Prosoria Medicated Shampoo
44eaaeb0-1400-4697-85d0-e8c2282c1cba
34390-5
HUMAN OTC DRUG LABEL
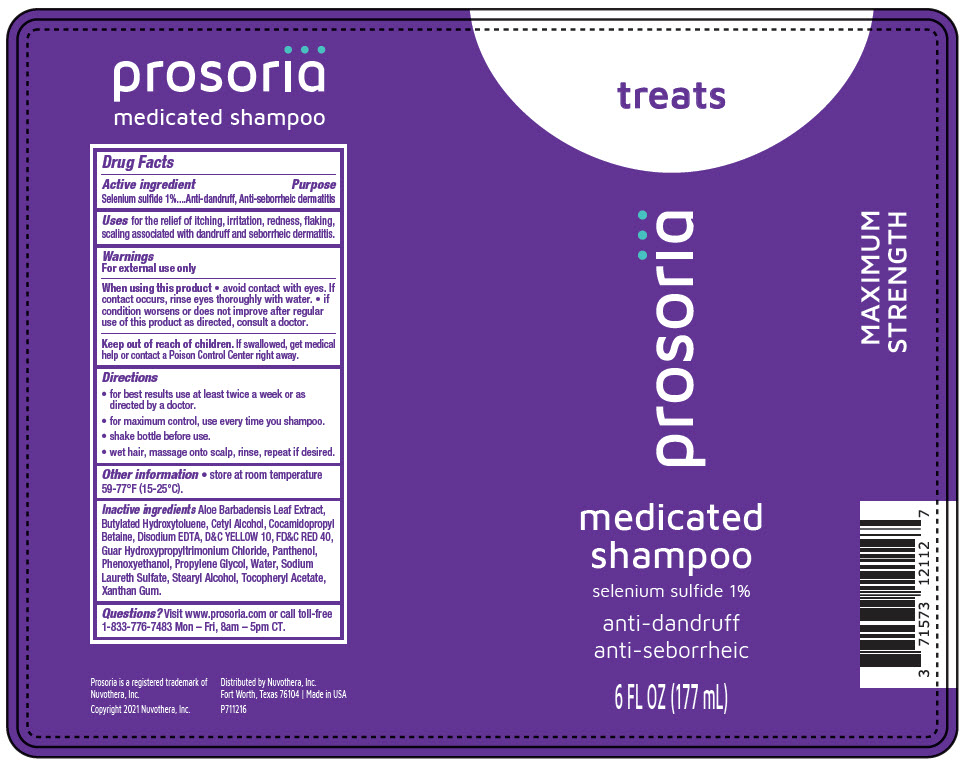
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Anti-dandruff, Anti-seborrheic dermatitis
Medication Information
Purpose
Anti-dandruff, Anti-seborrheic dermatitis
Description
Drug Facts
Uses
for the relief of itching, irritation, redness, flaking, scaling associated with dandruff and seborrheic dermatitis.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Warnings
For external use only
Directions
- for best results use at least twice a week or as directed by a doctor.
- for maximum control, use every time you shampoo.
- shake bottle before use.
- wet hair, massage onto scalp, rinse, repeat if desired.
Questions?
Visit www.prosoria.com or call toll-free 1-833-776-7483 Mon – Fri, 8am – 5pm CT.
Active Ingredient
Selenium sulfide 1%
Other Information
- store at room temperature 59-77°F (15-25°C).
Inactive Ingredients
Aloe Barbadensis Leaf Extract, Butylated Hydroxytoluene, Cetyl Alcohol, Cocamidopropyl Betaine, Disodium EDTA, D&C YELLOW 10, FD&C RED 40, Guar Hydroxypropyltrimonium Chloride, Panthenol, Phenoxyethanol, Propylene Glycol, Water, Sodium Laureth Sulfate, Stearyl Alcohol, Tocopheryl Acetate, Xanthan Gum.
When Using This Product
- avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water.
- if condition worsens or does not improve after regular use of this product as directed, consult a doctor.
Principal Display Panel 177 Ml Bottle Label
treats
prosoriä
medicated
shampoo
selenium sulfide 1%
anti-dandruff
anti-seborrheic
6 FL OZ (177 mL)
Structured Label Content
Uses
for the relief of itching, irritation, redness, flaking, scaling associated with dandruff and seborrheic dermatitis.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Anti-dandruff, Anti-seborrheic dermatitis
Warnings
For external use only
Directions
- for best results use at least twice a week or as directed by a doctor.
- for maximum control, use every time you shampoo.
- shake bottle before use.
- wet hair, massage onto scalp, rinse, repeat if desired.
Questions?
Visit www.prosoria.com or call toll-free 1-833-776-7483 Mon – Fri, 8am – 5pm CT.
Active Ingredient (Active ingredient)
Selenium sulfide 1%
Other Information (Other information)
- store at room temperature 59-77°F (15-25°C).
Inactive Ingredients (Inactive ingredients)
Aloe Barbadensis Leaf Extract, Butylated Hydroxytoluene, Cetyl Alcohol, Cocamidopropyl Betaine, Disodium EDTA, D&C YELLOW 10, FD&C RED 40, Guar Hydroxypropyltrimonium Chloride, Panthenol, Phenoxyethanol, Propylene Glycol, Water, Sodium Laureth Sulfate, Stearyl Alcohol, Tocopheryl Acetate, Xanthan Gum.
When Using This Product (When using this product)
- avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water.
- if condition worsens or does not improve after regular use of this product as directed, consult a doctor.
Principal Display Panel 177 Ml Bottle Label (PRINCIPAL DISPLAY PANEL - 177 mL Bottle Label)
treats
prosoriä
medicated
shampoo
selenium sulfide 1%
anti-dandruff
anti-seborrheic
6 FL OZ (177 mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:40.699362 · Updated: 2026-03-14T23:02:21.986500