and then relapses into respiratory depression, additional doses of Naloxone HCl Nasal Spray may be given every 2 to 3 minutes until emergency medical assistance arrives. (2.2)
-
•Additional supportive and/or resuscitative measures may be helpful while awaiting emergency medical assistance. (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Naloxone HCl Nasal Spray is supplied as a single-dose intranasal spray containing 4 mg of naloxone hydrochloride in 0.1 mL.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient and family members or caregivers to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Product: 50090-5908
NDC: 50090-5908-0 .1 mL in a VIAL, SINGLE-DOSE / 2 in a CARTON
2.1 Important Administration Instructions
Naloxone HCl Nasal Spray is for intranasal use only.
No additional device assembly is required.
Because treatment of suspected opioid overdose must be performed by someone other than the patient, instruct the prescription recipient to inform those around them about the presence of Naloxone HCl Nasal Spray and the Instructions for Use.
Instruct the patient or caregiver to read the Instructions for Use at the time they receive a prescription for Naloxone HCl Nasal Spray. Emphasize the following instructions to the patient or caregiver:
-
•Administer Naloxone HCl Nasal Spray as quickly as possible because prolonged respiratory depression may result in damage to the central nervous system or death. Since the duration of action of most opioids exceeds that of naloxone hydrochloride and the suspected opioid overdose may occur outside of supervised medical settings, seek immediate emergency medical assistance, keep the patient under continued surveillance until emergency personnel arrive, and administer repeated doses of Naloxone HCl Nasal Spray, as necessary. Always seek emergency medical assistance in the event of a suspected, potentially life-threatening opioid emergency after administration of the first dose of Naloxone HCl Nasal Spray.
-
•Additional doses of Naloxone HCl Nasal Spray may be required until emergency medical assistance becomes available.
-
•Do not attempt to reuse Naloxone HCl Nasal Spray. Each Naloxone HCl Nasal Spray contains a single dose of naloxone and cannot be reused.
-
•Re-administer Naloxone HCl Nasal Spray, using a new nasal spray, every 2 to 3 minutes if the patient does not respond or responds and then relapses into respiratory depression.
-
•Administer Naloxone HCl Nasal Spray in alternate nostrils with each dose.
-
•Administer Naloxone HCl Nasal Spray according to the printed instructions on the device label and the Instructions for Use.
-
•Place the patient in the supine position. Prior to administration, be sure the device nozzle is inserted in either nostril of the patient, and provide support to the back of the neck to allow the head to tilt back. Do not prime or test the device prior to administration.
-
•To administer the dose press firmly on the device plunger.
-
•Remove the device nozzle from the nostril after use.
-
•Turn patient on their side as shown in the Instructions for Use and call for emergency medical assistance immediately after administration of the first dose of Naloxone HCl Nasal Spray.
5.3 Precipitation of Severe Opioid Withdrawal
The use of Naloxone HCl Nasal Spray in patients who are opioid-dependent may precipitate opioid withdrawal characterized by the following signs and symptoms: body aches, diarrhea, tachycardia, fever, runny nose, sneezing, piloerection, sweating, yawning, nausea or vomiting, nervousness, restlessness or irritability, shivering or trembling, abdominal cramps, weakness, and increased blood pressure. In neonates, opioid withdrawal may be life-threatening if not recognized and properly treated and may include the following signs and symptoms: convulsions, excessive crying, and hyperactive reflexes. Monitor the patient for the development of the signs and symptoms of opioid withdrawal.
There are limited data to inform if the 2 mg dose of Naloxone HCl Nasal Spray will avoid precipitation of severe opioid withdrawal in the setting of opioid dependence. However, the 2 mg dose may not provide an adequate and timely reversal in persons who may be exposed to an overdose of a potent or very high dose of opioids.
Abrupt postoperative reversal of opioid depression after using naloxone hydrochloride may result in nausea, vomiting, sweating, tremulousness, tachycardia, hypotension, hypertension, seizures, ventricular tachycardia and fibrillation, pulmonary edema, and cardiac arrest. Death, coma, and encephalopathy have been reported as sequelae of these events. These events have primarily occurred in patients who had pre-existing cardiovascular disorders or received other drugs that may have similar adverse cardiovascular effects. Although a direct cause and effect relationship has not been established, after use of naloxone hydrochloride, monitor patients with pre-existing cardiac disease or patients who have received medications with potential adverse cardiovascular effects for hypotension, ventricular tachycardia or fibrillation, and pulmonary edema in an appropriate healthcare setting. It has been suggested that the pathogenesis of pulmonary edema associated with the use of naloxone hydrochloride is similar to neurogenic pulmonary edema, i.e., a centrally mediated massive catecholamine response leading to a dramatic shift of blood volume into the pulmonary vascular bed resulting in increased hydrostatic pressures.
There may be clinical settings, particularly the postpartum period in neonates with known or suspected exposure to maternal opioid use, where it is preferable to avoid the abrupt precipitation of opioid withdrawal symptoms. In these settings, consider use of an alternative, naloxone-containing product that can be titrated to effect and, where applicable, dosed according to weight. [see Use in Specific Populations (8.4)].
5.1 Risk of Recurrent Respiratory and Central Nervous System Depression
The duration of action of most opioids may exceed that of Naloxone HCl Nasal Spray resulting in a return of respiratory and/or central nervous system depression after an initial improvement in symptoms. Therefore, it is necessary to seek emergency medical assistance immediately after administration of the first dose of Naloxone HCl Nasal Spray and to keep the patient under continued surveillance. Administer additional doses of Naloxone HCl Nasal Spray if the patient is not adequately responding or responds and then relapses back into respiratory depression, as necessary [see Dosage and Administration (2.2)]. Additional supportive and/or resuscitative measures may be helpful while awaiting emergency medical assistance.
2.3 Dosing Modifications Due to Partial Agonists Or Mixed Agonist/antagonists (2.3 Dosing Modifications due to Partial Agonists or Mixed Agonist/Antagonists)
Reversal of respiratory depression by partial agonists or mixed agonist/antagonists, such as buprenorphine and pentazocine, may be incomplete and require higher doses of naloxone hydrochloride or repeated administration of Naloxone HCl Nasal Spray using a new nasal spray [see Warnings and Precautions (5.2)].
5.2 Risk of Limited Efficacy With Partial Agonists Or Mixed Agonist/antagonists (5.2 Risk of Limited Efficacy with Partial Agonists or Mixed Agonist/Antagonists)
Reversal of respiratory depression by partial agonists or mixed agonist/antagonists such as buprenorphine and pentazocine, may be incomplete. Larger or repeat doses of naloxone hydrochloride may be required to antagonize buprenorphine because the latter has a long duration of action due to its slow rate of binding and subsequent slow dissociation from the opioid receptor [see Dosage and Administration (2.3)]. Buprenorphine antagonism is characterized by a gradual onset of the reversal effects and a decreased duration of action of the normally prolonged respiratory depression.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:30.095416 · Updated: 2026-03-14T21:58:01.008833